Abstract
Purpose
Mechanical complications during assisted ventilation can evolve due to worsening lung disease or problems in airway management. These complications affect lung compliance or airway resistance, which in turn affect the chest wall dynamics. The objective of this study was to explore the utility of continuous monitoring of the symmetry and dynamics of chest wall motion in the early detection of complications during mechanical ventilation.
Methods
The local tidal displacement (TDi) values of each side of the chest and epigastrium were measured by three miniature motion sensors in 18 rabbits. The TDi responses to changes in peak inspiratory pressure (n = 7), induction of one-lung intubation (n = 7), and slowly progressing pneumothorax (PTX) (n = 6) were monitored in parallel with conventional respiratory (SpO2, EtCO2, pressure and flow) and hemodynamic (HR and BP) indices. PTX was induced by injecting air into the pleural space at a rate of 1 mL/min.
Results
A strong correlation (R 2 = 0.99) with a slope close to unity (0.94) was observed between percent change in tidal volume and in TDi. One-lung ventilation was identified by conspicuous asymmetry development between left and right TDis. These indices provided significantly early detection of uneven ventilation during slowly developing PTX (within 12.9 ± 6.6 min of onset, p = 0.02) almost 1 h before the SpO2 dropped (77.3 ± 27.4 min, p = 0.02). Decreases in TDi of the affected side paralleled the progression of PTX.
Conclusions
Monitoring the local TDi is a sensitive method for detecting changes in tidal volume and enables early detection of developing asymmetric ventilation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mechanically ventilated patients are at risk of mechanical complications that can lead to inappropriate ventilation. Mechanical complication can evolve due to worsening lung disease, developing pulmonary complications (e.g., pneumothorax, atelectasis) or problems in airway management (e.g., tube displacement, obstruction by secretions). Premature infants are especially prone to develop respiratory complications while mechanically ventilated. The narrow endotracheal tube can be easily obstructed by secretions, and the risk of tube displacement and one-lung ventilation is high. The incidence of pneumothorax (PTX) in very low birth weight infants (below 1,500 g) is between 3 and 9% [1, 2]. Deteriorating ventilation and hypoxia in premature infants can lead to hemodynamic instability and intracranial hemorrhage [3, 4].
Interestingly, 25–44% of the life-threatening respiratory events in neonatal and pediatric intensive care units (NICU, PICU) are detected by attending staff inspections and not by monitoring equipment [5, 6]. In the particular case of PTX, McIntosh [7, 8] reported that the time to diagnosis in a NICU ranged from 45 to 660 min from the initial onset, with a median of 127 min. Therefore, the currently monitored modalities have two main limitations, due to physiological shortcomings in the monitored parameters. The first is a prolonged delay from the onset of a significant mechanical problem in ventilation to the appearance of detectable changes. The second is their poor ability to characterize the nature and location of the underlying problem out of the various plausible causes.
The working hypothesis of this study is that bilateral, continuous monitoring of the symmetry and dynamics of chest wall motion provides additional crucial information that can expedite the detection of deteriorating ventilation. This method was recently tested in premature newborn infants during high frequency oscillatory ventilation (HFOV) [9]. The present study aimed at exploring the utility of bilateral monitoring of chest wall dynamics in three simulated, relatively common situations during conventional mechanical ventilation: decrease in peak inspiratory pressure (PIP), one-lung intubation, and slowly progressing PTX.
Methods
The experiments were carried in 18 adult male New Zealand rabbits weighing 2.1 ± 0.35 kg. The study was approved by the Institutional Ethics Committee for the Care and Use of Animals. Rabbits were anesthetized by intramuscular injection of xylazine (5 mg/kg), ketamine (35 mg/kg), and acepromazine (1 mL/kg). Additional doses of one-third of the aforementioned amounts were added every 20 min. A 3-mm endotracheal tube was placed by direct laryngoscopy or tracheostomy and conventional intermittent mandatory ventilation (CMV) was instituted (SLE 2000, SLE, Surrey, UK). The ventilatory parameters were adjusted to maintain normal arterial blood gas tensions utilizing a blood gas and electrolyte analyzer (Roche OPTI CCA, Mannheim, Germany). A percutaneous catheter was placed in the ear artery for blood sampling and recording blood pressure (BP). A pulse oximeter was attached to the ear or legs. BP, heart rate (HR), and end-tidal expiratory carbon dioxide concentration (EtCO2) were acquired by a standard system (Datex Ohmeda Inc., Wisconsin, USA). Endotracheal tube pressure (TP) was measured by a pressure transducer (Millar Instruments Inc., Houston, Texas, USA), and tidal volume (V T) by a flow-meter (TSD137, Fleish Pneumotachograph, Harvard Apparatus, Holliston, MA). The experimental setup is described schematically in Fig. 1 in the online supplementary material.
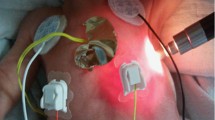
Two miniature (<1 g, 5 mm diameter) motion sensors were attached to both sides of the chest, at the mid-clavicular lines and the fifth or sixth intercostal space, and a third sensor was attached to the epigastrium (Figs. 2 and 3 in the supplementary material). These accelerometers enable one to measure the local subsonic (below 20 Hz) wall motion, as well as the vibrations from the breath and heart sounds, as presented in Fig. 4 in the supplementary material. The data were acquired at a sampling rate of 5 kHz (Pneumedicare system, Pneumedicare, Yokneam, Israel). The sounds’ contributions were filtered. The signals were used to measure the amplitude of the local tidal angular displacement at each site, denoted as the local tidal displacement index (TDi), of the right (TDiR) and left (TDiL) sides of the chest and the epigastrium (TDiE). The ratio of the right to left chest wall displacement (R/L) characterized the ventilation symmetry.
The sensitivity of each sensor was calibrated by utilizing a high resolution (1 μM) computerized lever arm system (Model 310C, Aurora Scientific Inc., ON, Canada). The system imposed oscillations at various known frequencies and amplitudes that were measured by the motion sensors. A linear relationship (R 2 = 0.99) between imposed oscillation amplitudes and measured tidal displacement was found, with small (3.7%) variability in sensitivity between the sensors (Fig. 5 in the supplementary material).
Study protocols
Three types of complications were simulated: (1) reduction in PIP (7 rabbits, 50 events), (2) one-lung intubation (7 rabbits, 20 events), and (3) slowly progressing right PTX (6 rabbits).
The PIP was reduced by 11, 22, 33, and 66%. These reductions were imposed in random order, with every event repeated at least twice per rabbit. One-lung intubation was induced by advancing the endotracheal tube until significant asymmetric ventilation was confirmed by auscultation. Slowly progressing PTX was created by inserting a chest tube (10 Fr) into the right pleural space, in the tenth intercostal space at the mid-axillary line. The incision was sutured, and the tube was connected to a syringe pump (Graseby 3100, SIMS Graseby Ltd., UK) which injected air at a constant rate of 1 mL/min.
Drops in PIP and one-lung intubations were imposed for at least 100 s, or until severe hemodynamic deterioration or hypoxemia (SpO2 < 90%) was observed, before restoring normal ventilation. For PTX, baseline recordings were made for at least 5 min before starting air injection into the pleural space. Injection continued until a persistent decrease in SpO2 (SpO2 < 90%) developed. At this point, the air in the pleural cavity was aspirated in order to measure its volume. The chest was transilluminated and photographed for qualitative control at baseline and twice during the perturbation. Arterial blood gas tensions were measured at baseline and after injecting 40, 60, and 80 mL of air.
Data analysis
Deviation was defined as a 20% change from baseline in TDiR, TDiL, or TDiE, whereas desaturation was defined as a decrease of SpO2 below 90%.
Statistical analysis
Results are presented as mean ± SD, and median and range. Differences between specific data means were compared using the Wilcoxon signed-rank test. Statistical significance was defined as p < 0.05. The linear correlation between TDi, PIP, and V T was evaluated using the Pearson coefficient of mean-square contingency (R 2).
Results
The rabbits (n = 18) were ventilated at a respiratory rate of 19.9 ± 3.2 mL/min, PIP of 16.7 ± 2.0 cmH2O, with a positive end-expiratory pressure (PEEP) of 3.0 cmH2O. Measured V T was 10.91 ± 0.69 mL/kg. Similar respiratory rates and V T were used by other authors [10]. No spontaneous breaths were detected during baseline measurements, as presented in Fig. 7 in the supplementary material.
Changes in PIP
The relative decreases in ΔP, where ΔP = PIP − PEEP, were larger than the relative decreases in PIP, because PEEP was equal to 3 cmH2O. The PIP reductions by 11, 22, 33, and 66% were associated with measured decreases in ΔP of 17.4 ± 2.2, 27.6 ± 0.8, 46.6 ± 3.0, and 82.2 ± 1.9%, respectively.
Typical responses to 33 and 11% decreases in the PIP are displayed in Fig. 6 in the supplementary material. Clear decreases in the TDi were observed within the first or second ventilation following the perturbations, even in the smallest imposed decreases in the PIP.
Figure 1 compiles all the results and presents detection success rates for four reductions in PIP. All the 66% decreases in PIP (13 events) were detected by the TDi as well as by changes in SpO2 and EtCO2. The 33% decreases in the PIP (14 events) were easily detected in all cases solely by the TDi. EtCO2 and SpO2 sensitivities were 78.6 ± 39.3 and 52.4 ± 41.3%, respectively. The 22% (17 events) and 11% (16 events) reductions in PIP were clearly detected in all events only by changes in the TDi (100% sensitivity), and hardly detected by other parameters. The TDi decreased within one or two breaths (after 1.8 ± 4.65 and 2.1 ± 2.71 s for 22 and 11% reductions, respectively). There were significant differences in the time to detection between the various indices. For simplicity, only the times to detection in response to the largest PIP reductions (−66%) are presented (Fig. 1e). Detectable changes in TDi occurred immediately in the first ventilation, followed by detectable decreases in EtCO2 (5.7 ± 6.8 s) and in SpO2 (22.4 ± 5.4 s).
Tidal displacement index (TDi) provided fast response to reduction in the peak inspiratory pressure (PIP). The figure compiles the rate of successful detection of an imposed a 66%, b 33%, c 22%, and d 11% decrease in PIP, as well as e the time to detection of the event by each monitored parameter in the imposed 66% reduction of PIP. Detectable changes in TDi always occurred within one to two breath cycles
Most importantly, changes in the TDi (ΔTDi) were proportional to changes in PIP (R 2 = 0.99) and the resulting reductions in V T. A strong correlation with a slope close to unity (0.99) was observed between the relative changes in ΔP and ΔTDi, as shown in Fig. 2a. A strong correlation with a slope close to unity (0.94) was also obtained between the changes in the tidal volume (ΔVT) and the changes in ΔTDi (Fig. 2b). A decrease of 33% in PIP yielded a 46.6 ± 3.0% decrease in ΔP, a 36.8 ± 3.5% decrease in V T and a 43.9 ± 9.3% decrease in TDi.
One-lung intubation
Figure 3 displays typically observed responses, characterized by a marked and rapid decrease in TDiL and an increase in R/L. Thereafter, the EtCO2 and SpO2 decreased, later followed by mild changes in HR and BP. Figure 4 compiles the results of one-lung intubation (7 rabbits). Sensitivity in detecting asymmetric ventilation (R/L) and decrease in the left lung ventilation (TDiL) by the motion sensors was 100%. The perturbations were detected within 8.8 ± 9.16 s by the changes in TDi and R/L indices. The time to detection using TDi was significantly shorter than for the other indices (P < 0.01).
Right-side PTX
Figure 5 represents one experiment of slowly progressing right PTX. A progressive decrease in TDiR was observed with the onset of PTX. TDiR decreased by 20% within 15.6 min of initiating the slow air injection into the right pleural space, and a prominent (20%) change in asymmetric ventilation was observed, as reflected by the R/L index after 13.1 min. Mean arterial blood pressure (MABP) decreased after 21.2 min. Interestingly, a decline in the SpO2 appeared at a very late stage: only after 69.0 min.
Figure 6 summarizes the sensitivity and the time to detection by the various indices (6 rabbits). The motion sensors easily detected all PTXs (100% sensitivity) as early as 12.9 ± 6.6 min after the onset of the slow (1 mL/min) air injection, and provided the first sign for deteriorating ventilation in all cases. Deteriorating ventilation was detected by both the decrease in TDiR on the affected right side and by the development of asymmetric ventilation. The significant (20%) decrease in BP was also very sensitive (100%), but appeared later (after 35.0 ± 23.9 min). The mean time until desaturation occurred was very long, after 77.3 ± 27.4 min. Hemodynamic changes preceded the changes in gas-exchange indices, i.e., EtCO2 and SpO2 and arterial blood gas tensions. The blood gas analyses are shown in Table 1.
Discussion
Continuous monitoring of chest wall dynamics presents the following three clinical advantages: (a) high sensitivity to changes in the tidal volume and precise quantification of the severity of these changes, as demonstrated by the strong correlation between the decrease in TDi and tidal volume; (b) assessment of ventilation symmetry, as demonstrated with one-lung intubation and PTX; and (c) early detection of deteriorating ventilation, as demonstrated by the early response to the induction of slowly progressing PTX.
The sensitivity of the TDi to changes in the ventilation was tested by the imposed reductions in PIP. All events, including mild decreases in PIP, were detected. The changes in TDi also detected all one-lung intubations and PTX events. PTX was detected when a relatively small amount of air (12.9 ± 6.6 mL) was injected into the pleural space. Changes in TDi preceded the changes in all other monitored parameters because the motion sensors, which are attached to the chest wall, directly monitor the changes in lung ventilation. The other physiological indices are consequential measures of changes in ventilation, which are influenced by various compensatory mechanisms.
The TDi also allows one to assess the severity of the ventilation changes. Decreases in TDi were proportional to decreases in inspiratory pressure and in the tidal volume. Furthermore, there was a progressive decrease in TDi with time during slowly progressive PTX as the amount of intrapleural air increased.
Assessment of ventilation symmetry is of particular importance. This crucial parameter can facilitate the diagnosis and localization of the underlying cause. The information can be used to differentiate between mechanical problems in the central airways, and problems below the bifurcation of the two main bronchi. The former yields symmetrical changes in the monitored TDiR and TDiL, whereas the latter produces asymmetrical changes. Tension PTX is usually detected in the NICU by a decrease in SpO2 and blood pH, and increase in PaCO2. However, these changes appear after a prolonged delay, after PTX reaches a critical mass [7, 8, 11]. It is instructive to note that changes in SpO2 were the last to occur in our study, whereas the TDI and R/L indices were able to identify the gradual development of the PTX even in its initial phase.
Numerous studies have explored the use of auscultation for ventilation monitoring, based on the characterization of breath sound patterns in various diseases [12–14]. However, the analysis of breath sounds and comparison with sounds in databanks have inherent limitations because human breath sounds vary among different individuals [14–17]. The method described here detects changes in ventilation dynamics relative to the individual’s baseline measurements. Furthermore, it characterizes the subsonic chest wall dynamics and does not use the breath sounds.
Simultaneous auscultation at multiple sites by large arrays of sensors over the chest has been suggested as a method for creating three-dimensional images of air flow through the lungs [18]. However, the use of large arrays of microphones for prolonged monitoring is cumbersome and inconvenient in the NICU setting.
Endotracheal tube air flow and pressure can be continuously measured in new ventilators [19, 20]. There are several explanations for the prolonged delay in the detection of complications [5–8] despite the close monitoring of the endotracheal tube flow and pressure. Monitoring air flows with sensors at the endotracheal tube inlet is a method insensitive to small differences between the inhaled and exhaled volumes, which gradually accumulate into large intrathoracic lesions, such as in PTX. A 1% decrease in the exhaled volume due to a leak into the pleural space cannot be detected today. However, this small air leak can develop into a tension PTX within 30 min. Measurements of endotracheal tube flow and pressure are unable to assess the symmetry of ventilation—an early sign of PTX. Moreover, the high resistance to air flow in the narrow endotracheal tube, used in pediatric patients, limits the ability to sense progressive changes in lung compliance and resistance.
Several noninvasive modalities have been developed for assessing respiratory mechanics in infants, such as respiratory inductance plethysmography [21–24], elastomeric plethysmography, optic respiratory plethysmography [25], and electrical impedance tomography (EIT) [26, 27]. The plethysmographic modalities are not easy to use for continuous monitoring in the setting of a NICU because they utilize belts that must be tight and encircle the chest and abdomen, and are bulky [22]. These plethysmographic techniques may chafe the skin, especially in the more immature of patients. None of these modalities are presently used routinely in the clinical NICU setup. The suggested continuous TDi monitoring was recently tested in premature infants during high frequency oscillatory ventilation [9]. Monitoring the deviations in TDi enabled the detection of deteriorating ventilation 22.4 ± 18.7 min before the obstruction by secretions evolved into hypoxemia requiring treatment [9].
Limitations
Measurement of TDi cannot accurately assess the absolute tidal volume. A strong correlation between the changes in the TDi and V T was presented. However, assessing the absolute V T from the TDi requires complex calibration. The aim of this new method is to monitor changes in the symmetry and dynamics of lung ventilation, rather than assessing the absolute V T.
The recorded chest wall dynamics in patients is more complex because the breathing pattern in small babies is rather irregular, with small tidal volumes, and includes sporadic body movements. In this preclinical study the breathing pattern during CMV was quite regular and no significant motion artifacts were encountered. We have validated that the system response is not affected by the breathing frequency in vitro (Fig. 5 in the supplementary material). The feasibility of the system in newborn infants ventilated with HFOV, where the signal amplitude is very small, has been tested [9]. The effects of irregular spontaneous breathing and motion artifact should be tested in a clinical study.
Detection of asymmetric ventilation will be limited in adults with a stiff chest. The study highlights that significant information is available from monitoring a compliant chest’s wall dynamics. Under relaxed conditions the chest wall compliance is larger than lung compliance in neonates and infants [28, 29].
Conclusions
Continuous monitoring of the symmetry and dynamics of the local chest wall displacement (TDi) by miniature body-surface sensors has the potential to provide early detection of complications during mechanical ventilation. The decrease in TDi is proportional to the severity of the decrease in ventilation during PIP reductions and PTX. Uneven ventilation due to one-lung intubation and PTX can be detected when the chest is flexible. The TDi provided early detection of PTX significantly (almost 1 h) before SpO2 dropped. The potential clinical utility in pediatric patients receiving respiratory support, in the presence of spontaneous breathing and body motions, should be explored in a clinical study.
References
Horbar JD, Badger GJ, Carpenter JH, Fanaroff AA, Kilpatrick S, LaCorte M, Phibbs R, Soll RF, for the Members of the Vermont Oxford Network (2002) Trends in mortality and morbidity for very low birth infants 1991–1999. Pediatrics 110:143–151
Morley CJ, Davis PG, Doyle LW, Brion LP, Hascoet JM, COIN Carlin JB, Investigators Trial (2008) Nasal CPAP or intubation at birth for very preterm infants. N Engl J Med 14:700–708
Linder N, Haskin O (2003) Risk factors for intraventricular hemorrhage in very low birth weight premature infants: a retrospective case-control study. Pediatrics 111:e590–e595
Gammon CM, Wiswell TE, Spitzer AR (1998) Volutrauma, PaCO2 levels, and neuro-developmental sequelae following assisted ventilation. Clin Perinatol 25:75–159
Frey B, Kehrer B, Losa M, Braun H, Berweger L, Micallef J, Ebenberger M (2000) Comprehensive critical incident monitoring in a neonatal-pediatric intensive care unit: experience with the system approach. Intensive Care Med 26:69–74
Szekely SM, Webb RK, Williamson JA, Russell WJ (1993) The Australian Incident Monitoring Study. Problems related to the endotracheal tube: an analysis of 2000 incident reports. Anaesth Intensive Care 21:611–616
McIntosh N (2002) Intensive care monitoring: past, present and future. Clin Med 2:349–355
McIntosh N, Becher JC, Cunningham S, Stenson B, Laing IA, Lyon AJ, Badger P (2000) Clinical diagnosis of pneumothorax is late: use of trend data and decision support might allow preclinical detection. Pediatr Res 48:408–415
Waisman D, Levy C, Faingersh A, Klotzman FIC, Konyukhov E, Kessel I, Rotschild A, Landesberg A (2011) A new method for continuous monitoring of the chest wall movement to characterize hypoxemic episodes during HFOV. Intensive Care Med 37:1174–1181
Piccin VS, Calciolari C, Yoshizaki K, Gomes S, Albertini-Yagi C, Dolhnikoff M, Macchione M, Caldini EG, Saldiva PH, Negri EM (2011) Effects of different mechanical ventilation strategies on the mucociliary system. Intensive Care Med 37:132–140
Watkinson M, Tiron I (2001) Events before the diagnosis of a pneumothorax in ventilated neonates. Arch Dis Child Fetal Neonatal Ed 85:F201–F203
Kiyokawa H, Pasterkamp H (2002) Volume-dependent variations of regional lung sound, amplitude, and phase. J Appl Physiol 93:1030–1038
Gross V, Dittmar A, Penzel T, Schüttler F, von Wichert P (2000) The relationship between normal lung sounds, age, and gender. Am J Respir Crit Care Med 162:905–909
Royston TJ, Mansy HA (2002) Modeling sound transmission through the pulmonary system and chest with application to diagnosis of a collapsed lung. J Acoust Soc Am 111:1931–1946
Munakata M, Ukita H, Doi I, Ohtsuka Y, Masaki Y, Homma Y, Kawakami Y (1991) Spectral and waveform characteristics of fine and coarse crackles. Thorax 46:651–657
Kraman SS, Wodicka GR, Pressler GA, Pasterkamp H (2006) Comparison of lung sound transducers using a bioacoustic transducer testing system. J Appl Physiol 101:469–476
Kraman SS, Pressler GA, Pasterkamp H, Wodicka GR (2006) Design, construction, and evaluation of a bioacoustic transducer testing (BATT) system for respiratory sounds. IEEE Trans Biomed Eng 53:1711–1715
Cinel I, Jean S, Tay C, Gratz I, Deal E, Parrillo JE, Dellinger RP (2008) Case report: vibration response imaging findings following inadvertent esophageal intubation. Can J Anesth 55:172–176
Cannon ML, Cornell J, Tripp-Hamel DS, Gentile MA, Hubble CL, Meliones JN, Cheifetz IM (2000) Tidal volumes for ventilated infants should be determined with a pneumotachometer placed at the endotracheal tube. Am J Respir Crit Care Med 162:2109–2012
Keszler M (2009) State of the art in conventional mechanical ventilation. J Perinatol 29:262–275
Tingay DG, Mills JF, Morley CJ, Pellicano A, Dargaville PA (2006) The deflation limb of the pressure-volume relationship in infants during high-frequency ventilation. Am J Respir Crit Care Med 173:414–420
Musante G, Schulze A, Gerhardt T, Everett R, Claure N, Schaller P, Bancalari E (2001) Proportional assist ventilation decreases thoracoabdominal asynchrony and chest wall distortion in preterm infants. Pediatr Res 49:175–180
Gappa M, Pillow JJ, Allen J, Mayer O, Stocks J (2006) Lung function tests in neonates and infants with chronic lung disease: lung and chest-wall mechanics. Pediatr Pulmonol 41:291–317
Copnell B, Tingay DG, Kiraly NJ, Sourial M, Gordon MJ, Mills JF, Morley CJ, Dargaville PA (2007) Effects of open endotracheal suction on lung volume in infants receiving HFOV. Intensive Care Med 33:689–693
Davis C, Mazzolini A, Mills J, Dargaville P (1999) A new sensor for monitoring chest wall motion during high-frequency oscillatory ventilation. Med Eng Phys 21:619–623
Dunlop S, Hough J, Riedel T, Fraser JF, Dunster K, Schibler A (2006) Electrical impedance tomography in extremely prematurely born infants and during high frequency oscillatory ventilation analyzed in the frequency domain. Physiol Meas 27:1151–1165
Frerichs I, Dargaville PA, van Genderingen H, Morel DR, Rimensberger PC (2006) Lung volume recruitment after surfactant administration modifies spatial distribution of ventilation. Am J Respir Crit Care Med 174:772–779
Colin AA, McEvoy C, Castile RG (2010) Respiratory morbidity and lung function in preterm infants of 32 to 36 weeks’ gestational age. Pediatrics 126:115–128
Papastamelos C, Panitch HB, England SE, Allen JL (1995) Developmental changes in chest wall compliance in infancy and early childhood. J Appl Physiol 78:179–184
Acknowledgments
This work was supported in part by the Dr. Y. Rabinovitz Research Fund and the Ruth and Allen Ziegler Fund for Pediatric Research (DW), and by a grant for the promotion of research at Technion (AL). Chest wall dynamics were measured by the equipment provided by Pneumedicare Ltd. The authors thank Idit Lavi, MA, for her kind advice on the statistical analysis; and Prof. Eduardo Bancalari for his critical review of our work.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is discussed in the editorial available at: doi:10.1007/s00134-011-2399-6.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Waisman, D., Faingersh, A., Levy, C. et al. Early detection of deteriorating ventilation by monitoring bilateral chest wall dynamics in the rabbit. Intensive Care Med 38, 120–127 (2012). https://doi.org/10.1007/s00134-011-2398-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-011-2398-7