Abstract
Background
It remains uncertain why immunocompetent patients with bacterial community-acquired pneumonia (CAP) die, in spite of adequate antibiotics.
Methods
This is a secondary analysis of the CAPUCI database which was a prospective observational multicentre study. Two hundred and twelve immunocompetent patients admitted to 33 Spanish ICUs for CAP were analyzed. Comparisons were made for lifestyle risk factors, comorbidities and severity of illness. ICU mortality was the principal outcome variable.
Results
Bacteremic CAP (43.3 vs. 21.1%) and empyema (11.5 vs. 2.2%) were more frequent (P < 0.05) in patients with Streptococcus pneumoniae CAP. Higher rates of adequate empiric therapy (95.8 vs. 75.5%, P < 0.05) were observed in patients with S. pneumoniae CAP. Patients with non-pneumococcal CAP experienced more shock (66.7 vs. 50.8%, P < 0.05), and need for mechanical ventilation (83.3 vs. 61.5%, P < 0.05). ICU mortality was 20.7 and 28% [OR 1.49(0.74–2.98)] among immunocompetent patients with S. pneumoniae (n = 122) and non-pneumococci (n = 90), in spite of initial adequate antibiotic. Multivariable regression analysis in these 184 immunocompetent patients with adequate empirical antibiotic treatment identified the following variables as independently associated with mortality: shock (HR 13.03); acute renal failure (HR 4.79), and APACHE II score higher than 24 (HR 2.22).
Conclusions
Mortality remains unacceptably high in immunocompetent patients admitted to the ICU with bacterial pneumonia, despite adequate initial antibiotics and comorbidities management. Patients with shock, acute renal failure and APACHE II score higher than 24 should be considered for inclusion in trials of adjunctive therapy in order to improve CAP survival.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Community-acquired pneumonia (CAP) is an important cause of morbidity, mortality, and increased health care costs [1, 2]. CAP is considered as severe when admission to the intensive care unit (ICU) is necessary. This may be indicated because of severe respiratory distress, sepsis or septic shock, and occurs in about 9–16% of hospitalized CAP patients [3, 4]. In patients with severe CAP mortality rates range from 23 to 50% according to the presence of shock, accuracy of processes of care, need for mechanical ventilation, and type of underlying disease [1, 5–9]. Prompt initiation of appropriate antibiotic therapy prior to the availability of culture results is recommended as it potentially benefits patients’ prognosis [8, 10, 11]. In the CAPUCI cohort, our group described significantly better survival rates and lower period of mechanical ventilation requirements when empiric antibiotic regimen was in accordance with the 2003 guidelines of the Infectious Diseases Society of America [12]. These recommendations were based on etiology, associated susceptibility, and data regarding specific risk factors such as underlying disease.
In adults, Streptococcus pneumoniae is by far the most common pathogen in CAP and empiric therapy should always cover this microorganism. Yet, few data are available about why immunocompetent patients still die in spite of adequate antibiotic agents and comorbidities management.
The primary aim of this study was set to estimate mortality in ICU patients admitted by documented bacterial pneumonia who received appropriate antibiotic therapy from hospital admission. A secondary objective was to provide understanding of factors that can be used to predict outcomes in immunocompetent patients with bacterial CAP requiring ICU admission and receiving appropriate antibiotic therapy. In addition, we evaluated possible effects of etiology on outcomes, independently of antibiotic adequacy. We hypothesized that further improvements in survival may require characterization of a subgroup of patients which may benefit from adjuvant therapy, as antibiotic treatment alone is not enough to improve survival in CAP patients admitted to ICU.
Materials and methods
Data were collected from all patients admitted for CAP to the ICU of 33 participating hospitals in Spain (1 December 2000 to 28 February 2002). One investigator at each hospital prospectively recorded variables in a previously designed database. The study was approved by the institutional research board and informed consent was waived by the ethics committee because of the observational nature of the study.
Study design
This study is a secondary analysis based on a database used to explore the impact of compliance with IDSA guidelines on survival. Therefore, details on setup of this observational study can be found elsewhere [12].
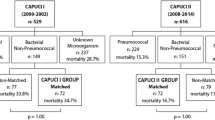
Immunocompromised patients were excluded for analysis (Fig. 1). Cases presumed to be caused by non-aureus Staphylococci or enterococci were considered contaminants and classified as unknown. Cases with non-bacterial pneumonia (virus, tuberculosis) were excluded.
The study cohort was divided in two groups according to the causative etiology: S. pneumoniae versus non-pneumococci. A sub-analysis was performed considering only patients with adequate initial antibiotic treatment. Finally, an additional (intent-to-treat) analysis was also performed including patients with unknown etiology. Relationships between etiology and outcomes were assessed using univariate and multivariable analysis.
Variables
Relationships with mortality were evaluated for the following variables: age, gender, life style risk factors, pre-existing comorbidities (alcoholism, active tobacco use, chronic obstructive pulmonary disease (COPD), cardiovascular disease, diabetes mellitus, and neurologic illness). Disease severity as assessed by the acute physiology and chronic health evaluation (APACHE) II score based on the first 24 h of ICU observation, the development of shock and acute renal failure, rapid radiographic spread of pulmonary infiltrates, ICU mortality, duration of mechanical ventilation and ICU stay. For the latter two variables only data from ICU survivors were considered.
Definitions
Community-acquired pneumonia was defined as an acute lower respiratory tract infection characterized by: (1) an acute pulmonary infiltrate evident on chest radiographs and compatible with pneumonia; (2) confirmatory findings of clinical examination, and (3) acquisition of the infection outside a hospital, long-term care facility, or nursing home.
Community-acquired pneumonia was considered severe when it required ICU admission. Patients were admitted to the ICU either because they were potential candidates for mechanical ventilation or because they were judged to be in an unstable condition requiring intensive medical or nursing care [13, 14].
Patients with HIV infection, neoplasia, those taking cytotoxic drugs or long-term oral steroid therapy, such as daily dose of >20 mg of prednisolone or the equivalent for >2 weeks were considered immunocompromised [8] and excluded.
Appropriate therapy was defined as the use of the at least one antibiotic to which all isolates were susceptible in vitro or (for Legionella pneumophila) was expected to be susceptible [8].
Definitions of lifestyle risk factors and comorbidities are defined elsewhere [12]. Specifically, shock was defined as the need for vasopressors for >4 h after adequate fluid replacement. Therapy of shock was consistent with available guidelines [15, 16] when the study was conducted. Acute renal failure was defined as an urine output of <20 mL/h or a total urine output of <80 mL in 4 h. Management of acute renal failure was in accordance with general guidelines and these patients received continuous veno-venous hemodialysis hemofiltration (CVVHD) techniques. All these variables were searched within the first 24 h of ICU admission. Other definitions of variables detailed in tables are provided as electronic supplemental material (ESM). ICU mortality was the principal outcome variable.
Statistical analysis
Discrete variables were expressed as counts (%) and continuous variables as mean and standard deviation (SD), unless stated otherwise; all statistical tests were two-sided. Differences in categorical variables were calculated using two-sided likelihood ratio Chi-square test or Fisher exact test and the Mann–Whitney U test or Kruskal–Wallis test were used for continuous variables, when appropriate.
Cox proportional-hazards regression analysis was used to assess the impact of independent variables on ICU mortality across the time. Variables significantly associated with mortality in the univariate analysis were entered in the model. Patients were classified in quartiles, based on APACHE II score at ICU admission. Results are presented as hazards ratios (HR) and 95% confidence intervals (CI). Logistic regression analysis was used to evaluate the variables independently associated with ICU length of stay (LOS). Results are presented as Odds Ratio (OR) and 95% confidence intervals (CI). In order to avoid spurious associations, variables entered in the regression models were those with a relationship in univariate analysis (P < 0.05) or a plausible relationship with the dependent variable. Potential explanatory variables were checked for collinearity prior to inclusion in the regression models using Tolerance and Variance Inflation Factor. Data analysis was performed using SPSS for Windows 12.0.0 (SPSS, Chicago, IL, US).
Results
The CAPUCI database included 529 patients with severe CAP. Seventy (13.2%) patients were excluded because they were immunocompromised. In these patients ICU mortality rate was 25.3%. Among 459 immunocompetent patients, 5 (1.0%) patients with tuberculosis and 9 (1.9%) patients with fungal or viral agents were excluded. As detailed in Fig. 1, 212 immunocompetent patients with documented microorganism were analyzed. Causative microorganisms are listed in Table 1. There were 122 patients with S. pneumoniae CAP and 90 patients with non-pneumococcal CAP. Legionella pneumophila (n = 20), Haemophilus influenzae (n = 19), Staphylococcus aureus (n = 15; three methicillin-resistant) and Pseudomonas aeruginosa (n = 16) accounted for 77.7% of non-pneumococcal isolates.
Diagnostic examinations performed are shown in electronic supplemental material (ESM, Table A). Patient characteristics for S.pneumoniae and non-pneumococcal CAP are summarized in Table 2. The mean age of patients was 61.8 years and 63.2% (n = 134) were ≥60 year old. There were no differences in age, gender, and comorbidities between the groups. Pneumococcal CAP was more often associated with bacteremia and empyema. A higher rate of appropriate empiric antibiotic therapy was found (95.8 vs. 75.5%, P < 0.05) among patients with pneumococcal CAP. Patients with non-pneumococcal CAP experienced more shock, bilateral infiltrates on chest X-ray, acute renal failure, and had a higher need for mechanical ventilatory support (Table 2). Most patients with P. aeruginosa isolates were COPD (n = 14; 87.5%) or have active tobacco uses (n = 6; 37.5%).
After excluding patients with inappropriate initial antibiotic agents (n = 28), mortality remained high (28 vs. 20.7%), with a trend to worse outcome (OR for death 1.49; 95%CI, 0.74–2.98) in patients with non-pneumococcal CAP. Duration of mechanical ventilation was similar in both groups, but ICU stay remained significantly longer in non-pneumococcal CAP (Table 2).
Characteristics of these 184 immunocompetent patients with etiological diagnosis and adequate empirical antibiotic treatment are shown in Table 3. APACHE II score, bacteremic CAP, shock, bilateral infiltrates, need of mechanical ventilation and acute renal failure were variables associated with ICU mortality in univariate analysis and were included in a multivariable model. When patients were stratified according to APACHE II score quartiles, we found significant differences in risk factors between patients with a cutoff on the higher quartile (Table 4). We performed a Cox hazard proportional analysis including these variables in the model and found, as detailed in Table 5, that Shock (HR 9.25), acute renal failure (HR 3.38), and APACHE II score higher than 24 (HR 2.11) at ICU admission remained independently associated with mortality. Inclusion of etiology (pneumococcal or non-pneumococcal pneumonia), combination antibiotic therapy or delay to first antibiotic dose longer than 4 h did not affect this model.
An “intent-to-treat” analysis was performed, including 233 additional patients with unknown organisms (Table 6). This was done on the basis that microbiologic confirmation is extremely uncommon at ICU admission. There are no differences in the mortality rate (censored at 28 days) between patients with bacterial and unknown etiology (Fig. 2). We performed a Cox hazard regression analysis including significantly variables in the model (Table 6) and the following variables were independently identified as associated with ICU death: APACHE II score >24 (HR, 2.51; 95%CI 1.65–3.82; P < 0.001), shock (HR, 2.79; 95%CI 1.42–5.47; P = 0.003) and acute renal failure (HR, 2.99; 95%CI 1.82–4.92; P < 0.001). When excluding patients that died within the first 48 h of ICU admission, the model did not significantly change (data no shown).
Finally, ICU length of stay (LOS) for survivors has been classified in four quartiles (see ESM, Table B). Factors significantly associated with ICU LOS in the last quartile (≥18 days) in univariate analysis were: higher APACHE II score, shock, bilateral infiltrates, acute renal failure, and need of mechanical ventilation (Table 7). When these variables were included in a multivariable analysis (logistic regression), only need of mechanical ventilation (OR = 19.41; 95%CI 8.23–45.78; P < 0.001) and APACHE II score higher than 24 (OR = 1.79; 95%CI 1.04–3.06; P = 0.03) were variables independently associated with ICU LOS in the last quartile.
Discussion
An outstanding finding of this study is that it confirms that mortality exceeds 20% in immunocompetent patients admitted to the ICU by CAP with adequate therapy, suggesting that antibiotics alone are not enough. Indeed, some reports suggested the use of activated C-protein, steroids or other agents [17–21] as adjuvant therapy to improve survival. Unfortunately, it remains unknown whether specific conditions can be used to identify patients with potential benefit [20]. Our findings suggest that variables remaining independently associated with mortality in patients with an appropriate antibiotic therapy were shock, acute renal failure, and an APACHE II score higher than 24.
The natural course of CAP is determined by host factors as well as by the causative pathogen [20]. In many cases of severe CAP, the clinical picture continues to deteriorate despite of an appropriate management of host comorbidities and microorganism. Non-resolution places an important emphasis on patient reassessment with the clinician analyzing the history, examination, prescription chart and investigation results, in order to narrow down the cause of failure to improve. A number of questions will be asked by the clinician: is the therapy appropriate [22, 23]? Is there a resistant organism or unusual pathogen present, further complicating the disease process [24]? Are the host factors appropriately controlled [20]? When all these elements are considered the clinician is left at a therapeutic crossroad. Additional or alternative antibiotics [25], higher dose of antimicrobials agents [26, 27], and more aggressive management of the host’s comorbidities are all options which may improve the patient’s condition. However, each has its long- and short-term consequences which may or may not be favorable to the patient or to the population.
A unique aspect of the current study was that we focus in the subset of patients with documented bacterial pneumonia and appropriate empiric antibiotic therapy. Therefore, if the antimicrobial agent is appropriate and comorbidities are controlled why does the patient’s condition still deteriorate? In our cohort of ICU patients, 43 (23.4%) patients ultimately died, in spite of these conditions. Etiology was not a factor associated with this excess of mortality. By the time severe sepsis is established, the inflammatory pathway has already had a hand in the irreversible damage to the patient’s organs, and aggressive host treatment or alteration of the antibiotic regimen dose remains as the only feasible options. Therefore, further improvement in survival of these patients depends on identifying subsets which might benefit from adjuvant therapy. PROWESS [28] study evaluated the use of activated C-protein for sepsis treatment and found benefit in a subgroup of patients with APACHE II score higher than 25, a cutoff very close to that we find in this analysis as an independent risk factor for mortality in severe CAP patients. Whether this is a subset of patients who benefit more from adjuvant therapy should be assessed in prospective studies.
An interesting finding is noted on Table 4. Prior antibiotic use was associated with a significantly higher survival. As a consequence, receipt of antimicrobial therapy prior to randomization may affect the results of trials of treatment of CAP. It suggests that prior therapy would dilute the treatment effect and potentially result in a false negative conclusion. This finding was also documented in a large Medicare database reported by Houck et al. [29]; therefore, timely antibiotic administration in all patients with CAP should be recommended. In addition, based on our findings, earlier identification of renal failure and shock, such as using RIFLE criteria for acute kidney injury [30] or lactate determination in CAP patients at the emergency department [31] might improve identification of potential patient candidates for adjunctive therapy. Further studies using RIFLE criteria, biomarkers for acute kidney injury [30] or lactate assessment [31] should be performed.
Finally, our analysis in immunocompetent patients with documented bacterial CAP is of real academic interest to improve the design of clinical trials. Unfortunately, many patients at ICU admission did not have an etiologic diagnosis. For this reason, an intent-to-treat analysis considering both patients with confirmed and unknown microorganism was performed. Lack of differences in setting variables associated with mortality, reinforce our findings.
This study suffers several limitations. First, hospital mortality was not recorded and some differences may account when prolonging the follow-up [32]. Second, the cohort was inadequately powered to evaluate impact of specific pathogens within the non-pneumococci group. This is because the data have been gathered for another principal research question [12]. Third, some variables, such as anticoagulant status, glycemic control or confusion, were not recorded, and may have additional influence on the outcomes. Fourth, there are several obstacles to obtain an optimal antibiotic therapy in ventilated critically ill patients. These patients often present an increased volume of distribution, a higher cardiac output, hypoalbuminemia and altered creatinine clearance. These factors compromise antibiotic effectiveness evaluation making it too unpredictable [33]. Finally, it is unclear whether our findings can be extrapolated to countries with other criteria for ICU admission or patients in medical wards. However, as pointed by Shorr and Wunderink [34] decision to triage a patient to ICU cannot be solely based on a pneumonia severity score. Available pneumonia severity scores are not adequately validated for ICU admitted CAP patients and clinicians base ICU admission decisions on risk factors that are not included in scores. So, uniform ICU admission criteria are not available in clinical practice and clinical judgment remains a factor considered in ICU admission decision [34, 35]. Further studies should investigate these issues.
In conclusion, our findings confirm that mortality remains unacceptably high in immunocompetent patients admitted to the ICU with documented bacterial CAP, in spite of adequate antibiotic therapy and management of comorbidities. These differences in survival are not explained by etiology or delay in using adequate antibiotic agents. Our findings suggest the need to explore the effect of adjuvant therapy in selected patients, beyond of antibiotic therapy, to improve survival.
References
Restrepo MI, Jorgensen JH, Mortensen EM, Anzueto A (2001) Severe community-acquired pneumonia: current outcomes, epidemiology, etiology and therapy. Curr Opin Infect Dis 14:703–709
Bauer TT, Welte T, Ernen C, Schlosser BM, Thate-Waschke I, de Zeeuw J, Schultze-Werninghaus G (2005) Cost analyses of community-acquired pneumonia from the hospital perspective. Chest 128:2238–2246
Restrepo MI, Mortensen EM, Velez JA, Frei C, Anzueto A (2008) A comparative study of community-acquired pneumonia patients admitted to the ward and the ICU. Chest 133:610–617
Lujan M, Gallego M, Rello J (2006) Optimal therapy for severe pneumococcal community-acquired pneumonia. Intensive Care Med 32:971–980
Rello J, Rodriguez R, Jubert P, Alvarez B (1996) Severe community-acquired pneumonia in the elderly: epidemiology and prognosis. Study Group for Severe Community-Acquired Pneumonia. Clin Infect Dis 23:723–728
Restrepo MI, Anzueto A (2006) The role of new therapies for severe community-acquired pneumonia. Curr Opin Infect Dis 19:557–564
Frei CR, Restrepo MI, Mortensen EM, Burgess DS (2006) Impact of guideline-concordant empiric antibiotic therapy in community-acquired pneumonia. Am J Med 119:854–871
Rello J, Rodriguez A, Torres A, Roig J, Sole-Violan J, Garnacho-Montero J, de la Torre MV, Sirvent JM, Bodi M (2006) Implications of COPD in patients admitted to the intensive care unit by community-acquired pneumonia. Eur Respir J 27:1210–1216
Blot SI, Rodriguez A, Sole-Violan J, Blanquer J, Almirall J, Rello J (2007) Effects of delayed oxygenation assessment on time to antibiotic delivery and mortality in patients with severe community-acquired pneumonia. Crit Care Med 35:2509–2514
Rello J, Catalan M, Diaz E, Bodi M, Alvarez B (2002) Associations between empirical antimicrobial therapy at the hospital and mortality in patients with severe community-acquired pneumonia. Intensive Care Med 28:1030–1035
Waterer GW, Kessler LA, Wunderink RG (2006) Delayed administration of antibiotics and atypical presentation in community-acquired pneumonia. Chest 130:11–15
Bodi M, Rodriguez A, Sole-Violan J, Gilavert MC, Garnacho J, Blanquer J, Jimenez J, de la Torre MV, Sirvent JM, Almirall J, Doblas A, Badia JR, Garcia F, Mendia A, Jorda R, Bobillo F, Valles J, Broch MJ, Carrasco N, Herranz MA, Rello J (2005) Antibiotic prescription for community-acquired pneumonia in the intensive care unit: impact of adherence to Infectious Diseases Society of America guidelines on survival. Clin Infect Dis 41:1709–1716
Rello J, Bodi M, Mariscal D, Navarro M, Diaz E, Gallego M, Valles J (2003) Microbiological testing and outcome of patients with severe community-acquired pneumonia. Chest 123:174–180
Bartlett JG, Dowell SF, Mandell LA, File TM Jr, Musher DM, Fine MJ (2000) Practice guidelines for the management of community-acquired pneumonia in adults. Infectious Diseases Society of America. Clin Infect Dis 31:347–382
Ognibene FP (1996) Hemodynamic support during sepsis. Clin Chest Med 17:279–287
Marik P, Varon J (1998) The hemodynamic derangements in sepsis: implications for treatments strategies. Chest 114:854–860
Laterre PF, Garber G, Levy H, Wunderink R, Kinasewitz GT, Sollet JP, Maki DG, Bates B, Yan SC, Dhainaut JF, PROWESS Clinical Evaluation Committee (2005) Severe community-acquired pneumonia as a cause of severe sepsis: data from the PROWESS study. Crit Care Med 33:952–961
Garcia-Vidal C, Calbo E, Pascual V, Ferrer C, Quintana S, Garau J (2007) Effects of systemic steroids in patients with severe community-acquired pneumonia. Eur Respir J 30:951–956
Gorman SK, Slavik RS, Marin J (2007) Corticosteroid treatment of severe community-acquired pneumonia. Ann Pharmacother 41:1233–1237
Wunderink RG, Waterer GW (2004) Community-acquired pneumonia: pathophysiology and host factors with focus on possible new approaches to management of lower respiratory tract infections. Infect Dis Clin North Am 18:743–759
Glück T, Opal SM (2004) Advances in sepsis therapy. Drugs 64:837–859
Waterer GW, Somes GW, Wunderink RG (2001) Monotherapy may be suboptimal for severe bacteremic pneumococcal pneumonia. Arch Intern Med 161:1837–1842
Waterer GW, Rello J (2006) Choosing the right combination therapy in severe community-acquired pneumonia. Crit Care 10:115
Waterer GW, Buckingham SC, Kessler LA, Quasney MW, Wunderink RG (2003) Decreasing beta-lactam resistance in Pneumococci from the Memphis region: analysis of 2,152 isolates from 1996 to 2001 Chest 124:519–525
Rodríguez A, Mendia A, Sirvent JM, Barcenilla F, de la Torre-Prados MV, Solé-Violán J, Rello J, CAPUCI Study Group (2007) Combination antibiotic therapy improves survival in patients with community-acquired pneumonia and shock. Crit Care Med 35:1493–1498
Wunderink RG (2006) A CAP on antibiotic duration. Am J Respir Crit Care Med 174:3–5
Dunbar LM, Wunderink RG, Habib MP, Smith LG, Tennenberg AM, Khashab MM, Wiesinger BA, Xiang JX, Zadeikis N, Kahn JB (2003) High-dose, short-course levofloxacin for community-acquired pneumonia: a new treatment paradigm. Clin Infect Dis 37:752–760
Bernard GR, Vincent JL, Laterre PF, LaRosa SP, Dhainaut JF, Lopez-Rodríguez A, Steingrub JS, Garber GE, Helterbrand JD, Ely EW, Fisher CJ Jr, Recombinant human protein C worldwide evaluation in severe sepsis (PROWESS) study group (2001) Efficacy and safety of recombinant human activated protein C for severe sepsis. N Engl J Med 344:699–709
Houck PM, Bratzler DW, Nsa W, Ma A, Bartlett JG (2004) Timing of antibiotic administration and outcomes for medicare patients hospitalized with community-acquired pneumonia. Arch Intern Med 164:637–644
Hoste EAJ, Clermont G, Kersten A, Venkataraman R, Angus DC, De Bacquer D, Kellum JA (2006) RIFLE criteria for acute kidney injury are associated with hospital mortality in critically ill patients: a cohort analysis. Crit Care 10:R73
Rodríguez A, Rello J (2008) Assessment of severity in patients with community-acquired pneumonia. Intensive Care Med 34:968
Waterer GW, Kessler LA, Wunderink RG (2004) Medium-term survival after hospitalization with community-acquired pneumonia. Am J Respir Crit Care Med 169:910–914
Roberts JA, Lipman J (2006) Antibacterial dosing in intensive care: pharmacokinetics, degree of disease and pharmacodynamics of sepsis. Clin Pharmacokinet 45:755–773
Shorr AF, Wunderink R (2008) There is no “CAP” on the importance of community-acquired pneumonia in the ICU. Chest 133:590–592
Restrepo MI, Bienen T, Mortensen EM, Anzueto A, Metersky ML, Escalante P, Wunderink RG, Mangura BT (2008) Evaluation of ICU admission criteria and diagnostic methods for patients with severe community-acquired pneumonia: current practice survey. Chest 133:828–829
Acknowledgments
We are indebted to Montserrat Olona for statistical advice and critical review of the manuscript. This study has been supported in part by Health Research Founds (FISS PI 04/1500 and FISS 05/2401), Generalitat de Catalunya Grant (SGR 05/920), and CIBER Enfermedades Respiratorias (CIBERES) by Carlos III Health Institute Grant.
Conflict of interest statement
Dr Rello serves as consultant and in the speakers Bureau for Novartis. The remaining authors declare that they have no competing interest.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
This article is discussed in the editorial available at: doi:10.1007/s00134-008-1366-3.
Electronic supplementary material
Below is the link to the electronic supplementary material.
134_2008_1363_MOESM1_ESM.doc
Table A Diagnostic examinations performed in 212 immunocompetent patients with bacterial community-acquired pneumonia distributed according to the causative aetiology. (supplementary electronic material) (DOC 47 kb)
The CAPUCI study investigators
The CAPUCI study investigators
J. Solé-Violan (Dr. Negrin Hospital, Gran Canaria), J. Blanquer (Clinic Hospital, Valencia), J. Jiménez (Virgen Del Rocio Hospital, Seville), V. de la Torre (Virgen De La Victoria Hospital, Malaga), J. M. Sirvent (Josep Trueta Hospital, Girona), M. Bodí (Joan XXIII Hospital, Tarragona), J. Almirall (Mataró Hospital, Mataró, Barcelona), A. Doblas (Juan Ramon imenez Hospital, Huelva), J. R. Badía (Clinic Hospital, Barcelona), F. García (General Hospital, Albacete), A. Mendia (Nuestra Señora de Aranzazu Hospital, San Sebastian), R. Jordá (Son Dureta Hospital, Palma de Mallorca), F. Bobillo (Clinico Hospital, Valladolid), J. Vallés (Hospital Parc Tauli, Sabadell, Barcelona), M. J. Broch (Sagunto Hospital, Valencia), N. Carrasco (Princesa Hospital, Madrid), M. A. Herranz (Rio Hortega Hospital, Valladolid), F. Alvarez Lerma (Del Mar Hospital, Barcelona), E. Messales (Trias i Pujol Hospital, Badalona, Barcelona), B. Alvarez (General Hospital, Alicante), J. C. Robles (Reina Sofia Hospital, Córdoba), E. Maravi (Virgen Del Camino Hospital, Pamplona), F. Barcenilla (Arnau de Vilanova Hospital, Lleida), M. A. Blasco (Peset Aleixandre Hospital, Valencia), G. Masdeu (Verge de la Cinta Hospital, Tortosa, Tarragona), M. J. López Pueyo (General Yague Hospital, Burgos), A. Margarit (Virgen Meritxell Hospital, Andorra), J. Fierro (Poniente Hospital, Almeria), F. Renedo (Leon Hospital, Leon), A. Lores (Bellvitge Hospital, Barcelona), R. Alonso (General de Asturias Hospital, Oviedo), M. J. Huertos (Puerto Real Hospital, Cadiz), and M. J. López (General Hospital, Segovia).
Rights and permissions
About this article
Cite this article
Rodriguez, A., Lisboa, T., Blot, S. et al. Mortality in ICU patients with bacterial community-acquired pneumonia: when antibiotics are not enough. Intensive Care Med 35, 430–438 (2009). https://doi.org/10.1007/s00134-008-1363-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-008-1363-6