Abstract
Harvesting both the semitendinosus and gracilis tendons for anterior cruciate ligament (ACL) reconstruction has a negative impact on muscle strength as well as knee function and stability. With a new “All-inside” technique, using only one hamstrings tendon (semitendinosus or gracilis) is possible because of a reduction in length requirements. The research question of this in vitro study was whether the use of only one hamstrings tendon (semitendinosus or gracilis) could restore knee kinematics and in situ force in the ACL to the level of an intact knee. Ten human cadaveric knees were tested in the following conditions: (1) intact, (2) ACL-deficient, and (3) ACL reconstruction with the “All-inside” technique using the (a) single semitendinosus tendon graft, or (b) single gracilis tendon graft. Using a robotic testing system, external loads, i.e. (1) an anterior tibial load of 134-N and (2) combined rotatory loads of 10-Nm valgus and 5-Nm internal tibial torques, were applied. The multiple degrees of freedom knee kinematics and the in situ forces in the ACL and ACL grafts were determined. In response to a 134-N anterior tibial load, the use of either graft could restore anterior tibial translation to within 1.3 mm of the intact knee. The in situ forces in the two grafts were not significantly different from those of the intact ACL. Under the combined rotatory loads, both grafts could restore knee kinematics as well as the in situ force in the grafts to the level of the intact ACL. The “All-inside” technique using either the semitendinosus or gracilis tendon for ACL reconstruction could satisfactorily restore time-zero knee kinematics and the in situ forces in either graft to those for the intact ACL, supporting clinical findings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anterior cruciate ligament (ACL) reconstruction using hamstrings tendons, typically both the semitendinosus and gracilis tendons as a quadrupled (QSTG) autograft, has gained popularity because these grafts are thought to provide sufficient initial stiffness and offer advantages over the bone-patellar tendon-bone graft in terms of reducing inferior patellar contracture, quadriceps weakness, extension deficits, anterior knee pain, as well as donor site morbidity [7, 10, 18, 25, 32, 34, 35]. However, harvesting both tendons for use in the QSTG autograft also presents new issues that include the loss of active knee flexion at deeper flexion angles and lower internal tibial torque as well as muscular weakness that directly impacts knee function and stability [29, 31, 50]. Even at 9 months after surgery, the strength of the hamstrings muscles are reduced [50]. Such a reduction would be detrimental to athletic performance, especially in sports, such as wrestling, football, and soccer, which require extensive use of the hamstrings muscles [3, 5, 8, 11]. Further, the semitendinous tendon is needed to prevent excessive anterior tibial translation when the knee is near full extension [37]. Therefore, it would be desirable to preserve as much of the hamstrings tendon as possible for ACL reconstruction, particularly for younger and more athletic patients [20].
Recently, a clinical alternative, namely the “All-inside” technique, has been adopted for ACL reconstruction [9, 24, 38]. In this technique, the femoral and tibial tunnels are manually drilled only halfway through the bones for graft fixation. Thus, the effective length of the graft required is significantly less than traditional full tunnel methods. As a result, only one hamstrings tendon (either the semitendinosus or gracilis), folded in triple or quadruple strands, is needed as an ACL replacement autograft, and thus, one tendon could be spared. A clinical study of 622 patients at 19–48 months postoperatively found 96% patient satisfaction [9]. There were no graft failures and all could return to pre-injury levels of activity, with good muscle function and knee stability. Nevertheless, it is necessary to back up these positive results with supporting scientific data.
Thus, the objective of this in vitro study was to evaluate the use of one hamstring tendon (semitendinosus or gracilis) graft in the “All-inside” technique for ACL reconstruction in terms of the restoration of knee kinematics and in situ force of the ACL to the level of an intact knee under applied loads simulating clinical exams. As the reduced tunnel length would allow a semitendinosus tendon graft folded in triple form or a gracilis tendon graft folded in quadruple form, we hypothesized that ACL reconstruction using only the semitendinosus or gracilis tendon autograft could restore the normal knee kinematics and the in situ forces in the ACL to the levels of the intact knee. Secondly, since the semitendinosus and gracilis tendon grafts have similar cross-sectional dimensions and biomechanical properties [14, 43, 51], we further hypothesized that there would be no significant differences between reconstructions using either the semitendinosus or the gracilis grafts in terms of knee kinematics and in situ forces in the graft. To test our hypotheses, the intact, ACL-deficient, and reconstructed knees were tested on a robotic/universal force moment sensor (UFS) testing system between full extension and 120° of knee flexion, and the 5 degrees of freedom (DOF) knee kinematics as well as the in situ forces in the intact ACL and the ACL replacement grafts could be measured and compared. This unique system is capable of obtaining data accurately without contacting the ligament or its replacement graft [12, 21].
Methods
Ten fresh-frozen human cadaveric knees (54 ± 8.7 years of age; range, 42–64 years) were used in this study. On the basis of our previous data, a power analysis was performed (power = .80, significance level = .05), so that differences of 2 mm for anterior tibial translation and 20 N for in situ force measurements could be detected. It was determined that 10 knees were required for this study. Each specimen was screened for a history of osteoarthritis, lower limb trauma, and neurologic disease. Specimens were stored in airtight plastic bags at −20°C until 24 h before testing, when they were thawed at room temperature [27, 45]. Clinical and arthroscopic examination of the knee joint was performed to confirm the presence of an intact and functional ACL, as well as the presence of any bony abnormalities and osteoarthritis. The semitendinosus and gracilis tendons were harvested from each knee to be used as the ACL replacement grafts, using a tendon stripper. The grafts were then wrapped in saline-soaked gauze to prevent dehydration.
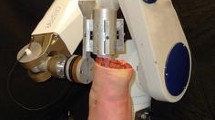
In preparation for testing, the femur and tibia were then cut approximately 20 cm from the joint line and all soft tissues were removed approximately 10 cm away from the joint line on both the femur and tibia, while leaving the joint intact. The fibula was rigidly fixed to the tibia with a cortical screw to maintain its anatomic position. The femur and tibia were each secured within custom-made aluminum cylinders by using an epoxy compound (Fibre Glass-Evercoat, Cincinnati, Ohio) with transfixing bolts. The specimen was then mounted in a robotic/UFS testing system [12, 13, 21, 33]. The femoral side was rigidly mounted to the base of the robotic manipulator (KUKA Model KR 210), while the tibial side was attached to the end-effector of the robotic manipulator via a load cell (Model Theta, ATI Industrial Automation) (Fig. 1). The robotic manipulator has a position and orientation accuracy and repeatability of less than 0.1 mm and 0.1°, respectively. The UFS is capable of measuring 3 forces and 3 moments in a Cartesian coordinate system fixed with respect to the sensor. Using the robotic/UFS testing system, the knee kinematics and the in situ forces in the intact ACL and the ACL replacement grafts were obtained. Throughout the experiment, the specimens were kept moist with 0.9% saline solution.
The sequence of tests performed and the data acquired are outlined in Table 1. The path of passive flexion–extension of the intact knee from full extension to 120° of flexion was first determined by the robotic/UFS testing system in 1° increments by means of minimizing all the external forces and moments. This path serves as the reference position from which external loads are applied and kinematics data are collected [17, 22, 42, 47]. Two external loading conditions were applied to the knee: (1) a 134-N anterior tibial load with the knee at full extension, 15, 30, 60, 90, and 120° of flexion; and (2) a combined 10 N m of valgus torque and 5 N m of internal–external tibial torque at 15° and 30° of knee flexion. The anterior tibial load simulates clinical examinations such as the anterior drawer and the Lachman tests, which are commonly used for the diagnosis of ACL deficiency. The combined rotatory load (CRL) statically simulates the pivot-shift test, which is another examination used to determine knee instability, especially rotatory instability due to ACL injury [30, 40, 41]. The 5 DOF kinematics of the intact knee (i.e., anterior–posterior, medial–lateral, and proximal–distal translations, as well as internal–external and varus-valgus rotations) were recorded at each flexion angle. The in situ force in the ACL could be determined by carefully transecting the ACL through a medial mini-arthrotomy. The medial mini-arthrotomy was done during specimen preparation; therefore, any effect of performing this procedure was consistent throughout the experiment. Then, the 5 DOF kinematics of the intact knee were repeated by the robotic manipulator in position-control mode for the ACL-deficient knee, while the UFS measured the new forces and moments. On the basis of the principle of superposition, the vector difference in forces measured between the intact and the ACL-deficient states represented the in situ force in the ACL [12, 33]. To assess changes in knee kinematics associated with ACL deficiency, the same external loading conditions previously applied to the intact knee were again applied to the ACL-deficient knee, and the resulting 5 DOF kinematics were determined.
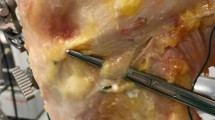
Subsequently, the ACL reconstructions were performed by one orthopedic surgeon (GZ) using the “All-inside” technique, using the (1) semitendinosus tendon alone and (2) the gracilis tendon alone. Each reconstruction required approximately 50 min, and the order of the reconstructions was randomized among knees. The ACL reconstruction was performed using a single femoral tunnel and a single tibial tunnel. The femoral tunnel was drilled using a free-hand technique using an inside-out technique through the mini-arthrotomy and was placed as posterior as possible within the intercondylar notch (without breaking the posterior wall of the femoral condyle) at approximately the 10 o’clock position in the right knee and at approximately the 2 o’clock position in the left knee. The optimal placement of the tunnel was marked with a 90° awl. A guide wire was placed at the marked optimal position and drilled through the femoral condyle at 120° of knee flexion. The tunnel was initially drilled completely through the bone with a 4.5-mm-diameter cannulated drill (EndoButton Drill, Acufex, Smith and Nephew, Andover, MA) over the guide wire. The tibial tunnel was made in the middle of the footprint of the tibial insertion of the ACL using a Protac tibial guide set (Acufex, Smith and Nephew, Andover, MA) at 60°, and was initially drilled using an outside-in technique with a 4.5-mm-diameter cannulated drill over the guide wire. The final drilling of the tunnels was made with a special manual retro-drill that has “drill-wings” which are turned out inside the joint and used to create the final half-tunnels starting from the inside of the joint. The diameter of the manually drilled half-tunnels were typically in range of 7–9 mm, the depth of the femoral tunnel was in range of 30–40 mm and the depth of tibial tunnel was 20–30 mm. The dimensions of the tripled semitendinosus tendon graft and quadruplicated gracilis tendon used for each All-inside reconstruction are provided in Table 2.
The femoral side was first fixed using an EndoButton CL (Smith and Nephew). The graft was then pulled through the tibial tunnel, and the knee was preconditioned by moving the knee through 5 cycles of the full range of knee flexion while applying a 22-N pretension. Finally, the tibial side was fixed using a cortical screw and washer as a post. For both reconstructions, the graft was fixed at 30° of knee flexion. Each graft was fixed while a 67-N posterior tibial load and 22-N of initial graft tension were maintained. Previous studies have shown that applying 67-N of posterior load during the fixation of the graft can more closely restore the knee kinematics and in situ force in the ACL graft to those of intact knee [15].
After each reconstruction, the same external loading conditions applied to the intact knee were repeated on the ACL-reconstructed knee to obtain the corresponding 5-DOF knee kinematics. The forces in the grafts were obtained using the superposition principle by releasing the grafts and replaying the knee kinematics of the reconstructed knee to measure the changes in forces.
Statistical methods
Because all variables were measured on the same specimen, statistical analysis of knee kinematics and in situ forces was performed using a 1-factor repeated measures analysis of variance (ANOVA), with knee state as the factor. Statistical significance was set at P < 0.05. A Bonferroni posthoc analysis was done to evaluate the effects of ACL reconstruction at specific angles of knee flexion.
Results
In response to a 134-N anterior tibial load, the anterior tibial translation for the intact knee increased from full extension to 30° of flexion, and then remained roughly level through 120° of flexion (Table 3). After ACL transection, the values for anterior tibial translation significantly increased by two to threefold throughout the range of flexion angles tested, and were 5.2–9.8 mm higher than the intact knee (P < 0.05). After ACL reconstruction, the anterior tibial translation was restored to within 1.3 mm of the intact knee using either the single semitendinosus or single gracilis autograft. For example, the values at full extension were only 0.5 and 0.4 mm higher than the intact knee, respectively (P > 0.05). These values were also 3.9–9.3 mm lower than the ACL-deficient knee throughout knee flexion (P < 0.05). There were no significant differences between the two reconstructions (P > 0.05). In fact, the maximum difference between the graft protocols was only 0.8 mm at 120° of flexion (P > 0.05).
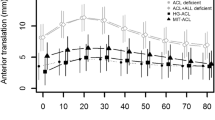
The resultant in situ forces in the intact ACL in response to a 134-N anterior tibial load increased slightly from full extension to 15° and 30° of knee flexion (137 ± 10 N) before decreasing steadily until 120° of knee flexion (76 ± 21 N) (Fig. 2). After ACL reconstruction, the trends for in situ force of the replacement grafts were similar to those for the intact ACL throughout the range of knee flexion. For the single semitendinosus graft, the corresponding values for the graft were 121 ± 30 N at 30° of knee flexion and 54 ± 22 N at 120° of knee flexion and were not statistically different compared with the intact ACL (P > 0.05) (Fig. 2). The same was true for the single gracilis graft where the corresponding values were 125 ± 29 N to 66 ± 19 N, respectively (P > 0.05). Further, no statistical differences were detected between the two autografts (P > 0.05) with the only exception at 120° of knee flexion where the in situ force of the single semitendinosus graft was on average 12 N lower (P < 0.05).
In response to the combined rotatory loads, values for the coupled anterior tibial translation increased from 15 to 30° of knee flexion under all experimental conditions (Table 4). After transection of the ACL, the anterior tibial translation values increased by three to eightfold compared to the intact knee at 15 and 30° (P < 0.05). With reconstruction, the coupled anterior tibial translation significantly decreased by 3.1–3.5 mm in comparison with the ACL-deficient knee (P < 0.05). After ACL reconstruction, the anterior tibial translation was restored to within 0.4 mm of the intact knee using either the single semitendinosus or single gracilis autograft (Table 4). No significant differences were detected when comparing the coupled anterior tibial translation of either reconstruction procedure to the intact knee or when comparing the two reconstruction procedures to each other (P > 0.05).
Under all experimental conditions, the internal tibial rotation increased from 15 to 30° of knee flexion (Table 4). After transection of the ACL, the internal tibial rotation values significantly increased by 1.8 at 15° (P < 0.05) but not at 30° of knee flexion (P > 0.05). Values for internal tibial rotation were restored to within 0.7 and 1.1° of the intact state at 15 and 30° of knee flexion, respectively, for both the single semitendinosus graft (P > 0.05) and single gracilis graft (P > 0.05). When comparing the two graft protocols, no statistical differences could be shown (P > 0.05).
Under the combined rotatory load, the resultant in situ force of the intact ACL was 83 ± 34 N at 15° and 73 ± 36 N at 30° of knee flexion (Fig. 3). The corresponding values for the single semitendinosus graft were within 9 N of the intact knee (74 ± 30 N and 67 ± 35 N, respectively), while the values for the single gracilis graft were within 2 N (85 ± 28 N and 75 ± 30 N, respectively). No statistically significant differences could be found between the in situ force of the grafts and the intact ACL (P > 0.05). Further, there were no statistically significant differences between the grafts, with a maximum difference of 11 N (P > 0.05).
Discussion
In this controlled laboratory study, quantitative data on knee kinematics as well as the in situ forces in the single hamstrings tendon autografts used for ACL reconstruction in an “All-inside” technique were determined in human cadaver knees by means of a robotic/UFS testing system. With an anterior tibial load and combined rotatory loads designed to mimic clinical exams, it was found that either autograft, i.e. semitendinosus or gracilis, could restore the knee kinematics to within 1.3 mm of those of the intact knee. Further, the resultant in situ forces in the grafts were not significantly different to those of the intact ACL, supporting our hypothesis. In addition, the knee kinematics and in situ forces of the intact ACL obtained in our study were similar to those published in the literature [17, 22, 36, 44, 47]. Our data suggest that only one hamstrings tendon in triple or quadruple strands could be used as an autograft for ACL reconstruction because the length of the needed graft is shortened using “All-inside” procedures, while the cross-sectional area remained similar to standard QSTG grafts.
Further, both the kinematics of the reconstructed knee and in situ forces of the single semitendinosus and single gracilis grafts were not different from each other under these loading conditions, except at 120° of flexion in response to the anterior tibial load. These results are not surprising since the semitendinosus and gracilis tendons have similar biomechanical properties [14, 43, 51], and the final single semitendinosus tendon graft and single gracilis tendon graft are folded in multiple strands such that their dimensions are comparable (Table 2). As such, in this time-zero study, there is no clear biomechanical advantage for harvesting one hamstrings tendon over another in terms of restoring knee kinematics and in situ force of the graft near the levels of the normal ACL.
Using our robotic/UFS testing system, our research center has previously published data on ACL reconstruction using a QSTG autograft and has found it could restore knee kinematics and in situ force in the ACL [44, 47, 48]. When the autograft was placed in the femoral insertion site of the PL bundle [48], the anterior tibial translation under a 134-N anterior tibial load was found to be within 2 mm of the intact knee from full extension to 90° of knee flexion. Additionally, the QSTG graft was able to restore the in situ force of the ACL graft to within 17 N throughout the range of knee flexion studied. Similarly, in response to combined rotatory loads, the QSTG graft was able to restore coupled anterior tibial translation and internal tibial rotation, as well as the in situ force of the QSTG graft to within 6 N of the intact ACL at 15 and 30° of knee flexion. Interestingly, the data in the current study using one hamstrings tendon were similarly effective compared to those of the QSTG tendon autograft in terms of restoring knee kinematics as well as in situ force of the replacement graft to the levels of the intact knee.
In more recent years, double bundle ACL reconstruction procedures have gained in popularity, as they are thought to better recreate the natural anatomy of the ACL [1, 4, 6, 16, 19, 26, 28, 29, 39, 46, 47, 49]. Our research center has compared an anatomic double-bundle reconstruction to a single-bundle reconstruction at the femoral insertion site of the PL bundle [48]. In this case, in response to anterior tibial and combined rotatory loads, both reconstructions were able to restore anterior tibial translation and in situ force in the ACL graft near those for the intact knee at flexion angles near knee extension. However, the double-bundle procedure was able to better restore anterior tibial translation in deeper flexion. Thus, to reproduce the complex function of the ACL throughout the range of knee flexion, reproducing both bundles of the ACL may have biomechanical advantages. On the other hand, a more laterally placed single-bundle reconstruction, such as the one used in the current study, may also work quite well, especially with the knee near extension where the ACL is most needed. This is especially important since long-term clinical data on double-bundle ACL reconstruction are not yet available, and better clinical outcomes with double bundle ACL reconstruction procedures has yet to be demonstrated [23].
Due to concerns about harvesting both the semitendinosus and gracilis tendons and the resulting impact on knee function and stability [2, 29, 31], it would be important to preserve one of the two hamstring tendons by the “All-inside” technique. Moreover, this novel surgical approach may have other apparent advantages. Since the tunnels are drilled manually from the inside-out, there is a decrease in length of the bone tunnels, and as a result, reduced bone loss. Also, the tunnel lengths can be adjusted for optimum fixation.
There are limitations of this study, however. First, cadaveric knees were tested in vitro, and thus, only represent a time-zero analysis that does not capture the effects of the in vivo healing response or additional cyclic loading. Second, the relatively simple external loading conditions in this study are a simulation of those used in clinical examinations. In vivo loading conditions, such as those during the pivot movements, which involve the iliotibial band as well as other muscle loads, were not included. Third, the level of externally applied loads used in this study is lower than those experienced while performing activities of daily living, thus forces in the grafts were less than that could potentially fail the graft [48]. Future studies will be performed on our robotic/UFS testing system using in vivo loading conditions and higher load levels in order to fully evaluate the “All-inside” technique.
Nevertheless, this study provided new and quantitative information. By measuring knee kinematics while the knee undergoes unrestricted motion in multiple degrees of freedom, the in situ force in the ACL and ACL replacement grafts were determined in a non-contact manner. More importantly, since a common reference position could be established, the experimental data collected for an intact knee, ACL-deficient knee, and ACL-reconstructed knee with single semitendinosus or single gracilis graft protocols were from the same cadaveric specimen, thus eliminating the large inter-specimen variation, and yielding significant increases in the statistical power [12].
In the end, the findings of this controlled laboratory study give confidence to those in the clinical setting that ACL reconstruction using the “All-inside” technique with either the semitendinosus and gracilis tendons was sufficient to maintain initial postoperative knee stability. The data also show that the in situ force in the grafts is also at the appropriate levels, i.e. compared to the intact ACL as well as those found for the commonly used QSTG tendon graft. Thus, we were able to positively answer our research question that at time-zero the “All-inside” technique could restore knee kinematics and in situ force of the ACL to the level of an intact knee and may have merit for continued use in ACL reconstruction procedures.
Conclusions
An “All-inside” technique using either the semitendinosus or gracilis tendon for ACL reconstruction could satisfactorily restore initial knee kinematics and the in situ forces in either graft to those for the intact ACL. These data also compared favorably to published results using both hamstrings as a quadrupled tendon autograft. These results in human cadaveric knees support the clinical findings using the “All-inside” procedure with only a single hamstrings tendon graft for ACL reconstruction.
References
Adachi N, Ochi M, Uchio Y, Iwasa J, Kuriwaka M, Ito Y (2004) Reconstruction of the anterior cruciate ligament. Single- versus double-bundle multistranded hamstring tendons. J Bone Joint Surg Br 86:515–520
Adachi N, Ochi M, Uchio Y, Sakai Y, Kuriwaka M, Fujihara A (2003) Harvesting hamstring tendons for ACL reconstruction influences postoperative hamstring muscle performance. Arch Orthop Trauma Surg 123:460–465
Agel J, Ransone J, Dick R, Oppliger R, Marshall SW (2007) Descriptive epidemiology of collegiate men’s wrestling injuries: National Collegiate Athletic Association Injury Surveillance System, 1988–1989 through 2003–2004. J Athl Train 42:303–310
Aglietti P, Giron F, Cuomo P, Losco M, Mondanelli N (2007) Single-and double-incision double-bundle ACL reconstruction. Clin Orthop Relat Res 454:108–113
Arnason A, Andersen TE, Holme I, Engebretsen L, Bahr R (2008) Prevention of hamstring strains in elite soccer: an intervention study. Scand J Med Sci Sports 18:40–48
Asagumo H, Kimura M, Kobayashi Y, Taki M, Takagishi K (2007) Anatomic reconstruction of the anterior cruciate ligament using double-bundle hamstring tendons: surgical techniques, clinical outcomes, and complications. Arthroscopy 23:602–609
Bonamo JJ, Krinick RM, Sporn AA (1984) Rupture of the patellar ligament after use of its central third for anterior cruciate reconstruction. A report of two cases. J Bone Joint Surg Am 66:1294–1297
Brophy RH, Barnes R, Rodeo SA, Warren RF (2007) Prevalence of musculoskeletal disorders at the NFL Combine–trends from 1987 to 2000. Med Sci Sports Exerc 39:22–27
Cerulli G, Caraffa A, Zamarra G, Antinolfi P, Vercillo F (2006) Mid-term follow-up of ACL reconstruction with All-inside technique. Presented at 12th ESSKA Congress, Innsbruck, Austria
Christen B, Jakob RP (1992) Fractures associated with patellar ligament grafts in cruciate ligament surgery. J Bone Joint Surg Br 74:617–619
Feeley BT, Kennelly S, Barnes RP, Muller MS, Kelly BT et al (2008) Epidemiology of National Football League training camp injuries from 1998 to 2007. Am J Sports Med 36:1597–1603
Fujie H, Livesay GA, Woo SL-Y, Kashiwaguchi S, Blomstrom G (1995) The use of a universal force-moment sensor to determine in situ forces in ligaments: a new methodology. J Biomech Eng 117:1–7
Fujie H, Mabuchi K, Woo SL-Y, Livesay GA, Arai S, Tsukamoto Y (1993) The use of robotics technology to study human joint kinematics: a new methodology. J Biomech Eng 115:211–217
Handl M, Drzik M, Cerulli G, Povysil C, Chlpik J et al (2007) Reconstruction of the anterior cruciate ligament: dynamic strain evaluation of the graft. Knee Surg Sports Traumatol Arthrosc 15:233–241
Hoher J, Kanamori A, Zeminski J, Fu FH, Woo SL-Y (2001) The position of the tibia during graft fixation affects knee kinematics and graft forces for anterior cruciate ligament reconstruction. Am J Sports Med 29:771–776
Jarvela T (2007) Double-bundle versus single-bundle anterior cruciate ligament reconstruction: a prospective, randomize clinical study. Knee Surg Sports Traumatol Arthrosc 15:500–507
Kanamori A, Woo SL-Y, Ma CB, Zeminski J, Rudy TW et al (2000) The forces in the anterior cruciate ligament and knee kinematics during a simulated pivot shift test: A human cadaveric study using robotic technology. Arthroscopy 16:633–639
Kartus J, Stener S, Lindahl S, Engstrom B, Eriksson BI, Karlsson J (1997) Factors affecting donor-site morbidity after anterior cruciate ligament reconstruction using bone-patellar tendon-bone autografts. Knee Surg Sports Traumatol Arthrosc 5:222–228
Kondo E, Yasuda K, Azuma H, Tanabe Y, Yagi T (2008) Prospective clinical comparisons of anatomic double-bundle versus single-bundle anterior cruciate ligament reconstruction procedures in 328 consecutive patients. Am J Sports Med 36:1675–1687
Liu W, Maitland ME (2000) The effect of hamstring muscle compensation for anterior laxity in the ACL-deficient knee during gait. J Biomech 33:871–879
Livesay GA, Fujie H, Kashiwaguchi S, Morrow DA, Fu FH, Woo SL-Y (1995) Determination of the in situ forces and force distribution within the human anterior cruciate ligament. Ann Biomed Eng 23:467–474
Loh JC, Fukuda Y, Tsuda E, Steadman RJ, Fu FH, Woo SL-Y (2003) Knee stability and graft function following anterior cruciate ligament reconstruction: Comparison between 11 o’clock and 10 o’clock femoral tunnel placement. 2002 Richard O’Connor Award paper. Arthroscopy 19:297–304
Longo UG, King JB, Denaro V, Maffulli N (2008) Double-bundle arthroscopic reconstruction of the anterior cruciate ligament: does the evidence add up? J Bone Joint Surg Br 90:995–999
Lubowitz JH (2006) No-tunnel anterior cruciate ligament reconstruction: the transtibial all-inside technique. Arthroscopy 22: 900 e901–e911
Maletis GB, Cameron SL, Tengan JJ, Burchette RJ (2007) A prospective randomized study of anterior cruciate ligament reconstruction: a comparison of patellar tendon and quadruple-strand semitendinosus/gracilis tendons fixed with bioabsorbable interference screws. Am J Sports Med 35:384–394
Mastrokalos DS, Springer J, Siebold R, Paessler HH (2005) Donor site morbidity and return to the preinjury activity level after anterior cruciate ligament reconstruction using ipsilateral and contralateral patellar tendon autograft: a retrospective, nonrandomized study. Am J Sports Med 33:85–93
Moon DK, Woo SL-Y, Takakura Y, Gabriel MT, Abramowitch SD (2006) The effects of refreezing on the viscoelastic and tensile properties of ligaments. J Biomech 39:1153–1157
Muneta T, Koga H, Mochizuki T, Ju YJ, Hara K et al (2007) A prospective randomized study of 4-strand semitendinosus tendon anterior cruciate ligament reconstruction comparing single-bundle and double-bundle techniques. Arthroscopy 23:618–628
Nakamura N, Horibe S, Sasaki S, Kitaguchi T, Tagami M et al (2002) Evaluation of active knee flexion and hamstring strength after anterior cruciate ligament reconstruction using hamstring tendons. Arthroscopy 18:598–602
Nielsen S, Helmig P (1985) Instability of knees with ligament lesions. Cadaver studies of the anterior cruciate ligament. Acta Orthop Scand 56:426–429
Ohkoshi Y, Inoue C, Yamane S, Hashimoto T, Ishida R (1998) Changes in muscle strength properties caused by harvesting of autogenous semitendinosus tendon for reconstruction of contralateral anterior cruciate ligament. Arthroscopy 14:580–584
Risberg MA, Holm I, Tjomsland O, Ljunggren E, Ekeland A (1999) Prospective study of changes in impairments and disabilities after anterior cruciate ligament reconstruction. J Orthop Sports Phys Ther 29:400–412
Rudy TW, Livesay GA, Woo SL-Y, Fu FH (1996) A combined robotic/universal force sensor approach to determine in situ forces of knee ligaments. J Biomech 29:1357–1360
Sachs RA, Daniel DM, Stone ML, Garfein RF (1989) Patellofemoral problems after anterior cruciate ligament reconstruction. Am J Sports Med 17:760–765
Sajovic M, Vengust V, Komadina R, Tavcar R, Skaza K (2006) A prospective, randomized comparison of semitendinosus and gracilis tendon versus patellar tendon autografts for anterior cruciate ligament reconstruction: five-year follow-up. Am J Sports Med 34:1933–1940
Sakane M, Fox RJ, Woo SL-Y, Livesay GA, Li G, Fu FH (1997) In situ forces in the anterior cruciate ligament and its bundles in response to anterior tibial loads. J Orthop Res 15:285–293
Shelburne KB, Torry MR, Pandy MG (2005) Effect of muscle compensation on knee instability during ACL-deficient gait. Med Sci Sports Exerc 37:642–648
Smith PA (2007) An alternative method for “all-inside” anterior cruciate ligament reconstruction. Arthroscopy 23:451
Streich NA, Friedrich K, Gotterbarm T, Schmitt H (2008) Reconstruction of the ACL with a semitendinosus tendon graft: a prospective randomized single blinded comparison of double-bundle versus single-bundle technique in male athletes. Knee Surg Sports Traumatol Arthrosc 16:232–238
Sullivan D, Levy IM, Sheskier S, Torzilli PA, Warren RF (1984) Medical restraints to anterior-posterior motion of the knee. J Bone Joint Surg Am 66:930–936
Torzilli PA, Greenberg RL, Insall J (1981) An in vivo biomechanical evaluation of anterior-posterior motion of the knee. Roentgenographic measurement technique, stress machine, and stable population. J Bone Joint Surg Am 63:960–968
Tsuda E, Fukuda Y, Loh JC, Debski RE, Fu FH, Woo SL-Y (2002) The effect of soft-tissue graft fixation in anterior cruciate ligament reconstruction on graft-tunnel motion under anterior tibial loading. Arthroscopy 18:960–967
Woo SL-Y, Abramowitch SD, Kilger R, Liang R (2006) Biomechanics of knee ligaments: injury, healing, and repair. J Biomech 39:1–20
Woo SL-Y, Kanamori A, Zeminski J, Yagi M, Papageorgiou C, Fu FH (2002) The effectiveness of reconstruction of the anterior cruciate ligament with hamstrings and patellar tendon. A cadaveric study comparing anterior tibial and rotational loads. J Bone Joint Surg Am 84A:907–914
Woo SL-Y, Orlando CA, Camp JF, Akeson WH (1986) Effects of postmortem storage by freezing on ligament tensile behavior. J Biomech 19:399–404
Yagi M, Kuroda R, Nagamune K, Yoshiya S, Kurosaka M (2007) Double-bundle ACL reconstruction can improve rotational stability. Clin Orthop Relat Res 454:100–107
Yagi M, Wong EK, Kanamori A, Debski RE, Fu FH, Woo SL-Y (2002) Biomechanical analysis of an anatomic anterior cruciate ligament reconstruction. Am J Sports Med 30:660–666
Yamamoto Y, Hsu WH, Woo SL-Y, Van Scyoc AH, Takakura Y, Debski RE (2004) Knee stability and graft function after anterior cruciate ligament reconstruction: a comparison of a lateral and an anatomical femoral tunnel placement. Am J Sports Med 32:1825–1832
Yasuda K, Kondo E, Ichiyama H, Tanabe Y, Tohyama H (2006) Clinical evaluation of anatomic double-bundle anterior cruciate ligament reconstruction procedure using hamstring tendon grafts: comparisons among 3 different procedures. Arthroscopy 22:240–251
Yasuda K, Tsujino J, Ohkoshi Y, Tanabe Y, Kaneda K (1995) Graft site morbidity with autogenous semitendinosus and gracilis tendons. Am J Sports Med 23:706–714
Zhang X, Curran M, Abramowitch SD, Woo SL-Y (2006) The Viscoelastic Properties of Human Hamstring Tendons. Presented at 5th World Congress on Biomaterials, Munich, Germany
Acknowledgments
Financial support of National Institutes of Health (AR39683, T32 EB000392), Innovazione Medica, and LARS Company, which developed the equipment and instrumentation for the “All-inside” technique, are acknowledged. The support and collaboration of Prof. Giuliano Cerulli (inventor of “All-inside” technique) and Let People Move are recognized.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zamarra, G., Fisher, M.B., Woo, S.LY. et al. Biomechanical evaluation of using one hamstrings tendon for ACL reconstruction: a human cadaveric study. Knee Surg Sports Traumatol Arthrosc 18, 11–19 (2010). https://doi.org/10.1007/s00167-009-0911-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-009-0911-0