Abstract
Introduction
Corticosteroids have been proposed to decrease morbidity and mortality in patients with septic shock. An impact on morbidity should be anticipated to be earlier and more easily detected than the impact on mortality.
Methods
Prospective, randomized, double-blind, placebo-controlled study of 28-day mortality in patients with septic shock for <72 h who underwent a short high-dose ACTH test in 52 centers in 9 European countries. Patients received 11-day treatment with hydrocortisone or placebo. Organ dysfunction/failure was quantified by the use of the sequential organ failure assessment (SOFA) score.
Results
From March 2002 to November 2005, 499 patients were enrolled (hydrocortisone 251, placebo 248). Both groups presented a similar SOFA score at baseline (hydrocortisone 10.8 ± 3.2 vs. placebo 10.7 ± 3.1 points). There was no difference in 28-day mortality between the two treatment groups (hydrocortisone 34.3% vs. placebo 31.5%). There was a decrease in the SOFA score of hydrocortisone-treated patients from day 0 to day 7 compared to the placebo-treated patients (p = 0.0027), driven by an improvement in cardiovascular organ dysfunction/failure (p = 0.0005) and in liver failure (p < 0.0001) in the hydrocortisone-treated patients.
Conclusion
Patients randomized to treatment with hydrocortisone demonstrated a faster decrease in total organ dysfunction/failure determined by the SOFA score, primarily driven by a faster improvement in cardiovascular organ dysfunction/failure. This organ dysfunction/failure improvement was not accompanied by a decreased mortality.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The use of 28-day mortality has constituted the most commonly chosen endpoint in sepsis studies. Mortality is easy to define and measure, and represents a clinically very relevant endpoint. Some authors, such as Petros, have questioned the adequacy of all-cause mortality as an endpoint [1]. In the case of sepsis and multiple organ failure, studies require large numbers of patients with their associated costs. Patients in intensive care, even with strict inclusion criteria for sepsis or septic shock, do not constitute a homogeneous population. Patients are heterogeneous with different diagnoses, time courses, ages, co-morbidities, sites of infection and invading microorganisms. In addition, patients have different degrees of physiological dysfunction resulting in diverse mortality risks that are difficult to adjust for post-hoc by general severity scores such as the APACHE II [2] or the SAPS II [3]. Only one multicenter randomized clinical trial in patients with severe sepsis and septic shock demonstrated a decreased 28-day all-cause mortality [4]. The study also showed an improved morbidity evidenced by decreases in Sequential Organ Failure (SOFA) scores [5]. Despite this fact the controversy over the appropriate endpoint for clinical trials continues [6]. Morbidity and organ failure-free days in addition to mortality have recently been proposed as endpoints [7]. The resolution of organ failure may represent a reasonable outcome because it results in a reduction in morbidity with less need for life support [8] and perhaps even costs [9].
The Corticosteroid Therapy of Septic Shock (CORTICUS) study’s primary endpoint was 28-day all-cause mortality in corticotropin non-responders. The analysis of the presence, amount and evolution of organ failure was a pre-planned secondary outcome. The hypothesis was that patients treated with hydrocortisone when compared to placebo would have a faster resolution of organ dysfunction/failure. The objective of this paper is to present the results of this analysis and to discuss their implications.
Materials and methods
Study design
The CORTICUS study was a multicenter, randomized, double-blind, placebo-controlled study of hydrocortisone therapy in patients with septic shock in 52 intensive care units (ICUs) [10]. This article reports a preplanned analysis of the presence, amount and evolution of organ dysfunction.
Study population
Patients were enrolled from March 2002 until 30 November 2005. All patients 18 years of age or above were prospectively enrolled in the study if they met all eligibility criteria including: (1) clinical evidence of infection, (2) evidence of a systemic response to infection, (3) evidence of shock within the previous 72 h defined by a systolic blood pressure (SBP) <90 mmHg despite adequate fluid replacement OR need for vasopressors for at least 1 h, (4) hypoperfusion or organ dysfunction attributable to sepsis and (5) informed consent according to local regulations. Main exclusion criteria included underlying disease with a poor prognosis, immunosuppression and prior administration of corticosteroids [7].
Study treatment
The study drug was administered as a 50-mg intravenous bolus every 6 h for 5 days, then tapered to 50 mg intravenously every 12 h for days 6–8, 50 mg every 24 h for days 9–11 and then stopped. In the control group, a matching placebo was used at similar times.
Data collection
Patient data included: (1) general characteristics including demographics, diagnoses and recent surgery, (2) severity of illness assessed by vital signs, Simplified Acute Physiology Score (SAPS) II [11], and (3) interventions including type and doses of vasopressors. Laboratory variables included hematological, chemistry and blood gas determinations. Cultures of blood and other potential sites of infection were recorded. A short corticotrophin (ACTH) test was performed using blood samples taken immediately before and 60 min after an intravenous bolus of 0.25 mg tetracosactrin (Novartis, Nuremberg, Germany or Alliance, Chippenham, UK).
During the 28-day period post-randomization, data were collected for vital signs, laboratory results, cultures and any major intervention. Mortality at 28 days was recorded.
Definitions
Organ system dysfunction/failure was assessed by the Sequential Organ Failure Assessment (SOFA) score [12], and computed at study baseline (day 0) and at days 1–7, 14 and 28. A score of 1 or 2 points in each of the six organ/systems was considered as evidence of organ dysfunction, and a score of 3 or 4 points was considered as evidence of organ failure. Organ failure reversal was defined as a score or sub-score below 3 in patients with an initial score of ≥3. Maximum and delta SOFA scores were calculated as described previously [13]. Reversal of shock was defined as the maintenance of a SBP ≥90 mmHg without vasopressor support for ≥24 h. A new septic shock episode was defined as a new episode of septic shock after reversal of the initial septic shock. Non-responders to the corticotropin test were defined by a cortisol increase ≤9 μg/dl.
Study outcomes
The main outcome of this trial was all-cause mortality at day 28. This specific study targeted the secondary endpoint of organ system failure reversal for each organ, especially shock.
Statistical analysis
All analyses were performed according to a pre-established plan. The population was analyzed by an “intention to treat” principle. All results of organ dysfunction/failure are presented as mean ± standard deviation with minimum and maximum values indicated by brackets.
Since data were gathered over time on the same patient, mixed effects models that are appropriate for clustered and dependent data were used to study the relationship between the treatment arms and the course of SOFA scores [14]. SOFA analyses were restricted to day 0–7 measurements since no consecutive daily data were available thereafter. Normal distributions and a linear relationship with time were assumed. To test for time by treatment interaction on the SOFA components (measured on categorical scales ranging from 0 to 4), multinomial regression models were used [15]. To assess the underlying assumption of randomly missing data, differences in available data were checked over time across randomized groups by generalized linear models with binomial link. Finally, to account for the potential competing risks of death in the ICU, the effect of treatment was assessed on the cumulative incidence of organ failure reversal, taking into account death prior to resolution as a competing event; cumulative incidence curves were then compared by the Gray test. All tests and p-values presented were two-sided. All statistical analyses and model fits were based on standard statistical packages (R and SAS).
Results
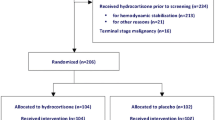
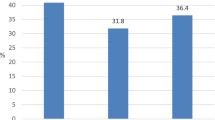
During the study period, 499 patients were analyzed (251 in the hydrocortisone group and 248 in the placebo group). At baseline, the two groups were well balanced for demographics, clinical characteristics, the type and site of infection, and infecting microorganisms as previously reported [10]. There were 233 (46.7%) corticotropin nonresponders (hydrocortisone 125; placebo 108) and 254 (50.9%) responders (hydrocortisone 118; placebo 136). As reported previously, there was no difference in 28-day mortality between patients assigned to hydrocortisone or placebo, respectively, in the overall population (34.3 vs. 31.5%) or in patients responding (28.8 vs. 28.7%) or not responding to corticotropin (39.2 vs. 36.1%) [7].
The course of the total SOFA score over the first week in the two treatment groups is displayed in Fig. 1. No evidence of any difference between available data across randomized groups was observed (data not shown). The total SOFA score was similar at baseline in both groups [hydrocortisone: 10.8 ± 3.2 (4–21); placebo: 10.7 ± 3.1 (3–21); p = 0.55; Table 1]. Thereafter, a significant time effect was observed (p < 0.0001) together with a time by treatment interaction (p = 0.0025). The rate of decrease in SOFA score from day 0 to day 7 in the placebo group was approximately 75% of that observed in the hydrocortisone group. The hydrocortisone patients had a greater and faster decrease in the cardiovascular component (p < 0.0001) as well as the liver component (p < 0.0001) (Fig. 2). Hydrocortisone-treated patients were more likely to become vasopressor-free (having a SOFA sub-score of 0 or 1) (p = 0.0024) with a higher mean number of vasopressor-free days in the first 7 days (2.5 ± 2.4) compared to the placebo group (1.4 ± 2.4) (p < 0.0001). In addition, hydrocortisone-treated patients more rapidly reached bilirubin levels below 6 mg/dl as compared to those in the placebo group (p = 0.0006). There were no differences over time between the two treatment groups for the coagulation (p = 0.95), renal (p = 0.76), or central nervous system components (p = 0.34). There were no differences in the pulmonary component between the treatment groups in the evolution over time of PaO2/FiO2 (p = 0.74) or the numbers of ventilator-free days during the first 7 days (0.5 ± 2.1 days in the hydrocortisone group vs. 0.5 ± 2.5 days in the placebo group, p = 0.27).
The subsets of patients with initial organ failures were evaluated taking into account those deaths priors to organ failure resolution within the 28 days as competing events. As shown in Table 2, the cumulative incidence of resolution of cardiovascular failure was shortened in the hydrocortisone-treated patients, with 67.1% with organ failure reversal at day 28 versus 58.2% in the placebo-treated patients (p = 0.041 by the Gray test; Fig. 3a), with no evidence of an increased mortality prior to the resolution (p = 0.48). This was similarly observed for renal failure, with 60.5% of organ failure reversal by day 28 in the hydrocortisone-treated patients as compared to 44.3% in the placebo-treated patients (p = 0.039; Fig. 3b).
Although the proportion of shock reversals was similar in non-responders [96/125 (76.8%) hydrocortisone, 76/108 (70.4%) placebo, p = 0.34]; responders [100/118 (84.7%) hydrocortisone, 105/136 (77.2%) placebo, p = 0.17] or all patients [202/251 (80.5%) hydrocortisone, 185/248 (74.6%) placebo, p = 0.14], the time for the cardiovascular component of the SOFA score was significantly shorter in patients receiving hydrocortisone, for the overall group (p = 0.003), responders (p = 0.003) and nonresponders (p < 0.05). No consistent pattern was seen regarding other components of the SOFA score (ESM, Tables E1–E6). There were no differences in the 234 hydrocortisone-treated patients compared to the 232 patients receiving placebo for adverse events including stroke (3 vs. 1), acute myocardial infarction (14 vs. 13) and peripheral limb ischemia (0 vs. 1), nor in the 28-day mortality from multiple system organ failure [41 of 86 (48%) vs. 38 of 78 (49%)], respectively. No patient had a severe adverse event with bowel infarction.
Discussion
Hydrocortisone treatment failed to improve 28-day mortality in the CORTICUS study for all patients or for non-responders and responders to ACTH, but did improve organ function as reflected by a faster decrease in SOFA scores. The cardiovascular and liver effects made the greatest contributions to decreasing the total SOFA score. This is in line with earlier reports [16].
Although there was no difference in overall shock reversal, patients receiving hydrocortisone reversed their shock faster than patients receiving placebo because of faster weaning of vasopressor support. This more rapid shock reversal with steroid therapy is consistent with previous reports [16–19]. This may be related to the fact that hydrocortisone can improve the hemodynamic response to noradrenaline, an effect independent of adrenal insufficiency [20], which seems dependent on the severity of illness as recently demonstrated by Minneci et al. [21]. The improvement in liver function with a decrease in bilirubin in the hydrocortisone-treated patients was probably related to the improved hemodynamics, but is also consistent with previous reports of improvement in bilirubin clearance with hydrocortisone after liver resection [22]. Of note, in patients with acute renal failure at baseline, hydrocortisone accelerated the recovery of renal function.
Although resolution of organ failure might be a more appropriate outcome for ICU studies, it should occur only when there is a consistent reversal of several organ systems with a concomitant trend (even if nonsignificant) towards an improved mortality. Unfortunately, this was not the case in this study. There was no consistent reversal of several organ systems, and if anything the mortality did not decrease but increased, albeit nonsignificantly.
Strengths of this study include the use of data from a prospective, randomized, controlled, multicenter study with analysis of a pre-specified secondary outcome. Limitations include the fact that the study was underpowered and had slow recruitment; organ system data collection did not occur during all 28 study days, and this is a substudy of the original study [10]. Thus, time was modeled by treatment interaction in SOFA scores and sub-scores by using mixed effects models that allow the analysis of such incomplete longitudinal data. This required the assumption of data missing at random, which was checked by testing for time by treatment interaction in the available data. There was no evidence of any difference in the frequency with which data were missing over time between randomized groups. However, deaths during study days 0–7 were first considered as non-informative dropouts in the analysis of SOFA course over time. This could be an issue when dealing with the whole longitudinal process, and thus, the analysis of a competing risks model of resolution of organ failure and prior death within the first 28 days is also reported. This analysis showed a delayed resolution of cardiovascular failure in the placebo arm as compared to the hydrocortisone arm (p = 0.04), with no evidence of increased mortality prior to the resolution (p = 0.48). There are also limitations in the use of the SOFA score in evaluating clinical trials. For each organ the parameters used may not be indicative of all of that organ’s function. For respiratory function positive end-expiratory pressure is not included, and for the cardiovascular system treatment-related adrenergic support is included. Despite these limitations, the SOFA score is the most commonly used organ dysfunction/failure score in practice [5].
The question arises as to whether hydrocortisone should be used to reverse septic shock earlier to replace vasopressor therapy despite the fact that the steroids do not improve overall survival. Although physicians will evaluate the risks and benefits of vasopressor versus steroid therapy for each individual patient, the present guidelines [23] recommend giving hydrocortisone only to the patients “in septic shock after blood pressure is identified to be poorly responsive to fluid and vasopressor therapy” as steroids reversed shock and improved survival only in refractory septic shock patients [17], but not in the Corticus study. Although there are doctors who will continue to use steroids for patients with vasopressor responsive (not refractory) septic shock to hasten shock reversal, this practice is not consistent with the current guidelines [23]. The faster reversal of shock in patients receiving hydrocortisone did not improve survival and was associated with more superinfections and the occurrence of new sepsis and septic shock episodes [10]. Although continued therapy with vasopressors can lead to complications, the present study did not demonstrate any evidence of increased bowel infarction or a greater mortality from multiple organ failure in the placebo group who received vasopressor therapy for longer periods of time. Therefore, the danger of superinfections and new sepsis appears to outweigh that of continued vasopressor therapy. On the basis of these findings, hydrocortisone cannot be recommended as a general adjuvant therapy for vasopressor responsive septic shock, even though they hasten shock reversal.
References
Petros AJ, Marshall JC, van-Saene HK (1995) Should morbidity replace mortality as an endpoint for clinical trials in intensive care? Lancet 345:369–371
Knaus WA, Wagner DP, Harrell FE, Draper EA (1994) What determines prognosis in sepsis? Evidence for a comprehensive individual patient risk assessment approach to the design and analysis of clinical trials. In: Reinhart K, Eyrich K, Sprung C (eds) Sepsis current perspectives in pathophysiology and therapy. Springer-Verlag, Berlin Heidelberg, pp 23–37
Le Gall J-R, Lemeshow S, Leleu G, Klar J, Huillard J, Rué M, Teres D, Artigas A (1995) Customized probability models for early severe sepsis in adult intensive care patients. JAMA 273:644–650
Bernard GR, Vincent J-L, Laterre P-F, LaRosa SP, Dhainaut J-F, Lopez-Rodriguez A, Steingrub JS, Garber GE, Helterbrand JD, Ely EW, Fisher CD Jr, for the Recombinant Human Activated Protein C Worldwide Evaluation in Severe Sepsis (PROWESS) Study Group (2001) Efficacy and safety of recombinant human activated protein C for severe sepsis. N Engl J Med 344:699–709
Vincent J-L, Angus DC, Artigas A, Kalil A, Basson BR, Jamal HH, Johnson G III, Bernard GR, for the Recombinant Human Activated Protein C Worldwide Evaluation in Severe Sepsis (PROWESS) Study Group (2003) Effects of drotrecogin alfa (activated) on organ dysfunction in the PROWESS trial. Crit Care Med 31:834–840
Carlet J (2006) Prescribing indications based on successful clinical trials in sepsis: a difficult exercise. Crit Care Med 34:525–529
Marshall JC, Vincent JL, Guyatt G, Angus DC, Abraham E, Bernard G, Bombardier C, Calandra T, Jørgensen HS, Sylvester R, Boers M (2005) Outcome measures for clinical research in sepsis: A report of the 2nd Cambridge Colloquium of the international sepsis forum. Crit Care Med 33:1708–1716
The Acute Respiratory Distress Syndrome Network (2000) Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med 342:1301–1308
Lilja HE, Leppäniemi A, Kemppainen E (2008) Utilization of intensive care unit resources in severe acute pancreatitis. JOP 8:179–184
Sprung CL, Annane D, Keh D, Moreno R, Singer M, Freivogel K, Weiss YG, Benbenishty J, Kalenka A, Forst H, Laterre P-F, Reinhart K, Cuthbertson BH, Payen D, Briegel J, for the CORTICUS Study Group (2008) Hydrocortisone Therapy for Patients with Septic Shock. N Engl J Med 358:111–124
Le Gall JR, Lemeshow S, Saulnier F (1993) A new simplified acute physiology score (SAPS II) based on a European/North American multicenter study. JAMA 270:2957–2963
Vincent J-L, Moreno R, Takala J, Willats S, De Mendonça A, Bruining H, Reinhart CK, Suter PM, Thijs LG (1996) The SOFA (Sepsis-related organ failure assessment) score to describe organ dysfunction/failure. Intensive Care Med 22:707–710
Moreno R, Vincent J-L, Matos R, Mendonça A, Cantraine F, Thijs L, Takala J, Sprung C, Antonelli M, Bruining H, Willatts S, on behalf of the Working Group on “Sepsis-related problems” of the European Society of Intensive Care Medicine (1999) The use of maximum SOFA score to quantify organ dysfunction/failure in intensive care. Results of a prospective, multicentre study. Intensive Care Med 25:686–696
Laird NM, Ware JH (1982) Random-effects models for longitudinal data. Biometrics 38(4):963–974
Lipsitz SH, Kim K, Zhao L (1994) Analysis of repeated categorical data using generalized estimating equations. Stat Med 13:1149–1163
Briegel J, Forst H, Haller M, Schelling G, Kilger E, JKuprat G, Hemmer B, Lenhart A, Heyduck M, Stoll C, Peter K (1999) Stress doses of hydrocortisone reverse hyperdynamic septic shock: a prospective, randomized, double-blind, single-center study. Crit Care Med 27:723–732
Annane D, Sébille V, Charpentier C, Bollaert PE, François B, Korach J-M, Capellier G, Cohen Y, Azoulay E, Troché G, Chaumet-Riffaut P, Bellissant E (2002) Effect of treatment with low doses of hydrocortisone and fludrocortisone on mortality in patients with septic shock. JAMA 288:862–871
Bollaert PE (2000) Stress doses of glucocorticoids in catecholamine dependency: a new therapy for a new syndrome? Intensive Care Med 26:3–5
Keh D, Boehnke T, Weber-Cartens S, Schulz C, Ahlers O, Bercker S, Volk H-D, Doecke W-D, Falke KJ, Gerlach H (2003) Immunologic and hemodynamic effects of “Low-Dose” hydrocortisone in septic shock. A double-blind, randomized, placebo-controlled, crossover study. Am J Respir Crit Care Med 167:512–520
Saito T, Takanashi M, Gallagher E, Fuse A, Suzaki S, Inagaki O, Yamada K, Ogawa R (1995) Corticosteroid effect on early beta-adrenergic down-regulation during circulatory shock: hemodynamic study and beta-adrenergic receptor assay. Intensive Care Med 21:204–210
Minneci PC, Deans KJ, Banks SM, Eichacker PQ, Natanson C (2004) Meta-Analysis: the effect of steroids on survival and shock during sepsis depends on the dose. Ann Intern Med 141:47–56
Hayashi Y, Takayama T, Yamazaki S, Moriguchi M, Ohkubo T, Nakayama H, Higaki T (2011) Validation of perioperative steroids administration in liver resection: a randomized controlled trial. Ann Surg 253:50–55
Dellinger RP, Levy MM, Carlet JM, Bion J, Parker MM, Jaeschke R, Reinhart K, Angus DC, Brun-Buisson C, Beale R, Calandra T, Dhainaut J-F, Gerlach H, Harvey M, Marini JJ, Marshall J, Ranieri M, Ramsay G, Sevransky J, Thompson BT, Townsend S, Vender JS, Zimmerman JL, Vincent J-L (2008) Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2008. Intensive Care Med 34:17–60
Acknowledgments
The CORTICUS study was supported by the European Commission contract QLK2-CT-2000-00589, the European Society of Intensive Care Medicine (ESICM), the International Sepsis Forum (ISF) and the Gorham Foundation. Roche Diagnostics GmbH, Mannheim/Penzberg, Germany, provided the Elecsys® Cortisol immunoassay. The EU Commission and other sponsors had no role in the design and conduct of the study; collection, management, analysis and interpretation of the data; or in the preparation, review or approval of the manuscript. Thanks also to Prof. Dr. Wolfgang Hartl for suggestions that greatly improved the quality of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
On behalf of the CORTICUS investigators
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix
Appendix
Steering Committee: C. Sprung (Chairman); D. Annane; J. Briegel; D. Keh; R. Moreno; D. Pittet; M. Singer; Y. Weiss.
Safety and Efficacy Monitoring Committee: J. Cohen (Chairman); C. Dore; T. Evans; N. Soni, F. Sorenson (Analytica International).
Study Coordinating Center: C. Sprung (Physician Coordinator); J. Benbenishty (Nurse Coordinator); A. Avidan, E. Ludmir; J. Kabiri; K. Furmanov; B. Hain; O. Kalugin; I. Zack.
Clinical Evaluation Committee: Y Weiss (Chairman); D. Annane; J. Briegel; S. Goodman; D. Keh; R. Moreno; M. Singer; C. Sprung
Berlin Coordinating Center: D. Keh (Chairman); A. Goessinger.
French Coordinating Center: D. Annane (Chairman); N. Zinsou, D. Friedman.
Munich Central Laboratory Harmonization: J. Briegel (Chairman); M. Vogeser.
Statistical Analyses: Analytica International- F. Sorenson, K. Freivogel.
CORTICUS Investigators: Austria: LKH Feldkirch, Feldkirch (P. Fae); Krankenhaus Barmherzige Schwestern, Linz (J. Reisinger); Universitaetsklinik fuer Innere Medizin II, Wien (G. Heinz); Belgium: Hopital St. Joseph, Arlon (M. Simon); Department of Critical Care Medicine, St Luc University Hospital, UCL, Brussels (P–F. Laterre, X. Wittebole, MN France); University Hospital Erasme, Université de Bruxelles, Brussels (J.L. Vincent, D. DeBacker); CHU Charleroi, Charleroi (P. Biston). France: Hopital de Caen, Caen (C. Daubin); Hopital Raymond Poincare, Garches (D. Annane, D. Lipiner, V. Maxime); Hopital Huriez, Lille (PA. Rodie Talbere, B. Vallet); Hopital Caremeau, Nimes (J.Y. Lefrant); Hopital Saint-Antoine, Paris (G. Offenstadt); Hopital Lariboisiere, Paris (D. Payen, A.C. Lukaszewicz). Germany: Zentralklinikum Augsburg, Augsburg (H. Forst, G. Neeser, Y. Barth); Charite Universitaetsmedizin Berlin, Campus Virchow-Klinikum (D. Keh, J.Langrehr, M.Oppert, C.Spies), Campus Mitte (C. Spies, S.Rosseau), Campus Benjamin Franklin (J. Weimann); Evangelisches Waldkrankenhaus Spandau, Berlin (M. Reyle Hahn); St. Joseph-Krankenhaus, Berlin (M. Schmutzler); Vivantes Klinikum Spandau, Berlin (K.J. Slama), Vivantes Klinikum Neukoelln, Berlin (H.Gerlach), Vivantes Klinikum im Friedrichshain,Berlin (S. Veit); Inst. For Anaesthesia & Operative Intensive Care Medicine, Darmstadt (M. Welte, L. Von Beck); University Hospital Carl Gustav Carus, Dresden (C. Marx); Krankenhaus Hennigsdorf, Hennigsdorf (A. Lange); Friedrich-Schiller Universitaet, Jena (K. Reinhart, F. Bloos, F. Brunkhorst); Klinikum Kempten-Oberallgaeu, Kempten (M. Haller); Klinikum of Landshut, Landshut (U. Helms); Klinikum Mannheim, Mannheim (A. Kalenka, F. Fiedler); Universitaetsklinikum Marburg, Marburg (M. Max); Klinik fuer Anaesthesiologie, Klinikum der Universitaet, Ludwig-Maximilians-Universitaet, Munich (J. Briegel); Department of Surgery, Klinikum der Universitaet-Grosshadern, Munich (W. Hartl); Staedtisches Krankenhaus Muenchen-Harlaching, Munich (M. Klimmer, T. Helmer); Universitaet Erlangen-Namberg, Nuernberg (M. Baumgaertel); Klinikum Ernst von Bergman, Potsdam (D. Pappert). Israel: Haemek Hospital, Afula (A. Lev); Hadassah Medical Organization, Jerusalem (Y. Weiss, C. Sprung, J. Benbenishty, O. Shatz); Belinson Medical Centre, Petach Tikva (P. Singer); Ichilov Hospital, Tel Aviv (A. Nimrod). Italy: Policlinico di Tor Vergata, Rome (S. Natoli); Centro di Rianimazione, Ospedale S. Eugenio, Rome (F. Turani). Netherlands: Erasmus University Medical Center, Rotterdam (B. Van der Hoven). Portugal: Hospital de St. Antonio do Capuchos, Lisbon (R. Moreno, R. Matos). United Kingdom: Aberdeen Royal Infirmary, Aberdeen (B.H. Cuthbertson, S. Roughton); The Ipswich Hospital NHS, Ipswich (M. Garfield); The General Infirmary at Leeds, Leeds (A. Mallick); University College London Hospitals NHS Foundation Trust, London (M. Singer, M McKendry); Southampton General Hospital, Southampton (T. Woodcock).
Rights and permissions
About this article
Cite this article
Moreno, R., Sprung, C.L., Annane, D. et al. Time course of organ failure in patients with septic shock treated with hydrocortisone: results of the Corticus study. Intensive Care Med 37, 1765–1772 (2011). https://doi.org/10.1007/s00134-011-2334-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-011-2334-x