Abstract
Purpose
Systemic antifungal therapy (SAT) of invasive candidiasis needs to be initiated immediately upon clinical suspicion. Controversies exist about adequate time and potential harm of antifungal de-escalation (DE) in documented and suspected candidiasis in ICU patients. Our objective was to investigate whether de-escalation within 5 days of antifungal initiation is associated with an increase of the 28-day mortality in SAT-treated non-neutropenic adult ICU patients.
Methods
From the 835 non-neutropenic adults recruited in the multicenter prospective observational AmarCAND2 study, we selected the patients receiving systemic antifungal therapy for a documented or suspected invasive candidiasis in the ICU and who were still alive 5 days after SAT initiation. They were included into two groups according to the occurrence of observed SAT de-escalation before day 6. The average causal SAT de-escalation effect on 28-day mortality was evaluated by using a double robust estimation.
Results
Among the 647 included patients, early de-escalation at day 5 after antifungal initiation occurred in 142 patients (22 %), including 48 (34 %) patients whose SAT was stopped before day 6. After adjustment for the baseline confounders, early SAT de-escalation was the solely factor not associated with increased 28-day mortality (RR 1.12, 95 % CI 0.76–1.66).
Conclusion
In non-neutropenic critically ill adult patients with documented or suspected invasive candidiasis, SAT de-escalation within 5 days was not related to increased day-28 mortality but it was associated with decreased SAT consumption. These results suggest for the first time that SAT de-escalation may be safe in these patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Candida species are one of the most frequently recovered pathogens in patients with hospital-acquired bloodstream infections and the most common cause of invasive fungal infection [1–3], which is associated with a mortality rate from 30 % to more than 60 % in the case of septic shock [4–8].
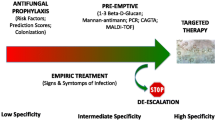
Early treatment of invasive candidiasis (IC) improves patients’ prognosis [4, 6, 8, 9]. Given the poor sensitivity of blood culture to diagnose IC [10], guidelines recommend to initiate systemic antifungal therapy (SAT), mostly an echinocandin, for critically ill patients with risk factors for IC and no other known cause of fever. This approach is considered valid by many experts while waiting for further evidence. The general opinion is that the administration of SAT should be guided by the evaluation of risk factors, the use of clinical prediction rules, culture data from non-sterile sites, and biological markers [11].
Unfortunately, no diagnostic tests are available to firmly confirm or discard the diagnosis of IC in the absence of positive blood cultures or non-contaminated positive sample from a sterile site. Therefore the management of antifungal treatment in suspected non-proven invasive fungal infection is speculative.
A cross-sectional multicenter study showed that SAT was administered to 7.5 % of ICU patient-days, although two-thirds of them had no documented invasive fungal infection [12]. Possible consequences of these practices are an increase of cost and selection of more resistant yeasts [13, 14].
Similarly, the positive predictive value for IC of the prediction rules in a general ICU population was lower than 20 % in the most recent studies [15–17] and systematic pre-emptive strategies in such predetermined patients failed to improve patients’ prognosis [15, 18].
Echinocandins are the first-line therapeutic option for IC, because of fungicidal activity, good tolerance, and a broad-spectrum activity [19, 20]. For IC, the European Society for Clinical Microbiology and Infectious Diseases (ESCMID) and Infectious Diseases Society of America (IDSA) guidelines recommend a de-escalation strategy (3 days in stabilized patients, as per IDSA, and 10 days overall, as per ESCMID) to limit the emergence of resistant strains and to reduce treatment costs [19, 20]. However, the level of recommendation is poor because of the lack of available data. The safety of de-escalation in the case of proven candidiasis has been recently suggested by prospective non-comparative studies [21, 22], but no comparative study exists for proven or probable IC treated in the ICU.
The objective of this study is to investigate whether de-escalation or stopping of SAT within 5 days of initiation is associated with an increase in 28-day mortality in SAT-treated non-neutropenic adult ICU patients. We used a marginal structural model (MSM) to assess the causal relationship of de-escalation on day-28 prognosis using the prospective multicenter observational French study AmarCAND2.
Materials and methods
Study design
The patients were selected from AmarCAND2, a multicenter, prospective, observational study conducted in French ICUs during 1 year (2012–2013). The investigating centers were ICUs having managed at least one IC case within the past year and willing to participate in the study. Investigators enrolled patients according to the study protocol and managed them according to their own clinical judgment, independently from the sponsor. The Ethics Committee of the French Intensive Care Society and the French National Committee for Data Protection and Freedom of Information approved the study. Such an observational study does not require patients to sign an informed consent according to French regulations; however, written information was provided and oral consent was obtained from all participating patients whenever possible, or their family.
Patients
Investigators enrolled consecutive adult patients hospitalized in ICU and requiring SAT for documented or suspected invasive Candida infection during their ICU stay. Patients receiving prophylactic SAT, those with neutropenia (absolute neutrophil count at most 500/mm3), those who had undergone solid organ transplant within the previous 15 days, or those receiving SAT for a mold infection were excluded.
Clinical and mycological data collection and definition
Data were collected for each patient using an electronic case report at study inclusion and throughout the study. The following data were collected: demographics data, severity scores (at inclusion, at SAT administration, at day 7 after SAT administration), clinical data at SAT administration, mycological data for confirmed invasive Candida infection, and information on the treatment. If the initial treatment was modified, the investigator was asked to record the date and reason(s) for the modification and information about the modification. At the end of the SAT, the following data were recorded: the SAT end date and the outcome of the Candida infection. The dates of ICU and hospital discharges were also collected, as was the vital status of all patients 28 days after the SAT initiation (alive, dead, or lost to follow-up after ICU discharge).
Studied population
AmarCAND2 patients who were still alive in the ICU at day 5 after the first SAT administration were included in the present study. Patients were divided into two groups according to the SAT strategy observed within 5 days following SAT initiation: (1) de-escalation group (DE) and (2) absence of de-escalation group (NoDE). SAT de-escalation was defined as either a switch from initial SAT drugs (except fluconazole) to fluconazole or termination of initial SAT drugs within 5 days following SAT initiation.
Study outcomes
The primary outcome was to evaluate whether SAT de-escalation within 5 days of SAT initiation was, or not, associated with the worsening of the 28-day mortality as compared to the mortality of adult non-neutropenic ICU patients who received SAT without any de-escalation. Subgroup analyses of the primary objective were performed for (1) patients with SAT de-escalation observed within 7 days after SAT initiation (allowing variation of SOFA score between SAT day and day 7 to be added in the model as an adjustment covariate); (2) patients who had a documented IC at day 5 (including a distinction between patients with C. albicans IC or patients with C. non-albicans IC); (3) patients who did not have a documented IC at day 5; (4) Patients with echinocandins as initial SAT; (5) patients with fluconazole as initial SAT; (6) patients without secondary location for Candida; (7) ICUs belonging to large hospitals with more than 1040 hospitalization-beds. Secondary objectives were to evaluate (1) if stopping SAT before day 5 is, or not, associated with the worsening of the 28-day mortality as compared to the mortality of adult non-neutropenic ICU patients without proven invasive candidiasis at day 5 and whose SAT was not stopped; (2) to compare the effect of SAT de-escalation for adults ICU patients on (a) SOFA score at day 7; (b) the length of ICU stay; (c) the number of days alive after ICU discharge; (d) the duration of SAT administration; (e) the number of days alive after the end of SAT.
Statistical analysis
A descriptive analysis of the patient’s characteristics was performed using median and interquartile range for quantitative data and frequencies and percent for qualitative data. The baseline characteristics of groups (DE vs. NoDE) were compared by means of the Chi-squared test for qualitative data and Mann–Whitney test for quantitative data. To estimate the average causal effect of DE on 28-day mortality, a double robust (DR) inverse probability of treatment weight (IPTW) estimator was used. The DR-IPTW estimator is an extension of the IPTW estimator [23]. The general principle of IPTW is to balance the distribution of baseline confounders across treatment groups, in order to reach the condition of a randomized controlled trial [24]. Two modeling steps are required. The first step is to model the treatment assignment, i.e., the propensity which is needed to compute the weights. The second step is to model the outcome as a function of the treatment in the weighted sample. When the treatment and the outcome are both binary, each modeling step usually relies on logistic regression models. Such regression models rely on strong assumptions about the underlying data distribution and may therefore be misspecified. DR-IPTW estimators were developed to prevent the consequence of model misspecification. This adaptation of the IPTW estimators guaranties consistency if only one of the two models is correctly specified and efficiency if they are both correctly specified [25]. The DR-IPTW estimator has a marginal interpretation, which corresponds to the average treatment effect, i.e., the difference in outcome had all patients being exposed to SAT de-escalation versus all patients being free from SAT de-escalation after adjustment for all measured confounders. The final results were expressed as relative risk (RR) for 28-day mortality between the two exposed pseudo-populations with bootstrapped standard errors and 95 % confidence intervals. Post hoc power analyses were performed for primary and secondary objectives. The robustness of the results was confirmed by IPTW models. Statistical analyses were performed using SAS v9.3 (SAS Institute Inc., Cary, NC, USA). The SAS Macro developed by Funk et al. was used for the DR estimation [26]. A p value less than 0.05 was considered significant. Details on the power calculation are given in the Electronic Supplementary Material (ESM).
Results
Patient characteristics
From 835 patients enrolled in the AmarCAND2 study, 647 (77.5 %) who were still alive in the ICU 5 days after SAT initiation were included in the present study. Of the 647 included patients, 142 (22 %) experienced a SAT de-escalation or a SAT stop within 5 days of SAT initiation and 505 (78 %) were in the NoDE group (Fig. 1). Patients in the DE group were younger and had a shorter previous ICU stay but their SAPS II or SOFA scores at ICU admission were similar (Table 1). The rate of proven IC was not different between DE and NoDE groups (Table 2). Seven days after SAT initiation, SOFA score was not different between DE and NoDE groups (Table 3).
Overall flow chart (primary objective). *SAT stop: N = 48. ICU intensive care unit, SAT systemic antifungal therapy, D5 day 5 after SAT initiation, DE SAT de-escalation group at day 5 included patients with a SAT de-escalation or a SAT stopping observed between D0 and D5 after SAT initiation, NoDE absence of SAT de-escalation at day 5, Death 28-day mortality
Initial SAT and SAT de-escalation
The use of echinocandins as first SAT was more frequent in the DE group (89 %) (Table 2). In the DE group, the median delay before SAT modification was 3 days (IQR 2; 4). A total of 94 patients (66 %) experienced a SAT de-escalation to fluconazole at a median dose of 800 mg (IQR 400; 800) and 48 patients (34 %) experienced a SAT stop within 5 days of initial SAT. Removal of a central catheter on the SAT initiation day was observed for 15 patients (58 %) in the DE group and for 23 patients (29 %) in the NoDE group (p < 0.01) (more details on removal of the possible source of infection are given Table E1 in ESM). The median duration of SAT was 11 days (IQR 7; 16) in the 323 remaining patients for whom the reason for SAT discontinuation was not recorded. Finally, antifungal prescriptions were defined into written procedures in 47 (61 %) ICUs and the de-escalation procedure was declared to follow international guidelines in 58 (75 %) ICUs. The existence of both types of procedures was not associated with the decision to de-escalate (Table 1).
Characteristics of patients with an invasive candidiasis at day 5
There was no difference between both groups regarding the characteristics of the patients and the evolution of the invasive candidiasis for the patients with an IC at day 5. In particular, the rates of clinical failure, or of death, were not different between DE and NoDE groups in both documented and non-documented IC (more details on the characteristics of the patients with an IC are given in Tables E2 and E3 in ESM).
Primary outcome
On the basis of the double robust estimation, the de-escalation strategy within 5 days had no significant impact on the 28-day mortality compared to non de-escalation strategy (relative risk 1.12, 95 % CI 0.76–1.66). Details on variables used for the double robust estimation are provided in Tables E4 and E5 in ESM. The sensitivity analysis of patients with a SAT de-escalation before day 7 gave similar results (RR 0.95 [0.66; 1.36]). Results remained unchanged in prespecified subgroups, more particularly for documented IC, non-documented IC, and first-line treatment with candins (Fig. 2). Moreover, subgroup analysis excluding suspected IC with C. glabrata or C. krusei showed no difference. The results using an IPTW estimator confirmed the double robust estimation model that we used (Table E6 in ESM).
Summary of results for primary and secondary outcomes. DE de-escalation, NoDE no de-escalation, ICU intensive care unit, D5 day 5, D7 day 7, SAT systemic antifungal treatment, IC invasive Candida infection. *RR relative risk. If RR < 1, de-escalation is protective for 28-day mortality. If RR > 1, de-escalation is not protective for 28-day mortality. †De-escalation was considered within 7 days after SAT initiation. The estimate was adjusted for SOFA score variation between the SAT start and day 7. §Fluconazole: All patients in the DE group had only stop treatment. ¶Exclusion of patients with suspected invasive candidiasis with C. glabrata and C. krusei. **Candida spp. for the 8 patients in the DE group were C. glabrata (N = 4), C. tropicalis (N = 2), C. parapsilosis (N = 1), and C. lusitaniae (N = 1). ††DE concerned only patients which switched from all initial SAT to SAT stop within 5 days
Secondary outcomes
Early SAT de-escalation was associated with a decrease in the length of ICU stay (14 days [9; 28] vs. 19 days [11; 35] group p < 0.01), and a decrease in the SAT duration. The number of days alive without SAT at day 28 was higher in the DE group (14 days [5; 23]) than in the NoDE group (10 days [2; 17] p < 0.01). On the basis of the cost of echinocandin agents in France, we calculated median costs in each DE group. It was 2835 € in the NoDE group (IQR 171; 7371) and 1743 € in the DE group (IQR 1134; 2382). The improvement of the SOFA score at day 7 was similar in both groups (Table 3). Clinical and microbiological failure rates were strictly similar between DE and NoDE groups (Table E3 in ESM).
Post hoc power analysis
The sample size for the primary objective allowed us to conclude the non-inferiority of SAT de-escalation, in the case of an absolute risk difference of 9 % with a power of 0.8. Details of the post hoc power are given in Table E7 in ESM.
Discussion
Our causal analysis based on a large prospective observational multicenter study showed that SAT de-escalation in case of suspected or documented IC in non-neutropenic ICU patients occurred in only 22 % of the cases, and did not impact day-28 mortality. DE led to a subsequent and significant decrease in the antifungal consumption. These results remained valid with different subgroups and were confirmed by sensitivity analysis.
Incidence of IC is increasing in ICU patients [27]. Despite the increasing amount of data on IC management, and the development of less toxic antifungal agents, the day-30 and early (before day 8) mortality rates are still increasing over time.
In order to save patients, physicians started antifungals early in case of severe sepsis or shock, without waiting for the definite proof of infection, as it is often delayed. This strategy naturally increased the number of SAT initiated in ICU and is certainly beneficial for ICU patients with documented fungal infections [12]. However, three out of four of the SAT are given without definite proof of fungal infection; this may lead to antifungal overuse with deleterious consequences [12]. Finally, the benefit of this empiric strategy has not been demonstrated in the general ICU population [15, 18]. Furthermore, the overuse of antifungal agents is associated with increased MICs of Candida spp. to antifungal drugs [13, 28–31]. In addition, antifungal pre-exposure is associated with clinical [27] and microbiological failures [13, 32] and with breakthrough infections [29, 33]. Antifungal streamlining strategies are therefore fundamental to try to curb the trends.
In the ESCMID guidelines, step-down therapy is recommended only after 10 days for documented infection. In the IDSA guidelines, step-down therapy to fluconazole is considered reasonable for patients who have clinically improved after initial therapy with an echinocandin or polyenes and who are infected with a Candida spp. that is likely to be susceptible to fluconazole [20]. However, both the de-escalation rules for the most severe critically ill patients and the stopping rules for SAT started early without proven infection are not defined [19, 20].
For proven invasive candidiasis, three other studies already found that de-escalation is safe in the case of Candida spp. sensitive to fluconazole. In a small cost-effectiveness study, de-escalation to oral fluconazole was encouraged for candidemia. Of the 37 episodes of documented candidemia, 27 were commenced on an echinocandin or voriconazole and 19 (70.3 %) were de-escalated to fluconazole on the basis of the intravenous oral switch therapy (IVOST) policy after a mean of 4.6 days, with good results and important cost saving [34].
In the recent ACTION project performed by the Japanese Mycosis Group, clinical failure rate and mortality rate were lower in patients with adherence to step-down oral therapy [22].
An open-label, non-comparative study evaluated an intravenous (i.v.) to oral step-down strategy. Patients with candidiasis were treated intravenously with anidulafungin. After 5 days of i.v. therapy, investigators had the option to step-down to oral azole therapy (fluconazole or voriconazole) if patients met prespecified criteria (ability to tolerate oral therapy; not febrile for more than 24 h; hemodynamically stable; not neutropenic; and documented clearance of Candida from the bloodstream). Of the 250 patients enrolled in the mITT population, 102 were switched to oral therapy after a median of 6 days, and clinical response relapse and mortality were similar between patients with early switch and the other patients [21].
Our results confirm previous observations that early de-escalation to azole is possible and safe. We also showed that empiric treatment could be safely stopped after 5 days of SAT therapy in the absence of proven invasive candidiasis.
De-escalation strategy is not explained by severity of organ dysfunction, shock, surgical type of admission, or by the use of other antimicrobial treatments. It occurred less frequently in older patients, for patients in ICU for a longer period of time, or those still under parenteral nutrition.
This study had several limitations. First, it was calibrated to reach a power of 80 % to show an absolute risk difference (ARD) of 9 % between both groups. The study is therefore underpowered to show a smaller ARD, although still of important clinical relevance. The post hoc power analysis showed that the sample size necessary to reach a sufficient power for subgroup analysis or to show a smaller ARD would need thousands of patients enrolled. This would be unrealistic for a clinical trial.
Secondly, the causal model based on observational data was conducted under the unverifiable assumption of the absence of immeasurable confounders. In this study, there were no measurable time-dependent confounders during the SAT administration. This was a limitation, because SAT de-escalation could be related to the evolution of the patient’s state of health during time.
As an example, the beta-d-glucan was not routinely measured within the AmarCAND2 study. However, the potential usefulness of repeated measurements of beta-d-glucan is only suggested in very particular subgroups of ICU patients with complicated abdominal surgery abdominal leakage or acute pancreatitis [35] and lack of specificity and sensitivity in the general ICU population [36].
Third, the reasons leading the investigators to stop SAT were not recorded, although it may have influenced the final results. However, the guidelines for de-escalation were equally followed between DE and NoDE groups.
Last, we cannot be sure that measured confounders occurring between SAT initiation and day 5 that were making physicians prone to de-escalate have been fully taken into account. However, neither the characteristics of the invasive candidiasis nor the SOFA at day 7 or the delta SOFA between SAT initiation and day 7 were different between groups. Furthermore, we analyzed de-escalation at day 7 as an exposure variable; it had no significant effect on the 28-day mortality.
We concluded that, in non-neutropenic critically ill adult patients with proven or suspected IC, SAT de-escalation within 5 days was associated with a decrease in antifungal consumption without apparent deleterious effect on day-28 mortality. De-escalation to fluconazole may be recommended for stabilized patients, with negative blood culture and absence of secondary location for candidiasis. The latter has to be confirmed in randomized control trials.
References
Rangel-Frausto MS, Wiblin T, Blumberg HM, Saiman L, Patterson J, Rinaldi M, Pfaller M, Edwards JE, Jarvis W, Dawson J, Wenzel RP (1999) National epidemiology of mycoses survey (NEMIS): variations in rates of bloodstream infections due to Candida species in seven surgical intensive care units and six neonatal intensive care units. Clin Infect Dis 29:253–258
Tabah A, Koulenti D, Laupland K, Misset B, Valles J, Bruzzi de Carvalho F, Paiva JA, Cakar N, Ma X, Eggimann P, Antonelli M, Bonten MJM, Csomos A, Krueger WA, Mikstacki A, Lipman J, Depuydt P, Vesin A, Garrouste-Orgeas M, Zahar J-R, Blot S, Carlet J, Brun-Buisson C, Martin C, Rello J, Dimopoulos G, Timsit J-F (2012) Characteristics and determinants of outcome of hospital-acquired bloodstream infections in intensive care units: the EUROBACT International Cohort Study. Intensive Care Med 38:1930–1945
Blumberg HM, Jarvis WR, Soucie JM, Edwards JE, Patterson JE, Pfaller MA, Rangel-Frausto MS, Rinaldi MG, Saiman L, Wiblin RT, Wenzel RP (2001) Risk factors for candidal bloodstream infections in surgical intensive care unit patients: the NEMIS Prospective Multicenter Study. Clin Infect Dis 33:177–186
Bassetti M, Righi E, Ansaldi F, Merelli M, Trucchi C, Cecilia T, De Pascale G, Diaz-Martin A, Luzzati R, Rosin C, Lagunes L, Trecarichi EM, Sanguinetti M, Posteraro B, Garnacho-Montero J, Sartor A, Rello J, Rocca GD, Antonelli M, Tumbarello M (2014) A multicenter study of septic shock due to candidemia: outcomes and predictors of mortality. Intensive Care Med 40:839–845
Garrouste-Orgeas M, Timsit JF, Tafflet M, Misset B, Zahar J-R, Soufir L, Lazard T, Jamali S, Mourvillier B, Cohen Y, De Lassence A, Azoulay E, Cheval C, Descorps-Declere A, Adrie C, Costa de Beauregard M-A, Carlet J, OUTCOMEREA Study Group (2006) Excess risk of death from intensive care unit-acquired nosocomial bloodstream infections: a reappraisal. Clin Infect Dis 42:1118–1126
Kollef M, Micek S, Hampton N, Doherty JA, Kumar A (2012) Septic shock attributed to Candida infection: importance of empiric therapy and source control. Clin Infect Dis 54:1739–1746
Pittet D, Li N, Woolson RF, Wenzel RP (1997) Microbiological factors influencing the outcome of nosocomial bloodstream infections: a 6-year validated, population-based model. Clin Infect Dis 24:1068–1078
Wisplinghoff H, Seifert H, Tallent SM, Bischoff T, Wenzel RP, Edmond MB (2003) Nosocomial bloodstream infections in pediatric patients in United States hospitals: epidemiology, clinical features and susceptibilities. Pediatr Infect Dis J 22:686–691
Grim SA, Berger K, Teng C, Gupta S, Layden JE, Janda WM, Clark NM (2012) Timing of susceptibility-based antifungal drug administration in patients with Candida bloodstream infection: correlation with outcomes. J Antimicrob Chemother 67:707–714
Köck R, Eißing LC, Boschin MG, Ellger B, Horn D, Idelevich EA, Becker K (2013) Evaluation of bactec mycosis IC/F and Plus Aerobic/F blood culture bottles for detection of Candida in the presence of antifungal agents. J Clin Microbiol 51:3683–3687
Eggimann P, Pittet D (2014) Candida colonization index and subsequent infection in critically ill surgical patients: 20 years later. Intensive Care Med 40:1429–1448
Azoulay E, Dupont H, Tabah A, Lortholary O, Stahl J-P, Francais A, Martin C, Guidet B, Timsit J-F (2012) Systemic antifungal therapy in critically ill patients without invasive fungal infection. Crit Care Med 40:813–822
Alexander BD, Johnson MD, Pfeiffer CD, Jiménez-Ortigosa C, Catania J, Booker R, Castanheira M, Messer SA, Perlin DS, Pfaller MA (2013) Increasing echinocandin resistance in Candida glabrata: clinical failure correlates with presence of FKS mutations and elevated minimum inhibitory concentrations. Clin Infect Dis 56:1724–1732
Saraya T, Tanabe K, Araki K, Yonetani S, Makino H, Watanabe T, Tsujimoto N, Takata S, Kurai D, Ishii H, Miyazaki Y, Takizawa H, Goto H (2014) Breakthrough invasive Candida glabrata in patients on micafungin: a novel FKS gene conversion correlated with sequential elevation of MIC. J Clin Microbiol 52:2709–2712
Ostrosky-Zeichner L, Shoham S, Vazquez J, Reboli A, Betts R, Barron MA, Schuster M, Judson MA, Revankar SG, Caeiro JP, Mangino JE, Mushatt D, Bedimo R, Freifeld A, Nguyen MH, Kauffman CA, Dismukes WE, Westfall AO, Deerman JB, Wood C, Sobel JD, Pappas PG (2014) MSG-01: a randomized, double-blind, placebo-controlled trial of caspofungin prophylaxis followed by preemptive therapy for invasive candidiasis in high-risk adults in the critical care setting. Clin Infect Dis 58:1219–1226
León C, Ruiz-Santana S, Saavedra P, Galván B, Blanco A, Castro C, Balasini C, Utande-Vázquez A, González de Molina FJ, Blasco-Navalproto MA, López MJ, Charles PE, Martín E, Hernández-Viera MA, Cava Study Group (2009) Usefulness of the “Candida score” for discriminating between Candida colonization and invasive candidiasis in non-neutropenic critically ill patients: a prospective multicenter study. Crit Care Med 37:1624–1633
Ostrosky-Zeichner L, Sable C, Sobel J, Alexander BD, Donowitz G, Kan V, Kauffman CA, Kett D, Larsen RA, Morrison V, Nucci M, Pappas PG, Bradley ME, Major S, Zimmer L, Wallace D, Dismukes WE, Rex JH (2007) Multicenter retrospective development and validation of a clinical prediction rule for nosocomial invasive candidiasis in the intensive care setting. Eur J Clin Microbiol Infect Dis 26:271–276
Bailly S, Bouadma L, Azoulay E, Orgeas MG, Adrie C, Souweine B, Schwebel C, Maubon D, Hamidfar-Roy R, Darmon M, Wolff M, Cornet M, Timsit J-F (2015) Failure of empirical systemic antifungal therapy in mechanically ventilated critically ill patients. Am J Respir Crit Care Med 191:1139–1146
Cornely OA, Bassetti M, Calandra T, Garbino J, Kullberg BJ, Lortholary O, Meersseman W, Akova M, Arendrup MC, Arikan-Akdagli S, Bille J, Castagnola E, Cuenca-Estrella M, Donnelly JP, Groll AH, Herbrecht R, Hope WW, Jensen HE, Lass-Flörl C, Petrikkos G, Richardson MD, Roilides E, Verweij PE, Viscoli C, Ullmann AJ, ESCMID Fungal Infection Study Group (2012) ESCMID guideline for the diagnosis and management of Candida diseases 2012: non-neutropenic adult patients. Clin Microbiol Infect 18(Suppl 7):19–37
Pappas PG, Kauffman CA, Andes D, Benjamin DK, Calandra TF, Edwards JE, Filler SG, Fisher JF, Kullberg B-J, Ostrosky-Zeichner L, Reboli AC, Rex JH, Walsh TJ, Sobel JD, Infectious Diseases Society of Americia (2009) Clinical practice guidelines for the management of candidiasis: 2009 update by the Infectious Diseases Society of America. Clin Infect Dis 48:503–535
Vazquez J, Reboli AC, Pappas PG, Patterson TF, Reinhardt J, Chin-Hong P, Tobin E, Kett DH, Biswas P, Swanson R (2014) Evaluation of an early step-down strategy from intravenous anidulafungin to oral azole therapy for the treatment of candidemia and other forms of invasive candidiasis: results from an open-label trial. BMC Infect Dis 14:97
Takesue Y, Ueda T, Mikamo H, Oda S, Takakura S, Kitagawa Y, Kohno S, ACTIONs Project (2015) Management bundles for candidaemia: the impact of compliance on clinical outcomes. J Antimicrob Chemother 70:587–593
Bang H, Robins JM (2005) Doubly robust estimation in missing data and causal inference models. Biometrics 61:962–973
Bailly S, Pirracchio R, Timsit JF (2015) What’s new in the quantification of causal effects from longitudinal cohort studies: a brief introduction to marginal structural models for intensivists. Intensive Care Med. doi:10.1007/s00134-015-3919-6
Zhang M, Schaubel DE (2012) Contrasting treatment-specific survival using double-robust estimators. Stat Med 31:4255–4268
Funk MJ, Westreich D, Wiesen C, Stürmer T, Brookhart MA, Davidian M (2011) Doubly robust estimation of causal effects. Am J Epidemiol 173:761–767
Lortholary O, Renaudat C, Sitbon K, Madec Y, Denoeud-Ndam L, Wolff M, Fontanet A, Bretagne S, Dromer F, French Mycosis Study Group (2014) Worrisome trends in incidence and mortality of candidemia in intensive care units (Paris area, 2002–2010). Intensive Care Med 40:1303–1312
Andes D, Forrest A, Lepak A, Nett J, Marchillo K, Lincoln L (2006) Impact of antimicrobial dosing regimen on evolution of drug resistance in vivo: fluconazole and Candida albicans. Antimicrob Agents Chemother 50:2374–2383
Fekkar A, Dannaoui E, Meyer I, Imbert S, Brossas JY, Uzunov M, Mellon G, Nguyen S, Guiller E, Caumes E, Leblond V, Mazier D, Fievet MH, Datry A (2014) Emergence of echinocandin-resistant Candida spp. in a hospital setting: a consequence of 10 years of increasing use of antifungal therapy? Eur J Clin Microbiol Infect Dis 33:1489–1496
Khan Z, Ahmad S, Joseph L, Al-Obaid K (2014) Isolation of cholesterol-dependent, multidrug-resistant Candida glabrata strains from blood cultures of a candidemia patient in Kuwait. BMC Infect Dis 14:188
Lewis JS, Wiederhold NP, Wickes BL, Patterson TF, Jorgensen JH (2013) Rapid emergence of echinocandin resistance in Candida glabrata resulting in clinical and microbiologic failure. Antimicrob Agents Chemother 57:4559–4561
Shields RK, Nguyen MH, Press EG, Updike CL, Clancy CJ (2013) Caspofungin MICs correlate with treatment outcomes among patients with Candida glabrata invasive candidiasis and prior echinocandin exposure. Antimicrob Agents Chemother 57:3528–3535
Bizerra FC, Jimenez-Ortigosa C, Souza ACR, Breda GL, Queiroz-Telles F, Perlin DS, Colombo AL (2014) Breakthrough candidemia due to multidrug-resistant Candida glabrata during prophylaxis with a low dose of micafungin. Antimicrob Agents Chemother 58:2438–2440
Bal AM, Shankland GS, Scott G, Imtiaz T, Macaulay R, McGill M (2014) Antifungal step-down therapy based on hospital intravenous to oral switch policy and susceptibility testing in adult patients with candidaemia: a single centre experience. Int J Clin Pract 68:20–27
Tissot F, Lamoth F, Hauser PM, Orasch C, Flückiger U, Siegemund M, Zimmerli S, Calandra T, Bille J, Eggimann P, Marchetti O (2013) Beta-glucan antigenemia anticipates diagnosis of blood culture-negative intraabdominal candidiasis. Am J Respir Crit Care Med 188:1100–1109
Poissy J, Sendid B, Damiens S, Ichi Ishibashi K, François N, Kauv M, Favory R, Mathieu D, Poulain D (2014) Presence of Candida cell wall derived polysaccharides in the sera of intensive care unit patients: relation with candidaemia and Candida colonisation. Crit Care 18:R135
Acknowledgments
Members of the independent scientific committee: Olivier Leroy (MD, medical intensivist), Jean-François Timsit (MD, PhD, medical intensivist, epidemiologist), Jean-Pierre Gangneux (MD, PhD, mycologist), Elie Azoulay (MD, PhD, medical intensivist), Jean-Michel Constantin (MD, PhD, anesthesiologist and intensivist), Hervé Dupont (MD, PhD, anesthesiologist and intensivist), Olivier Lortholary (MD, PhD, infectious diseases specialist), Jean-Paul Mira (MD, PhD, medical intensivist), Philippe Montravers (MD, PhD, anesthesiologist and intensivist), Pierre-François Perrigault (MD, anesthesiologist and intensivist). Manuscript preparation: the authors thank Celine Feger (MD; EMIBiotech), who provided assistance in preparing and editing the manuscript and Romain Pirrachio (MD, PhD; University of California, San Francisco) who reviewed statistical analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by MSD France, which was the sponsor.
Conflicts of interest
Authors had full access to the database, and the statistical analyses were conducted by an academic team: Biostatistics Unit, Lille University Hospital, Lille, France, which worked in full independence from MSD.
EA has been a consultant to Astellas, Alexion, Cubist, Gilead, and MSD, and has benefited from grants to his research unit from Gilead and Pfizer. J-MC has been a consultant to MSD. HD has been a consultant to Astellas, Gilead, Cubist, Astrazeneca, Merck, and Pfizer. J-PG has been a consultant to Astellas, Gilead, Merck, and Pfizer. DG has benefited from grants of the Principality of Monaco to his research unit. O Leroy has been consultant to Astellas, Gilead, Merck, Novartis, Pfizer, and Sanofi. O Lortholary has been consultant to Gilead Sciences and Novartis and member of the speaker’s bureau of Astellas, Basilea, Merck, Pfizer, and Sanofi. J-PM has been a consultant to Astellas, Gilead, MSD, and LFB. PM has been a consultant to Astra-Zeneca, Cubist, MSD, Pfizer, and TMC. P-FP has been a consultant to MSD and Pfizer. JFT has given lectures for symposiums set up by Astellas, Pfizer, MSD, 3M, Novartis, and Gilead; has benefited from unrestricted research grants to his research unit from 3M, MSD, and Astellas; and has been a consultant involved in scientific boards for MSD, 3M, and Bayer. SB has no conflict of interest.
Additional information
Take-home message: Early de-escalation of systemic antifungal treatments is not consensual. A causal analysis based on a multicenter prospective cohort in 87 French ICUs has shown that antifungal de-escalation within a 5-day interval is safe in SAT-treated non-neutropenic adult intensive care unit patients.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bailly, S., Leroy, O., Montravers, P. et al. Antifungal de-escalation was not associated with adverse outcome in critically ill patients treated for invasive candidiasis: post hoc analyses of the AmarCAND2 study data. Intensive Care Med 41, 1931–1940 (2015). https://doi.org/10.1007/s00134-015-4053-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-015-4053-1