Abstract
Objective
To determine whether severity and organ failure scores over the first 3 days in an ICU predict in-hospital mortality in onco-hematological malignancy patients.
Design and setting
Retrospective study in a 22-bed medical ICU.
Patients
92 consecutive patients with onco-hematological malignancies including 20 hematopoietic stem cell transplantation (HSCT) patients (11 with allogenic HSCT).
Measurements
Simplified Acute Physiology Score (SAPS) II, Organ Dysfunction and/or Infection (ODIN) score, Logistic Organ Dysfunction System (LODS), and Sequential Organ Failure Assessment (SOFA) score were recorded on admission. The change in each score (Δ score) during the first 3 days in the ICU was calculated as follows: severity or organ failure score on day 3 minus severity or organ failure score on day 1, divided by severity or organ failure score on day 1.
Results
In-hospital mortality was 58%. Using multivariate analysis in-hospital mortality was predicted by all scores on day 1 and all Δ scores. Areas under the receiver operating characteristics curves were similar for SAPS II (0.78), ODIN (0.78), LODS (0.83), and SOFA (0.78) scores at day 1. They were also similar for ΔSAPS II, ΔODIN, ΔLODS, and ΔSOFA. Similar results were observed when excluding patients with allogenic HSCT.
Conclusion
Severity and three organ failure scores on day 1 and Δ scores perform similarly in predicting in-hospital mortality in ICU onco-hematological malignancy patients but do not predict individual outcome. Decision to admit such patients to the ICU or to forgo life-sustaining therapies should not be based on these scores.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Admission of onco-hematological patients to the intensive care unit (ICU) frequently involves extensive technological and costly resources. Therefore the decision over such admission may present an ethical dilemma to oncologists and ICU physicians [1, 2]. Several studies investigating selection decisions for ICU admission report that critically ill cancer patients are less likely to be admitted to the ICU [3, 4]. Nevertheless the introduction of new treatment protocols, including bone marrow transplantation and early ICU admission, could result in better survival in onco-hematological patients [5, 6, 7, 8]. Moreover, recent studies show that predictive factors for mortality are unrelated to neutropenia, bone marrow transplantation, or underlying onco-hematological malignancy [7, 9, 10, 11, 12, 13].
Severity scores and organ failure scores could be useful in predicting the outcome of onco-hematological patients admitted in the ICU and thus help the medical decision making. Some studies have shown severity scores at admission to be valuable prognostic factors for ICU mortality [8, 14] while other studies report that these scores do not accurately predict outcome [7, 15, 16]. ICU mortality has also been shown to be due primarily to the extent of organ failure rather than to the underlying disease in neutropenic critically ill cancer patients [12, 13]. As in the general ICU population [17, 18], sequential assessment of severity and organ failure scores during the first few ICU days may be of interest to improve their predictive value for outcome [19, 20, 21]. Based on the uncertain prognosis of onco-hematological patients in whom a 4-day trial of full life-support therapy in ICU may be proposed [22], such a sequential scoring approach could therefore be more relevant.
This study investigated whether severity and organ failure scores at admission and the changes in these over the first 3 days in the ICU are reliable predictive factors of outcome in a specific onco-hematological malignancy population admitted in the ICU.
Patients and methods
Study design and data collection
The study was performed in the medical ICU of Charles Nicolle Hospital of Rouen University, including 22 acute care and 6 post-acute care beds. A mean of 900 patients per year are admitted in the ICU, including 30 patients with onco-hematological disorders usually referred from the Henri Becquerel Regional Cancer Hospital. The ICU policy is to admit unreservedly all patients who are proposed by onco-hematologists, and for whom a life-expectancy for more than 6 months is expected.
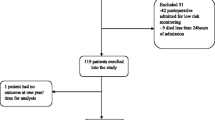
Table 1 presents the characteristics of the 92 consecutive patients with onco-hematological malignancies admitted to the ICU between January 2000 and July 2003. The main onco-hematological malignancies were acute leukemia and lymphoma. Twenty patients (22%) benefited from hematopoietic stem cell transplantation (HSCT), including 10 autologous (11%) and 11 (12%) allogenic (one benefited from both). More than one-half of the patients were admitted for acute respiratory failure. Two-thirds of the patients required mechanical ventilation on day 1, including 7 of the 28 patients (25%) who were switched from noninvasive ventilation (NIV) to invasive mechanical ventilation for worsening. One-half of the patients received vasopressor agents on day 1, and 30 (33%) needed renal replacement therapy during ICU stay.
Patients' medical flowcharts were reviewed retrospectively; we did not consider readmissions. The following clinical data were collected: clinical characteristics at admission including gender and age; type of onco-hematological malignancy with or without neutropenia (neutrophil count < 1,000 /mm3) or (HSCT); reasons for admission including acute respiratory failure, shock, sepsis, acute renal failure or coma. We also collected supportive therapies at admission or during ICU stay including mechanical ventilation, vasopressors use, renal replacement therapy, and clinical outcome including length of ICU stay; ICU mortality and in-hospital mortality. Three organ failure scores and one general severity score were calculated on admission (day 1) and on day 3: Organ Dysfunction and/or Infection (ODIN) score [23], Logistic Organ Dysfunction System (LODS) [24], Sequential Organ Failure Assessment (SOFA) score [25], and Simplified Acute Physiology Score (SAPS) II [26]. The change in each of these scores during the first three ICU days was defined as the delta score (ΔSAPSII, ΔODIN, ΔLODS, and ΔSOFA) and calculated as follows: severity or organ failure score on day 3 minus severity or organ failure score on day 1, divided by severity or organ failure score on day 1. Each Δ score was constructed as a ratio, rather than a difference, to avoid grouping together patients with a same absolute score change but with a widely different degree of severity or organ failure at admission.
Statistical analysis
The values of continuous variables with normal distribution are presented as mean ± SD and those of variables with nonnormal distribution as median and ranges. We performed a logistic regression analysis to determine the predictive factors for in-hospital mortality. Odds ratio (OR) values for scores and Δ scores are all expressed as point value. A subgroup analysis was performed in patients without allogenic HSCT. We tested the validity of the logistic regression model by the Hosmer-Lemeshow goodness of fit test [27]. For seven of the variables—SAPSII, ODIN, SOFA, ΔSAPSII, ΔODIN, ΔLODS, ΔSOFA—the linear model fitted quite well. The p values were 0.86 for ΔLODS, 0.84 for SAPSII, 0.81 for ΔSAPSII, 0.42 for SOFA, 0.34 for ODIN, 0.19 for ΔSOFA, and 0.07 for ΔODIN. For LODS, as the linear model did not fit (p = 0.03), we added a quadratic term into the logistic regression. The model then fitted, as the goodness of fit test then yielded p = 0.74.
Characteristics of the underlying hematological disease and reasons for admission with p value less than 0.20 in the univariate analysis were entered into the stepwise logistic regression analysis to define a base model. Each score and Δ score were then added into the base model. For each logistic model we report the OR and associated 95% confidence interval (CI) as well as p value from the goodness of fit test. For LODS we report the β parameters of the LODS and LODS2 logistic regression equations, and we report OR for a 1 unit increase in LODS for a patient at the 7.7 score (mean of LODS scores for our patients).
Discrimination between scores on day 1 and between Δ scores during the first 3 days in the ICU for predicting in-hospital mortality was assessed by constructing receiver operating characteristic (ROC) curves. Areas under the ROC curves (AUC) were calculated to achieve a global measure of the score discrimination. The AUC values were then compared pairwise using the contrasts on the basis of a nonparametric Mann-Whitney U test [28]. In-hospital mortality was the outcome variable of interest. All tests were two-tailed, and p values less than 0.05 were considered statistically significant. Analyses were performed using the Stata 8.0 (Stata, College Station, Tex., USA).
Results
Outcome data, scores at baseline, and Δ scores over the first 3 days in the ICU are shown in Table 1. Eleven patients died before the third day, and 12 were discharged from the ICU before day 3. Therefore information on severity and organ failure scores was not available on day 3 for 23 patients, and Δ scores were assessed for 69 patients in the overall population. The overall ICU and in-hospital mortality rates were 50% and 58%, respectively. Allogenic HSCT patients had the highest in-hospital mortality rate (91%). On admission SAPS II and organ failure scores (ODIN, LODS, SOFA) were significantly lower in survivors than in nonsurvivors (p < 0.0001): 48 ± 12 vs. 69 ± 23, 2 ± 1 vs. 3 ± 1, 5 ± 3 vs. 10 ± 6, 7 ± 3 vs. 11 ± 5, respectively. In general, ΔSAPSII, ΔODIN, ΔLODS, and ΔSOFA worsened in nonsurvivors and improved in survivors: –12 ± 18% vs. +13 ± 37%, –5 ± 41% vs. +48 ± 96%, –8 ± 56% vs. +56 ± 155%, –1 ± 4% vs. +20 ± 35%, respectively. The individual distribution of ΔSAPSII, ΔODIN, ΔLODS, and ΔSOFA values for survivors and nonsurvivors is displayed in Fig. 1.
Table 2 presents the results of univariate analysis of prognostic factors regarding in-hospital mortality in the overall population. Admission for severe sepsis or septic shock (p = 0.01), neutropenia (p = 0.03), and presence of HSCT (p = 0.006) or allogenic HSCT (p = 0.009) were found to predict in-hospital mortality. Among supportive treatments, in-hospital mortality was significantly associated with the need for vasopressors at admission (78% of patients who needed vasopressors died vs. 40% of patients who did not, p < 0.0001) and for invasive mechanical ventilation at admission (82% of patients who needed invasive mechanical ventilation died vs. 41% who did not, p = 0.0001). Use of NIV on day 1 was not associated with poor prognosis (54% of patients who needed NIV died vs. 59% who did, p = 0.65). All severity and organ failure scores at admission were found to be significant predictive factors for in-hospital mortality (p < 0.0001) as well as all Δ scores (p < 0.01) in patients who were still present on day 3 in the ICU.
Table 3 shows the logistic models used for multivariate analysis. In the overall population SAPSII (p < 0.0001), ODIN (p < 0.0001), LODS (p < 0.0001), and SOFA (p < 0.0001) scores at ICU admission and every Δ score (p < 0.05) were found to be independent predictive factors for in-hospital mortality. Discrimination between the different scores to predict in-hospital mortality was similar since AUCs for baseline scores at admission as well as for their Δ scores (Table 4; see Electronic Supplementary Material Fig. S2).
The subgroup analysis in patients without allogenic HSCT involved 81 patients on day 1 and 63 on day 3. Univariate analysis showed the following baseline scores on day 1 to be significant predictive factors of in-hospital mortality: SAPSII (OR 1.07, 95%CI 1.03–1.11, p < 0.0001, goodness-of-fit p = 0.92), ODIN (OR 2.69, 95%CI 1.69–4.29, p < 0.001, goodness-of-fit p = 0.48), LODS (OR 1.55, 95%CI 1.16–2.08, p < 0.0001, goodness-of-fit p = 0.99), and SOFA (OR 1.35, 95%CI 1.17–1.56, p < 0.0001, goodness-of-fit p = 0.32). The following Δ scores were found to be significant predictive factors: ΔSAPS II (OR 134.95, 95%CI 5.08–3586.04, p = 0.003, goodness-of-fit p = 0.66), ΔODIN (OR 3.87, 95%CI 1.17–12.76, p = 0.03, goodness-of-fit p = 0.24), ΔLODS (OR 2.71, 95%CI 0.96–7.65, p = 0.06, goodness-of-fit p = 0.32), and ΔSOFA (OR 6.63 95%CI 1.18–37.21, p = 0.03, goodness-of-fit p = 0.53). Multivariate analysis (Table 5) showed that scores and Δ scores were independent predictive factors for in-hospital mortality. In this subset of patients none of the score at admission or of the Δ score was better than the others to predict death based on comparison between AUC values.
Discussion
To our knowledge, this is the first study using one severity (SAPS II) and three organ failure (ODIN, LODS, SOFA) scores at admission and over the first 3 days in the ICU to identify predictive factors for in-hospital mortality in a selective onco-hematological malignancy population. Our results show that these scores at admission and their change in patients still present on day 3 are independent predictive factors of in-hospital mortality. None of these scores appears to be superior to the others in predicting outcome. Furthermore, similar results were observed after excluding patients with allogenic HSCT. The Δ scores provided no further outcome information in patients with onco-hematological malignancies.
Since the prognosis of the underlying disease per se does not have a significant impact on mortality in neutropenic critically ill cancer patients [12, 13], general severity and organ failure scores have been proposed to predict outcome in these patients. Discordant results have been reported either for these general severity [7, 8, 14, 15, 16] and organ failure scores [12, 13]. However, meaningful comparisons between these studies are difficult due to their varying designs, populations, admission policies, and statistical analyses. One more specific and complex severity of illness score has also been developed. The ICU Cancer Mortality Model (CMM), a multivariable logistic regression model, was found to provide accurate prediction of in-hospital mortality in critically ill cancer patients with solid and hematological malignancies after ICU admission [29]. More recent studies, however, have found the CCM to be either inaccurate in predicting in-hospital mortality [15] or to not provide improved prediction over that of other severity scores [19, 30] such as SAPS II and Acute Physiology and Chronic Health Evaluation II [31].
Only a few studies have evaluated sequential assessment of severity or organ failure scores during the first ICU days to predict outcome in critically ill patients [17, 18, 20, 21]. In a general ICU population a composite score using daily SAPS II and LODS scores was found to accurately predict in-hospital mortality in patients hospitalized longer than 3 days [17]. This accuracy was found as good and similar when using daily LODS and SOFA scores [19]. In 94 neutropenic cancer patients most of whom were suffering from onco-hematological malignancies (55%), multiple assessments of SAPS, SAPS II, and number of organ system failure (OSF) measures provided accurate assessment of severity of illness and risk of death [20]. In addition, change in OSF measurements over the first 3 days in the ICU provided further information, allowing classification of patients into groups with different probability of in-hospital mortality. More recently the ΔLODS over the first 3 days in the ICU has been demonstrated to be an independent predictive factor of 30-day mortality in cancer patients with septic shock whereas LODS at admission was not a predictive factor [21].
Our overall in-hospital mortality rate (58%) was similar to that reported in previous studies investigating changes in scores among critically ill cancer patients [20, 21]. One finding of the present study is that Δ scores provide no additional information for outcome evaluation in this specific onco-hematological population over that of baseline scores. This finding is not consistent with previous studies [17, 20, 21] showing that changes in organ failure scores during the first few ICU days predicted survival more accurately than did these scores when measured at ICU admission. Several explanations can be considered. First, from a methodological point of view, we did not strictly compare scores on day 1 with Δ scores on day 3 using AUC values since the Δ score calculation was closely derived from that of baseline scores. Nevertheless, global discrimination to accurately predict outcome as reflected by AUC values was found in the overall population to be better for the different scores (0.78–0.83) than for Δ scores (0.67–0.72). Second, the critically ill cancer population that we studied did not include patients with solid tumor, in contrast to the case with other investigators [20]. Third, our onco-hematological population included many cases of HSCT (22%), 11 of them allogenic. None of the previous studies assessing changes in scoring systems in ICU cancer patients [12, 19] included allogenic HSCT, a specific population with a well known poor prognosis [7, 9]. This was confirmed in our study since the in-hospital mortality rate was 91% in this subset of patients, and since one-half of them died in the ICU before the third day. To minimize the effect of this potential bias on our results we performed a subgroup analysis by excluding patients with allogenic HSCT. However, the value of baseline scores and Δ scores to predict outcome was found to be similar to that obtained in the overall population. Another striking finding of the present study is that the outcome predictive value of SAPS II on admission or of its change over the first 3 days in the ICU was of similar significance than that obtained for three organ failure scores (ODIN, LODS, SOFA) or of their changes over this period. Finally, to assess prognosis of onco-hematological patients at ICU admission intensivists have the choice between one severity of illness score (SAPS II) and one or more organ failure scores (ODIN, LODS, SOFA). Based on our results the reassessment of these scores on day 3 would be unnecessary, and if reassessment is performed, only one of the scores need be used.
Several limitations of the present study must be considered. First, the retrospective design of the study is a limitation per se, although there were no missing data for the calculation of any scoring systems. Second, we enrolled a relatively limited number of patients on admission (n = 92), and therefore only 69 patients were still present in the ICU on day 3 to calculate Δ scores. Our findings are nevertheless similar to those reported in previous studies [20, 21]. Third, we used severity and organ dysfunction scores originally developed in a general ICU population [23, 24, 25, 26]. These scores have been also widely used in most studies involving critically ill cancer patients [7, 12, 13, 14, 15, 16, 20, 21]. Fourth, since this was a single-center study, we cannot formally exclude a selection bias due to admission, do-not-resuscitate orders, and end-of-life decisions policy as well as treatment strategies. Fifth, we did not find evidence of superiority of Δ scores over scores calculated at admission. However, we chose to reassess scores on day 3, and we cannot exclude that reassessment at a later period (e.g., day 4 or 5) would have led to different results.
In conclusion, SAPS II, ODIN, LODS, and SOFA on day 1 as well as their respective change over the first 3 days in the ICU are independent predictive factors for in-hospital mortality of onco-hematological malignancy patients with or without allogenic HSCT, admitted in the ICU. All the studied scores on day 1 and their respective changes over the first three ICU days seem to perform similarly to predict in-hospital survival. However, performance of scores at admission and on day 3 are of limited value to help in the decision making process in critically ill onco-hematological malignancy patients. A decision to forgo life sustaining therapies in each individual patient should rely on the clinical context, the reversibility of the acute medical disease, and the discussion among the multidisciplinary team, taking into account patient's preferences and values.
References
Task Force of the American College of Critical Care Medicine, Society of Critical Care Medicine (1999) Guidelines for intensive care unit admission, discharge, and triage. Crit Care Med 26:633–638
Society of Critical Care Medicine Ethics Committee (1994) Consensus statement on the triage of critically ill patients. JAMA 271:1200–1203
Azoulay E, Pochard F, Chevret S, Vinsonneau C, Garrouste M, Cohen Y, Thuong M, Paugam C, Apperre C, De Cagny B, Brun F, Bornstain C, Parrot A, Thamion F, Lacherade JC, Bouffard Y, Le Gall JR, Herve C, Grassin M, Zittoun R, Schlemmer B, Dhainaut JF, Protocetic Group (2001) Compliance with triage to intensive care recommandations. Crit Care Med 29:2132–2136
Sprung CL, Geber D, Eidelman LA, Baras M, Pizov R, Nimrod A, Oppenheim A, Epstein L, Cotev S (1999) Evaluation of triage decisions for intensive care admission. Crit Care Med 27:1073–1079
Azoulay E, Recher C, Alberti C, Soufir L, Leleu G, Le Gall JR, Fermand JP, Schlemmer B (1999) Changing use of intensive care for hematological patients: the example of multiple myeloma. Intensive Care Med 25:1395–1401
Kress JP, Christenson J, Pohlman AS, Linkin DR, Hall JB (1999) Outcomes of critically ill cancer patients in a university hospital setting. Am J Respir Crit Care Med 160:1957–1961
Staudinger T, Stoiser B, Mullner M, Locker GJ, Laczika K, Knapp S, Burgmann H, Wilfing A, Kofler J, Thalhammer F, Frass M (2000) Outcome and prognostic factors in critically ill cancer patients admitted to the intensive care unit. Crit Care Med 28:1322–1328
Kroschinsky F, Weise M, Illmer T, Haenel M, Bornhaeuser M, Hoeffken G, Ehninger G, Schuler U (2002) Outcome and prognostic features of intensive care unit treatment in patients with haematological malignancies. Intensive Care Med 28:1294–1300
Price KJ, Thall PF, Kish SK, Shannon VR, Andersson BS (1998) Prognostic indicators for blood and marrow transplant patients admitted to an intensive care unit. Am J Respir Crit Care Med 158:876–884
Darmon M, Azoulay E, Alberti C, Fieux F, Moreau D, Le Gall JR, Schlemmer B (2002) Impact of neutropenia duration on short-term mortality in neutropenic critically ill cancer patients. Intensive Care Med 28:1775–1780
Khassawneh BY, White P Jr, Anaissie EJ, Barlogie B, Hiller FC (2002) Outcome from mechanical ventilation after autologous peripheral blood stem cell transplantation. Chest 121:185–188
Blot F, Guiguet M, Nitenberg G, Leclercq B, Gachot B, Escudier B (1997) Prognostic factors for neutropenic patients in an intensive care unit: respective roles of underlying malignancies and acute organ failures. Eur J Cancer 33:1031–1037
Massion PB, Dive AM, Doyen C, Bulpa P, Jamart J, Bosly A, Installe E (2002) Prognosis of hematologic malignancies does not predict intensive care unit mortality. Crit Care Med 30:2260–2270
Sculier JP, Paesmans M, Markiewicz E, Berghmans T (2000) Scoring systems in cancer patients admitted for an acute complication in a medical intensive care unit. Crit Care Med 28:2786–2792
Soares M, Fontes F, Dantas J, Gadelha D, Cariello P, Nardes F, Amorim C, Toscano L, Rocco JR (2004) Performance of six severity-of-illness scores in cancer patients requiring admission to the intensive care unit: a prospective observational study. Crit Care 8:R194–R203
Benoit DD, Vandewoude KH, Decruyenaere JM, Hoste EA, Colardyn FA (2003) Outcome and early prognostic indicators in patients with a hematologic malignancy admitted to the intensive care unit for a life-threatening complication. Crit Care Med 31:104–112
Timsit JF, Fosse JP, Troche G, De Lassence A, Alberti C, Garrouste-Orgeas M, Azoulay E, Chevret S, Moine P, Cohen Y (2001) Accuracy of a composite score using daily SAPS II and LOD scores for predicting hospital mortality in ICU patients hospitalized for more than 72 h. Intensive Care Med 27:1012–1021
Timsit JF, Fosse JP, Troche G, De Lassence A, Alberti C, Garrouste-Orgeas M, Bornstain C, Adrie C, Cheval C, Chevret S, for the OUTCOMEREA Study Group, France (2002) Calibration and discrimination by daily Logistic Organ Dysfunction scoring comparatively with daily Sequential Organ Failure Assessment scoring for predicting hospital mortality in critically ill patients. Crit Care Med 30:2003–2013
Schellongowski P, Benesch M, Lang T, Traunmuller F, Zauner C, Laczika K, Locker GJ, Frass M, Staudinger T (2004) Comparison of three severity scores for critically ill cancer patients. Intensive Care Med 30:430–436
Guiguet M, Blot F, Escudier B, Antoun S, Leclercq B, Nitenberg G (1998) Severity of 2illness scores for neutropenic cancer patients in an intensive care unit: which is the best predictor? Do multiple assessment times improve the predictive value? Crit Care Med 26:488–493
Larche J, Azoulay E, Fieux F, Mesnard L, Moreau D, Thiery G, Darmon M, Le Gall JR, Schlemmer B (2003) Improved survival of critically ill cancer patients with septic shock. Intensive Care Med 29:1688–1695
Rubenfeld GD, Crawford SW (1996) Withdrawing life support from mechanically ventilated recipients of bone marrow transplants: a case for evidence-based guidelines. Ann Intern Med 125:625–633
Fagon JY, Chastre J, Novara A, Medioni P, Gibert C (1993) Characterization of intensive care unit patients using a model based on the presence or absence of organ dysfunctions and/or infection: the ODIN model. Intensive Care Med 19:137–144
Le Gall JR, Klar J, Lemeshow S, Saulnier F, Alberti C, Artigas A, Teres D (1996) The Logistic Organ Dysfunction System. A new way to assess organ dysfunction in the intensive care unit. JAMA 276:802–810
Vincent JL, Moreno R, Takala J, Willatts S, De Mendonca A, Bruining H, Reinhart CK, Suter PM, Thijs LG (1996) The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure: on behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 22:707–710
Le Gall JR, Lemeshow S, Saulnier F (1993) A new simplified acute physiologic score (SAPS II) based on a European/North American multicenter study. JAMA 270:2957–2964
Lemeshow S, Hosmer DW Jr (1982) A review of goodness-of-fit statistics for use in the development of logistic regression models. Am J Epidemiol 115:92–106
DeLong ER, DeLong DM, Clarke-Pearson DL (1988) Comparing the areas under two or more correlated receiver operating characteristics curves: a non parametric approach. Biometrics 44:837–845
Groeger JS, Lemeshow S, Price K, Nierman DM, White P Jr, Klar J, Granovsky S, Horak D, Kish SK (1998) Multicenter outcome study of cancer patients admitted to the intensive care unit: a probability of mortality model. J Clin Oncol 16:761–770
Berghmans T, Paesmans M, Sculier JP (2004) Is a specific oncological scoring system better at predicting the prognosis of cancer patients admitted for an acute medical complication in an intensive care unit than general gravity scores? Support Care Cancer 12:234–239
Knaus WA, Draper EA, Wagner DP, Zimmerman JE (1985) APACHE II: a severity of disease classification system. Crit Care Med 13:818–829
Acknowledgements
We thank Jean-Louis Teboul for his fruitful comments for reviewing the manuscript and Mary Kenny for her valuable advice for editing the manuscript. Part of this study was presented in abstract form at the American Thoracic Society International Conference at San Diego in May 2005 (American Journal of Respiratory and Critical Care Medicine, vol. 2, A430).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Lamia, B., Hellot, MF., Girault, C. et al. Changes in severity and organ failure scores as prognostic factors in onco-hematological malignancy patients admitted to the ICU. Intensive Care Med 32, 1560–1568 (2006). https://doi.org/10.1007/s00134-006-0286-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-006-0286-3