Abstract
Purpose
The association between conflicts of interest (COI) and study results or article conclusions in goal-directed hemodynamic therapy (GDHT) research is unknown.
Methods
Randomized controlled trials comparing GDHT with usual care were identified. COI were classified as industry sponsorship, author conflict, device loaner, none, or not reported. The association between COI and study results (complications and mortality) was assessed using both stratified meta-analysis and mixed effects meta-regression. The association between COI and an article’s conclusion (graded as GDHT-favorable, neutral, or unfavorable) was investigated using logistic regression.
Results
Of the 82 eligible articles, 43 (53%) had self-reported COI, and 50 (61%) favored GDHT. GDHT significantly reduced complications on the basis of the meta-analysis of studies with any type of COI, studies declaring no COI, industry-sponsored studies, and studies with author conflict but not on studies with a device loaner. However, no significant relationship between COI and the relative risk (GDHT vs. usual care) of developing complications was found on the basis of meta-regression (p = 0.25). No significant effect of GDHT was found on mortality. COI had a significant overall effect (p = 0.016) on the odds of having a GDHT-favorable vs. neutral conclusion based on 81 studies. Eighty-four percent of the industry-sponsored studies had a GDHT-favorable conclusion, while only 27% of the studies with a device loaner had the same conclusion grade.
Conclusions
The available evidence does not suggest a close relationship between COI and study results in GDHT research. However, a potential association may exist between COI and an article’s conclusion in GDHT research.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Goal-directed hemodynamic therapy (GDHT) is the management of global and/or regional blood flow guided by predetermined hemodynamic parameters with specified goals for intervention. Since pioneering work in 1988 [1] and 1995 [2], GDHT has revolutionized hemodynamic care in patients receiving anesthesia and surgery or admitted to the intensive care unit.
The efficacy of GDHT among different randomized controlled trials (RCTs) has been inconsistent. For example, in patients with sepsis, one GDHT protocol comprised of maintaining the central venous pressure between 8 and 12 mmHg, the mean arterial pressure between 65 and 90 mmHg, and the central venous blood oxygen saturation ≥ 70% significantly reduced mortality from 46.5% to 30.5% (n = 263) [3]; however, one decade later, three different RCTs failed to replicate this result using almost the same GDHT protocol in the same patient population with a much larger sample size (n = 1260–1600) [4,5,6]. Similar discrepancies are widespread in GDHT research [7].
The cause of these inconsistencies across GDHT research is unclear but may be related to factors such as the heterogeneity of the patient population [8, 9], the parallel use of the Enhanced Recovery After Surgery (ERAS) protocol [10, 11], the different GDHT protocols used by different studies [12,13,14], and the timing and type of antibiotics administered to septic patients [15]. However, other causes likely exist that contribute to the inconsistency of the results.
Modern GDHT is characterized by the use of innovative technologies that are noninvasive or minimally invasive and emphasize the monitoring of intravascular volume and cardiac output. These monitors are manufactured by competing companies and incur definite costs. For reasons including but not limited to the competition among different manufacturers and the costs incurred by these novel devices, different types of conflicts of interest (COI) are widespread in GDHT research.
The recent editorials published in Science [16] and Intensive Care Medicine [17] highlight the concern over the potential confounding effect of COI on biomedical research. A robust body of literature demonstrates that industry-sponsored studies tend to have proindustry results and/or conclusions [18,19,20,21]. However, these previous investigations primarily focused on research related to drugs, smoking, alcoholism, and nutrition [18,19,20], while the association between COI and research related to medical devices has not been adequately studied. Given the rapid implementation of contemporary hemodynamic monitors in acute care, an urgent need exists to understand the influence of COI on GDHT research.
We hypothesize that GDHT research is confounded by COI. RCTs that had specifically compared GDHT with usual care in adult patients under acute care were identified and analyzed to understand the association between COI and GDHT research. We herein differentiate between study results and article conclusions because results are based on objective data, while conclusions can be influenced by personal opinions.
Methods
Literature search
A systematic literature search of published RCTs comparing GDHT with usual care was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [22]. A medical librarian (A.B.) performed the systematic search of multiple databases after consultation with lead authors and a medical subject heading (MeSH) analysis of key articles provided by the research team. The formal search used relevant controlled vocabulary terms and synonymous free-text words and phrases to capture the concepts of RCT and GDHT. The electronic databases OVID Medline, OVID Embase, PubMed, and Cochrane Trials were searched on October 12, 2017 and July 6, 2018. The strategy used for the last search is presented in Supplemental file 1. Additional studies were identified nonsystematically by screening the reference lists of relevant articles and searching Google Scholar and PubMed.
Study selection
Two investigators (L.Z. and L.M.) independently screened identified references and then performed full-article reviews; conflicts were resolved by consulting a third investigator (Y.A.). The inclusion criteria were as follows: (1) adult patients (≥ 18 years old); (2) comparison between GDHT and usual care; (3) complications, mortality, or length of hospital stay reported as outcome; (4) perioperative or critical care setting; and (5) randomized controlled trial. A study was excluded if it (1) was not a randomized study, (2) was not published in a full-text article, (3) compared two different forms of GDHT instead, and (4) did not report the outcome of interest. GDHT was defined as the management of global and/or regional blood flow or oxygen delivery guided by predetermined hemodynamic parameters with specified goals for intervention. Usual care was defined as the hemodynamic management that is widely accepted as the standard of care but without guidance based on advanced volume or flow monitoring.
Definition of outcomes
Complication was defined as any deviation from the normal postoperative course or organ dysfunction [23,24,25]. Organ-specific complications include myocardial infarction, congestive heart failure, cardiac arrest, atrial fibrillation or other types of arrhythmia, pulmonary embolus, pneumonia treated with antibiotics, respiratory failure requiring intubation, respiratory insufficiency requiring physiotherapy or oxygen therapy, stroke, transient ischemic attack, postoperative delirium or cognitive decline, renal insufficiency requiring dialysis, acute kidney injury, urinary tract infection requiring antibiotics, hepatic insufficiency, gut hypoperfusion, ileus, disseminated intravascular coagulation, and sepsis. Surgery-related complications refer to surgical site bleeding, infection, anastomotic leakage, stenosis, ischemia, or tissue necrosis. In-study mortality was defined as the mortality reported by the study, referring to the rate of death in the hospital or at any time point specified by the study. The longest follow-up was used in the meta-analysis when different mortalities at different time points were reported. The length of hospital stay was defined as the total days from admission until the actual day of discharge or the day the patient was deemed fit for discharge.
Classification of conflicts of interest
All eligible articles were independently investigated by two investigators (L.Z. and L.M.) to determine the presence and type of self-reported COI in each article, with special attention focused on disclosure, acknowledgments, and the author’s work place. The COI was classified as industry sponsorship, author conflict, device loaner, none, or not reported (Table 1). If an article had different types of COI, the following priority order was used for classification: industry sponsorship > author conflict > device loaner.
Grading of an article’s conclusion
The conclusion of each eligible article was graded as GDHT-favorable, neutral, or unfavorable by two investigators (L.Z. and L.M.) independently. An article was determined to have a favorable conclusion if it favored GDHT over usual care, an unfavorable conclusion if it favored usual care over GDHT, and a neutral conclusion otherwise.
Data extraction
The following data were extracted from each eligible article: (1) setting of acute care, (2) number of patients, (3) classification of COI, (4) protocols involving hemodynamic parameters and specific goals, (5) complications, (6) mortality, (7) length of hospital stay, (8) article conclusion grade, (9) study origin, and (10) monitoring device used.
Quality assessment of selected studies
The risk of bias of each study was assessed by the tool established by the Cochrane Collaboration [26]. The following domains were assessed: (1) random sequence generation (selection bias), (2) allocation concealment (selection bias), (3) blinding of participants and personnel (performance bias), (4) blinding of outcome assessment (detection bias), (5) incomplete data outcome (attrition bias), (6) selective reporting (reporting bias), and (7) other bias. A study was rated as having a high risk of bias overall if more than one domain was rated as having a high risk of bias. Publication bias was assessed by visual inspection of the funnel plot, with a symmetrical plot indicating the absence of bias, and an asymmetrical plot indicating the presence of bias.
Synthesis of evidence
The effects of GDHT on complications and mortality were assessed by meta-analysis using RevMan 5.3 software (The Nordic Cochrane Centre, The Cochrane Collaboration, 2014). The I2 statistic [27] was calculated to measure the extent of heterogeneity, and the Cochrane Q test statistic was used to assess the statistical significance. A random effects model was used if significant heterogeneity was identified, and a fixed effects model was used otherwise. To quantify the magnitude of the effect size for dichotomous outcomes of complications and mortality, the risk ratio (RR) with a 95% confidence interval (CI) was calculated. A two-sided p value less than 0.05 was considered statistically significant. Forest plots were constructed to help visualize both the result of a single study and the pooled result.
The association between COI and study results, or more specifically whether the therapeutic effect of GDHT varies with different classes of COI, was investigated by both stratified meta-analysis and mixed effects meta-regression analysis. The metafor package [28] implemented in R software was used to perform meta-regression analysis, in which we investigated whether the heterogeneity of complications or mortality among eligible studies (log RR as the dependent outcome variable) is explainable by the COI classification or other study-level factors, including the study publication year, study setting, patient number, device used for hemodynamic monitoring, GDHT protocol, and origin of the study.
The association between COI and an article’s conclusion was investigated by logistic regression, in which COI classifications and other study-level factors were treated as independent variables, and the article’s conclusion was treated as the dependent outcome variable. The results are expressed in odds ratios (OR) and 95% CIs to indicate the effect of COI on the conclusion (favorable vs. neutral).
Results
Results of literature search
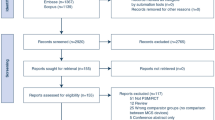
The systematic search yielded 2174 references, and the nonsystematic search identified 15 additional records. Following de-duplication and screening of the title/abstract, 139 studies were retained and underwent a subsequent full-text review. On the basis of the selection criteria, 82 articles were retained for the final synthesis of evidence [2,3,4,5,6, 24, 29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104]. The selection process is detailed in Fig. 1.
Characteristics of eligible articles
The setting, number of patients, monitor used, intervention protocol, and main conclusion of each study are presented in Table 2. These 82 eligible articles were published between 1993 and 2018, with 71 studies being conducted in the perioperative setting, and 11 studies being conducted in the critical care setting. Most studies originated from Europe (n = 46, 56%). The risks of bias, expressed as the percentages of low, unclear, and high risks of the different domains of all studies included in the meta-analysis, are presented in Fig. 2, while the risks of bias of different domains of each study based on review authors’ judgments are detailed in Supplemental file 2. We detected no obvious evidence of publication bias in the studies included in the meta-analysis based on visual assessment of the funnel plots (Supplemental files 3 and 4).
Prevalence of COI
Of the 82 eligible articles, 43 (53%) reported COI (industry sponsorship = 19; author conflict = 13; device loaner = 11), 33 (40%) declared no COI, and 6 (7%) did not include a COI statement (Supplemental file 5). None of the coauthors among these 82 articles were employed by a related industry.
Effects of GDHT on complications per COI classification
Fifty-one studies reported the number of patients with complications, with 3555 patients being managed by GDHT, and 3592 patients being managed by usual care (Supplemental file 5). Compared with usual care, GDHT significantly reduced the risk of developing complications based on these 51 studies (RR = 0.81, 95% CI 0.74–0.88; p = 0.0001; Fig. 3a), based on 31 studies with any type of COI (RR = 0.85, 95% CI 0.77–0.93; p = 0.006; Fig. 3b), based on 17 studies declaring no COI (RR = 0.76, 95% CI 0.64–0.90; p = 0.002; Fig. 3c), based on 12 industry-sponsored studies (RR = 0.83, 95% CI 0.75–0.92; p = 0.006; Fig. 3d), and based on 11 studies with author conflict (RR = 0.77, 95% CI 0.64–0.93; p = 0.007; Fig. 3e) but not based on eight studies with a device loaner (RR = 1.01, 95% CI 0.86–1.20; p = 0.90; Fig. 3f). These effects are comparable as indicated by the overlapping 95% CI ranges.
Forest plots showing the risk ratios of having a patient with complications in all eligible studies (a), studies with any type of conflict of interest (b), studies declaring no conflicts of interest (c), industry-sponsored studies (d), studies with author conflict (e), and studies with a device loaner (f). The size of the diamond reflects the weight of the trial in the pooled analysis. The horizontal bars represent the 95% confidence interval (CI)
Effects of GDHT on mortality per COI classification
Forty-six studies reported in-study mortality, with 5942 patients being managed by GDHT, and 6003 patients being managed by usual care (Supplemental file 5). No statistically significant heterogeneity was identified among the various studies. Compared with usual care, GDHT led to a statistically significant change in mortality based on these 46 studies (RR = 0.91, 95% CI 0.85–0.99; p = 0.02; Fig. 4a) but not based on 25 studies with any type of COI (RR = 0.93, 95% CI 0.85–1.03; p = 0.15; Fig. 4b), 17 studies declaring no COI (RR = 0.89, 95% CI 0.78–1.01; p = 0.07; Fig. 4c), eight industry-sponsored studies (RR = 0.92, 95% CI 0.75–1.12; p = 0.39; Fig. 4d), seven studies with author conflict (RR = 0.77, 95% CI 0.47–1.26; p = 0.30; Fig. 4e), or 10 studies with a device loaner (RR = 0.94, 95% CI 0.85–1.03; p = 0.20; Fig. 4f). These effects are comparable as indicated by the overlapping 95% CI ranges.
Forest plots showing the risk ratios of mortality in all eligible studies (a), studies with any type of conflict of interest (b), studies declaring no conflicts of interest (c), industry-sponsored studies (d), studies with author conflict (e), and studies with a device loaner (f). The size of the diamond reflects the weight of the trial in the pooled analysis. The horizontal bars represent the 95% confidence interval (CI)
Results of meta-regression analysis
The raw data used for meta-regression analysis are presented in Supplemental file 6. As there was no significant heterogeneity in the effect of GDHT on mortality among the 46 eligible studies (I2 = 0%, Fig. 4a), meta-regression was performed to analyze only the effects of GDHT on complications based on the pooled evidence from 51 eligible studies (I2 = 53%, Fig. 3a). The results (Supplemental file 7) did not identify a significant effect of the COI classification (p = 0.25), study setting (p = 0.55), patient number (p = 0.40), device used (p = 0.94), GDHT protocol (p = 0.99), or study origin (p = 0.20) on the observed study heterogeneity. The only factor that had a significant correlation with the RR of GDHT vs. usual care was the year of study publication (p = 0.0012, Fig. 5). As indicated by the significant p values from the tests of residual heterogeneity, it is highly likely that other study-level factors exist that were not considered in our meta-regression but influence the effect of GDHT on complications.
Association between COI and article conclusions
Among the 82 eligible articles, 50 (61%) had a GDHT-favorable conclusion, 31 (38%) had a GDHT-neutral conclusion, and 1 (1%) had a GDHT-unfavorable conclusion (Supplemental file 5). The percentages of articles with specific conclusion grades per COI classification are presented in Fig. 6. Industry-sponsored studies had the highest percentage (84%) of GDHT-favorable conclusions, followed by studies with author conflict (77%), studies declaring no COI (55%), studies including no COI disclosure (50%), and studies with a device loaner (27%). Logistic regression analysis showed that only COI (p = 0.016) and the GDHT protocol (p = 0.022) were significantly associated with an article’s conclusion (GDHT-favorable vs. neutral) in GDHT research (Supplemental file 8).
Discussion
Our study demonstrated that (1) more than half (53%) of RCTs comparing GDHT with usual care have COI; (2) GDHT reduces complications in studies with any type of COI, studies declaring no COI, industry-sponsored studies, and studies with author conflict but not in studies with a device loaner; (3) the heterogeneity of complications among eligible studies cannot be explained by COI, i.e., the therapeutic effect of GDHT on complications does not appear to vary with different classes of COI; (4) GDHT has no effect on mortality on the basis of the meta-analysis of studies with different COI; (5) COI might have a significant overall effect on the odds of having a GDHT-favorable vs. neutral conclusion; and (6) the majority of industry-sponsored studies have a GDHT-favorable conclusion (84%), while the majority of studies with a device loaner have a GDHT-neutral conclusion (73%). Taken together, the available evidence does not suggest a close relationship between COI and study results; however, it does suggest a potential association between COI and an article’s conclusion in GDHT research.
The influence of industry relationships on the outcomes of original research has been scrutinized in various fields of biomedical research [18, 19]. Although the findings diverge, these efforts do highlight concern regarding the potential confounding effect of industry relationships on biomedical research. This concern is corroborated by one recent cumulative meta-analysis concluding that compared with nonindustry-related studies, industry-related studies are more likely to have both favorable efficacy results, based on 25 papers that included 2923 studies (RR = 1.27, 95% CI 1.17–1.37), and favorable conclusions, based on 29 papers that included 4583 studies (RR = 1.34, 95% CI 1.19–1.51) [18]. Aggregation of the results of eight systematic reviews also concluded that the odds of industry-sponsored studies having a proindustry conclusion is 3.60 (95% CI 2.63–4.91) [19].
However, the majority of these previous investigations were based on drug studies, while only a few reports were based on device studies [105,106,107]. The influence of industry relationships on GDHT research, a field dependent on advanced hemodynamic monitoring devices, has not been reported. One difference between drug and device studies is that devices and reusable supplies can be loaned, which generates a COI different from those of industry sponsorship and author conflict. On the basis of this consideration, we classified COI into industry sponsorship, author conflict, and device loaner in our investigation, an approach that differs from that used in previous investigations in which studies were dichotomized into only the industry-sponsored and unsponsored categories. This differentiation is important because our findings suggest that different types of COI may have different associations with the results and conclusions of GDHT research.
Study results and article conclusions are different. Conclusions can be influenced by personal opinions and may or may not be supported by results [21], and differentiating results and conclusions is prudent when investigating the influence of COI on biomedical research. Methods for analysis also differ. In our study, the association between COI and the study results of GDHT research (i.e., complications and mortality) was assessed by stratified meta-analysis and meta-regression, whereas the association between COI and a GDHT research article’s conclusion was assessed by logistic regression, which is in accordance with the fact that study results are quantitative, while an article’s conclusions are qualitative.
Our investigation revealed that COI are widespread in GDHT research. Although GDHT is a landmark event in intensive hemodynamic care, the inconsistent results and conclusions of GDHT research as well as the associated costs hinder its wide clinical adoption [7]. In addition to industry influence, resource constraints, and the pressure of academic productivity, the urgent need for more evidence may be responsible for the high prevalence of COI in GDHT research. Our investigation found a self-reported incidence of 53%; however, the true incidence might be higher because of underreporting [108]. In our investigation, 55% of the studies declaring no COI had a GDHT-favorable conclusion, which was lower than that of industry-sponsored studies (84%) and studies with author conflict (77%) but higher than that of studies with a device loaner (27%). Although the cause of this discrepancy remains to be elucidated, the possibility of unreliable COI disclosure in GDHT research is suggested.
We found that GDHT can reduce complications but has only a marginal effect on mortality based on the overall evidence. The exact cause of this discrepancy is unknown but may be partially attributable to the difference between outcome measures (i.e., an outcome-dependent effect). The reporting of a complication not only depends on its definition but also on the accuracy and completeness of the information needed for the diagnosis. The diagnosis of a complication made by one investigator may not be made by a different investigator. This potential discrepancy does not exist when using death or survival as the end point, suggesting that the use of objective measures, such as mortality, may result in fewer inconsistencies.
Our meta-analysis did not include studies that reported complications as total events per patient or group instead of the number or percentage of patients in whom complications occurred. Moreover, we were not able to perform a meta-analysis of the length of hospital stay because of the diverse reporting methods (e.g., median vs. mean, whole range vs. interquartile range vs. 95% CI). The differing criteria for the length of hospital stay, variably defined as the time from admission to the actual day of discharge vs. the day the patient was deemed fit for discharge, added another source of heterogeneity.
There are a number of limitations in this study. It should first be noted that the cause–effect relationship between COI and the results or conclusions of GDHT research cannot be determined by this investigation. With the use of meta-analysis, we are able to calculate the pooled estimate of the therapeutic effect with improved precision compared to that of an individual study; however, we cannot guarantee that our estimates have improved accuracy (i.e., less bias) because the number of eligible studies was limited, and we had no access to the raw data from these studies. Our investigation could not determine the influence of nonfinancial COI, such as strongly held beliefs, personal relationships, and desire for career advancement, on GDHT research [109]. This factor in addition to the limited number of quality studies and the potentially missing or inaccurate disclosure of COI may confound the estimation of the association between COI and GDHT research.
It should be noted that multiple tests were performed in our investigation, and we recognize that the family-wise error rate in our study was not necessarily controlled at the 0.05 level, as we did not adjust for raw p values from multiple meta-analyses, meta-regression, or logistic regression. As a result, the statistical significance should be interpreted with caution. Nevertheless, measures such as RRs and 95% CIs should be relied upon to interpret the magnitude of any effects identified in the current study.
In summary, more than half of the RCTs comparing GDHT with usual care are related to industry in the forms of industry sponsorship, author conflict, or device loaner. The available evidence does not suggest a close relationship between COI and study results; however, it does suggest a potential association between COI and an article’s conclusion in GDHT research.
References
Shoemaker WC, Appel PL, Kram HB, Waxman K, Lee TS (1988) Prospective trial of supranormal values of survivors as therapeutic goals in high-risk surgical patients. Chest 94:1176–1186
Mythen MG, Webb AR (1995) Perioperative plasma volume expansion reduces the incidence of gut mucosal hypoperfusion during cardiac surgery. Arch Surg 130:423–429
Rivers E, Nguyen B, Havstad S, Ressler J, Muzzin A, Knoblich B, Peterson E, Tomlanovich M (2001) Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med 345:1368–1377
ARISE Investigators, ANZICS Clinical Trials Group, Peake SL, Delaney A, Bailey M, Bellomo R, Cameron PA, Cooper DJ, Higgins AM, Holdgate A, Howe BD, Webb SA, Williams P (2014) Goal-directed resuscitation for patients with early septic shock. N Engl J Med 371:1496–1506
Mouncey PR, Osborn TM, Power GS, Harrison DA, Sadique MZ, Grieve RD, Jahan R, Harvey SE, Bell D, Bion JF, Coats TJ, Singer M, Young JD, Rowan KM, Pro MTI (2015) Trial of early, goal-directed resuscitation for septic shock. N Engl J Med 372:1301–1311
Pro CI, Yealy DM, Kellum JA, Huang DT, Barnato AE, Weissfeld LA, Pike F, Terndrup T, Wang HE, Hou PC, LoVecchio F, Filbin MR, Shapiro NI, Angus DC (2014) A randomized trial of protocol-based care for early septic shock. N Engl J Med 370:1683–1693
Meng L, Heerdt PM (2016) Perioperative goal-directed haemodynamic therapy based on flow parameters: a concept in evolution. Br J Anaesth 117:iii3–iii17
Hamilton MA, Cecconi M, Rhodes A (2011) A systematic review and meta-analysis on the use of preemptive hemodynamic intervention to improve postoperative outcomes in moderate and high-risk surgical patients. Anesth Analg 112:1392–1402
Cecconi M, Corredor C, Arulkumaran N, Abuella G, Ball J, Grounds RM, Hamilton M, Rhodes A (2013) Clinical review: goal-directed therapy-what is the evidence in surgical patients? The effect on different risk groups. Crit Care 17:209
Jones C, Kelliher L, Dickinson M, Riga A, Worthington T, Scott MJ, Vandrevala T, Fry CH, Karanjia N, Quiney N (2013) Randomized clinical trial on enhanced recovery versus standard care following open liver resection. Br J Surg 100:1015–1024
Una Orejon R, Huercio Martinez I, Mateo Torres E, Jofre Escudero C, Gomez Rivas J, Diez Sebastian J, Ureta Tolsada MP (2017) Impact of a goal directed therapy in the implementation of an ERAS (enhanced recovery after surgery) protocol in laparoscopic radical cystectomy. Arch Esp Urol 70:707–714
van der Zee E, Egal M, Gommers D, Groeneveld A (2017) Targeting urine output and 30-day mortality in goal-directed therapy: a systematic review with meta-analysis and meta-regression. BMC Anesthesiol 17:22
Parker MM (2007) Goals for fluid resuscitation: a real challenge. Crit Care Med 35:295–296
Vincent JL, Weil MH (2006) Fluid challenge revisited. Crit Care Med 34:1333–1337
Kalil AC, Johnson DW, Lisco SJ, Sun J (2017) Early goal-directed therapy for sepsis: a novel solution for discordant survival outcomes in clinical trials. Crit Care Med 45:607–614
Piller C (2018) Hidden conflicts? Science 361:16–20
Tobin MJ (2018) Conflicts of interest and the patient-doctor covenant. Intensive Care Med. https://doi.org/10.1007/s00134-018-5282-x
Lundh A, Lexchin J, Mintzes B, Schroll JB, Bero L (2017) Industry sponsorship and research outcome. Cochrane Database Syst Rev 2:MR000033
Bekelman JE, Li Y, Gross CP (2003) Scope and impact of financial conflicts of interest in biomedical research: a systematic review. JAMA 289:454–465
Chartres N, Fabbri A, Bero LA (2016) Association of industry sponsorship with outcomes of nutrition studies: a systematic review and meta-analysis. JAMA Intern Med 176:1769–1777
Als-Nielsen B, Chen W, Gluud C, Kjaergard LL (2003) Association of funding and conclusions in randomized drug trials: a reflection of treatment effect or adverse events? JAMA 290:921–928
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 339:b2535
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
Boyd O, Grounds RM, Bennett ED (1993) A randomized clinical trial of the effect of deliberate perioperative increase of oxygen delivery on mortality in high-risk surgical patients. JAMA 270:2699–2707
Chong MA, Wang Y, Berbenetz NM, McConachie I (2018) Does goal-directed haemodynamic and fluid therapy improve peri-operative outcomes? A systematic review and meta-analysis. Eur J Anaesthesiol 35:469–483
Jorgensen L, Paludan-Muller AS, Laursen DR, Savovic J, Boutron I, Sterne JA, Higgins JP, Hrobjartsson A (2016) Evaluation of the Cochrane tool for assessing risk of bias in randomized clinical trials: overview of published comments and analysis of user practice in Cochrane and non-Cochrane reviews. Syst Rev 5:80
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560
Viechtbauer W (2010) Conducting meta-analyses in R with the metafor package. J Stat Softw 36:48
Bender JS, Smith-Meek MA, Jones CE (1997) Routine pulmonary artery catheterization does not reduce morbidity and mortality of elective vascular surgery: results of a prospective, randomized trial. Ann Surg 226:229–236 (discussion 236–227)
Sinclair S, James S, Singer M (1997) Intraoperative intravascular volume optimisation and length of hospital stay after repair of proximal femoral fracture: randomised controlled trial. BMJ 315:909–912
Bonazzi M, Gentile F, Biasi GM, Migliavacca S, Esposti D, Cipolla M, Marsicano M, Prampolini F, Ornaghi M, Sternjakob S, Tshomba Y (2002) Impact of perioperative haemodynamic monitoring on cardiac morbidity after major vascular surgery in low risk patients. A randomised pilot trial. Eur J Vasc Endovasc Surg 23:445–451
Gan TJ, Soppitt A, Maroof M, el-Moalem H, Robertson KM, Moretti E, Dwane P, Glass PS (2002) Goal-directed intraoperative fluid administration reduces length of hospital stay after major surgery. Anesthesiology 97:820–826
Conway DH, Mayall R, Abdul-Latif MS, Gilligan S, Tackaberry C (2002) Randomised controlled trial investigating the influence of intravenous fluid titration using oesophageal Doppler monitoring during bowel surgery. Anaesthesia 57:845–849
Venn R, Steele A, Richardson P, Poloniecki J, Grounds M, Newman P (2002) Randomized controlled trial to investigate influence of the fluid challenge on duration of hospital stay and perioperative morbidity in patients with hip fractures. Br J Anaesth 88:65–71
Sandham JD, Hull RD, Brant RF, Knox L, Pineo GF, Doig CJ, Laporta DP, Viner S, Passerini L, Devitt H, Kirby A, Jacka M (2003) A randomized, controlled trial of the use of pulmonary-artery catheters in high-risk surgical patients. N Engl J Med 348:5–14
Wakeling HG, McFall MR, Jenkins CS, Woods WG, Miles WF, Barclay GR, Fleming SC (2005) Intraoperative oesophageal Doppler guided fluid management shortens postoperative hospital stay after major bowel surgery. Br J Anaesth 95:634–642
Pearse R, Dawson D, Fawcett J, Rhodes A, Grounds RM, Bennett ED (2005) Early goal-directed therapy after major surgery reduces complications and duration of hospital stay. A randomised, controlled trial [ISRCTN38797445]. Crit Care 9:R687–R693
Noblett SE, Snowden CP, Shenton BK, Horgan AF (2006) Randomized clinical trial assessing the effect of Doppler-optimized fluid management on outcome after elective colorectal resection. Br J Surg 93:1069–1076
Lopes MR, Oliveira MA, Pereira V, Lemos I, Auler J, Michard F (2007) Goal-directed fluid management based on pulse pressure variation monitoring during high-risk surgery: a pilot randomized controlled trial. Crit Care 11(5):R100
Donati A, Loggi S, Preiser JC, Orsetti G, Munch C, Gabbanelli V, Pelaia P, Pietropaoli P (2007) Goal-directed intraoperative therapy reduces morbidity and length of hospital stay in high-risk surgical patients. Chest 132:1817–1824
Harten J, Crozier JE, McCreath B, Hay A, McMillan DC, McArdle CS, Kinsella J (2008) Effect of intraoperative fluid optimisation on renal function in patients undergoing emergency abdominal surgery: a randomised controlled pilot study (ISRCTN 11799696). Int J Surg 6:197–204
Buettner M, Schummer W, Huettemann E, Schenke S, van Hout N, Sakka SG (2008) Influence of systolic-pressure-variation-guided intraoperative fluid management on organ function and oxygen transport. Br J Anaesth 101:194–199
Kapoor P, Kakani M, Chowdhury U, Choudhury M, Lakshmy R, Kiran U (2008) Early goal-directed therapy in moderate to high-risk cardiac surgery patients. Ann Card Anaesth 11:27
Smetkin AA, Kirov MY, Kuzkov VV, Lenkin AI, Eremeev AV, Slastilin VY, Borodin VV, Bjertnaes LJ (2009) Single transpulmonary thermodilution and continuous monitoring of central venous oxygen saturation during off-pump coronary surgery. Acta Anaesthesiol Scand 53:505–514
Van der Linden PJ, Dierick A, Wilmin S, Bellens B, De Hert SG (2010) A randomized controlled trial comparing an intraoperative goal-directed strategy with routine clinical practice in patients undergoing peripheral arterial surgery. Eur J Anaesthesiol 27:788–793
Benes J, Chytra I, Altmann P, Hluchy M, Kasal E, Svitak R (2010) Intraoperative fluid optimization using stroke volume variation in high risk surgical patients: results of prospective randomized study. Crit Care 14(3):R118
Jammer I, Ulvik A, Erichsen C, Lodemel O, Ostgaard G (2010) Does central venous oxygen saturation-directed fluid therapy affect postoperative morbidity after colorectal surgery? A randomized assessor-blinded controlled trial. Anesthesiology 113:1072–1080
Mayer J, Boldt J, Mengistu AM, Rohm KD, Suttner S (2010) Goal-directed intraoperative therapy based on autocalibrated arterial pressure waveform analysis reduces hospital stay in high-risk surgical patients: a randomized, controlled trial. Crit Care 14:R18
Pillai P, McEleavy I, Gaughan M, Snowden C, Nesbitt I, Durkan G, Johnson M, Cosgrove J, Thorpe A (2011) A double-blind randomized controlled clinical trial to assess the effect of Doppler optimized intraoperative fluid management on outcome following radical cystectomy. J Urol 186:2201–2206
Cecconi M, Fasano N, Langiano N, Divella M, Costa M, Rhodes A (2011) Goal directed haemodynamic therapy during elective total hip arthroplasty under regional anaesthesia. Crit Care 15(3):R132
Brandstrup B, Svendsen PE, Rasmussen M, Belhage B, Rodt SA, Hansen B, Moller DR, Lundbech LB, Andersen N, Berg V, Thomassen N, Andersen ST, Simonsen L (2012) Which goal for fluid therapy during colorectal surgery is followed by the best outcome: near-maximal stroke volume or zero fluid balance? Br J Anaesth 109:191–199
Bartha E, Arfwedson C, Imnell A, Fernlund ME, Andersson LE, Kalman S (2013) Randomized controlled trial of goal-directed haemodynamic treatment in patients with proximal femoral fracture. Br J Anaesth 110:545–553
Scheeren TW, Wiesenack C, Gerlach H, Marx G (2013) Goal-directed intraoperative fluid therapy guided by stroke volume and its variation in high-risk surgical patients: a prospective randomized multicentre study. J Clin Monit Comput 27:225–233
Salzwedel C, Puig J, Carstens A, Bein B, Molnar Z, Kiss K, Hussain A, Belda J, Kirov MY, Sakka SG, Reuter DA (2013) Perioperative goal-directed hemodynamic therapy based on radial arterial pulse pressure variation and continuous cardiac index trending reduces postoperative complications after major abdominal surgery: a multi-center, prospective, randomized study. Crit Care 17:R191
Bisgaard J, Gilsaa T, Ronholm E, Toft P (2013) Optimising stroke volume and oxygen delivery in abdominal aortic surgery: a randomised controlled trial. Acta Anaesthesiol Scand 57:178–188
Bisgaard J, Gilsaa T, Ronholm E, Toft P (2013) Haemodynamic optimisation in lower limb arterial surgery: room for improvement? Acta Anaesthesiol Scand 57:189–198
Bundgaard-Nielsen M, Jans O, Muller RG, Korshin A, Ruhnau B, Bie P, Secher NH, Kehlet H (2013) Does goal-directed fluid therapy affect postoperative orthostatic intolerance? A randomized trial. Anesthesiology 119:813–823
Ramsingh DS, Sanghvi C, Gamboa J, Cannesson M, Applegate RL 2nd (2013) Outcome impact of goal directed fluid therapy during high risk abdominal surgery in low to moderate risk patients: a randomized controlled trial. J Clin Monit Comput 27:249–257
McKenny M, Conroy P, Wong A, Farren M, Gleeson N, Walsh C, O’Malley C, Dowd N (2013) A randomised prospective trial of intra-operative oesophageal Doppler-guided fluid administration in major gynaecological surgery. Anaesthesia 68:1224–1231
Srinivasa S, Taylor MH, Singh PP, Yu TC, Soop M, Hill AG (2013) Randomized clinical trial of goal-directed fluid therapy within an enhanced recovery protocol for elective colectomy. Br J Surg 100:66–74
Zakhaleva J, Tam J, Denoya PI, Bishawi M, Bergamaschi R (2013) The impact of intravenous fluid administration on complication rates in bowel surgery within an enhanced recovery protocol: a randomized controlled trial. Colorectal Dis 15:892–899
Zheng H, Guo H, Ye JR, Chen L, Ma HP (2013) Goal-directed fluid therapy in gastrointestinal surgery in older coronary heart disease patients: randomized trial. World J Surg 37:2820–2829
Goepfert MS, Richter HP, Zu Eulenburg C, Gruetzmacher J, Rafflenbeul E, Roeher K, von Sandersleben A, Diedrichs S, Reichenspurner H, Goetz AE, Reuter DA (2013) Individually optimized hemodynamic therapy reduces complications and length of stay in the intensive care unit: a prospective, randomized controlled trial. Anesthesiology 119:824–836
Zhang J, Chen CQ, Lei XZ, Feng ZY, Zhu SM (2013) Goal-directed fluid optimization based on stroke volume variation and cardiac index during one-lung ventilation in patients undergoing thoracoscopy lobectomy operations: a pilot study. Clinics 68:1065–1070
Phan TD, D’Souza B, Rattray MJ, Johnston MJ, Cowie BS (2014) A randomised controlled trial of fluid restriction compared to oesophageal Doppler-guided goal-directed fluid therapy in elective major colorectal surgery within an enhanced recovery after surgery program. Anaesth Intensive Care 42:752–760
Pearse RM, Harrison DA, MacDonald N, Gillies MA, Blunt M, Ackland G, Grocott MP, Ahern A, Griggs K, Scott R, Hinds C, Rowan K, Group OS (2014) Effect of a perioperative, cardiac output-guided hemodynamic therapy algorithm on outcomes following major gastrointestinal surgery: a randomized clinical trial and systematic review. JAMA 311:2181–2190
Peng K, Li J, Cheng H, Ji FH (2014) Goal-directed fluid therapy based on stroke volume variations improves fluid management and gastrointestinal perfusion in patients undergoing major orthopedic surgery. Med Princ Pract 23:413–420
Pestana D, Espinosa E, Eden A, Najera D, Collar L, Aldecoa C, Higuera E, Escribano S, Bystritski D, Pascual J, Fernandez-Garijo P, de Prada B, Muriel A, Pizov R (2014) Perioperative goal-directed hemodynamic optimization using noninvasive cardiac output monitoring in major abdominal surgery: a prospective, randomized, multicenter, pragmatic trial: POEMAS Study (PeriOperative goal-directed thErapy in Major Abdominal Surgery). Anesth Analg 119:579–587
van Beest PA, Vos JJ, Poterman M, Kalmar AF, Scheeren TW (2014) Tissue oxygenation as a target for goal-directed therapy in high-risk surgery: a pilot study. BMC Anesthesiol 14:122
Lai CW, Starkie T, Creanor S, Struthers RA, Portch D, Erasmus PD, Mellor N, Hosie KB, Sneyd JR, Minto G (2015) Randomized controlled trial of stroke volume optimization during elective major abdominal surgery in patients stratified by aerobic fitness. Br J Anaesth 115:578–589
Mikor A, Trasy D, Nemeth MF, Osztroluczki A, Kocsi S, Kovacs I, Demeter G, Molnar Z (2015) Continuous central venous oxygen saturation assisted intraoperative hemodynamic management during major abdominal surgery: a randomized, controlled trial. BMC Anesthesiol 15:82
Parke RL, McGuinness SP, Gilder E, McCarthy LW, Cowdrey KA (2015) A randomised feasibility study to assess a novel strategy to rationalise fluid in patients after cardiac surgery. Br J Anaesth 115:45–52
Fellahi JL, Brossier D, Dechanet F, Fischer MO, Saplacan V, Gerard JL, Hanouz JL (2015) Early goal-directed therapy based on endotracheal bioimpedance cardiography: a prospective, randomized controlled study in coronary surgery. J Clin Monit Comput 29:351–358
Moppett IK, Rowlands M, Mannings A, Moran CG, Wiles MD, Investigators N (2015) LiDCO-based fluid management in patients undergoing hip fracture surgery under spinal anaesthesia: a randomized trial and systematic review. Br J Anaesth 114:444–459
Ackland GL, Iqbal S, Paredes LG, Toner A, Lyness C, Jenkins N, Bodger P, Karmali S, Whittle J, Reyes A, Singer M, Hamilton M, Cecconi M, Pearse RM, Mallett SV, Omar RZ (2015) Individualised oxygen delivery targeted haemodynamic therapy in high-risk surgical patients: a multicentre, randomised, double-blind, controlled, mechanistic trial. Lancet Respir Med 3:33–41
Correa-Gallego C, Tan KS, Arslan-Carlon V, Gonen M, Denis SC, Langdon-Embry L, Grant F, Kingham TP, DeMatteo RP, Allen PJ, D’Angelica MI, Jarnagin WR, Fischer M (2015) Goal-directed fluid therapy using stroke volume variation for resuscitation after low central venous pressure-assisted liver resection: a randomized clinical trial. J Am Coll Surg 221:591–601
Kumar L, Kanneganti YS, Rajan S (2015) Outcomes of implementation of enhanced goal directed therapy in high-risk patients undergoing abdominal surgery. Indian J Anaesth 59:228–233
Funk DJ, HayGlass KT, Koulack J, Harding G, Boyd A, Brinkman R (2015) A randomized controlled trial on the effects of goal-directed therapy on the inflammatory response open abdominal aortic aneurysm repair. Crit Care 19:247
Stens J, de Wolf SP, van der Zwan RJ, Koning NJ, Dekker NA, Hering JP, Boer C (2015) Microcirculatory perfusion during different perioperative hemodynamic strategies. Microcirculation 22:267–275
Hand WR, Stoll WD, McEvoy MD, McSwain JR, Sealy CD, Skoner JM, Hornig JD, Tennant PA, Wolf B, Day TA (2016) Intraoperative goal-directed hemodynamic management in free tissue transfer for head and neck cancer. Head Neck 38(Suppl 1):E1974–E1980
Broch O, Carstens A, Gruenewald M, Nischelsky E, Vellmer L, Bein B, Aselmann H, Steinfath M, Renner J (2016) Non-invasive hemodynamic optimization in major abdominal surgery: a feasibility study. Minerva Anestesiol 82:1158–1169
Kapoor PM, Magoon R, Rawat R, Mehta Y (2016) Perioperative utility of goal-directed therapy in high-risk cardiac patients undergoing coronary artery bypass grafting: “a clinical outcome and biomarker-based study”. Ann Card Anaesth 19:638–682
Osawa EA, Rhodes A, Landoni G, Galas FR, Fukushima JT, Park CH, Almeida JP, Nakamura RE, Strabelli TM, Pileggi B, Leme AC, Fominskiy E, Sakr Y, Lima M, Franco RA, Chan RP, Piccioni MA, Mendes P, Menezes SR, Bruno T, Gaiotto FA, Lisboa LA, Dallan LA, Hueb AC, Pomerantzeff PM, Kalil Filho R, Jatene FB, Auler Junior JO, Hajjar LA (2016) Effect of perioperative goal-directed hemodynamic resuscitation therapy on outcomes following cardiac surgery: a randomized clinical trial and systematic review. Crit Care Med 44:724–733
Schmid S, Kapfer B, Heim M, Bogdanski R, Anetsberger A, Blobner M, Jungwirth B (2016) Algorithm-guided goal-directed haemodynamic therapy does not improve renal function after major abdominal surgery compared to good standard clinical care: a prospective randomised trial. Crit Care 20:50
Luo J, Xue J, Liu J, Liu B, Liu L, Chen G (2017) Goal-directed fluid restriction during brain surgery: a prospective randomized controlled trial. Ann Intensive Care 7:16
Stens J, Hering JP, van der Hoeven CWP, Boom A, Traast HS, Garmers LE, Loer SA, Boer C (2017) The added value of cardiac index and pulse pressure variation monitoring to mean arterial pressure-guided volume therapy in moderate-risk abdominal surgery (COGUIDE): a pragmatic multicentre randomised controlled trial. Anaesthesia 72:1078–1087
Weinberg L, Ianno D, Churilov L, Chao I, Scurrah N, Rachbuch C, Banting J, Muralidharan V, Story D, Bellomo R, Christophi C, Nikfarjam M (2017) Restrictive intraoperative fluid optimisation algorithm improves outcomes in patients undergoing pancreaticoduodenectomy: A prospective multicentre randomized controlled trial. PLoS One 12:e0183313
Kaufmann KB, Stein L, Bogatyreva L, Ulbrich F, Kaifi JT, Hauschke D, Loop T, Goebel U (2017) Oesophageal Doppler guided goal-directed haemodynamic therapy in thoracic surgery—a single centre randomized parallel-arm trial. Br J Anaesth 118:852–861
Elgendy MA, Esmat IM, Kassim DY (2017) Outcome of intraoperative goal-directed therapy using Vigileo/FloTrac in high-risk patients scheduled for major abdominal surgeries: A prospective randomized trial. Egypt J Anaesth 33:263–269
Kapoor PM, Magoon R, Rawat RS, Mehta Y, Taneja S, Ravi R, Hote MP (2017) Goal-directed therapy improves the outcome of high-risk cardiac patients undergoing off-pump coronary artery bypass. Ann Card Anaesth 20:83–89
Xu H, Shu S-H, Wang D, Chai X-Q, Xie Y-H, Zhou W-D (2017) Goal-directed fluid restriction using stroke volume variation and cardiac index during one-lung ventilation: a randomized controlled trial. J Thorac Dis 9:2992–3004
Gomez-Izquierdo JC, Trainito A, Mirzakandov D, Stein BL, Liberman S, Charlebois P, Pecorelli N, Feldman LS, Carli F, Baldini G (2017) Goal-directed fluid therapy does not reduce primary postoperative ileus after elective laparoscopic colorectal surgery: a randomized controlled trial. Anesthesiology 127:36–49
Kim HJ, Kim EJ, Lee HJ, Min JY, Kim TW, Choi EC, Kim WS, Koo BN (2018) Effect of goal-directed haemodynamic therapy in free flap reconstruction for head and neck cancer. Acta Anaesthesiol Scand 62:903–914
Szturz P, Folwarczny P, Kula R, Neiser J, Sevcik P, Benes J (2018) Multi-parametric functional hemodynamic optimization improves postsurgical outcome after intermediate risk open gastrointestinal surgery, a randomized controlled trial. Minerva Anestesiol. https://doi.org/10.23736/S0375-9393.18.12467-9
Gerent ARM, Almeida JP, Fominskiy E, Landoni G, de Oliveira GQ, Rizk SI, Fukushima JT, Simoes CM, Ribeiro U Jr, Park CL, Nakamura RE, Franco RA, Candido PI, Tavares CR, Camara L, Dos Santos Rocha Ferreira G, de Almeida EPM, Filho RK, Galas F, Hajjar LA (2018) Effect of postoperative goal-directed therapy in cancer patients undergoing high-risk surgery: a randomized clinical trial and meta-analysis. Crit Care 22:133
Corbella D, Toppin PJ, Ghanekar A, Ayach N, Schiff J, Van Rensburg A, McCluskey SA (2018) Cardiac output-based fluid optimization for kidney transplant recipients: a proof-of-concept trial. Can J Anaesth. 65:873–883
Calvo-Vecino JM, Ripolles-Melchor J, Mythen MG, Casans-Frances R, Balik A, Artacho JP, Martinez-Hurtado E, Serrano Romero A, Fernandez Perez C, Asuero de Lis S, Group FTI (2018) Effect of goal-directed haemodynamic therapy on postoperative complications in low-moderate risk surgical patients: a multicentre randomised controlled trial (FEDORA trial). Br J Anaesth 120:734–744
Holm C, Mayr M, Tegeler J, Horbrand F, Henckel von Donnersmarck G, Muhlbauer W, Pfeiffer UJ (2004) A clinical randomized study on the effects of invasive monitoring on burn shock resuscitation. Burns 30:798–807
Lin SM, Huang CD, Lin HC, Liu CY, Wang CH, Kuo HP (2006) A modified goal-directed protocol improves clinical outcomes in intensive care unit patients with septic shock: a randomized controlled trial. Shock 26:551–557
Chytra I, Pradl R, Bosman R, Pelnar P, Kasal E, Zidkova A (2007) Esophageal Doppler-guided fluid management decreases blood lactate levels in multiple-trauma patients: a randomized controlled trial. Crit Care 11:R24
Takala J, Ruokonen E, Tenhunen JJ, Parviainen I, Jakob SM (2011) Early non-invasive cardiac output monitoring in hemodynamically unstable intensive care patients: a multi-center randomized controlled trial. Crit Care 15:R148
Andrews B, Muchemwa L, Kelly P, Lakhi S, Heimburger DC, Bernard GR (2014) Simplified severe sepsis protocol: a randomized controlled trial of modified early goal-directed therapy in Zambia. Crit Care Med 42:2315–2324
Zhang Z, Ni H, Qian Z (2015) Effectiveness of treatment based on PiCCO parameters in critically ill patients with septic shock and/or acute respiratory distress syndrome: a randomized controlled trial. Intensive Care Med 41:444–451
Yu J, Zheng R, Lin H, Chen Q, Shao J, Wang D (2017) Global end-diastolic volume index vs CVP goal-directed fluid resuscitation for COPD patients with septic shock: a randomized controlled trial. Am J Emerg Med 35:101–105
Popelut A, Valet F, Fromentin O, Thomas A, Bouchard P (2010) Relationship between sponsorship and failure rate of dental implants: a systematic approach. PLoS One 5:e10274
Bartels RH, Delye H, Boogaarts J (2012) Financial disclosures of authors involved in spine research: an underestimated source of bias. Eur Spine J 21:1229–1233
DeGeorge BR Jr, Holland MC, Drake DB (2015) The impact of conflict of interest in abdominal wall reconstruction with acellular dermal matrix. Ann Plast Surg 74:242–247
Wang AT, McCoy CP, Murad MH, Montori VM (2010) Association between industry affiliation and position on cardiovascular risk with rosiglitazone: cross sectional systematic review. BMJ 340:c1344
Viswanathan M, Carey T, Belinson S, Berliner E, Chang S, Graham E, Guise J, Ip S, Maglione M, McCrory D, McPheeters M, Newberry S, Sista P, White C (2014) A proposed approach may help systematic reviews retain needed expertise while minimizing bias from nonfinancial conflicts of interest. J Clin Epidemiol 67:1229–1238
Acknowledgements
We thank Dr. Cary P. Gross from the Center for Outcomes Research & Evaluation, Department of Internal Medicine, Yale University School of Medicine, New Haven, Connecticut for his help with manuscript preparation.
Funding
Support was provided from institutional and/or departmental sources at Yale University and Central South University, China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
L. Meng is a consultant for CAS Medical Systems, Inc. The other authors declare no competing interests.
Additional information
Essential gist
Conflicts of interest may have a potential association with article conclusions in goal-directed hemodynamic therapy research.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, L., Dai, F., Brackett, A. et al. Association of conflicts of interest with the results and conclusions of goal-directed hemodynamic therapy research: a systematic review with meta-analysis. Intensive Care Med 44, 1638–1656 (2018). https://doi.org/10.1007/s00134-018-5345-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-018-5345-z