Abstract
Purpose
Posterior tibial slope (PTS) for cruciate-retaining (CR) total knee arthroplasty (TKA) is usually pre-determined by the surgeon. Limited information is available comparing different choices of PTS on the kinematics of the CR TKA, independent of the balancing of the extension gap. This study hypothesized that with the same balanced extension gap, the choice of PTS significantly impacts the intraoperatively measured kinematics of CR TKA.
Methods
Navigated CR TKAs were performed on seven fresh-frozen cadavers with healthy knees and intact posterior cruciate ligament (PCL). A custom designed tibial baseplate was implanted to allow in situ modification of the PTS, which altered the flexion gap but maintained the extension gap. Knee kinematics were measured by performing passive range of motion (ROM) tests from full extension to 120° of flexion on the intact knee and CR TKAs with four different PTSs (1°, 4°, 7°, and 10°). The measured kinematics were compared across test conditions to assess the impact of PTS.
Results
With a consistent extension gap, the change of PTS had significant impact on the anteroposterior (AP) kinematics of the CR TKA knees in mid-flexion range (45°–90°), but not so much for the high-flexion range (90°–120°). No considerable impacts were found on internal/external (I/E) rotation and hip–knee–ankle (HKA) angle. However, the findings on the individual basis suggested the impact of PTS on I/E rotation and HKA angle may be patient-specific.
Conclusions
The data suggested that the choice of PTS had the greatest impact on the mid-flexion AP translation among the intraoperatively measured kinematics. This impact may be considered while making surgical decisions in the context of AP kinematics. When using a tibial component designed with “center” pivoting PTS, a surgeon may be able to fine tune the PTS to achieve proper mid-flexion AP stability.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cruciate-retaining (CR) total knee arthroplasty (TKA), often performed with a fixed posterior tibial slope (PTS) [5], has been demonstrated to produce generally excellent outcomes [18, 28]. However, several fluoroscopic studies have revealed “unphysiological” anterior sliding of the femur relative to the tibia during flexion postoperatively [10, 26, 29]. The paradoxical sliding produces flexion instability leading to difficulty with stair climbing and ascending/descending inclines [3], earlier impingement of the posterior polyethylene insert that prevents a patient from achieving high-flexion [2], increased wear of the polyethylene insert [4, 24], as well as pain and swelling in the knee. It is of paramount importance to properly balance the soft-tissue environment during CR TKA, which facilitates physiological rollback, improved proprioception [9], and a quadriceps muscle friendly mechanism to avoid excessive shear at the implant-bone interface [11]. One key consideration during soft-tissue balancing is the tensioning of the posterior cruciate ligament (PCL). An overly tight PCL can result in postoperative stiffness, excessive femoral rollback [27], excessive polyethylene wear [12], and even rupture of the ligament itself. On the contrary, a loose PCL will compromise proper femoral rollback and AP translation.
With a balanced extension gap, the selection of PTS during CR TKA may produce varying flexion space. Fujimoto et al. compared postoperative kinematics between knees with small PTS (~ 5°) and those with large PTS (~ 10°), and found that the large PTS group had a significantly higher posterior displacement of the medial femoral condyle between the flexion range of 10°–90° [14]. Other studies have demonstrated that PTS is correlated with the maximum postoperative flexion angle [6, 20]. The studies mentioned above were all based on postoperative observations and provided great insight into the impact of PTS. However, they did not account for the inter-patient variations as the comparisons were done between patient groups with different PTSs. Recent studies extended the investigation by modifying the PTS intraoperatively, therefore enabling the assessment of the impact of PTS using the same subject as its own control. Following this method, Okazaki et al. reported that every 5° change of PTS resulted in a 2 mm variation in the flexion gap [22]. Additionally, maximum flexion angle and posterior tibiofemoral translation were found to increase significantly with higher PTS [7]. Both studies utilized customized tibial articular inserts with various angles of inclination for the intraoperative adjustment of the PTS. However, the effect of repeatedly removing and assembling the inserts on the biomechanical integrity of the soft tissues was not assessed in the investigations. Furthermore, modification of the PTS in these studies inevitably altered extension gap to some degree, due to the inclination of the custom tibial inserts were designed to pivot around their most anterior border and the bony resection was not adjusted accordingly for each PTS to maintain the joint line at full extension. Nevertheless, most studies mentioned above measured kinematics based on fluoroscopic or radiographic assessment [6, 7, 14], which is prone to human error and the analyses may be limited by the resolution of the images. To date, there is a paucity of data on the impact of the choice in PTS on the CR TKA kinematics, compared within the same group of subjects with well controlled, consistent extension gap balance to remove its confounding effect.
The purpose of this cadaveric study was to determine, with a balanced and consistent gap maintained in full extension, the impact of PTS on CR TKA kinematics. It was hypothesized that independent of extension gap balancing, the choice of PTS influenced the intraoperatively measured kinematics of CR TKA.
Materials and methods
Specimen preparation and test setup
Seven full cadaveric specimens were used in this study (3 female, 4 male, age 78 ± 13 years), all from non-arthritic donors. The specimens were fresh-frozen during transportation and storage, and thawed to room temperature for 24 h before the lab session. Preoperatively, the specimens were strapped to the table at the level of the pelvis. The mid shaft of the thigh was then attached to a transversal horizontal bar (Fig. 1a). As a result, the femur was oriented at approximately 45° relative to the table and immobilized during the kinematic test. Four threaded stainless-steel hooks were rigidly fixed to each leg at medial and lateral sides of the tibial diaphysis (40 cm distal to the joint line), and medial and lateral femoral epicondyles. A 50 N compressive load was applied to the knee via the hooks with low modulus springs (25 N on each side) to simulate the condition of a standard clinical knee examination. The choice of femoral epicondyles for femoral hook placement minimized the impact from femoral flexion/extension on the spring load. Furthermore, the low spring modulus reduced the sensitivity of compressive force to small variation in spring displacement during the kinematic evaluations.
Kinematic testing
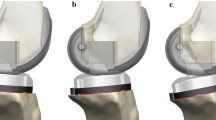
Kinematics of each intact knee were measured and recorded with the assistance of an image-free surgical navigation system (ExactechGPS®, Blue-Ortho, Gieres, France) [1]. The surgeon authors (JYJ and MC) performed passive ROM test three times on each knee specimen, from full extension to high-flexion (intact knee kinematics). A navigated TKA for Optetrak Logic CR® system (Exactech Inc, Gainesville, FL, USA) was then initiated. First, a medial parapatellar incision was made to expose the knee joint, followed by an inspection performed by the surgeons to rule out the presence of macroscopic chondral damage and/or PCL insufficiency. Next, bony landmarks were acquired by the navigation system to establish the anatomical references for both the TKA surgery and the later kinematic evaluation. Bony resections were then generated targeting neutral varus/valgus alignment and 0° flexion/extension in the femur referencing the femoral mechanical axis. For the tibia, the resection was made at neutral varus/valgus and 4° PTS referencing the tibial mechanical axis. The navigated CR TKA was then completed with gap balancing performed at the initial PTS (4°) with accurately controlled internal/external rotation of the tibial baseplate by aligning it with the line connecting medial third of the tubercle and the PCL attachment site. The implanted baseplate was modified from the original Logic CR tibial design to allow easy and precise intraoperative adjustment of PTS by an anterior screw, avoiding potential damage to the soft tissues due to repeated removal and assembly of tibial inserts of varying inclinations (Fig. 1b). The change of slope pivots around the AP “center” of the baseplate, which corresponded to the tibiofemoral contact location of the Logic CR design at full extension. By design, it maintains the extension gap size while modifying the flexion gap. The test method and the special tibial baseplate were described in detail and validated through a previous study, which reported sub-degree/mm error and high repeatability in the measurement of AP displacement, ROM, and HKA [8].
After the CR TKA, the surgeons adjusted the tibial baseplate to 4 PTSs in the order of: 10°, 7°, 4°, and 1° with the guidance of the navigation system. The ROM test was repeated 3 times at each PTS with the patella reduced into the trochlear groove and the arthrotomy closed with surgical sutures. Passive kinematics were recorded by the navigation system during the ROM tests, including:
-
Knee flexion: the angle between femoral and tibial mechanical axes, projected onto the femoral sagittal plane;
-
Internal/external (I/E) rotation: the angle between the femoral and tibial sagittal planes, projected onto the tibial transverse plane;
-
Hip–knee–ankle (HKA) angle: the angle between femoral and tibial sagittal planes, projected onto the tibial coronal plane;
-
Femoral AP translation: AP location of the distal femoral center (mechanical axis exit point) relative to the proximal tibial center (center of the tibial spines), projected onto the tibial transverse plane.
Data processing and statistical analysis
Four knees (2 from each surgeon’s tested subjects) were randomly selected to quantify the repeatability of the measurements. The root mean square error (RMSE) of the 3 repeated tests at intact condition (with a closed arthrotomy) were calculated at 7 sampled flexion angles with 10° intervals (range 30°–90°).
As anatomical landmarks were not yet acquired by the navigation system at the time of ROM testing on the intact knees, the intact kinematics measured before arthrotomy were generated by interpreting the data using the later intraoperatively acquired anatomical references. At each test condition (intact, PTSs of 10°, 7°, 4°, and 1°), the three repeated measurements were averaged for analysis. I/E rotation, HKA angle, and femoral AP translation were compared in the mid-flexion (45°–90°) and high-flexion (90°–120°) ranges, respectively, to detect differences between the kinematic curves from various test conditions (2-sample Kolmogorov–Smirnov test). The kinematic differences between test conditions were further assessed at 7 selected flexion angles sampled from 30° to 120° with 15° increments (Paired t test on all 7 knees at each sampled flexion angle). Analyses were performed in Minitab 16 (Minitab Inc., State College, PA, USA) and R-package (R Core Team, 2017), with statistical significance defined as p < 0.05. Post hoc sample size analysis was performed for the given sample size to investigate the power of the found significant differences.
Institutional approval
Prior to this study, institutional approval has been granted by Hôpitaux Universitaires de Strasbourg, Illkirch, 67400, FR.
Results
High repeatability was found for the ROM test, with low RMSEs in the measurements (2.0 ± 0.7°, 0.3 ± 0.1°, and 0.7 ± 0.2 mm for I/E rotation, HKA, and AP displacement, respectively). In general, the kinematics averaged from the seven knees followed a similar trend as those of the intact knees (Fig. 2). Significant differences between different test conditions were found in the femoral AP translation curves in mid-flexion range, but not observed in high-flexion range (Figs. 2c, 3). Similarly, at the 7 sampled flexion angles, a lower PTS demonstrated significant higher anterior femoral AP translation at early- to mid-flexion (30°, 45°, 60°, and 75°), while no difference was found at higher flexion angles (90°, 105°, and 120°) (Fig. 4). Greater femoral anterior translations by 4.8 mm, 3.1 mm, and 1.9 mm were found in the 1° PTS than that of the 10° PTS at the flexion angles of 30°, 45°, and 60°, respectively (Fig. 4). Even though some significant differences between the curves were found in mid- and high-flexion ranges in I/E rotation and HKA angle (2-sample Kolmogorov–Smirnov test), the magnitude of differences observed was too small to be considered as clinically impactful (Fig. 2a, b). The paired t test at all seven sample flexion angles did not show significant differences for either I/E rotation or HKA angle (non-significant, denoted as n.s.). However, on the individual basis, considerable magnitude of impact was observed from PTS on I/E rotation and HKA angle (Fig. 5). Post hoc sample size analysis demonstrated that with the sample size of 7, the majority of the significant differences found had sufficient power of 80% or higher (range 80–100%). The two exceptions were the differences detected in the AP translation between 10° PTS and 7° PTS at flexion angle of 45° (power = 67%), and between 10° PTS and 1° PTS at flexion angle of 60° (power = 73%), indicating potential insignificance due to Type I error.
Discussion
The most important finding from the present study was that while maintaining the extension gap size, the choice of PTS significantly impacted the mid-flexion AP kinematics of the CR TKA, whereas the same significance was not observed in high-flexion. The sensitivity of the mid-flexion AP kinematics to the choice of PTS may be due to the level of tension in PCL, as a reduced PTS would tighten the flexion gap, resulting in a more posterior positioning of the femoral relative to the tibia. Specifically, for the CR design and component sizes implanted in this study, a change in PTS from 10° to 1° resulted in a reduction of ~ 3 mm in the size of the flexion gap, leading to approximately 5 mm more anterior femoral translation during mid-flexion. Under the same method for modifying PTS, a computational biomechanics study demonstrated PCL force was impacted by PTS during mid-flexion [17], which may partially explain the observation from this present study. The Optetrak Logic CR® system offers multiple tibial inserts with varying inclinations pivoting around the neutral tibiofemoral component contact location at full extension (Fig. 1b). A recently published musculoskeletal simulation study demonstrated that with increased knee flexion in CR TKA, this method for PTS variation had significantly less impact on knee laxity, kinematics and force during a squatting activity, compared to adjusting PTS referencing the anterior border of the tibial baseplate [21]. Although no significant differences were found in tibiofemoral I/E rotation and HKA angle, PTS was shown to have a drastic influence on the knee kinematics on the individual basis (Fig. 5), suggesting that the impact of PTS on I/E rotation and HKA may be patient-specific.
Our findings aligned with previous publications, which suggested that AP displacement was significantly affected by PTS. A change of 10° in PTS was found to be associated with 4 mm of anterior translation of the femur in a computational biomechanics study on functional activities, including standing, squatting, and walking [25]. Compared to the reported value, this study demonstrated a comparable impact of PTS on the anterior femoral translation (maximum differences of ~ 5 mm detected between 1° and 10° of PTS). In addition, the results from the present study agreed with a previously published investigation reporting a more posterior condylar position in the small PTS group (~ 5°) compared to that of the large PTS group (~ 10°) [14].
This study has several strengths. During the tests, each knee specimen served as its own control, enabling direct comparison between different PTSs without the interference of inter-subject and/or inter-surgeon variability. Furthermore, several methods have been applied in previous investigations to adjust the PTS for testing, which included using inter-changeable tibial inserts [7, 22] or applying an anterior open wedge high tibial osteotomy with special plate fixation [15]. The impact of frequent change of tibial inserts on the soft tissue integrity was not assessed in those studies and remained unknown. In addition, adjusting PTS via osteotomy inevitably involves surgical variability, therefore, accurate adjustment of the PTS to the target values may not be achieved. Nevertheless, the above published methods may alter the extension joint line while adjusting the PTS, therefore introducing additional variability to the results as the result of altered extension gap. In contrast, the custom tibial baseplate used in this study allowed continuous adjustment of the PTS in situ without affecting extension gap, and has been demonstrated in a separate study to be soft-tissue preserving [8]. Lastly, the application of computer navigation during the test not only assisted in accurate and precise adjustment of the PTS to the predefined investigation target, but also offered advantages over fluoroscopic or radiographic assessment by providing accurate measurements of knee kinematics in a consistent anatomical reference system.
The present study also has several limitations: first, the cadaveric tests did not account for weight-bearing condition, muscle forces, nor physiological activities. However, it still offered clinical relevance as it represented a typical clinical examination on ROM during an office visit or TKA surgery. In addition, results were comparable to reported values from clinical measurement or simulated functional activities. Second, the native PTS was not quantified in this study due to the lack of a robust measurement. The quantification of the native PTS can be sensitive to the type of medical imaging modality assessed, the measurement method, and the selection of anatomical reference, as well as the specific (medial or lateral) tibial compartment the measurement is performed on [13, 19, 30]. Nevertheless, PTS measurement based on the bony surface may not fully represent the “true” tibial slope at the level of articulation when considering geometry and degeneration of the cartilage layer [16, 23]. It has been reported that an average of a 6° difference exists between the PTS measured based on the bony surface and the meniscal slope of the proximal tibial [16]. As such, the authors did not have high confidence in reporting native PTS. Finally, the results may be design-specific and may not be generalized to all CR designs.
The clinical relevance of the study may suggest that, a CR TKA design that enables intraoperative change of PTS following an AP “center” pivoting mechanism may provide the surgeon an opportunity to manage mid-flexion stability by trialing and choosing a proper insert slope. The management of stability may be further enhanced by quantitative measurements on intraoperative kinematics using a CAOS system.
Conclusions
Proper flexion/extension balance during CR TKA is crucial for the success of the surgery. This study demonstrated that with a balanced and consistent extension gap, the choice of PTS significantly influenced the mid-flexion AP kinematics of a CR TKA. Decreasing PTS resulted in greater anterior translation of the femur during mid-flexion, with no impact in higher flexion. Furthermore, individual consideration may need to be taken on the HKA angle and I/E rotation to properly restore patient-specific kinematics. Intraoperative kinematic evaluation by surgical navigation, or the availability of multiple tibial articular surface inclinations for trialing and implantation, may provide more insights into the assessment of mid-flexion AP kinematics, as well as inform proper HKA angle and I/E rotation kinematics on an individual basis.
References
Angibaud LD, Dai Y, Liebelt RA, Gao B, Gulbransen SW, Silver XS (2015) Evaluation of the accuracy and precision of a next generation computer-assisted surgical system. Clin Orthop Surg 7(2):225–233
Bellemans J, Banks S, Victor J, Vandenneucker A, Moemans A (2002) Fluoroscopic analysis of the kinematics of deep flexion in total knee arthroplasty. J Bone Jt Surg Br 84-B:50–53
Blaha JD (2004) The rationale for a total knee implant that confers anteroposterior stability throughout range of motion. J Arthroplasty 19(4 Suppl 1):22–26
Blunn GW, Walker PS, Joshi A, Hardinge K (1991) The dominance of cyclic sliding in producing wear in total knee replacements. Clin Orthop Relat Res 273:253–260
Brooks P (2009) Seven cuts to the perfect total knee. Orthopedics 32(9). https://doi.org/10.3928/01477447-20090728-27
Catani F, Fantozzi S, Ensini A, Leardini A, Moschella D, Giannini S (2006) Influence of tibial component posterior slope on in vivo knee kinematics in fixed-bearing total knee arthroplasty. J Orthop Res 24(4):581–587
Chambers AW, Wood AR, Kosmopoulos V, Sanchez HB, Wagner RA (2016) Effect of posterior tibial slope on flexion and anterior–posterior tibial translation in posterior cruciate-retaining total knee arthroplasty. J Arthroplasty 31(1):103–106
Dai Y, Angibaud LD, Jenny JY, Hamad C, Jung A, Cross MB (2016) A soft-tissue preserving method for evaluating the impact of posterior tibial slope on kinematics during cruciate-retaining total knee arthroplasty: a validation study. Knee 23(6):1074–1082
Del Valle ME, Harwin SF, Maestro A, Murcia A, Vega JA (1998) Immunohistochemical analysis of mechanoreceptors in the human posterior cruciate ligament: a demonstration of its proprioceptive role and clinical relevance. J Arthroplasty 13(8):916–922
Dennis DA, Komistek RD, Mahfouz MR (2003) In vivo fluoroscopic analysis of fixed-bearing total knee replacements. Clin Orthop Relat Res 410:114–130
Dorr LD, Ochsner JL, Gronley J, Perry J (1988) Functional comparison of posterior cruciate-retained versus cruciate-sacrificed total knee arthroplasty. Clin Orthop Relat Res 236:36–43
Emodi GJ, Callaghan JJ, Pedersen DR, Brown TD (1999) PCL function following total knee arthroplasty: the effect of joint line elevation. Iowa Orthop J 19:82–92
Faschingbauer M, Sgroi M, Juchems M, Reichel H, Kappe T (2014) Can the tibial slope be measured on lateral knee radiographs? Knee Surg Sports Traumatol Arthrosc 22(12):3163–3167
Fujimoto E, Sasashige Y, Tomita T, Iwamoto K, Masuda Y, Hisatome T (2014) Significant effect of the posterior tibial slope on the weight-bearing, midflexion in vivo kinematics after cruciate-retaining total knee arthroplasty. J Arthroplasty 29(12):2324–2330
Giffin JR, Vogrin TM, Zantop T, Woo SLY, Harner CD (2004) Effects of increasing tibial slope on the biomechanics of the knee. Am J Sports Med 32(2):376–382
Jenny JY, Rapp E, Kehr P (1997) Proximal tibial meniscal slope: a comparison with the bone slope. Rev Chir Orthop Reparatrice Appar Mot 83(5):435–438
Kang KT, Koh YG, Son J, Kwon OR, Lee JS, Kwon SK. (2017) Influence of increased posterior tibial slope in total knee arthroplasty on knee joint biomechanics: a computational simulation study. J Arthroplasty. https://doi.org/10.1016/j.arth.2017.09. 025
Li N, Tan Y, Deng Y, Chen L (2014) Posterior cruciate-retaining versus posterior stabilized total knee arthroplasty: a meta-analysis of randomized controlled trials. Knee Surg Sports Traumatol Arthrosc 22(3):556–564
Lipps DB, Wilson AM, Ashton-Miller JA, Wojtys EM (2012) Evaluation of different methods for measuring lateral tibial slope using magnetic resonance imaging. Am J Sports Med 40(12):2731–2736
Malviya A, Lingard EA, Weir DJ, Deehan DJ (2009) Predicting range of movement after knee replacement: the importance of posterior condylar offset and tibial slope. Knee Surg Sports Traumatol Arthrosc 17(5):491–498
Marra MA, Strzelczak M, Heesterbeek PJC, van de Groes SAW, Janssen DW, Koopman BFJM, Wymenga AB, Verdonschot NJJ (2017) Anterior referencing of tibial slope in total knee arthroplasty considerably influences knee kinematics: a musculoskeletal simulation study. Knee Surg Sports Traumatol Arthrosc. https://doi.org/10.1007/s00167-017-4561-3
Okazaki K, Tashiro Y, Mizu-uchi H, Hamai S, Doi T, Iwamoto Y (2014) Influence of the posterior tibial slope on the flexion gap in total knee arthroplasty. Knee 21(4):806–809
Puthumanapully PK, Harris SJ, Leong A, Cobb JP, Amis AA, Jeffers J (2014) A morphometric study of normal and varus knees. Knee Surg Sports Traumatol Arthrosc 22(12):2891–2899
Reinders J, Sonntag R, Kretzer JP (2014) Wear behavior of an unstable knee: stabilization via implant design?. Biomed Res Int 2014:821475
Shelburne KB, Kim HJ, Sterett WI, Pandy MG (2011) Effect of posterior tibial slope on knee biomechanics during functional activity. J Orthop Res 29(2):223–231
Stiehl JB, Komistek RD, Dennis DA, Paxson RD, Hoff WA (1995) Fluoroscopic analysis of kinematics after posterior-cruciate-retaining knee arthroplasty. J Bone Jt Surg Br 77(6):884–889
Takatsu T, Itokazu M, Shimizu K, Brown TD (1998) The function of posterior tilt of the tibial component following posterior cruciate ligament-retaining total knee arthroplasty. Bull Hosp Jt Dis 57(4):195–201
Verra WC, Boom LG, Jacobs WC, Schoones JW, Wymenga AB, Nelissen RG (2015) Similar outcome after retention or sacrifice of the posterior cruciate ligament in total knee arthroplasty. Acta Orthop 86(2):195–201
Yoshiya S, Matsui N, Komistek RD, Dennis DA, Mahfouz M, Kurosaka M (2005) In vivo kinematic comparison of posterior cruciate-retaining and posterior stabilized total knee arthroplasties under passive and weight-bearing conditions. J Arthroplasty 20(6):777–783
Zhang Y, Wang J, Xiao J, Zhao L, Li ZH, Yan G, Shi ZJ (2014) Measurement and comparison of tibial posterior slope angle in different methods based on three-dimensional reconstruction. Knee 21(3):694–698
Acknowledgements
The authors gratefully acknowledge Charlotte Bolch for providing support in statistical methods and sample size analysis.
Funding
No external source of funding was used.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors disclose the following conflict of interest: Jean-Yves Jenny: (1) royalties Aesculap; (2) paid consultant Exactech Inc; (3) unpaid consultants FH Orthopedics; (4) Board member/committee appointments French society for Hip and Knee Surgery, CAOS-international. Michael Cross: (1) paid consultant for Acelity, Acelity Surgical Board, Exactech Inc, Intellijoint, Link Orthopaedics, Smith and Nephew, Thervance Biopharma, and Zimmer Biomet; (2) medical/orthopaedic publications editorial/governing board Journal of Orthopaedics and Traumatology, Techniques in Orthopaedics, and Bone and Joint Journal 360; (3) Stock or stock options Intellijoint. Yifei Dai and Laurent Angibaud: paid employee and stock/stock options Exactech Inc. Cyril Hamad and Amaury Jung: paid employee Exactech Inc subsidiary (Blue Ortho).
Ethical approval
All procedures performed in this study involving human cadavers were in accordance with the ethical standards of Hôpitaux Universitaires de Strasbourg, Illkirch, 67400, FR.
Informed consent
For this type of study, formal consent is not required.
Rights and permissions
About this article
Cite this article
Dai, Y., Cross, M.B., Angibaud, L.D. et al. Posterior tibial slope impacts intraoperatively measured mid-flexion anteroposterior kinematics during cruciate-retaining total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc 26, 3325–3332 (2018). https://doi.org/10.1007/s00167-018-4877-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-018-4877-7