Abstract
This paper reviews the functional anatomy of the anterior cruciate ligament (ACL), which has a parallel array of collagen fascicles that have usually been divided into two ‘fibre bundles’: anteromedial (AM) and posterolateral (PL), according to their tibial attachment sites. The PL bundle has shorter fibres, and so it is subjected to greater tensile strains than the AM bundle when the whole ACL is stretched; its oblique orientation in the coronal plane imbues it with greater ability to resist tibial rotation than the more vertical AM fibre bundle. Most studies have found that the AM bundle is close to isometric when the knee flexes, while the PL bundle slackens approximately 6 mm. There is little evidence of significant fibre bundle elongation in response to tibial rotation. Selective bundle cutting studies have been performed, allowing both the bundle tensions and their contributions to resisting tibial anterior translation and tibial rotation to be calculated. These show that the function of the PL bundle was dominant near knee extension in some studies, particularly when resisting anterior drawer and that its contribution reduced rapidly with knee flexion through 30 degrees. There has been little study of the contributions of the fibre bundles in control of tibial internal–external rotation or the pivot shift: one study found that the AM bundle had larger tensions than the PL bundle during a simulated pivot shift, but another study found that cutting the PL bundle allowed a larger increase in coupled tibial anterior translation than cutting the AM bundle. It was concluded that the AM bundle is most important for resisting tibial anterior drawer—the primary function of the ACL—while the PL bundle is tight near knee extension, when it has a role in control of tibial rotational laxity. There is a clear need for further study of dynamic knee instability, to gain better understanding of how best to reconstruct the ACL and associated tissues.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Biomechanical, anatomical and surgical research on anterior cruciate ligament (ACL) reconstruction has focused mainly on single-bundle autogenous tendon reconstructions and factors such as graft choice, positioning, tensioning and fixation have been shown to be important. Unfortunately, close replication of normal knee function is still not achieved in many cases, and so research on the ideal ACL reconstruction continues. Reconstruction of the ACL may be justified because delay in surgery leads to an increasing prevalence of meniscal damage [22], and preservation of the menisci via ACL reconstruction may be the principal factor in avoiding later cartilage damage [35]. However, not all studies have shown that ACL reconstruction protects the knee joint from developing osteoarthritis [20]. A persistent pivot-shift sign is a prognostic factor for late osteoarthrosis [24] and is a good indicator of a patient’s subjective instability [26]. The importance of a persisting ‘mini pivot’ [31] is not yet known, yet it is not uncommon: Aglietti et al. [1] detected a residual minimal rotatory instability (pivot shift) in almost one-fifth of the cases, and Freedman et al. [13], in a metaanalysis of thirty-four ACL reconstruction studies, found that a pivot-shift glide was present in 15% of knees with single-bundle patellar tendon grafts and 14% of those with hamstrings grafts.
The persistence of traces of knee instability in some knees with single-bundle ACL reconstruction has led to efforts to develop double-bundle reconstruction methods. However, increasing appreciation of rotational laxity has also led to a move to place single-bundle grafts more laterally in the femoral intercondylar notch and that reduces the prevalence of pivot-shift laxity [30, 38]. However, although some biomechanical studies in vitro have found that a laterally placed (anatomical) single-bundle ACL graft can successfully eliminate the mini pivot [27], others have reported that it does not always manage to do so [31]. Studies in vivo, which have included fluoroscopic [48], magnetic resonance imaging [33] and optical gait analysis systems [8, 41] to compare ACL-reconstructed patients to matched controls in pivoting and cutting activities, have found that single-bundle ACL reconstruction surgery did not completely restore rotational kinematics and stability.
This situation is encouraging continued work to optimise ACL reconstruction methods, particularly the use of double-bundle (‘anatomical’) grafts, which are an attempt to reproduce the natural ACL fibre bundle anatomy more closely than is possible with a single-bundle graft. Double-bundle ACL reconstruction was pioneered by Mott [36] and Zaricznyj [54], but their methods were not taken up by others at that time, and there has been little reporting of their results. Noting this situation, this paper sets out the present knowledge of the functions of the fibre bundles of the ACL, particularly in controlling tibial rotation, as a basis for work on reconstructing them.
Functional consequences of the anatomy of the ACL
When the intercondylar synovial tissue has been dissected away to reveal the load-bearing structure of the ACL, a band of collagen fibres is seen, which are oriented distal–anterior–medial from the posterior area of the medial aspect of the lateral femoral condyle (i.e., the lateral wall of the femoral intercondylar notch) to the anterior-central interspinous area of the tibial plateau. Because of the orientations of the bone attachments, the distance between them varies from anterior to posterior, and the anterior fibres are longer than those that are posterior in the cross-section [51, 52]. The consequence of this is that the posterior fibres will suffer a greater strain (i.e., elongation as a percentage of their original length) than the longer anterior fibres, for a given bone–bone movement, rendering the shorter posterior fibres more vulnerable to being ruptured; this is generally true for ligaments, which fail at a strain limit [10, 42]. In addition, the ACL attachments are quite large, and so individual fibre attachments have differing relationships to the axis of knee flexion–extension, leading to a range of tightening–slackening behaviour across the range of motion.
The length change patterns of the fibres of the ACL are controlled principally by their femoral attachment sites. Patterns of tightening or slackening behaviour have been measured in order to define the area with the least deviation from zero length change, or ‘isometry’ [4, 14, 19, 45]. The ‘isometric’ area is close to the posterior end of Blumensaat’s line, and past practise was to try to reproduce this behaviour, thus minimising cyclic elongation patterns during knee motion. However, it is now realised that the resulting ‘high-noon’ graft placements were neither anatomical nor mechanically efficient. The concept of fibre attachments moving around the isometric area during knee flexion–extension explains the varying contributions of the fibre bundles of the ACL during knee flexion. The anterior fibres attach to the femur relatively proximal/anterior, near to the isometric area, so they have little length change, while the posterior fibres attach posterior/distal away from the isometric area, so they slacken when the knee flexes but retighten in deep knee flexion (Fig. 1).
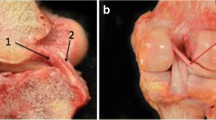
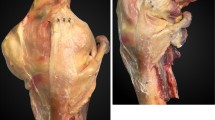
In order to help to distinguish between the behaviours of fibres in different parts of the cross-section of the ACL, it has proven convenient to artificially divide it into ‘fibre bundles’. While these divisions are usually artefactual, there is an anatomical basis for doing so. The human foetus has separate fibre bundles in the ACL during embryogenesis [11], and this separation may persist into old age, when synovial tissue planes may sometimes be found between the bundles [37, 46]. Similarly, quadrupedal mammals, such as the sheep [40], have two separate, well-defined bundles in their ACL. Most authors work on the basis of two main fibre bundles: one anteromedial (AM bundle) and one posterolateral (PL bundle), which were named by Girgis et al. [17] on the basis of the relative positions of their attachments to the tibial plateau. Other authors have divided the ACL into a larger number of fibre bundles: Norwood and Cross [37] isolated three main fibre bundles, which were distinguished at their tibial attachments: AM, intermediate and PL bundles. The tibial attachment was described as a triangle with a posterior apex for the PL bundle attachment and two anterior corners, occupied by the AM and intermediate bundles. The femoral AM bundle attachment was described as being posterior and superior in the notch, while the PL was ‘anterior and inferior’ (presumably described when viewing the intercondylar notch at 90° knee flexion); the intermediate bundle was between the previous two. Amis and Dawkins [3] also described an intermediate bundle, and that three-bundle structure was reproduced by Tanaka et al. [47] as a triple-bundle reconstruction; they hoped that the diverging anterior fibre bundles would help to control tibial rotation. Taken to its logical limit, this approach may lead to mapping of the many natural fascicles as a basis for an ‘anatomical scaffold’. Friederich et al. [14] showed that, when ACL fibres were traced across from femur to tibia, they created a parallel array in the extended knee (Fig. 2). Hara et al. [18] have shown that many small fascicular structures can be traced: they are twisted around each other when the knee flexes, leading to much greater complexity (Fig. 3). However, at present, there is no data to support such complexity during ACL reconstruction surgery.
Length changes of the ACL bundles during knee motion
The length changes of the ACL fibre bundles provide an important guide to their functional behaviour in controlling both anterior drawer and internal–external rotational laxity. Ligaments function within a small range of tensile elongation and typically rupture at 20% strain [39, 49]. This means that, if there are large changes in the distances between the attachments when the knee flexes, much of that length change must cause slackening of the fibre bundle and that indicates an arc of motion when the load-bearing contribution of that bundle is likely to be reduced or absent.
Girgis et al. [17] observed that while in extension the whole ACL was taut, in flexion, only a small anterior band was in tension and the posterior bulk of the ligament was slack. This observation has since been measured: several studies in vitro have instrumented the fibre bundles of the ACL, usually by threading sutures along them from one attachment to the other and then out to a displacement transducer [3, 29, 44]. They found that the PL bundle was taut in full knee extension, slackened by 5–6 mm in mid-flexion, then re-tightened somewhat beyond 90 degrees knee flexion. Contrastingly, the AM bundle was taut across the range of flexion–extension, tending to be tighter in the flexed knee, with only 2–3 mm length changes (Fig. 1). Bach et al. [5] implanted strain gauges in the AM and PL bundles and measured the strain changes during range of motion. The AM bundle exhibited a quasi-isometric behaviour from 10 to 90 degrees knee flexion, with changes of less than 1%. At 8 degrees hyperextension and full flexion, the AM bundle was stretched 4%. The PL bundle on the contrary was relaxed from 40 degrees onward while in 8 degrees hyperextension it elongated by 10% of its initial length.
Several more recent studies have produced similar findings by using 3D imaging methods in vivo. Yoo et al. [51] used reconstructions from CT scans to measure the distance between the femoral and tibial attachments at a range of knee flexion angles. The AM bundle slackened 4 mm by 90 degrees flexion, and the PL bundle by 7 mm; both bundles were longest in full extension. Jordan et al. [25] used dual-beam fluoroscopy to measure the distance between the femoral and tibial bundle attachments during lunging postures: the AM bundle slackened 3 mm by 90 degrees knee flexion and 6 mm by 135 degrees, while the PL bundle was longest in knee extension and slackened 6 mm when the knee flexed 90 degrees and 8 mm by 135 degrees. Iwahashi et al. [23] used open-access MRI to estimate the lengths of three fibre bundles: AM, PL and intermediate (IM), across 0–150 degrees knee flexion. All three bundles were at their greatest length when the knee was in extension: AM 34 mm, IM 33 mm and PL 27 mm, and all were at the shortest at 100 degrees flexion, having slackened by 3, 5 and 6 mm, respectively. Thus, the AM bundle was closest to isometric and the PL bundle, having the shortest fibres, had the largest per cent slackening. These data relate to the clinical use of ‘triple-bundle’ ACL reconstruction [47].
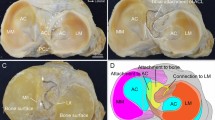
The reader should note that these studies did not actually measure the length changes of fibre bundles in the ACL but calculated the changing straight-line distances between the femoral and tibial attachments. Noting that the ACL fibres are not stretched greatly during unloaded knee flexion–extension motion, the relatively large ‘length changes’ reported are largely made up of buckling collapse of the slack ligament. When this approach is extended to include computer modelling, it enables the theoretical variation of ACL fibre lengths to be displayed across the attachment areas [2] (Fig. 4).
Computed prediction of the distribution of ACL fibre length changes throughout the cross-section of the fibres [2]. The isometry lines show the elongation as a ratio of the length at full extension, across the tibial attachment (anterior at top, medial at right) when the knee model was flexed 90 degrees (Reproduced from Amiri et al. [2] with permission from Elsevier Limited)
Despite the detailed differences between the results of the studies reviewed above, they did all agree that both the AM and PL fibre bundles (which means the whole of the cross-section of the ACL) were longest/tightest when the knee was extended, and even longer if it was forced into hyperextension.
Although recent work on the ACL has become more concerned with the rotational component of instability, much of the earlier experimental work did not examine how tibiofemoral rotation affected the lengths of the fibre bundles. Amis and Dawkins [3] found that tibial internal or external rotation in vitro had no significant effect on bundle lengths. This implies that the ACL will have only a small effect in controlling internal–external rotation, but the torque was only 1 Nm. In contrast, Brophy et al. [6] imposed ‘manual maximum’ loads to their cadaveric knees, for both anterior translation at 30 and 90 degrees knee flexion, and for internal rotation at 0 degrees flexion. The AM and PL bundles were both elongated similarly in all three loading conditions; however, the PL bundle is shorter than the AM, and so significantly larger fibre strains were calculated in the PL bundle. The reader should recall, however, that calculated length changes are not the same as tensile strains, because the PL bundle slackens significantly with knee flexion, and so part of the elongation is taking up the slack before the ligament itself is stretched, unless the knee is in full extension. In addition, the PL bundle is shorter than the AM bundle, so it will be subjected to greater strains (percentage elongation) than the AM bundle after the slackness has been taken up.
Resistance to tibial anterior translation (drawer)
This is the primary function of the ACL, and so the contributions of the fibre bundles have been studied mostly in this mode. In addition, interest in rotational laxity measurement has only been strong comparatively recently. Selective cutting of the fibre bundles of the ACL, in order to show how they controlled tibial anterior translation laxity, was first performed by Furman et al. [15]. Although they only loaded their knees by hand, they did find significant differences in the increases of laxity following the transection of each fibre bundle. When the AM bundle was cut, the anterior laxity in the extended knee did not increase significantly, but there was a significant increase at 90 degrees knee flexion, when the laxity did not differ significantly from that of the ACL-deficient knee. Conversely, cutting the PL bundle had a significant effect in the extended knee, when the increased laxity was not different from that with complete ACL deficiency, but cutting the PL bundle at 90 degrees flexion had no measureable effect. Thus, they found reciprocal actions, between flexion and extension. This classic paper set a foundation of understanding of the roles of the fibre bundles which remains.
The role of the whole ACL as the ‘primary restraint’ to anterior translation of the tibia was first quantified by Butler et al. [9], who used a materials testing machine to apply carefully controlled displacements and to measure the resulting forces. When the force after ACL transection was deducted from the force with the ACL intact (at the same anterior drawer position), the reduction in force represented the load resisted by the ACL. This method was later used by Amis and Dawkins, [3] with selective cutting of the fibre bundles. The AM bundle was dominant at 90 degrees knee flexion and the PL bundle at 20 degrees. This pattern has since been duplicated by Yagi et al. [50] when they measured the tensions in their double-bundle ACL reconstruction grafts. Norwood and Cross [37] divided the ACL into three fibre bundles: the AM and intermediate bundles were described as primary restraints to anterior translation while, when the PL bundle was selectively cut, the tibial external rotation and recurvatum test increased.
Tensions in the fibre bundles when they resist tibial anterior translation or rotation
The tensions in each of the fibre bundles of the ACL can be discerned by using a robot with a six degree-of-freedom load cell attached to the knee in vitro: this allows the tension in the bundle to be estimated from the difference in the force between intact and bundle-cut states. Sakane et al. [43] examined the forces in the bundles in response to tibial anterior drawer forces ranging from 22 to 110 N. The AM bundle had a relatively constant in situ force, not changing with flexion angle, whereas the PL bundle showed larger in situ force between 0 and 45 degrees of knee flexion, with a peak at 15 degrees, and less when the knee was flexed. Similarly, Gabriel et al. [16] estimated the tensions in the bundles in two loading conditions: anterior drawer and under combined rotational loads. With an isolated tibial anterior drawer force of 134 N at different knee flexion angles, the PL bundle showed a greater tension towards extension while the AM bundle force increased with flexion and was maximal at 60 degrees flexion. At 0 degrees flexion, the PL bundle had significantly higher force than the AM bundle: 67 versus 48 N, respectively. As the knee flexed, the force in the PL bundle declined rapidly: by 60 degrees flexion, the force in the PL bundle had dropped significantly to 15 N, while the AM bundle tension had risen significantly, to 92 N.
Under a combined rotational load of 10 Nm valgus moment and 5 Nm tibial internal rotation torque at 15 and 30 degrees of flexion, Gabriel et al. [16] found that the PL bundle had a larger tension at 15 (21 N) than at 30 degrees knee flexion (14 N). The AM bundle had a similar tension at both angles, and both were significantly higher than in the PL bundle: 30 and 35 N, respectively (Fig. 5). This combination of torques had been found to induce a coupled anterior translation of the tibia and hence was used as a substitute for a pivot-shift test, although that is a dynamic instability that occurs while the knee is flexing–extending, and not at a fixed angle of knee flexion. The results suggest that the PL bundle has some role but that it is less important than the AM bundle when restraining the combined rotational loads used in this experiment, even allowing for differences in orientation of the fibre bundles. (The PL bundle slants across the intercondylar notch, and so its tension acts more directly against tibial rotation than does the tension in the AM bundle, which acts closer to a direction vertical to the plane of the tibial plateau.)
The forces induced in the fibre bundles of the ACL in response to loading the tibia with a combination of 10 Nm valgus moment plus 5 Nm internal rotation torque, intended to simulate the pivot-shift test (Reproduced from Gabriel et al. [16] with permission from John Wiley and Sons)
Thus, these studies found that the PL bundle was tensed near knee extension and relaxed rapidly as the knee flexed. Conversely, the tension in the AM bundle tended to increase with knee flexion. The experiment on combined rotations intended to simulate a pivot shift found that the AM bundle was tensed by tibial rotation significantly more than the PL bundle. However, it should also be noted that the force in the whole ACL in response to rotational loading was less than 40% of that induced by anterior drawer, which implies that resisting tibial rotation is a secondary function.
Tibial rotational laxity and the pivot shift
It has been hypothesised that the AM and PL bundles have different roles in controlling rotational motion and stability of the knee [53]. The AM bundle is oriented nearly vertically in the intercondylar notch in the coronal plane, and so it is thought to have little ability to restrain tibial internal–external rotation, being aligned with, and close to the axis of rotation. Conventional single-bundle ACL reconstruction, with the femoral attachment relatively high in the intercondylar notch, usually replicates only the AM bundle and often results in persistence of a small abnormal rotational laxity that is documented clinically as a residual trace of the pivot shift [7, 31]. Conversely, the PL bundle slants across the notch to a more distal-lateral femoral attachment, and so it has a more horizontal orientation. It has also been assumed to be further from the axis of tibial internal-external rotation, and these factors imply that it should be able to control tibial rotations better than the AM bundle.
Tibial rotational laxity changes in response to complete ACL deficiency have been reported by several studies, with differing conclusions as to whether ACL deficiency has a clinically observable effect on rotational laxity [31]. There has been very little work to measure the effects of the individual fibre bundles of the ACL. Lorbach et al. [34] used an optical navigation system with trackers pinned to the bones in vitro to measure tibiofemoral rotation. At 10 Nm torque (which is above what can be imposed by one hand), cutting the PL bundle led to mean increases of internal and external rotation of 1 or 2 degrees, then cutting the whole ACL led to mean increases of 2 or 3 degrees above the intact levels. Thus, although a statistically significant effect was calculated for the PL bundle, that would not have been easily discernable on clinical examination.
Zantop et al. [53] and Diermann et al. [12] have studied the effects of combined 10 Nm valgus moment and 4 Nm internal rotation torque on tibiofemoral laxity using a robot, in a very similar manner to Gabriel et al. [16]. This loading was used to simulate the pivot shift, although the tests were performed at fixed angles of knee flexion, rather than being more dynamic tests. None of these three studies show data on the effect of cutting ACL fibre bundles on tibial rotational laxity, and only Diermann et al. [12] measured the rotational effect of cutting the whole ACL under these loads, and again they found only two degrees mean increase in tibial internal rotation (and 5 mm increase in anterior translation). Zantop et al. [53] found that isolated transection of the AM bundle had a non-significant effect (1 mm mean increase) on coupled tibial anterior translation in response to the combined rotational torques, whereas cutting the PL bundle led to increased anterior translation of 6 mm (Fig. 6).
When the tibia was loaded with a combination of 10 Nm valgus moment plus 5 Nm internal rotation torque, intended to simulate the pivot-shift test, it induced a coupled anterior translation, which was significantly larger after PL bundle transection than after AM bundle transection (Reproduced from Zantop et al. [53] with permission from Sage Publications)
Recent work in vitro in the author’s laboratory [28] found that cutting either of the ACL bundles individually had statistically significant effects, but these were very small, approximately one degree, and so would be difficult to detect clinically. Even cutting the whole ACL led to a maximum increase of tibial internal rotation of four degrees, with no change in external rotation. This work also measured kinematics during a simulated pivot-shift test, flexing–extending the knee while it was also loaded in a manner known to induce pivot-shift instability [31]: 5 Nm valgus moment, 1 Nm internal rotation torque and 50 N ilio-tibial tract tension. If either the AM or PL bundle were cut alone, tibial anterior translation and internal rotation did not change significantly; only when the whole ACL was cut was there a significant (3 mm) increase in tibial anterior translation, across the arc near knee extension.
Conclusion
The literature reviewed in this paper contains biomechanical data that demonstrate the roles of the two functional fibre bundles of the ACL. Their different patterns of length changes during knee motion, and the resulting variations in the tension, which are induced by loading the knee, lead to differing roles to control tibiofemoral joint laxity, with the PL bundle having a clearer role near knee extension and the AM bundle dominant in the flexed knee. It might then be argued that their different contributions to knee function could constitute a rationale for reconstructing both of these fibre bundles during ACL reconstruction. Although several studies [21, 28, 32] have concluded that the effects on knee laxity of individual ACL fibre bundle deficiencies are so small that they would be difficult to detect clinically, that is not the same as knowing that partial ACL ruptures (of one or other of the fibre bundles) will not cause instability symptoms. It is only recently that there has been much interest in tibial rotational instability, and there remains much scope for further work to understand pathological kinematics and to design the best methods for restoration of normal biomechanical behaviour.
References
Aglietti P, Giron F, Buzzi R, Biddau F, Sasso F (2004) Anterior cruciate ligament reconstruction: bone-patellar tendon-bone compared with double semitendinosus and gracilis tendon grafts. A prospective, randomized clinical trial. J Bone Joint Surg [Am] 86:2143–2155
Amiri S, Cooke TDV, Wyss UP (2011) A multiple-bundle model to characterize the mechanical behaviour of the cruciate ligaments. Knee 18:34–41
Amis AA, Dawkins GP (1991) Functional anatomy of the anterior cruciate ligament. Fibre bundle actions related to ligament replacements and injuries. J Bone Joint Surg [Br] 73B:260–267
Amis AA, Zavras TD (1995) Isometricity and graft placement during anterior cruciate ligament reconstruction. Knee 2:5–17
Bach JM, Hull ML, Patterson HA (1997) Direct measurement of strain in the posterolateral bundle of the anterior cruciate ligament. J Biomech 30:281–283
Brophy RH, Voos JE, Shannon FJ, Granchi CC, Wickiewicz TL, Warren RF, Pearle AD (2008) Changes in the length of virtual anterior cruciate ligament fibers during stability testing. A comparison of conventional single-bundle reconstruction and native anterior cruciate ligament. Am J Sports Med 36:2196–2203
Bull AMJ, Earnshaw PH, Smith A, Katchburian MV, Hassan ANA, Amis AA (2002) Intraoperative measurement of knee kinematics in reconstruction of the anterior cruciate ligament. J Bone Jt Surg 84B:1075–1081
Bush-Joseph CA, Hurwitz DE, Patel RR, Bahrani Y, Garretson R, Bach BR Jr, Andriacchi TP (2001) Dynamic function after anterior cruciate ligament reconstruction with autologous patellar tendon. Am J Sports Med 29:36–41
Butler DL, Noyes FR, Grood ES (1980) Ligamentous restraints to anterior-posterior drawer in the human knee. A Biomechanical study. J Bone Jt Surg [Am] 62:259–270
Butler DL, Guan Y, Kay MD, Cummings JF, Feder SM, Levy MS (1992) Location-dependent variations in the material properties of the anterior cruciate ligament. J Biomech 25:511–518
Chhabra A, Starman JS, Ferretti M, Vidal Af, Zantop T, Fu FH (2006) Anatomic, radiographic, biomechanical, and kinematic evaluation of the anterior cruciate ligament and its two functional bundles. J Bone Joint Surg [Am] 88A(Suppl 4):2–10
Diermann N, Schumacher T, Schanz S, Raschke MJ, Petersen W, Zantop T (2009) Rotational instability of the knee: internal tibial rotation under a simulated pivot shift test. Arch Orthop Trauma Surg 129:353–358
Freedman KB, D’Amato MJ, Nedeff DD, Kaz A, Bach BR Jr (2003) Arthroscopic anterior cruciate ligament reconstruction: a metaanalysis comparing patellar tendon and hamstring tendon autografts. Am J Sports Med 31:2–11
Friederich NF, O’Brien WR (1992) Functional anatomy of the cruciate ligaments. In: Jakob RP, Staeubli HU (eds) The knee and the cruciate ligaments. Springer-Verlag, Berlin, pp 78–91
Furman W, Marshall JL, Girgis FG (1976) The anterior cruciate ligament. A functional analysis based on post-mortem studies. J Bone Jt Surg (Am) 58-A:179–185
Gabriel MT, Wong EK, Woo SL, Yagi M, Debski RE (2004) Distribution of in situ forces in the anterior cruciate ligament in response to rotatory loads. J Orthop Res 22:85–89
Girgis FG, Marshall JL, Al-Monajem A (1975) The cruciate ligaments of the knee joint. Anatomical, functional and experimental analysis. Clin Orthop Relat Res 106:216–231
Hara K, Mochizuki T, Sekiya I, Yamaguchi K, Akita K, Muneta T (2009) Anatomy of normal human anterior cruciate ligament attachments evaluated by divided small bundles. Am J Sports Med 37:2386–2391
Hefzy MS, Grood ES, Noyes FR (1989) Factors affecting the region of most isometric femoral attachments. Part II: the anterior cruciate ligament. Am J Sports Med 17:208–216
Hogervorst T, Pels Rijcken TH, Rucker D, van der Hart CP, Taconis WK (2002) Changes in bone scans after anterior cruciate ligament reconstruction: a prospective study. Am J Sports Med 30:823–833
Hole RL, Lintner DM, Kamaric E, Moseley JB (1996) Increased tibial translation after partial sectioning of the anterior cruciate ligament: the posterolateral bundle. Am J Sports Med 24:556–560
Irvine GB, Glasgow MM (1992) The natural history of the meniscus in anterior cruciate insufficiency. Arthroscopic analysis. J Bone Jt Surg [Br] 74B:403–405
Iwahashi T, Shino K, Nakata K, Nakamura N, Yamada Y, Yoshikawa H, Sugamoto K (2008) Assessment of the “functional length” of the three bundles of the anterior cruciate ligament. Knee Surg Sports Traumatol Arthrosc 16:167–174
Jonsson H, Riklund-Ahlstrom K, Lind J (2004) Positive pivot shift after ACL reconstruction predicts later osteoarthrosis: 63 patients followed 5–9 years after surgery. Acta Orthop Scand 75:594–599
Jordan SS, DeFrate LE, Nha KW, Papannagari R, Gill TJ, Li G (2007) The in vivo kinematics of the anteromedial and posterolateral bundles of the anterior cruciate ligament during weightbearing knee flexion. Am J Sports Med 35:547–554
Kocher MS, Steadman JR, Briggs KK, Sterett WI, Hawkins RJ (2004) Relationships between objective assessment of ligament stability and subjective assessment of symptoms and function after anterior cruciate ligament reconstruction. Am J Sports Med 32:629–634
Kondo E, Merican AM, Yasuda K, Amis AA (2011) Biomechanical comparison of anatomic double-bundle, anatomic single-bundle and non-anatomic single-bundle anterior cruciate ligament reconstructions. Am J Sports Med 39:279–288
Kondo E, Merican AM, Tsai TY, Yasuda K, Amis AA (2012) Biomechanical analysis of partial tears of anteromedial and posterolateral bundles of the anterior cruciate ligament. Personal publication
Kurosawa H, Yamakoshi K, Yasuda K, Sasaki T (1991) Simultaneous measurement of changes in length of the cruciate ligaments during knee motion. Clin Orthop Relat Res 265:233–240
Lee MC, Seong SC, Lee S, Chang CB, Park YK, Jo H, Kim CH (2007) Vertical femoral tunnel placement results in rotational knee laxity after anterior cruciate ligament reconstruction. Arthroscopy 23:771–778
Lie DTT, Bull AMJ, Amis AA (2007) Persistence of the mini pivot shift after anatomically placed anterior cruciate ligament reconstruction. Clin Orthop Relat Res 457:203–209
Lintner DM, Kamaric E, Moseley JB, Noble PC (1995) Partial tears of the anterior cruciate ligament: are they clinically detectable? Am J Sports Med 123:111–118
Logan MC, Williams A, Lavelle J, Gedroyc W, Freeman MAR (2004) Tibiofemoral kinematics following successful anterior cruciate ligament reconstruction using dynamic magnetic resonance imaging. Am J Sports Med 32:984–992
Lorbach O, Pape D, Maas S, Zerbe T, Busch L, Kohn D, Seil R (2010) Influence of the anteromedial and posterolateral bundles of the anterior cruciate ligament on external and internal tibial rotation. Am J Sports Med 38:721–727
McDermott I, Amis AA (2006) Review article: the consequences of meniscectomy. J Bone Jt Surg [Br] 88B:1549–1556
Mott HW (1983) Semitendinosus anatomic reconstruction for cruciate ligament insufficiency. Clin Orthop Relat Res 172:90–92
Norwood LA, Cross MJ (1979) Anterior cruciate ligament: functional anatomy of its bundles in rotatory instabilities. Am J Sports Med 7:23–26
Pinczewski LA, Salmon LJ, Jackson WF, von Bormann RB, Haslam PG, Tashiro S (2008) Radiological landmarks for placement of the tunnels in single-bundle reconstruction of the anterior cruciate ligament. J Bone Jt Surg [Br] 90-B:172–179
Race A, Amis AA (1994) The mechanical properties of the two bundles of the human posterior cruciate ligament. J Biomech 27:13–24
Radford WJ, Amis AA, Kempson SA, Stead AC, Camburn M (1994) A comparative study of single- and double-bundle ACL reconstructions in sheep. Knee Surg Sports Traumatol Arthrosc 2:94–99
Ristanis S, Giakas G, Papageorgiou CD, Moraiti T, Stergiou N, Georgoulis AD (2003) The effects of anterior cruciate ligament reconstruction on tibial rotation during pivoting after descending stairs. Knee Surg Sports Traumatol Arthrosc 11:360–365
Robinson JR, Bull AMJ, Amis AA (2005) Structural properties of the medial collateral ligament complex of the human knee. J Biomech 38:1067–1074
Sakane M, Fox RJ, Woo SL, Livesay GA, Li G, Fu FH (1997) In situ forces in the anterior cruciate ligament and its bundles in response to anterior tibial loads. J Orthop Res 15:285–293
Sapega AA, Moyer RA, Schneck C, Komalahiranya N (1990) Testing for isometry during reconstruction of the anterior cruciate ligament. J Bone Jt Surg 72A:259–267
Sidles JA, Larson RV, Garbini JL, Downey DJ, Matsen FA (1988) Ligament length relationships in the moving knee. J Orthop Res 6:583–610
Steckel H, Vadala G, Davis D, Fu F (2006) 2D and 3D 3-Tesla magnetic resonance imaging of the double-bundle structure in anterior cruciate ligament anatomy. Knee Surg Sports Traumatol Arthrosc 14:1151–1158
Tanaka Y, Shino K, Horibe S, Nakamura N, Nakagawa S, Mae T, Otsubo H, Suzuki T (2011) Triple-bundle ACL grafts evaluated by second-look arthroscopy. Knee Surg Sports Traumatol Arthrosc. doi:10.1007/s00167-011-1551-8
Tashman S, Collon D, Anderson K, Kolowich P, Anderst W (2004) Abnormal rotational knee motion during running after anterior cruciate ligament reconstruction. Am J Sports Med 32:975–983
Woo SL, Debski RE, Withrow JD, Janaushek MA (1999) Biomechanics of knee ligaments. Am J Sports Med 27:533–543
Yagi M, Wong EK, Kanamori A, Debski RE, Fu FH, Woo SLY (2002) Biomechanical analysis of an anatomic anterior cruciate ligament reconstruction. Am J Sports Med 30:660–666
Yoo YK, Jeong WS, Shetty NS, Ingham SJM, Smolinski P, Fu F (2010) Changes in ACL length at different knee flexion angles: an in vivo biomechanical study. Knee Surg Sports Traumatol Arthrosc 18:292–297
Zantop T, Petersen W, Sekiya JK, Musahl V, Fu FH (2006) Anterior cruciate ligament anatomy and function relating to anatomical reconstruction. Knee Surg Sports Traumatol Arthrosc 14:982–992
Zantop T, Herbort M, Raschke MJ, Fu FH, Petersen W (2007) The role of the anteromedial and posterolateral bundles of the anterior cruciate ligament in anterior tibial translation and internal rotation. Am J Sports Med 35:223–227
Zaricznyj B (1987) Reconstruction of the anterior cruciate ligament of the knee using a doubled tendon graft. Clin Orthop Relat Res 220:162–175
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Amis, A.A. The functions of the fibre bundles of the anterior cruciate ligament in anterior drawer, rotational laxity and the pivot shift. Knee Surg Sports Traumatol Arthrosc 20, 613–620 (2012). https://doi.org/10.1007/s00167-011-1864-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-011-1864-7