Abstract
This prospective outcomes study was designed to prospectively investigate the outcome of the microfracture technique when applied to full thickness chondral lesions of the knee in a group of athletes. From 1991 to 1999, 109 patients were treated using the microfracture technique. We prospectively followed up 53 athletes who satisfied our inclusion criteria. Average age was 38 years (range 19–55) and mean follow-up was 72 months (range 36–120). Etiology, clinical signs, symptoms and activity level were noted preoperatively and at final follow-up. Lysholm, Tegner, IKDC and functional tests were utilized. Intraoperatively, location, size of the lesions and associated pathologies were recorded. Roentgenograms, MRI or CT scan were done before treatment and at final follow-up. Etiologic factors were mostly related to sports microtrauma (37.5%) and macrotrauma (21%), while 37.5% of our patients did not report any traumatic etiology and 4% showed patellar malalignment. The most common location was medial femoral condyle (61%). Knee pain and swelling improved in 70%, tibiofemoral crepitus in 60%. Hop test was normal in 70% at final follow-up. Subjective evaluation was 40/100 preoperatively and 70/100 at final follow-up. Lysholm was 56.8 preoperatively and 87.2 final. IKDC revealed: 0 A, 3 B, 40 C and 10 D preoperatively while at final follow-up 70% scored A or B. Tegner improved at 2 years from 3.2 to 6; however, at final follow-up 80% showed a decline in sport activity level (Tegner 5). Microfracture technique can offer clinical, functional and subjective improvement in athletically active patients. However because of the decline in sports participation over time, microfracture may not be the definitive procedure for the athlete’s knee and other procedures may be indicated in the future.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Articular cartilage has the capability of distributing loads across the knee joint and can tolerate a great amount of stress, thus protecting the subchondral bone. Due to its avascularity, injuries if left untreated have little or no potential to heal spontaneously with normal hyaline cartilage [6, 10, 19, 21, 23, 27, 29]. However, lesions that penetrate the subchondral bone can undergo some degree of repair resulting from fibrin clot formation, migration of stem cells, and vascular ingrowth [16, 18, 19].
Rodrigo et al. [26] reported their results using the microfracture technique for the treatment of full thickness cartilage lesions where multiple holes were made in defects. The authors believed that this technique helps to hold the marrow clot, allowing it to adhere firmly while preserving the integrity of the subchondral plate, and thereby producing a durable repair cartilage as it takes advantage of the body’s own healing capacities.
The simplicity and the low cost of the instruments utilized for this procedure prompted us to adopt this technique when we began this study in 1991. Furthermore, microfracture was attractive because of the minimal iatrogenic damage associated with its use, so that once performed it did not preclude the use of other future procedures: “Microfracture does not burn any bridges”.
In the last 10 years, several authors have adopted microfracture and reported good results [3, 10, 25, 28, 30].
The purpose of this study was prospectively to investigate the outcome of the microfracture technique when applied to full thickness chondral lesion of the knee in an athletic population.
Materials and methods
From July 1991 to June 1999, 109 consecutive patients underwent microfracture technique for full thickness osteochondral lesion of the knee. Among this group of patients, we prospectively followed up a group of 53 athletes with unilateral knee pain, clinical and imaging findings (MRI, CT or standard X-rays) suggestive of articular cartilage injury. The final diagnosis was confirmed during arthroscopy.
Patients included in our series were all athletic individuals, which we considered as those playing in competitive sports at regional or national levels or participating in active sports at least twice per week. We required consent for participation in this study, willingness to follow-up when requested, and feasibility of following a specific rehabilitation program in the same institution.
Patients with osteochondritis dissecans, partial thickness chondral defects, malalignment requiring osteotomy (>10°), total meniscectomy or significant degenerative arthritis (2–3° according to Ahlbach classification) were excluded from this series as well as those unable to use the contralateral leg during rehabilitation. The final group included 33 males and 20 females, with a mean age of 38 years (range 19–55). Among these, 26 were professional and 27 were recreational athletes. The summary sports activities of our patients are presented in Table 1. The average follow-up was 72 months (range 36–120).
All surgery was performed by the same surgeon and all patients followed the same postoperative rehabilitation program. During preoperative clinical examination of the knee, the following were recorded: pain, crepitus, swelling, and limitation of function.
Each patient was interviewed and was asked to fill up a questionnaire regarding the type and level of sports participation according to the Tegner scale, previous knee problems and/or surgery, with special attention given to determine when the problem arised and the etiology of the lesions. Preoperative IKDC and Lysholm scores were determined, as well as the Tegner activity level prior to the onset of symptoms and prior to surgery. Intraoperatively, the location and the size of the lesions as well as the associated pathologies were recorded.
At final follow-up, the patients were asked to fill up another questionnaire to assess subjective function and symptoms. They were asked to score the operated knee, considering the contralateral normal knee as 100%. Sport activity level prior to surgery, at 2 years postoperative, and at final follow-up were compared. Clinical examination was performed at final evaluation by an independent examiner to compare with the findings prior to surgery. IKDC knee form and Lysholm were utilized for final assessment. Functional test (one leg hop) was also performed comparing the operated with the normal leg with a series of three jumps.
In ten of the patients in this series, we were able to perform second look arthroscopy for the following reasons: seven subjects underwent arthroscopy because of meniscal problems or patellofemoral pain, two patients subsequently underwent autologous chondrocyte implantation due to persistent pain, and one patient had ACL reconstruction in the contralateral knee and consented to second look arthroscopy in the previously operated knee.
In all these ten subjects, intraoperative biopsies with 2 mm diameter instrument were performed for histopathologic studies. On each occasion, the aim was to take a border sample contained normal cartilage and regenerated tissue, with the exception of patients who underwent autologous chondrocyte implantation. In these cases, we took a few samples from the most representative places. The regenerated tissue samples were taken from previous post-traumatic lesions of femoral condyle (4) and tibial plateau (1), from non-traumatic lesions of trochlea (1), patella (1), tibia (2) and femur (1).
Surgical technique
The operations were performed according to the technique described by Steadman, except that we used only two portals for the arthroscope and the working instruments. We did not use a tourniquet routinely. An initial thorough diagnostic examination of the knee was done. After identifying the full-thickness chondral lesion, we removed all the unstable cartilage, including that loosely attached to the surrounding rim using a shaver and/or a hand held angled courette. When present, the calcified layer of cartilage was also taken out using a courette. Once the exposed subchondral bone plate was thoroughly debrided, we made multiple holes using a custom-angled arthroscopic awl (Fig.1). The holes were placed perpendicular to the joint surface approximately 3–4 mm apart and about 2–4 mm deep, taking care not to damage the subchondral plate between the holes (Fig. 2). Once the holes were completed, the irrigation fluid pump pressure was lowered to visualize the release of fat droplets and blood from the microfracture holes into the knee (Fig. 3). All instruments were then removed. We did not routinely use an intra-articular drain. Typically, other necessary intra-articular procedures were done first before doing microfracture, with the exception of anterior cruciate ligament reconstruction.
The postoperative rehabilitation program was carried out as suggested by Steadman. All patients were immediately placed on a continuous passive motion machine (CPM) in the recovery room and cold therapy was started. Patients with lesions in the weight bearing surfaces began CPM initially at a range of motion (ROM) of 10–70°. ROM was then increased as tolerated by 10–20° until full ROM was achieved. The CPM machine was utilized for 6–8 h daily. Isometric exercises and limited dynamic quadriceps training was started immediately after surgery.
Crutch assisted touchdown weight bearing ambulation was allowed from 4 to 6 weeks according to the size and location of the lesion. Thereafter, weight bearing loading gradually increased, as well as strength training, to achieve full weight bearing walk at 8 weeks.
Patients with lesions in the patellofemoral joint were placed in a brace set at 0–30° for 8 weeks, partial weight bearing was allowed after 7 days and isometric quadriceps exercises were introduced. The patients were allowed to remove the brace during passive range of motion exercise in a CPM and were also allowed to start hydrotherapy and bicycling at 2 weeks. After 8 weeks, the full weight bearing walk was allowed, the brace was discontinued and the strength training program was progressively advanced. From the beginning of the 9th week, active functional training was started. Symptoms of overloading (pain, effusion, tenderness) noticed by surgeon or physiotherapist were indications to slow down the rehabilitation protocol. From the 4th postoperative month, we gradually introduced specific sport training elements (running, squatting). Thereafter subjects were allowed return to sport. Associated treatment, included: 11 ACL reconstructions, three lateral releases, and three meniscal suturings performed simultaneously.
Results
There were no complications directly related to microfracture technique in this study. The average follow-up was 72 months (range 36–120). The medial femoral condyle was the most frequently involved area, representing 61% of the lesions, followed by the lateral femoral condyle. The most common etiology found was sports related microtrauma and non-traumatic causes consisting of 75% (Tables 2 and 3). The average size of the lesions was 400 mm2 (300–600 mm2). A summary of the different knee evaluation scores is presented in Table 4. There was a statistically significant (P< 0.05) improvement of knee function in our patients comparing preoperative and final follow–up, with 70% of these subjects considered normal or nearly normal at final follow-up.
Pain and swelling at activity levels 1 and 2 was present preoperatively in 47 patients. At final follow-up, 33 of these subjects showed complete pain relief, 12 were unchanged or minimally changed and two worsened.
Improvement was also noted in daily living activities in 65% of the patients with no modifications through 6 years. Preoperatively, tibiofemoral crepitus was present in 47 patients and was described as moderate in 40% (18) and severe and painful in 60% (29) of cases. At final follow-up, there was relief and improvement in 60% (29), no change in 30% (14) and worsening in 5% (4) of cases. Functional test (one leg hop) was normal or nearly normal in 70%, abnormal in 20%, and 10% were not able to jump more than 50% of the distance covered using the normal leg.
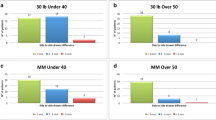
While strenuous sports activities improved in 80% of the subjects during the first 2 years, they gradually decreased to 55% at final follow-up. Tegner activity scale was 3.2 preoperatively, 6 at 2 years and 5 at final follow-up.
Separate analysis of the patients after ACL reconstruction (most common concomitant injury in our study) revealed ability to continue the same sport and activity level by seven of them (63%).
Results of imaging modalities at final follow-up including MRI, CT scan and X-ray compared with the preoperative results showed visible increase of degenerative changes on the knee joint in 30% of the subjects, with initial medial compartment arthritis in five patients.
On second look arthroscopies, we noted that the cartilage defects were covered with fibrocartilaginous hybrid tissue, which was level with the normal residual cartilage and was firm to hard when palpated with the probe (Fig. 4).
Histopathologic sections showed areas of fibromyxoid tissue with differentiation, a transition zone with some cartilage tissue and areas with initial hyaline transformation (hybrid tissue) (Fig. 5a–c).
Patients with traumatic, isolated lesions of medial femoral condyle showed better results compared with others. Poorer outcomes were observed in non-traumatic, multifocal lesions.
We did not find any statistically significant differences in clinical results between patients with or without concomitant ACL injuries or meniscus surgery, but there was a tendency for subsequent arthritis corresponding to the previous meniscus and cartilage lesion locations.
One of our patient developed superficial infection, which immediately resolved after oral antibiotic therapy. Another patient with a history of drug abuse developed deep infection and required three arthroscopic lavages and prolonged antibiotic therapy. This patient developed severe arthritis. As already mentioned two patients subsequently underwent autologous chondrocyte implantation due to persistent pain after 5 years.
Discussion
The natural history of chondral lesions may be difficult to predict, as some authors think that many lesions are non-progressive and remain asymptomatic, while others believe that even small asymptomatic lesions may increase in size and eventually become painful if left untreated [4, 8, 19]. Furthermore, some people who sustain articular surface injury do not seek treatment [21].
Several methods have been described to treat articular cartilage defects [1, 2, 4, 9, 12, 13, 14, 17, 22, 23, 26, 29, 31, 32]. The goal of treatment is to restore the integrity of joint surface, reduce effusion and inflammation, provide a pain-free full range of motion and temporize or eliminate the need for prosthetic replacement [19].
The marrow stimulating technique is one of the methods employed to treat articular cartilage injuries. This technique stimulates the vascular mediated elements such as fibrin clot containing pluripotent stem cells [1, 2, 10, 11, 16, 17, 19, 23, 24, 31].
In the early 1990s, Steadman started his trial with microfractures and in 1994, Rodrigo et al. [26] reported their results. Starting on July 1991, we decided to adopt their technique in athletes for the following reasons: minimally invasive, technically simple and low-cost procedure, preservation of the subchondral bone plate, and most important, possibility for future treatments.
Some authors [4, 10, 15, 16, 27] included among the disadvantages of microfracture the slow healing process, the long rehabilitation time and the fact that the repair tissue is fibrocartilage and not hyaline cartilage.
The quality of the repaired tissue is still a matter of discussion. According to Yoon [32], significant amounts of type II collagen formation were verified after microfracture in osteoarthritic knees. Frisbie et al. [7], in their study, have reported an increase in type II collagen mRNA expression 6 weeks after microfracture. However, Shapiro et al. [27] have shown that the repair tissue formed after microfracture is mostly fibrocartilage, and the collagen fibrils were not well integrated into the remaining cartilage, making this tissue vulnerable to shear stresses and prone to breaking down over time. Despite these concerns Rodkey and Steadman [25, 26, 29] still maintain that the tissue formed after microfracture can function similar to normal cartilage. In their basic science studies in animals, they observed that the newly formed tissue was made up of a combination of types I and II collagen, containing blocks of mature articular cartilage.
Similar to Rodkey’s studies, our histopathologic specimens in humans showed areas of fibromyxoid tissue with differentiation (hybrid tissue), a transition zone with some cartilage and areas with initiation of hyaline transformation.
Rodkey et al. [25] also noted an improvement of pain in 75% of patients with full thickness articular cartilage lesions 3 years after treatment, with a 68% improvement in activities of daily living. Steadman et al. [30] noted a mean postoperative Lysholm score of 83.1 and Tegner activity scale score of 4.5 in a group of patients with degenerative knee. Marder and Timmerman [18] demonstrated significant improvement in knee scores after microfracture of maximal 2 cm diameter lesions, regardless of the type of postoperative rehabilitation. In a study by Gill et al. [10], 86% of patients were able to go back to their previous level of sports after microfracture for traumatic cartilage defects.
Our data at 6 years showed IKDC A or B in 70% of patients with a mean satisfaction score of 70/100, as well as Lysholm and Tegner score of 87.2 and 5, respectively, and these results are consistent with the findings of similar studies [3, 10, 25, 30]. Our results show a better outcome compared with that of Harwin [12], who treated chondral lesions with debridement alone.
Another marrow stimulating technique
Drilling combined with autogenous periosteum transplantation was described by Alfredson et al. [2]. These authors reported good clinical improvement and successful return to sport after treatment of ACL tears combined with deep cartilage defects. Equivalent technique and equally satisfying results have been shown by Lorentzon et al. [17] regarding treatment of patellar cartilage full thickness lesions. The results are summarized in Table 5.
Knutsen et al. [16], in a prospective randomized multi-center study, reported no significant difference between macroscopic and histological findings comparing microfracture to ACI. The clinical improvement was significantly better in the microfracture group after the first 2 years; however, there was a tendency for ACI to result in more hyaline repair cartilage. Mosely et al. [20], on the other hand, showed that ACI patients had greater improvements in the overall condition score and reached a higher level of functioning than microfracture patients.
To improve natural repair response and mechanical and histological quality of regenerated tissue, some authors [13, 22] have used specific scaffolds for engineering of autologous chondrocytes or stimulating of stem cells. Investigators have reported promising results, but prospective randomized study are needed.
It still remains to be seen, however, if the better quality of repair will produce a significant difference in the long run. To date, there is no single technique that stands out as being better than another, as no long-term randomized studies have been published to confirm the benefit of replacing articular cartilage defects with hyaline rather than fibrocartilage [5, 11, 23].
The present study has the limitation of not having a randomized control design, making it very difficult to show the merits of this technique as opposed to other methods. However, our study has a long-term follow-up in a specific group of patients, while most other studies were done in mixed populations [3, 18, 30, 32].
At final follow-up, all of our patients claimed improvement in their subjective evaluation, although we noted a decline in sports participation over time. Initially we postulated that a possible reason could be the associated lesions. However, among the 11 patients operated on simultaneously for ACL reconstruction, seven (63%) were still playing the same sport at final follow-up. Furthermore, similar to Knutsen et al. [16], we did not find any statistically significant difference in clinical results between patients with or without concomitant ACL lesions. We should also take into consideration the natural decline in sport activity with age, especially when follow-up exceed 5 years. Finally, it is possible that the fibrocartilaginous repair tissue, unlike true hyaline cartilage, has diminished resilience, stiffness and wear resistance that could eventually deteriorate over time, creating pain during sport activities.
In conclusion, based on our results, microfracture technique coupled with a strict rehabilitation program can offer functional and subjective improvement in athletically active patients with full thickness chondral defect of the knee. Microfracture may be performed as a simple, minimally invasive method to promote cartilage healing with the hope of relieving symptoms. Despite the other factors not directly related to cartilage condition which naturally reduce sports activity, such as age, business and family duties, since there was a decline in level of sports participation with time, we consider that microfracture may not be the definitive treatment for the athlete’s knee and other procedures may be indicated in the future.
References
Alfredson H, Lorentzon R (1999) Superior results with continuous passive motion compared to active motion after periostal transplantation. Knee Surg Sports Traumatol Arthrosc 7:232–238
Alfredson H, Thorsen K, Lorentzon R (1999) Treatment of tear of the anterior cruciate ligament combined with localised deep cartilage defects in the knee with ligament reconstruction and autologous periosteum transplantation. Knee Surg Sports Traumatol Arthrosc 7:69–74
Blevins FT, Steadman J, Rodrigo JJ et al (1998) Treatment of articular cartilage defects in athletes: an analysis of functional outcome and lesion appearance. Orthopaedics 21:767–768
Browne JE, Branch TP (2000) Surgical alternatives for articular cartilage lesions. J Am Acad Orthop Surg 8:180–189
Cain EL, Clancy WG (2001) Treatment algorithm for osteochondral injuries of the knee. Clin Sports Med 20:321–342
Curl WW, Krome J, Gordon S et al (1997) Cartilage injuries: a review of 31,516 knee arthroscopies. Arthroscopy 13:456–460
Frisbie D, Oxford J, Southwood L, Trotter G et al (2003) Early events in cartilage repair after subchondral bone microfracture. Clin Orthop 407:215–227
Ghadially FN, Thomas I, Oryschak AF et al (1997) Long-term results of superficial defects in articular cartilage: a scanning electron microscope study. J Pathol 121:213–217
Gilbert J (1998) Current treatment options for the restoration of the articular cartilage. Am J Knee Surg 11:42–46
Gill TJ, Steadman JR, Briggs KK et al (1999) The treatment of full-thickness chondral defects in the knee: indications, techniques and long term results of microfracture. Presented at annual meeting of AAOS, Anaheim, Calif
Grana WA (2000) Healing of articular cartilage: a review. Am J Knee Surg 13:29–32
Harwin SF (1999) Arthroscopic debridement for osteoarthritis of the knee: predictor of patient satisfaction. Arthroscopy 15:142–146
Hoemann C, Hurtig M, Ranger P et al (2003) Full-thickness cartilage repair using an injectable chitosan scaffold, BST-Cargel. Presented at 7th International Orthopaedics, Biomechanics, Sports Rehabilitation Conference, Assisi
Hunziker EB (2002) Articular cartilage repair: basic science and clinical progress. A review of the current status and prospects. Osteoarthritis Cartilage 10:432–463
Jun-kyo S, Scherping S, Takashi M et al (1995) Basic science of articular cartilage injury and repair. Oper Tech Sports Med 3:78–86
Knutsen G, Engebretsen L, Ludvigsen T et al (2003) A randomized trial of chondrocyte implantation versus microfracture in the knee. Presented at 7th International Orthopaedics, Biomechanics, Sports Rehabilitation Conference, Assisi
Lorentzon R, Alfredson H, Hildingsson C (1998) Treatment of deep cartilage defects of patella with periostal transplantation. Knee Surg Sports Traumatol Arthrosc 6:202–208
Marder A, Timmerman LA (2003) Athroscopic microfracture of chondral defects of the knee: a comparison of two postoperative treatments. Presented at the Annual meeting Arthroscopy Association of North America, Phoenix, Ariz
Minas T, Nehrer S (1997) Review: current concepts in the treatment of articular cartilage defects. Orthopedics 20:525–538
Mosely JB, Anderson A, Browne JE (2003) A controlled study of autologous chondrocyte implantation versus microfracture for articular cartilage lesions of the femur. Arthoscopy Association of North America meeting, Phoenix, Ariz
Newman A (1998) Articular cartilage repair. Am J Sports Med 26:309–324
Ochi M (2003) Transplantation of tissue-engineered cartilage for repair of cartilage defects. Presented at 7th International Orthopaedics, Biomechanics, Sports Rehabilitation Conference, Assisi
O’Driscoll S (1998) Current concepts review: the healing and regeneration of articular cartilage. J Bone Joint Surg Am 80:1795–1812
Rae PJ, Noble J (1998) Arthroscopic drilling of osteochondral lesions of the knee. J Bone Joint Surg Br 71:534
Rodkey WG, Steadman JR, Rodrigo JJ et al (1999) The microfracture technique to treat full thickness articular cartilage defects: biological basis and long term clinical results. In: AANA Annual Meeting Instructional Course lecture #105 “Arthroscopic treatment of chondral injuries: Options and techniques”
Rodrigo JJ, Steadman JR, Silliman JF, Fulstone HA (1994) Improvement of full thickness chondral defect healing in the human knee after debridement and microfracture using continuous passive motion. Am J Knee Surg 7:109–116
Shapiro F, Koide S, Glimcher MJ (1993) Cell origin and differentiation in the repair of full thickness defects of articular cartilage. J Bone Joint Surg Am 75:532–553
Sledge SL (2001) Microfracture techniques in the treatment of osteochondral injuries. Clin Sports Med 20:365–377
Steadman JR, Rodkey W, Rodrigo J (2001) Microfracture: surgical technique and rehabilitation to treat chondral defects. Clin Orthop 391S:362–369
Steadman JR, Miller BS, Briggs KK, Rodkey WG (2002) Patient satisfaction and functional outcome after microfracture of the degenerative knee. Presented at the 4th International Cartilage Repair Society Symposium, Toronto
Xiaochun Wei (2002) Cartilage defects treated with periostal transplantation and drilling of subchondral bone. Presented at the 4th International Cartilage Repair Society Symposium, Toronto
Yoon KH, Kim HS, Lim CT, Bae DK (2003) Cartilage regeneration after microfracture in osteoarthritic knee. In: American Academy of Orthopedic Surgeons Meeting, New Orleans
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gobbi, A., Nunag, P. & Malinowski, K. Treatment of full thickness chondral lesions of the knee with microfracture in a group of athletes. Knee Surg Sports Traumatol Arthrosc 13, 213–221 (2005). https://doi.org/10.1007/s00167-004-0499-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-004-0499-3