Abstract
Objective
Increased expression of long pentraxin 3 (PTX3) has been found in patients with sepsis or acute respiratory distress syndrome. Tissue factor (TF) activation plays an important role in the pathogenesis of acute lung injury. The present study sought to determine the relationship between PTX3 expression and TF activation in acute lung injury.
Methods
Lung injury was induced by intratracheal instillation of lipopolysaccharide (LPS) in mice, and the PTX3 expression, TF activation and lung injury were determined. We also treated the lung injury with an anti-human tissue factor monoclonal antibody in human tissue factor knock-in (hTF-KI) mice.
Results
Balb/c mice were challenged with increasing doses of LPS. After 24 h, PTX3 protein in the bronchioalveolar lavage fluid was increased in parallel with the severity of lung injury, and correlated with tissue factor (TF) activity. The expression and distribution of PTX3 and TF were further documented in detail 6 h after LPS (5 mg/kg) instillation. Treatment with anti-human TF monoclonal antibody dramatically attenuated LPS-induced lung injury, alveolar fibrin deposition and inflammatory cell infiltration in “humanized” hTF-KI mice 6 h after LPS challenge. The PTX3 expression was significantly decreased by the anti-coagulant therapy.
Conclusion
These results support the clinical finding that PTX3 may be a useful biomarker to the reflect severity of lung injury and provide effective therapies. The interplay between PTX3 and TF could be a potential mechanism that mediates lung injury.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Despite many advances achieved in diagnosis and management, acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) still comprise one of the common causes of life-threatening respiratory failure [1]. Searching for biomarkers for diagnosis and prognosis, and developing novel therapies have been the focuses in ALI/ARDS research [2]. Application of bioinformatics, proteomics and other systems biology approaches has revealed several potential biomarkers for ALI/ARDS, such as LIX (LPS-induced CXC chemokine) [3], PBEF (Pre-B cell colony-enhancing factor) [4], MIF (macrophage migration inhibitory factor) [5] and Pre-Elafin [6]. One such molecule that we are interested in is long pentraxin 3 (PTX3), which is a soluble pathogen pattern recognition receptor for innate immunity [7, 8].
In our previous studies, we have found that PTX3 was one of the genes induced by TNFα in lung alveolar epithelial cells [9], mediated through JNK signaling pathway [10]. We further found induction of PTX3 in multiple ALI models in rats, including systemic administration of lipopolysaccharide (LPS), hemorrhagic shock/resuscitation and mechanical ventilation [11]. Clinical data have shown that the PTX3 level was dramatically increased in the plasma of patients with endotoxic shock and sepsis, which was correlated with the severity of the diseases [12]. Recently, a clinical study demonstrated that the plasma level of PTX3 in ARDS patients was correlated with lung function, systemic organ failure and clinical outcome [13], which suggests that PTX3 could be an early marker of ALI/ARDS [14]. A working group organized by NIH suggested that a good biomarker should reflect the pathogenic process of the disease and respond to a therapeutic intervention [15]. Animal studies with well-established disease models and controlled experimental conditions could be useful to verify whether PTX3 meets these criteria.
It has been reported that PTX3 could promote tissue factor (TF) expression in human endothelial cells and monocytes upon inflammatory stimuli, such as LPS, IL-1β and TNFα [16, 17]. TF is a key initiator of coagulation cascades and is highly expressed in patients with ALI [18]. The evolving understanding of ALI/ARDS indicates an extensive cross-talk between inflammatory responses and coagulation cascades, which leads to reciprocal modulation of the disease [19, 20]. The pro-coagulation state in both vascular and alveolar compartments ultimately leads to fibrin deposition in the lung parenchyma. Therefore, pulmonary coagulopathy has been suggested as a therapeutic target, and many anti-coagulant therapies have being tested in preclinical and clinical studies [21, 22]. Recently, we have demonstrated the therapeutic effects of an anti-human TF antibody on intestinal ischemia–reperfusion induced ALI in human TF knock-in (hTF-KI) mice [23].
In the current study, we first used intratracheal instillation of LPS as a model to determine whether the expression levels of PTX3 could reflect the severity of lung injury. We then used “humanized” hTF-KI transgenic mice to determine whether increased PTX3 expression can be reversed by an effective therapy.
Materials and methods
Animals, acute lung injury and treatment
The studies were approved by the Animal Use and Care Committee of the University Health Network. All animals received humane care. Balb/c mice and human TF knock-in (hTF-KI) transgenic mice were used in this study. All mice used were 6–10 weeks old and housed in a pathogen-free facility. The hTF-KI mice (129v/C57BL6 background, Centocor, Malvern, PA) were generated by replacing murine TF gene with the full length human TF coding sequence, which is expressed under control of murine TF promoter [23, 24].
Acute lung injury was induced by intratracheal instillation of LPS (Escherichia coli serotype 055:B5, Sigma, St. Louis, MO) [25, 26]. Briefly, mice were anesthetized with 5% isoflurane and suspended by their front teeth at a 60° angle. The tongue was gently pulled out to expose the larynx and to prevent the swallow reflex. An aliquot (100 μl) of saline with different doses of LPS (0, 1, 2.5 and 5 mg/kg) was instilled into the trachea with a pipette. The animals were killed by exsanguination under anesthesia 24 h later. To determine the early responses, another group of animals were given 5 mg/kg of LPS and killed after 6 h. To determine the PTX3 levels after anti-coagulant treatment, the monoclonal antibody against human TF (5 mg/kg in 100 μl of PBS; Centocor, Malvern, PA), which has been previously characterized [23, 24], was injected into the hTF-KI mice through the jugular vein 10 min after LPS instillation. The same volume of PBS was given to the control group. Mice were killed 6 h after LPS instillation.
Bronchoalveolar lavage (BAL)
After killing the animals, tracheotomy was conducted (four mice/group), and a 22-gauge catheter was inserted into the trachea. With the right bronchial ligated, the left lung was lavaged with 250 μl of ice cold saline twice. The recovered lavage fluids were centrifuged for 10 min at 5,000 rpm, and the supernatants were stored at −80°C until further analysis.
Histology and immunohistochemistry staining
In a sub-group of animals (four mice/group), the left lung was inflated at 20 cmH2O and fixed with 4% paraformaldehyde in PBS, and embedded in paraffin. Hematoxylin and eosin (H&E) staining was conducted with 5-μm tissue slides. The lung injury was assessed with a modified scoring system according to interstitial cellular infiltrate, alveolar edema, hemorrhage and cellular exudates [27–29] in a blinded fashion. Immunohistochemical staining for PTX3 and TF was conducted with a Vectastain ABC kit (Vector Laboratories, Burlingame, CA). Briefly, lung tissue slides (5 μm) were incubated with either a polyclonal antibody (Ab) for PTX3 (1:50 dilution) (Santa Cruz Biotechnology, Santa Cruz, CA), or for TF (1:200 dilution) (a gift of Dr. J. H. Morrissey, University of Illinois at Urbana-Champaign), and then a secondary antibody. The positive staining was visualized with Vector red for PTX3 and diaminobenzidine (DAB) for TF, and then the slides were counterstained with 1% methyl green and hematoxylin, respectively. The specificity of antibodies was determined by replacing the primary antibody with non-immunized IgG (Sigma). Martius Scarlet Blue (MSB) staining was performed for alveolar fibrin deposition as previously described [23, 30].
Quantitative real-time PCR
Total RNA from the snap-frozen right lungs (four mice/group) was isolated using TRIZol Reagent (Invitrogen, Burlington, Canada) and purified with an RNeasy Mini Kit (Qiagen, Mississauga, Canada). Synthesis of cDNA from total RNA was carried out using a TaqMan Reverse Transcription Reagent kit (Applied Biosystems, Foster City, CA). The primers (Supplemental Table 1) were designed using the Primer Express 1.5 software (Applied Biosystems) and synthesized by ACGT Corp. (Toronto, Canada). The amplification was performed in a real-time PCR machine (ABI PRISM 9700HT, Applied Biosystems, Foster City, CA). The relative expression levels of genes interested were normalized with a housekeeping gene, GAPDH.
ELISA for PTX3
PTX3 levels in the homogenized lungs, BAL fluid and plasma were determined in duplicate with an ELISA kit (R & D System, Minneapolis, MN). The optical density was read at 450 nm with an automatic plate reader (Thermo Labosystems, Chantilly, VA). The concentrations were calculated by converting the optical density readings against a standard curve [10, 11].
TF activity assay
TF activity in the homogenized lung tissues, BAL fluids and plasma was measured with a colorimetric assay (American Diagnostica, Stamford, CT). Lipidated human TF provided in the kit was used as positive control for a standard curve.
Statistical analysis
Data are presented as mean ± standard deviation (SD). The GraphicPad Prizm program was used for the statistics analysis following the standard protocols. The inter-group differences were tested by one-way analysis of variance (ANOVA) and Tukey’s test for post hoc multiple comparisons. The differences between two groups were tested with Student's t test. The correlation between PTX3 levels and TF activities was tested by Pearson’s correlation analysis. P values <0.05 are defined as significant.
Results
Intra-tracheal LPS-induced lung injury is associated with PTX3 expression and TF activation
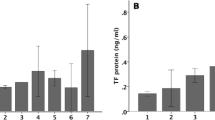
To determine whether the PTX3 expression could be associated with the severity of lung injury, we challenged Balb/c mice with different dosages of LPS (0, 1, 2.5 and 5 mg/kg) through intratracheal instillation. After 24 h, infiltration of inflammatory cells, hemorrhage and interstitial edema were observed in the lung in an LPS dose-dependent manner (Fig. 1). TF activation is a hallmark of lung injury [22]. TF activity and PTX3 concentration in the BAL fluids were increased with the incremental doses of LPS (Fig. 2). The concentration of PTX3 was positively correlated with the TF activity and the lung injury scores (Fig. 2).
LPS instillation induced mouse lung injury in a dose-dependent manner. Balb/c mice were challenged with different dosages of LPS via intratracheal instillation and killed 24 h later. The severity of lung injury increased with the incremental doses of LPS. The slides shown are representatives from four mice/group (H&E, 400×)
Increased PTX3 expression was associated with TF activation and injury severity induced by LPS in lung. LPS challenged Balb/c mice were killed 24 h later, and the lungs were subjected to BAL. TF activity and PTX3 concentration in the BAL fluid were increased as the LPS dose increased and were well correlated each other. PTX3 levels were correlated with lung injury scores. All data shown are mean ± SD from four mice/group. *p < 0.05 and **p < 0.01 versus control
It has been reported that PTX3 expression is rapidly enhanced at the early stage of infectious/inflammatory diseases [8, 13]. To determine the relationship between PTX3 and TF at the early stage of lung injury, another group of animals were killed 6 h after LPS (5 mg/kg) intratracheal instillation. The LPS-induced lung injury (Supplemental Fig. 1) was accompanied with significant increases in immunohistochemistry staining, mRNA level and activity of TF in the lung tissues (Fig. 3). The levels of PTX3 gene and protein (Fig. 4) in the lung tissues as well as in the serum were also significantly increased after LPS challenge. Immunostaining (Fig. 4) showed that the increased PTX3 was mainly found along the alveolar wall and airway epithelium. Some cells in the alveolar space were also stained PTX3 positive.
LPS challenge induced TF expression and activation at the early stage of lung injury. Balb/c mice were challenged with LPS (5 mg/kg, intratracheal instillation) for 6 h. TF protein expression and distribution in the lung were shown by immunohistochemistry (dark brown, 400×). TF gene expression in the lung was determined by quantitative RT-PCR. TF activity in the lung was measured with a colorimetric assay. TF expression and activation were significantly enhanced within 6 h after LPS instillation. Data shown are mean ± SD from four mice/group. **p < 0.01 versus saline control
PTX3 expression was enhanced early during LPS-induced lung injury. Balb/c mice were challenged with LPS (5 mg/kg, intratracheal instillation) for 6 h. PTX3 gene expression in the lung was determined by quantitative RT-PCR. PTX3 protein in the lung tissue homogenates and the sera collected from the mice were determined by ELISA. All data shown are mean ± SD from four mice/group. *p < 0.05 versus saline control. LPS-induced PTX3 protein expression and distribution are shown by immunohistochemistry staining in pink (400×)
Reducing ALI with anti-TF antibody decreased PTX3 expression in the lung
We then used transgenic mice in which the murine TF gene was replaced by knocking in human TF gene (hTF-KI) to determine whether the PTX3 expression could be decreased by anti-coagulant therapy with an anti-human TF antibody. We have recently shown that the anti-human TF monoclonal antibody ameliorated intestinal ischemia-reperfusion-induced lung injury in the hTF-KI mice [23]. In the present study, the antibody treatment dramatically attenuated the LPS-induced lung injury and alveolar fibrin deposition (Fig. 5). A significant reduction of lung injury score, total cell counts in the BAL fluid and inhibition of plasma TF activity was detected in the antibody-treated group in comparison with vehicle control (Fig. 6). Importantly, this antibody also significantly decreased PTX3 expression in the BAL fluid and in the lung tissues (Fig. 6), suggesting that PTX3 level does reflect the lung injury and is useful to monitor the therapeutic effects.
Anti-hTF mAb reduced LPS-induced lung injury and fibrin alveolar depositon. The hTF-KI mice were challenged with LPS (5 mg/kg) intratracheally, and then treated with 5 mg/kg of anti-hTF mAb (LPS + Ab group) or PBS (LPS only group) intravenously. The animals were killed 6 h later. The therapeutic effects of anti-TF antibody were determined by reduced tissue injury (H&E staining, 400×) and alveolar fibrin deposition (MSB staining, 400×) in the lung
Anti-hTF mAb administration improved lung injury score, and reduced TF activation and PTX3 expression. LPS (5 mg/kg) challenged hTF-KI mice were treated with 5 mg/kg of anti-hTF mAb (LPS + Ab group) or PBS (LPS only group) intravenously, and then killed 6 h later. The anti-hTF treatment significantly improved lung injury scores and reduced total cell counts in the BAL fluid and the plasma TF activity. The lung PTX3 levels, determined in the BAL fluids and lung tissue lysates, were also significantly lower in the antibody treated group. All data shown are mean ± SD from four to six mice/group. *p < 0.05
Discussion
PTX3 as a potential biomarker
PTX3 is an acute phase protein and the first defined member of long pentraxin family. Unlike the classic pentraxins, such as C-reactive protein and serum amyloid P that are mainly produced in liver, PTX3 is produced by a variety of tissue cells and inflammatory cells at the site of injury/inflammation [8]. For this reason, we used an intra-pulmonary injury model induced by intra-tracheal instillation of LPS in the present study. The expression of PTX3 in the lung tissue and BAL fluid increased in an LPS dose-dependent manner, in parallel with the severity of lung injury. ALI/ARDS could be induced by intra- and extra-pulmonary causes [31]. In another study, both intra-pulmonary (ventilator-induced lung injury) and extra-pulmonary (systemic administration of LPS, hemorrhagic shock/resuscitation) factors induced PTX3 expression in rats [11]. Moreover, the expression of PTX3 was also correlated with expressions of TNFα and IL-1β [11], as well as TF in the lungs [32]. Responding to a therapeutic intervention has been considered as one of the important features for potential biomarkers [14, 15]. It is difficult to provide clinical evidence to support this notion, whereas animal studies are more feasible for the proof in principle. One of the novel findings of the present study is that the anti-TF therapy reduced lung injury and PTX3 expression. Our studies provide experimental data to support the clinical observations [12, 13] that PTX3 may be a biomarker for ALI/ARDS. These data cannot be easily obtained in the clinical setting.
Therapeutic application of anti-human TF antibody in ALI
One of the well-recognized mechanisms of ALI/ARDS is the excessive inflammatory response in the lung [1]. A pro-coagulation state exists during the pathogenesis of ARDS, and alveolar fibrin deposition has been characterized as a hallmark of the early phase of ALI. Anti-coagulant therapy has been studied for ALI/ARDS treatment [21, 33, 34]. The production and activation of TF have been found to be significantly increased in human lung epithelial cells in vitro after stimulation with pro-inflammatory cytokines and in vivo in ARDS patients [35].
Recently, we have reported that blockade of TF using a monoclonal antibody specifically against human TF significantly attenuated the inflammation and lung tissue injury induced by intestinal ischemia-reperfusion in the hTF-KI transgenic mice [23]. In the present study, we further demonstrated the protective effects of the anti-TF antibody in the intrapulmonary ALI model induced by intratracheal LPS challenge. These studies provide examples of using “humanized” mice to test anti-human antibodies in the pre-clinical stage in both extra- and intra-pulmonary ARDS animal models.
Since the number of transgenic mice available was limited to us, the LPS dose-related studies and the relationship between PTX3 and TF expression and distribution studies at early phase of LPS stimulation were conducted with Balb/c mice, whereas hTF-KI mice are from 129v/C57BL6 background. Different strains of mice may have different inflammatory responses. On the other hand, the increased PTX3 expression and TF activation observed from different strains of mice suggest that this could be common responses.
Relationship between PTX3 and TF
It was reported that PTX3 enhanced TF expression/activation induced by LPS or inflammatory cytokines in cultured human endothelial cells [17] and in monocytes activated by LPS [16]. These observations led to the speculation that PTX3 may interact with coagulation cascades by modulating TF expression/activation. Indeed, we found a positive correlation between the expression of PTX3 and TF activity. Administration of anti-hTF antibody protected animals from severe lung injury and reduced PTX3 expression in the lung. This finding suggests a reciprocal relationship between TF and PTX3, which merits further investigation.
In summary, our results strongly support PTX3 as a biomarker of acute lung injury. Further studies are necessary to determine the role of PTX3/TF interaction in ALI/ARDS.
References
Ware LB, Matthay MA (2000) The acute respiratory distress syndrome. N Engl J Med 342:1334–1349
Liu M (2005) Searching for acute respiratory distress syndrome genes: aren’t we there yet? Am J Respir Crit Care Med 171:298–299
Jeyaseelan S, Chu HW, Young SK, Worthen GS (2004) Transcriptional profiling of lipopolysaccharide-induced acute lung injury. Infect Immun 72:7247–7256
Ye SQ, Simon BA, Maloney JP, Zambelli-Weiner A, Gao L, Grant A, Easley RB, McVerry BJ, Tuder RM, Standiford T, Brower RG, Barnes KC, Garcia JG (2005) Pre-B-cell colony-enhancing factor as a potential novel biomarker in acute lung injury. Am J Respir Crit Care Med 171:361–370
Gao L, Flores C, Fan-Ma S, Miller EJ, Moitra J, Moreno L, Wadgaonkar R, Simon B, Brower R, Sevransky J, Tuder RM, Maloney JP, Moss M, Shanholtz C, Yates CR, Meduri GU, Ye SQ, Barnes KC, Garcia JG (2007) Macrophage migration inhibitory factor in acute lung injury: expression, biomarker, and associations. Transl Res 150:18–29
Bastarache JA, Wang L, Wang Z, Albertine KH, Matthay MA, Ware LB (2008) Intra-alveolar tissue factor pathway inhibitor is not sufficient to block tissue factor procoagulant activity. Am J Physiol Lung Cell Mol Physiol 294:L874–881
Bottazzi B, Garlanda C, Salvatori G, Jeannin P, Manfredi A, Mantovani A (2006) Pentraxins as a key component of innate immunity. Curr Opin Immunol 18:10–15
He X, Han B, Liu M (2007) Long pentraxin 3 in pulmonary infection and acute lung injury. Am J Physiol Lung Cell Mol Physiol 292:L1039–1049
dos Santos CC, Han B, Andrade CF, Bai X, Uhlig S, Hubmayr R, Tsang M, Lodyga M, Keshavjee S, Slutsky AS, Liu M (2004) DNA microarray analysis of gene expression in alveolar epithelial cells in response to TNFalpha, LPS, and cyclic stretch. Physiol Genomics 19:331–342
Han B, Mura M, Andrade CF, Okutani D, Lodyga M, dos Santos CC, Keshavjee S, Matthay M, Liu M (2005) TNFalpha-induced long pentraxin PTX3 expression in human lung epithelial cells via JNK. J Immunol 175:8303–8311
Okutani D, Han B, Mura M, Waddell TK, Keshavjee S, Liu M (2007) High-volume ventilation induces pentraxin 3 expression in multiple acute lung injury models in rats. Am J Physiol Lung Cell Mol Physiol 292:L144–153
Muller B, Peri G, Doni A, Torri V, Landmann R, Bottazzi B, Mantovani A (2001) Circulating levels of the long pentraxin PTX3 correlate with severity of infection in critically ill patients. Crit Care Med 29:1404–1407
Mauri T, Coppadoro A, Bellani G, Bombino M, Patroniti N, Peri G, Mantovani A, Pesenti A (2008) Pentraxin 3 in acute respiratory distress syndrome: an early marker of severity. Crit Care Med 36:2302–2308
Cribbs SK, Martin GS (2008) Biomarkers in acute lung injury: are we making progress? Crit Care Med 36:2457–2459
Biomarkers Definitions Working Group (2001) Biomarkers and surrogate endpoints: preferred definitions and conceptual framework. Clin Pharmacol Ther 69:89–95
Napoleone E, di Santo A, Peri G, Mantovani A, de Gaetano G, Donati MB, Lorenzet R (2004) The long pentraxin PTX3 up-regulates tissue factor in activated monocytes: another link between inflammation and clotting activation. J Leukoc Biol 76:203–209
Napoleone E, Di Santo A, Bastone A, Peri G, Mantovani A, de Gaetano G, Donati MB, Lorenzet R (2002) Long pentraxin PTX3 upregulates tissue factor expression in human endothelial cells: a novel link between vascular inflammation and clotting activation. Arterioscler Thromb Vasc Biol 22:782–787
Gando S, Nanzaki S, Morimoto Y, Kobayashi S, Kemmotsu O (1999) Systemic activation of tissue-factor dependent coagulation pathway in evolving acute respiratory distress syndrome in patients with trauma and sepsis. J Trauma 47:719–723
Welty-Wolf KE, Carraway MS, Ortel TL, Piantadosi CA (2002) Coagulation and inflammation in acute lung injury. Thromb Haemost 88:17–25
Bastarache JA, Ware LB, Bernard GR (2006) The role of the coagulation cascade in the continuum of sepsis and acute lung injury and acute respiratory distress syndrome. Semin Respir Crit Care Med 27:365–376
Hofstra JJ, Juffermans NP, Schultz MJ, Zweers MM (2008) Pulmonary coagulopathy as a new target in lung injury–a review of available pre-clinical models. Curr Med Chem 15:588–595
Laterre PF, Wittebole X, Dhainaut JF (2003) Anticoagulant therapy in acute lung injury. Crit Care Med 31:S329–336
Stahel PF, Smith WR, Moore EE (2008) Hypoxia and hypotension, the “lethal duo” in traumatic brain injury: implications for prehospital care. Intensive Care Med 34:402–404
Snyder LA, Rudnick KA, Tawadros R, Volk A, Tam SH, Anderson GM, Bugelski PJ, Yang J (2008) Expression of human tissue factor under the control of the mouse tissue factor promoter mediates normal hemostasis in knock-in mice. J Thromb Haemost 6:306–314
Perl M, Chung CS, Lomas-Neira J, Rachel TM, Biffl WL, Cioffi WG, Ayala A (2005) Silencing of Fas, but not caspase-8, in lung epithelial cells ameliorates pulmonary apoptosis, inflammation, and neutrophil influx after hemorrhagic shock and sepsis. Am J Pathol 167:1545–1559
Takano H, Inoue K, Yanagisawa R, Sato M, Shimada A, Morita T, Sawada M, Nakamura K, Sanbongi C, Yoshikawa T (2004) Protective role of metallothionein in acute lung injury induced by bacterial endotoxin. Thorax 59:1057–1062
Mura M, Andrade CF, Han B, Seth R, Zhang Y, Bai XH, Waddell TK, Hwang D, Keshavjee S, Liu M (2007) Intestinal I/R-induced acute lung injury and oncotic cell death in multiple organs. Shock 28:227–238
Mura M, Han B, Andrade CF, Seth R, Hwang D, Waddell TK, Keshavjee S, Liu M (2006) The early responses of VEGF and its receptors during acute lung injury: implication of VEGF in alveolar epithelial cell survival. Crit Care 10:R130
Nishina K, Mikawa K, Takao Y, Maekawa N, Shiga M, Obara H (1997) ONO-5046, an elastase inhibitor, attenuates endotoxin-induced acute lung injury in rabbits. Anesth Analg 84:1097–1103
Singh I, Smith A, Vanzieleghem B, Collen D, Burnand K, Saint-Remy JM, Jacquemin M (2002) Antithrombotic effects of controlled inhibition of factor VIII with a partially inhibitory human monoclonal antibody in a murine vena cava thrombosis model. Blood 99:3235–3240
Liu M (2008) ARDS and knowledge translation. Intensive Care Med 34:589–591
dos Santos CC, Okutani D, Hu P, Han B, Crimi E, He X, Keshavjee S, Greenwood C, Slutsky AS, Zhang H, Liu M (2008) Differential gene profiling in acute lung injury identifies injury-specific gene expression. Crit Care Med 36:855–865
Chambers RC (2008) Procoagulant signalling mechanisms in lung inflammation and fibrosis: novel opportunities for pharmacological intervention? Br J Pharmacol 153(Suppl 1):S367–378
Schultz MJ, Haitsma JJ, Zhang H, Slutsky AS (2006) Pulmonary coagulopathy as a new target in therapeutic studies of acute lung injury or pneumonia—a review. Crit Care Med 34:871–877
Bastarache JA, Wang L, Geiser T, Wang Z, Albertine KH, Matthay MA, Ware LB (2007) The alveolar epithelium can initiate the extrinsic coagulation cascade through expression of tissue factor. Thorax 62:608–616
Acknowledgments
We thank Dr. Lily Li (Centocor Inc.) for the hTF-KI mice and anti-hTF monoclonal antibody, and Dr. J.H. Morrissey (University of Illinois at Urbana-Champaign) for the polyclonal anti-TF antibody. This work is supported by Canadian Institutes of Health Research (operating grants: MOP-13270 and MOP-42546) and the Ontario Thoracic Society.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
He, X., Han, B., Bai, X. et al. PTX3 as a potential biomarker of acute lung injury: supporting evidence from animal experimentation. Intensive Care Med 36, 356–364 (2010). https://doi.org/10.1007/s00134-009-1720-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-009-1720-0