Abstract
Purpose
The goal of this study was to present a 2- to 5-year prospective follow-up of an anatomical posterolateral corner reconstruction in a series of 16 patients with symptomatic instability and pain complaints of the knee.
Methods
All 16 patients underwent a posterolateral corner reconstruction as described by LaPrade et al. If cruciate ligament ruptures were present and had not been addressed earlier, these were reconstructed as well. Preoperatively and 2–5 years after surgery, multiple subjective knee outcome scores (VAS satisfaction score, Tegner, Lysholm, Noyes score and IKDC subjective knee form) were obtained, and the laxity of the joint was evaluated objectively by using bilateral varus stress radiographs to compare the injured with the uninjured knee.
Results
Eleven patients had concomitant ACL or PCL surgery or already had undergone surgery on this cruciate ligament. Mean varus laxity of the injured knee on varus stress radiographs improved significantly from 9.6° (6.6–17.1) to 6.3° (0.3–13.4), p = 0.0011. Post-operative varus laxity did not return to the level of the uninjured knee: 4.4° (1.5–7.7), p = 0.036. VAS satisfaction score, the Tegner, Lysholm, Noyes scores and the IKDC subjective knee form all improved significantly.
Conclusion
The anatomical reconstruction of the posterolateral corner does provide restoration of the external rotation stability in the majority of patients. However, the varus laxity could not be restored in all patients. Functional knee scores improved significantly, and most reconstructed knees had a laxity of <3° compared with the uninjured knee, but the reconstructed knee did not become as stable as the uninjured knee. The results of this study can assist surgeons and patients to have realistic expectations of this operation.
Level of evidence
Case series with no comparative group, Level IV.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anatomical studies have shown that the main stabilising structures of the posterolateral corner (PLC) of the knee can be considered to be the fibular (lateral) collateral ligament (LCL), the popliteofibular ligament (PFL) and the popliteus muscle and tendon (PM, PT) [2, 18, 36, 38]. Injuries to the PLC can lead to invalidating instability of the knee joint, although operative treatment is only recommended for severe injuries [4]. Untreated PLC injuries increase the failure rate of both anterior cruciate ligament (ACL) and posterior cruciate ligament (PCL) reconstructions [9, 13, 19, 30, 31]. In the past, several techniques to treat these instabilities have been described [12, 35]. LaPrade et al. [16, 21] described an anatomical reconstruction technique of the injured PLC. Multiple studies have shown that an anatomical reconstruction is superior to a repair [22, 23, 29, 37].
Posterolateral corner insufficiency often consists of a varus laxity component and a rotational laxity component. Several studies describe good results of a two-tailed technique in which a graft is passed through the head of the fibula and attached to the femoral epicondyle [15, 25, 26]. The LaPrade et al. technique addresses both laxity components by reconstructing/reinforcing the lateral collateral ligament, the popliteal tendon and the PFL with two grafts. The technique is based on the quantitative attachment anatomy of these three structures. However, only a few clinical studies are available that recognise the additional value of this technique or that consider whether the patients benefit from this extra reinforcement [27, 29, 39]. Only LaPrade et al. [7, 15, 16] have analysed this technique using prospective studies.
The main goal of this study was to investigate whether results could be achieved comparable with those reported by LaPrade et al. We hypothesised that this surgical technique would show an objective improvement in the stability of the knee joint. Our second hypothesis was that patients would show improved knee function.
Materials and methods
Between 2004 and 2010, 16 patients underwent a PLC reconstruction of the knee as described by LaPrade et al. [17] and were included in this study. The inclusion criteria were the following clinical findings: post-traumatic varus instability >5 mm, external tibial rotation measured clinically in 30° knee flexion >5° than that of the uninjured knee. Furthermore, there had to be a minimal lateral compartment opening of 5° during varus stress radiographs in extension.
Exclusion criteria were as follows: patients with grade 4 osteoarthritis according to the Outerbridge classification, patients with a previous osteotomy or an indication for an osteotomy for varus malalignment. A varus alignment of 3°–4° or greater and the presence of a varus thrust were considered to be an indication for an osteotomy.
Leg alignment was assessed on long leg radiographs. All patients underwent magnetic resonance imaging of the injured knee.
The median age of the subjects was 33 (18–58); in seven cases, the right knee was injured and in nine cases the left. Ten males and six females were operated. Fourteen patients had chronic instability complaints; two patients underwent a reconstruction in the acute stage (within 6 weeks).
Fourteen subjects had a clinical grade III PLC injury (>10 mm varus and/or >10° rotational instability), and two subjects had a severe grade II-PLC injury (varus instability of 5–10 mm and/or rotational instability of >10°) [11].
Eleven of the 16 patients had concomitant complex ligament injury of the involved knee (Table 1).
Three patients had peroneal nerve damage at presentation. This recovered completely in two patients; one patient had remaining damage.
Eight patients had either undergone a reconstruction of one of the cruciate ligaments before referral to our hospital or undergone a PLC operation that later failed (Table 1). In all 16 patients, we performed a reconstruction of the PLC as described by LaPrade et al. [17]; the ruptured cruciate ligaments were also reconstructed. In two cases, a partial PCL rupture was left untreated, since on testing these knees only showed minimal laxity (Table 1).
The surgical technique was as described by LaPrade et al. [17]. An Achilles tendon allograft was used and split lengthwise to create two grafts: the first graft to reconstruct the LCL and the PF; the second graft to reconstruct the PT (Figs. 1, 2). The grafts were fixed with interference screws into the femur, tibia and fibula. The allograft was not secured with a screw in the fibular head in all patients; sometimes the fibular head was small compared with the drill hole size needed for the graft. In such cases, we did not insert a screw in order to prevent a fracture of the soft bone. The thickness of the tunnels drilled and that of the interference screws sometimes differed from those used by LaPrade et al. [17], depending on the situation. The procedures were performed in a similar way by three surgeons.
At the first post-operative day, a varus/valgus stabilising brace was applied. Flexion and extension of the knee were limited during the first two post-operative months by the brace, with a range from 20° extension to 70° flexion. On the second post-operative day, the patient was mobilised with crutches with a maximal weight-bearing load of 5–10 kg for the next 2 months. After that period, under supervision of a physiotherapist, the load was gradually increased to full weight bearing on the operated knee, and maximum range of motion allowed was increased from 0° extension to 90° flexion in the brace until 4 months post-operatively. Low-molecular-weight heparin was prescribed for a 2-month period to prevent deep venous thrombosis.
Preoperative varus stress radiographs were performed as a standard diagnostic tool in the evaluation of the PLC injury. This has been previously described [6, 8, 10, 14, 34]: the method provides an objective and reproducible measure of lateral compartment opening. The radiographs were obtained using the Telos device (Fa Telos, Medizinisch-Technische GmbH, Griesheim, Germany) with the subject lying in a supine position with the leg in 0° of extension, while a 15-kg load was applied at the level of the joint line. Using the measurement tool included in the radiographic database program, the lateral compartment opening was determined as the angle between the tangent to the femoral condyles and the line through the deepest tibial joint surfaces. Measurements were made at the nearest 0.1 degree. A detailed description of the measurement technique and its accuracy was previously published by Heesterbeek et al. [10].
To test our hypothesis, 2–5 years after surgery, the lateral compartment opening was again measured on the stress radiographs and compared with the lateral compartment opening in the uninjured knee. We considered the varus laxity of the uninjured leg to be the baseline value.
Several subjective clinical score forms (VAS satisfaction score, the IKDC subjective form and the Noyes, Lysholm and Tegner scores) were completed preoperatively and 2–5 years after surgery.
The accredited ethics committee (Dutch acronym: METC, English: IRB) Slotervaartziekenhuis and Reade reviewed this study, registered under number P1312, by expedited review and determined, based on the Dutch Medical Research Involving Human Subjects Act (Dutch acronym: WMO), that the research activities described meet the requirements for exemption from METC review under the WMO.
The data obtained at the final follow-up were compared with the preoperative data using Wilcoxon signed-rank tests.
All patients were followed up prospectively, but because of some missing values, it was not possible to present the results for both the 2- and 5-year follow-up. Therefore, we took the scores collected at the 5-year follow-up when available, and if not, the scores at the 2-year follow-up were used.
For one patient, only stress radiographs were made 1 year post-operatively; another patient died after the 1-year post-operative follow-up appointment. For these two patients, we used the 1-year post-operative stress radiograph values in our post-operative evaluation.
p-values smaller than 0.05 were considered significant.
Results
Median follow-up was 60 (23.5–74.1) months, excluding the two patients who had missing values after the 1-year follow-up.
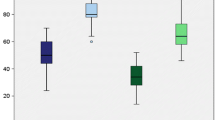
The median opening of the lateral compartment on the stress radiograph decreased significantly from 9.6° (6.6–17.1) to 6.3° (0.3–13.4), (p = 0.0011). However, it remained more than on the uninjured side: 4.4° (1.5–7.7), (p = 0.036) (Fig. 3).
All clinical scores increased significantly. The VAS satisfaction score improved from 33 (20–51) to 67 (9–92), (p = 0.0022).
Tegner score improved from 2.0 (0–4) to 3.5 (0–8), (p = 0.047). The post-operative Lysholm score improved from 58.5 (0–82) to 82 (16–100), (p = 0.0097). The Noyes score improved from 56 (20–65) to 76 (54–100), (p = 0.0022), and the IKDC subjective knee form showed an increase from 44 (11–55) to 67 (22–99), (p = 0.0033) (Fig. 4). One patient (patient number 9 in Table 1) had a low VAS (9), low Lysholm score (16) and low IKDC score (22). This patient had a preoperative pain syndrome that did not resolve post-operatively.
The external rotation in 30° of knee flexion was <5° for all patients 1 year after surgery and remained <5° after 2–5 years follow-up in all patients, except for three; in those patients external rotational laxity increased slightly to 6–10° (Table 1).
When a concomitant rupture of the ACL was addressed with a reconstruction, this led to a stable anterior drawer test in the majority of the patients. The PCL reconstructions, however, still showed some instability during clinical examination.
Discussion
The most important finding of the present study was that satisfactory results could be achieved with a PLC reconstruction technique as described by LaPrade et al. [17]. However, the operated knees did not become as stable as the uninjured knees. The studied group showed a significantly decreased median opening of the lateral compartment on stress radiographs at a follow-up of 2–5 years.
The VAS satisfaction score, the Tegner, Lysholm, Noyes scores and the IKDC subjective knee form score all improved significantly. To our knowledge, this is the first time that the results of a reconstruction technique according to LaPrade et al. [17] are described by another group.
Multiple techniques have been described for the treatment of chronic posterolateral knee injuries. In our view, of all described techniques, the LaPrade et al. [17] technique comes closest to the native situation. In theory, by reconstructing the LCL, PFL and PT based on their quantitative attachment anatomy, the surgeon is able to restore static laxity under varus and external rotation testing conditions [15]. Only few clinical studies have described this technique, with or without modifications. Yoon et al. [39] compared this technique with a non-anatomical technique and revealed better rotational and varus laxity with the more anatomical technique. Noyes et al. [29] operated on nine patients with this technique; however, these were evaluated together with a group of five patients with only a collateral ligament reconstruction. Nevertheless, they showed good results and good rotational stability. LaPrade et al. analysed 54 patients with PLC or combined injuries with an average follow-up of 4.3 years, showing a significant improvement in the post-operative IKDC objective scores for varus opening at 20°, external rotation at 30°, reverse pivot shift and the single leg hop [15]. The study population consisted of a very heterogeneous group of patients, as in the present study. Geeslin et al. analysed 26 knees in 25 patients following an acute PLC injury. Ten patients were treated with the anatomic reconstruction technique as in the present study, and the remaining 16 knees were treated with PLC repair or repair and reconstruction. Varus stress radiographs demonstrated a significant improvement in the side-to-side difference in the lateral compartment gap, from 6.2 mm preoperatively to 0.1 mm at the time of final follow-up, on average 2.4 years [7].
Several studies reported the varus laxity measurements in millimetres. In the present study, the laxity was measured in degrees on stress radiographs to prevent any discrepancies due to the magnification factor [10]. As a consequence, the results of the present study are difficult to compare with other studies.
When considering a lateral opening of <3° (operated vs uninjured knee) as being successful (surgeon’s opinion), then 10 out of 14 PLC reconstructions can be considered a success (two patients lacked values for the uninjured side and thus could not be evaluated). This success rate of 71 % is comparable with the results of Noyes et al. [27] who found a success rate of 76 % (16 out of 21 patients) at a mean follow-up of 42 months. Yoon et al. [39] reported post-operative residual varus laxity of more than 5 mm compared with the contralateral side in 14 % of the 21 cases. However, these results were based on clinical examination; there were no varus stress radiographs to enable an objective measurement. As mentioned earlier, in the study by Geeslin et al. [7] varus stress radiographs demonstrated an improvement in the side-to-side difference in the lateral compartment gap from 6.2 mm preoperatively to 0.1 mm post-operatively, making the operated knees almost as stable as the uninjured knees. Although this seems a better result than the results from the present study, it is difficult to compare as Geeslin et al. [7] investigated a group of patients who underwent either a reconstruction, a repair, or a repair and reconstruction.
It is not clear why in the present study some patients had a residual lateral collateral laxity. These were not the three patients with slightly increased external rotation. The fixation for all grafts was performed in the standard manner. The femoral fixation using the bone block seems reasonably secure. Theoretically, the tibial fixation of the graft might slip within the tunnel. In addition, the loosening of the lateral collateral ligament reconstruction might have been caused by a further slippage or stretching of the rather long graft passing through the fibular head and then the tibia. Furthermore, the allograft had not been secured in all patients with a screw in the fibular head; sometimes the fibular head was small and the bone soft. In only one patient with residual laxity, the allograft was not fixed with an interference screw in the fibular head, and we found no correlation between the usage of an interference screw in the fibular head to the laxity. Another theory could be that the rather soft bone of the fibular head might have been deformed during the rehabilitation, but in all cases the tunnel edges had been smoothened. A neutral hinged brace for 4 months was used during rehabilitation, and we consider this to be a rather conservative post-operative care.
In this study, 11 subjects had additional injuries like PCL and ACL ruptures, which is comparable with other studies [1, 7, 15, 24, 32, 33, 39] and is the reality in our practice. These additional injuries will have a negative impact on the clinical outcome and make an isolated evaluation of the PLC reconstruction difficult. In our view, the reconstruction of concurrent injuries, especially of the cruciate ligaments, is mandatory in order to obtain a good PLC reconstruction result. In the cases where the cruciate ligament ruptures were not addressed, the anterior-posterior stability was still rather poor, which strengthens the view that the extra restraint provided by intact or reconstructed cruciate ligaments plays a relevant role in the stability of the knee [25]. As stated by LaPrade et al., the PCL plays an important role both in restraining lateral and rotational laxity at 90° flexion. Therefore, simultaneous reconstruction of the PCL proved to be essential both in vitro and in vivo to assure improvement in the PLC stability [14, 20]. The consequence of missing a PLC injury in the presence of a known tear of the ACL or PCL could lead to failure of the reconstructed cruciate ligament [3, 33].
Although the results in this study show an overall improvement in functional scores, the median post-operative Tegner score was only 3.5. A study by Freeman et al. [5] reported improvement in Tegner activity from 2.25 to 4.5, which was significant, as in the present study. Noyes et al. reported an improvement in activity level post-surgery after the anatomical reconstruction. It was not reported whether they returned to their original activity level [27–29]. Considering the results mentioned above, it is important to advise patients that we aim for a fully weight-bearing leg and not to have them return to their former activity level.
A limitation of the present study is that it is a single cohort study and no comparison has been made with an alternative (less-anatomical) reconstruction technique. Instead, the result of the reconstruction was compared with the non-injured side. Furthermore, the size and the variety of the cohort can be considered as limitations of the present study, although this is inherent to the rarity and the nature of the injury. A further limitation of the study might be that AP and rotational laxity were assessed as part of the IKDC score.
Fourteen patients had chronic instability complaints and two patients were operated on during the acute stage, which might introduce bias since a reconstruction in patients with chronic instability is potentially more difficult considering possible tissue retraction, adhesions and entrapment of the common peroneal nerve in scar tissue [20, 37].
Conclusion
The anatomical reconstruction of the PLC does provide restoration of the external rotation stability in the majority of patients. However, the varus laxity could not be restored in all patients. Functional knee scores improved significantly and most reconstructed knees had a laxity of <3° compared with the uninjured knee, but the reconstructed knee did not become as stable as the uninjured knee. The results of this study can assist surgeon and patient to have realistic expectations of this operation.
References
Becker EH, Watson JD, Dreese JC (2013) Investigation of multiligamentous knee injury patterns with associated injuries presenting at a level I trauma center. J Orthop Trauma 27:226–231
Brinkman JM, Schwering PJ, Blankevoort L, Kooloos JG, Luites J, Wymenga AB (2005) The insertion geometry of the posterolateral corner of the knee. J Bone Jt Surg Br 87:1364–1368
Covey DC (2001) Injuries of the posterolateral corner of the knee. J Bone Jt Surg Am 83-A:106–118
Dhillon M, Akkina N, Prabhakar S, Bali K (2012) Evaluation of outcomes in conservatively managed concomitant Type A and B posterolateral corner injuries in ACL deficient patients undergoing ACL reconstruction. Knee 19:769–772
Freeman RT, Duri ZA, Dowd GS (2002) Combined chronic posterior cruciate and posterolateral corner ligamentous injuries: a comparison of posterior cruciate ligament reconstruction with and without reconstruction of the posterolateral corner. Knee 9:309–312
Garavaglia G, Lubbeke A, Dubois-Ferriere V, Suva D, Fritschy D, Menetrey J (2007) Accuracy of stress radiography techniques in grading isolated and combined posterior knee injuries: a cadaveric study. Am J Sports Med 35:2051–2056
Geeslin AG, LaPrade RF (2011) Outcomes of treatment of acute grade-III isolated and combined posterolateral knee injuries: a prospective case series and surgical technique. J Bone Jt Surg Am 93:1672–1683
Gwathmey FW Jr, Tompkins MA, Gaskin CM, Miller MD (2012) Can stress radiography of the knee help characterize posterolateral corner injury? Clin Orthop Relat Res 470:768–773
Harner CD, Vogrin TM, Hoher J, Ma CB, Woo SL (2000) Biomechanical analysis of a posterior cruciate ligament reconstruction. Deficiency of the posterolateral structures as a cause of graft failure. Am J Sports Med 28:32–39
Heesterbeek PJ, Verdonschot N, Wymenga AB (2008) In vivo knee laxity in flexion and extension: a radiographic study in 30 older healthy subjects. Knee 15:45–49
Hughston JC, Andrews JR, Cross MJ, Moschi A (1976) Classification of knee ligament instabilities. Part II. The lateral compartment. J Bone Jt Surg Am 58:173–179
Jung YB, Jung HJ, Kim SJ, Park SJ, Song KS, Lee YS, Lee SH (2008) Posterolateral corner reconstruction for posterolateral rotatory instability combined with posterior cruciate ligament injuries: comparison between fibular tunnel and tibial tunnel techniques. Knee Surg Sports Traumatol Arthrosc 16:239–248
Kannus P (1989) Nonoperative treatment of grade II and III sprains of the lateral ligament compartment of the knee. Am J Sports Med 17:83–88
LaPrade RF, Heikes C, Bakker AJ, Jakobsen RB (2008) The reproducibility and repeatability of varus stress radiographs in the assessment of isolated fibular collateral ligament and grade-III posterolateral knee injuries. An in vitro biomechanical study. J Bone Jt Surg Am 90:2069–2076
LaPrade RF, Johansen S, Agel J, Risberg MA, Moksnes H, Engebretsen L (2010) Outcomes of an anatomic posterolateral knee reconstruction. J Bone Jt Surg Am 92:16–22
LaPrade RF, Johansen S, Engebretsen L (2011) Outcomes of an anatomic posterolateral knee reconstruction: surgical technique. J Bone Jt Surg Am 93(Suppl 1):10–20
LaPrade RF, Johansen S, Wentorf FA, Engebretsen L, Esterberg JL, Tso A (2004) An analysis of an anatomical posterolateral knee reconstruction: an in vitro biomechanical study and development of a surgical technique. Am J Sports Med 32:1405–1414
LaPrade RF, Morgan PM, Wentorf FA, Johansen S, Engebretsen L (2007) The anatomy of the posterior aspect of the knee. An anatomic study. J Bone Jt Surg Am 89:758–764
LaPrade RF, Resig S, Wentorf F, Lewis JL (1999) The effects of grade III posterolateral knee complex injuries on anterior cruciate ligament graft force. A biomechanical analysis. Am J Sports Med 27:469–475
LaPrade RF, Wentorf F (2002) Diagnosis and treatment of posterolateral knee injuries. Clin Orthop Relat Res 402:110–121
LaPrade RF, Wentorf FA, Crum JA (2004) Assessment of healing of grade III posterolateral corner injuries: an in vivo model. J Orthop Res 22:970–975
Levy BA, Stuart MJ, Whelan DB (2010) Posterolateral instability of the knee: evaluation, treatment, results. Sports Med Arthrosc 18:254–262
Lunden JB, Bzdusek PJ, Monson JK, Malcomson KW, Laprade RF (2010) Current concepts in the recognition and treatment of posterolateral corner injuries of the knee. J Orthop Sports Phys Ther 40:502–516
Mariani PP, Santoriello P, Iannone S, Condello V, Adriani E (1999) Comparison of surgical treatments for knee dislocation. Am J Knee Surg 12:214–221
Mauro CS, Sekiya JK, Stabile KJ, Haemmerle MJ, Harner CD (2008) Double-bundle PCL and posterolateral corner reconstruction components are codominant. Clin Orthop Relat Res 466:2247–2254
McCarthy M, Camarda L, Wijdicks CA, Johansen S, Engebretsen L, Laprade RF (2010) Anatomic posterolateral knee reconstructions require a popliteofibular ligament reconstruction through a tibial tunnel. Am J Sports Med 38:1674–1681
Noyes FR, Barber-Westin SD (1995) Surgical reconstruction of severe chronic posterolateral complex injuries of the knee using allograft tissues. Am J Sports Med 23:2–12
Noyes FR, Barber-Westin SD (2007) Posterolateral knee reconstruction with an anatomical bone-patellar tendon-bone reconstruction of the fibular collateral ligament. Am J Sports Med 35:259–273
Noyes FR, Barber-Westin SD, Albright JC (2006) An analysis of the causes of failure in 57 consecutive posterolateral operative procedures. Am J Sports Med 34:1419–1430
Noyes FR, Barber-Westin SD, Roberts CS (1994) Use of allografts after failed treatment of rupture of the anterior cruciate ligament. J Bone Jt Surg Am 76:1019–1031
O’Brien SJ, Warren RF, Pavlov H, Panariello R, Wickiewicz TL (1991) Reconstruction of the chronically insufficient anterior cruciate ligament with the central third of the patellar ligament. J Bone Jt Surg Am 73:278–286
Pacheco RJ, Ayre CA, Bollen SR (2011) Posterolateral corner injuries of the knee: a serious injury commonly missed. J Bone Jt Surg Br 93:194–197
Ranawat A, Baker CL III, Henry S, Harner CD (2008) Posterolateral corner injury of the knee: evaluation and management. J Am Acad Orthop Surg 16:506–518
Rios CG, Leger RR, Cote MP, Yang C, Arciero RA (2010) Posterolateral corner reconstruction of the knee: evaluation of a technique with clinical outcomes and stress radiography. Am J Sports Med 38:1564–1574
Schechinger SJ, Levy BA, Dajani KA, Shah JP, Herrera DA, Marx RG (2009) Achilles tendon allograft reconstruction of the fibular collateral ligament and posterolateral corner. Arthroscopy 25:232–242
Shahane SA, Ibbotson C, Strachan R, Bickerstaff DR (1999) The popliteofibular ligament. An anatomical study of the posterolateral corner of the knee. J Bone Jt Surg Br 81:636–642
Stannard JP, Brown SL, Farris RC, McGwin G Jr, Volgas DA (2005) The posterolateral corner of the knee: repair versus reconstruction. Am J Sports Med 33:881–888
Terry GC, LaPrade RF (1996) The posterolateral aspect of the knee. Anatomy and surgical approach. Am J Sports Med 24:732–739
Yoon KH, Bae DK, Ha JH, Park SW (2006) Anatomic reconstructive surgery for posterolateral instability of the knee. Arthroscopy 22:159–165
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
van der Wal, W.A., Heesterbeek, P.J.C., van Tienen, T.G. et al. Anatomical reconstruction of posterolateral corner and combined injuries of the knee. Knee Surg Sports Traumatol Arthrosc 24, 221–228 (2016). https://doi.org/10.1007/s00167-014-3369-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-014-3369-7