Abstract
Objective
To assess the diagnostic value of a single determination of serum C-reactive protein as a marker of sepsis in critically ill patients.
Design
Prospective, observational study.
Setting
Intensive care unit of a university hospital.
Patients and participants
One hundred twenty-five adult patients with systemic inflammatory response syndrome (SIRS) (55 patients without evidence of infection and 70 patients with the diagnosis of sepsis confirmed by documented infection). Twenty-five patients with non-complicated acute myocardial infarctions (AMI) and 50 healthy volunteers were used as controls.
Interventions
None.
Measurements and results
Serum C-reactive protein concentration was measured within the first 24 h of SIRS onset. Healthy subjects, AMI and non-infectious SIRS patients showed lower C-reactive protein median values ([(0.21 [95% confidence intervals (95% CI), 0.21–0.4] mg/dl, 2.2 [95% CI, 2.1–4.9] mg/dl and 1.7 [95% CI, 2.4–5.5] mg/dl, respectively) than patients with sepsis (18.9 [95% CI, 17.1–21.8]), p<0.001. The presence of severe sepsis (rs=0.27; p=0.03), SOFA score (r s =0.25; p=0.03) and arterial lactate (r s =0.24; p=0.04) correlated significantly with C-reactive protein concentrations in sepsis cases. The best threshold value for C-reactive protein for predicting sepsis was 8 mg/dl (sensitivity 94.3%, specificity 87.3%). The area under the receiver-operating characteristic curve for C-reactive protein was 0.94 (95% CI, 0.89–0.98).
Conclusions
Determination of serum C-reactive protein can be used as an early indicator of infection in patients with SIRS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sepsis, clinically defined as the systemic inflammatory response syndrome (SIRS) secondary to infection [1, 2], is an important cause of death and increased morbidity and cost in the ICU [3]. Delay in initiating adequate treatment or source control increases the mortality rate [4, 5]. Therefore, early identification of sepsis should be a priority in clinical practice and constitutes a primary goal in the guidelines for the management of sepsis [6]. A classification scheme for sepsis (PIRO) [2] has recently been developed. Host response, although difficult to characterize, is currently considered a target in therapies for sepsis [2]. C-reactive protein (CrP), a pentraxin [7] released by the liver during the acute-phase response (APR) after insults such as significant inflammation or tissue damage [8, 9], is a potential marker of the presence of infection and severity of sepsis [2, 8]. An elevated blood CrP concentration is thought to be highly suggestive of bacterial infection, though other non-infectious conditions may also elevate CrP [8, 9]. Adding a single laboratory inflammatory marker like blood CrP to SIRS criteria may improve the diagnosis of sepsis and may help in the decision to start appropriate antimicrobial therapy.
The aim of this study was to assess the predictive value of a single measurement of serum CrP (sCrP) in patients with signs of SIRS, as a diagnostic test for sepsis. Our hypothesis was that a single sCrP value can be used to identify the presence of infection within 24 h of developing SIRS criteria, much in the same way as troponin is used for diagnosing acute myocardial infarction (AMI).
Material and methods
We undertook a prospective observational study in the intensive care unit (ICU) of a university teaching hospital.
Study subjects
Critically ill adult patients admitted to the ICU were included in the study. Four groups were defined: groups 1 and 2 included patients with early-onset SIRS, with a distinction made between infected (group 1, i.e. sepsis) and non-infected patients (group 2). Two other groups were designated as controls, a non-systemic inflammatory condition (group 3, patients with non-complicated AMI [48-h evolution]) and a healthy state (group 4, adult volunteers) that was used only to set normal marker values. Exclusion criteria were age under 14 years and pregnancy. The need for informed consent was waived by the ethics board which approved the study.
Measurements
Patients showing two or more signs of SIRS for between 4 h and 24 h were included in SIRS group 1 (if sepsis was diagnosed) or SIRS group 2 (if not). A thorough examination was performed of every patient with SIRS, looking for signs of infection. Only the first infectious episode was considered. Patients who were receiving antimicrobial therapy were not included. At the time of patient enrollment, vital signs were recorded and blood samples were simultaneously collected to determine sCrP, complete blood count, creatinine, arterial blood gases and lactate. A chest radiograph was obtained in all cases. Samples of blood (three aerobic and anaerobic media), urine and other fluids and tissues suspected of infection were collected for cultures and microbiological diagnosis. Scheduled antibiotics were given soon after the data and sample collection whenever an infection was suspected. All laboratory measurements were performed immediately and microbiological samples were promptly processed.
Definitions
Systemic inflammatory response syndrome was defined by at least two of the following criteria [1]: heart rate higher than 90 beats/min, respiratory rate more than 20 breaths/min or mechanical ventilation (MV), body temperature above 38°C or below 36°C, white blood cell counts (WBC) more than 12,000/mm3 or less than 4,000/mm3. Sepsis was defined as SIRS secondary to a documented infection confirmed by a microbiological diagnosis (i.e. infectious SIRS) [1]. Sepsis was considered as severe when its signs were associated with hypotension, hypoperfusion or organ dysfunction [1]. Ventilator-associated pneumonia (VAP) was clinically suspected, as reported elsewhere [10]. A microbiological diagnosis of VAP was considered positive when quantitative cultures yielded 106 colony forming units (cfu)/ml or more for endotracheal aspirates [11] or 103 cfu/ml or more for protected specimen brush sampling. Severity of illness at the time of ICU admission was measured by the Acute Physiology and Chronic Health Evaluation (APACHE) II score and additionally by the Injury Severity Score (ISS) in trauma patients. The presence of any organ dysfunction was documented in all patients using the Sequential Organ Failure Assessment (SOFA) scale [12].
Laboratory tests
The sCrP concentration was measured by a Boehringer Mannheim (BM)/Hitachi automated immunoturbidimetric (Tina-quant, BM, Germany) technique. The CrP assay precision was calculated from replicate determinations in sera from human control samples in a single assay. The mean intra-assay and inter-assay coefficients of variation were 1.3% and 2%, respectively. The lower detection limit was set at 0.2 mg/dl. The other biochemical parameters and blood counts were assessed by routine laboratory procedures. Samples obtained for microbiological analysis were processed following normal procedures and all microorganisms isolated were identified by standard laboratory methods.
Statistical analysis
The data are presented as median values and 95% confidence intervals (CI) for continuous variables unless otherwise indicated, and percentages for categorical variables. The statistics were computed with the Statistical Package for Social Sciences (SPSS) version 10.0 software (SPSS, Chicago, lL, USA). Data distribution was tested and appropriate tests were used to evaluate the differences between groups. The association between variables was assessed by Spearman’s correlation test (r s ). The logistic regression coefficient was used to estimate the odds ratio (OR) (with 95% CI) for the diagnostic marker with microbiological confirmation of infection as the dependent variable. The data were analyzed by diagnostic efficiency derived from the receiver operating characteristic (ROC) curve and area under the ROC curve (AUC). All statistical tests were two-tailed and p less than 0.05 was considered significant.
Results
Study population characteristics
Two hundred subjects assigned to four groups were studied: 70 infected patients with SIRS (sepsis), 55 non-infected patients with SIRS, 25 patients with non-complicated AMI diagnoses and 50 healthy volunteers. The last two groups served as two-step controls for sCrP values. So, 125 patients had SIRS for which a diagnosis of sepsis was sought.
Control groups
Healthy volunteers
Fifty healthy adults donated blood samples for analysis of sCrP. The mean age of the donors was 35 years (range 29–61); 64% were males and 36% females. Their sCrP concentrations were 0.21 (0.21–0.40) mg/dl, which were lower than those of the other groups (p<0.001) (Fig. 1).
Box-plot distribution with interquartile range of C-reactive protein values in the different study groups. The horizontal lines from the bottom to the top represent 10th, 25th, 50th, 75th and 90th percentiles. The 50th ones are median values. Outliers are represented by circles. Healthy volunteer controls (n=50), AMI acute myocardial infarction (n=25), SIRS non-infectious systemic inflammatory response syndrome (n=55), sepsis (n=70). p<0.001 between all groups except AMI versus SIRS groups with p=0.39
Non-complicated acute myocardial infarction patients
Twenty-five patients with a diagnosis of non-complicated AMI without SIRS were studied. The electrocardiographic localization of infarction was anterior in 10 cases and posterior and/or inferior in the other 15. Their mean age and APACHE II were 63.2 years (range 32–81) and 10.5 (95% CI, 7.8–13.1), respectively. Blood collected at 48 h from ICU admission showed sCrP concentrations of 2.2 (2.1–4.9) mg/dl (Fig. 1).
Systemic inflammatory response syndrome groups: infected (sepsis) and non-infected patients
The admission diagnoses of the 125 SIRS patients (separated into non-infected and infected cases) were: multiple trauma (n=38 [13+25]), head injury (n=32 [14+18]), early postoperative course of cardiovascular surgery with cardiopulmonary bypass (coronary artery bypass grafting or valve replacement; n=24 [18+6]), cerebrovascular accidents (n=16 [4+12]), congestive heart failure with coronary heart disease (n=6 [4+2]), acute respiratory failure (n=6 [3+3]) and acute meningitis (n=3 [0+3]). Other characteristics of the SIRS patients with and without infection are compared in Table 1. The mean interval of time between ICU admission and diagnosis of SIRS was 3.2 days (95% CI, 1.1–5.3). SIRS was detected during the first 48 h following ICU admission in 64 out of the 125 patients. A few documented infections (8%) were considered community-acquired, although SIRS onset occurred within the first 2 days of ICU stay in 53% of the infected cases. Seventy patients with SIRS had diagnoses of severe trauma. The quantity of infected and non-infected SIRS trauma patients was comparable (43 out of 70 vs 27 out of 55, respectively; p=0.34). ISS values were similar in the infected and non-infected SIRS trauma groups (28 [26.1–30.9] vs 26 [27.1–34.1]; p=0.57). Twenty-two (31.4%) patients in the sepsis group had severe sepsis syndrome. Severe sepsis patients showed higher arterial lactate concentrations than those in the non-severe sepsis subgroup (1.91 [1.17–4.11] mmol/l vs 1.28 [1.16–1.43] mmol/l; p=0.012).
Serum C-reactive protein values in systemic inflammatory response syndrome groups
Serum C-reactive protein concentrations were higher in the sepsis group (18.9 [17.1–21.8] mg/dl) than in the non-infectious SIRS group (1.7 [2.4–5.5] mg/dl; p<0.001; Fig. 1). SOFA scores and arterial lactate concentrations correlated significantly with sCrP concentrations in sepsis cases (r s =0.25; p=0.03 and r s =0.24; p=0.04, respectively).
Post hoc comparison of sepsis subgroups
No differences in sCrP levels were found within the SIRS subgroups with two, three and four signs of SIRS, respectively (Table 2).
Systemic inflammatory response syndrome patients were also divided in two age-related subgroups (under 60 years and 60 years or older) and the corresponding sCrP concentrations were compared (Table 3). sCrP levels did not differ significantly in SIRS patients aged under 60 and in the patients over this age. Besides, sCrP concentrations were also statistically similar in the age-related subgroups of septic patients. Among all 125 SIRS patients, sCrP values were higher in the patients who did not undergo surgery than in those who did, but sCrP concentrations were similar in those septic patients who had recently undergone surgery and those who had not (Table 3). Septic trauma patients had lower sCrP concentrations, although sCrP values in trauma patients were even lower when they had no sepsis (Table 3).
The presence of severe sepsis correlated with sCrP levels (r s =0.27; p=0.03). The severe sepsis subgroup showed higher sCrP concentrations (22.6 [18.3–30.4] mg/dl) than the other 48 sepsis cases (16.5 [15.2–19.2] mg/dl; p=0.03). Gram-positive and Gram-negative bacteria infections exhibited similar sCrP values (19.1 [16.6–22.1] mg/dl vs 16.6 [14.5–22.6] mg/dl, respectively; p>0.5). Bronchopulmonary infections had lower sCrP concentrations (16.5 [15–19.9] mg/dl) than infections at the other sites (21.3 [18.5–29.1] mg/dl; p<0.05), but the frequency of pneumonia was higher in trauma patients than in the non-trauma subgroup (48.6% vs 29.2%, respectively; p<0.01). Nevertheless, sCrP concentrations of trauma and non-trauma sepsis with pneumonia were not significantly different (13.9 [13.8–18.4] vs 20.5 [14.1–27.4] mg/dl, respectively; p=0.11). There were no statistical differences in sCrP values between bacteremic and non-bacteremic sepsis cases (20.7 [17–25.2] mg/dl vs 18.1 [15.6–21.7] mg/dl; p=0.16, respectively).
Infections: etiologies and sites
Eighty-six infections, all with bacterial isolates, were diagnosed in the 70 sepsis cases. Gram-positive bacteria were considered responsible for infections in 65.7% of the sepsis cases and Gram-negative bacteria for 42.9%. Both Gram-positive and Gram-negative bacteria were isolated in 8.6% of the septic patients.
The lungs were the most frequent site of infection. Bacteria were isolated in respiratory secretions in 68.6% of the cases of sepsis and the lungs were the sole infection location in 54.3%. Bloodstream and urinary tract infections (UTI) were the second and third most frequent sites of infection, respectively (Table 4). Three septic patients had acute bacterial meningitis (ABM). The main foci of the other four patients were acute cholecystitis, subphrenic abscess, infected abdominal wound (IAW) and pleural empyema. Infectious foci associated with bacteremia were VAP in six cases, UTI in three, ABM in one and IAW in one. Five septic patients had two concurrent non-related foci of infection. The type of bacteria and corresponding sites of infection are described in Table 4.
Prediction of sepsis. Diagnostic threshold and accuracy of serum C-reactive protein values
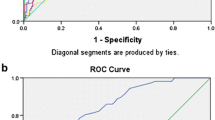
The OR was calculated for sCrP corresponding to a 1 mg/dl increase. The OR of sCrP for microbiologically confirmed sepsis was 1.33 (95% CI, 1.21–1.46). The best cut-off value for sCrP for predicting sepsis in the first 24 h of SIRS onset was 8 mg/dl. The corresponding sensitivity, specificity, predictive values and likelihood ratios of sCrP at that level are shown in Table 5. The AUC for sCrP was 0.94 (95% CI, 0.89–0.98; Fig. 2).
Discussion
This study demonstrates that within a short space of time sCrP levels are significantly higher in patients with sepsis than in patients with non-infectious SIRS. A sCrP value above 8 mg/dl was highly predictive of infection when performed within the first 24 h of developing signs of SIRS. SIRS criteria [1] have been reported to have high sensitivity but poor specificity [2]. Some studies have proposed CrP as a biological marker of infection and a diagnostic criterion for sepsis [2, 13–19], but others [20, 21–24] have drawn attention to its limitations: its slow kinetics and poor diagnostic specificity. Blood CrP is merely a complementary sign of the host response to an infection and it should be interpreted in the clinical setting in combination with SIRS signs [18], other clinical signs of infection and bacteriological data. CrP values cannot be used in isolation to obtain a specific diagnosis, although their value in infectious diseases is beyond doubt [8, 13, 14, 18, 19]. CrP induction requires interleukin (IL)-6 and either IL-1 or tumor necrosis factor-alpha [9]. Therefore, CrP synthesis and secretion usually reflect proinflammatory cytokine production [25, 26] and may be considered its surrogate marker.
Increased sCrP levels associated with infections and sepsis have been described in many studies [13–18, 21–24, 27]. The sCrP data and diagnostic value observed in our non-infected SIRS and sepsis cases are similar to those findings reported by other authors [13–15, 17, 18, 27]. The observation that the elderly have an altered APR, showing a more rapid increase in sCrP levels than younger patients [28], was not corroborated in our patients. No differences were found between sCrP values of patients with infections caused by Gram-positive and -negative bacteria, in spite of reports of differences in inflammatory responses [29]. Some researchers have studied whether sCrP may help to detect bacteremia [22, 30]. Similar to other findings [30], sCrP concentrations were not significantly different in bacteremic sepsis cases.
Some studies have suggested that procalcitonin (PCT) may be a better laboratory marker for the diagnosis of sepsis. The diagnostic performance of PCT appears to be higher than CrP in some studies and similar in others. Variable ranges of AUC, sensitivity and specificity have been described for CrP and PCT [14–18, 20, 21, 23, 24], although these studies present substantial methodological differences. Differences in our diagnostic performance of sCrP with respect to other studies may be related to several facts. First, the inclusion of sepsis cases should avoid the consideration of patients with non-detected sepsis as non-infected SIRS. Second, the times when infection is suspected and sCrP measured are not always reported. Sometimes the first sCrP value may be recorded after the first 24 h of SIRS onset [13, 14, 16–18, 31]. Data from our study were collected within the first day of SIRS onset. Lastly, precision and reproducibility of any diagnostic test result must be detailed in order to facilitate its replication. Characteristics of CrP and PCT assay validation as sepsis diagnostic markers have been described in only some studies [13, 15, 26].
Procalcitonin assay costs have been reported up to five-fold higher than CrP measurements [32, 33]. Moreover, sCrP can be automatically measured on an emergency basis and faster than PCT. Although the combination of CrP with PCT may help to increase the specificity of sCrP as an indicator of infection, it would increase the already high costs of sepsis even further [34].
Some researchers have rejected an association between sCrP and severity and presence of organ dysfunctions [23, 35]. Nevertheless, evidence of a relationship between risk of organ failure and high sCrP concentrations has recently been reported [27]. High circulating levels of IL-6 have been associated with higher mortality in VAP, indicating a greater inflammatory response [36]. In all likelihood, the more CrP the body produces, the sicker it gets [37]. Our severe sepsis cases exhibited higher sCrP concentrations. Furthermore, a relationship was found in the sepsis group between organ dysfunction (i.e. SOFA scores) and extent of APR (i.e. sCrP levels).
This study has some limitations. First, accurate times of SIRS onset and data collection were not recorded, although the 125 patients were enrolled during the first 24 h of SIRS onset. Second, about half of all SIRS patients had diagnoses of trauma. Surgery and trauma are prompters of APR, however sCrP values were not higher in such subgroups. Some studies have considered sCrP to be useful for diagnosing infections in trauma patients, too [16, 31]. We also observed significantly higher sCrP levels in trauma patients when they had sepsis. Third, only the first infectious episode was considered. Besides, most of the sepsis cases corresponded to ICU early-onset infections with a high proportion of lung infections. A considerable number of infections, mainly VAP, occur within the first 7 days of severe acute disease in critically ill patients [38].
An unexpected finding of this study was that sCrP concentrations in pneumonia cases were lower than in cases with other sites of infection. However, the higher proportion of trauma patients with lung infection makes that fact difficult to explain. It is not clear whether the lower sCrP increase in trauma patients with sepsis is related to the presence of pneumonia or it is an effect of trauma. Fourth, most non-infected SIRS and septic patients needed MV because their illness severity was rather high. Fifth, the majority of SIRS and septic patients had an altered level of consciousness, but this was not usually indicative of severe brain dysfunction. The brain induces an APR when injured, although the more severe the brain dysfunction, the greater is the extent of that response [39]. Lastly, the relatively small size of the study sample could also limit the generalizability of our results.
The verification of an episode of infection using culture is a time-consuming process which usually defers the diagnosis of sepsis and so the initiation of appropriate treatment may be delayed. Adding a single sCrP determination to prompt recognition of SIRS and signs of infection [34] appears to improve sepsis diagnosis. The test is cheap, easy to measure, available in many hospitals [19] and could be added to clinical guidelines for the treatment of VAP [4, 40] and other severe infections within early adequate management of sepsis.
References
Bone RC, Balk RA, Cerra FB, Dellinger RP, Fein AM, Knaus WA, Schein RM, Sibbald WJ (1992) Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest 101:1644–1655
Levy MM, Fink MP, Marshall JC, Abraham E, Angus D, Cook D, Cohen J, Opal SM, Vincent JL, Ramsay G; SCCM/ESICM/ACCP/ATS/SIS (2003) 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med 31:1250–1256
Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR (2001) Epidemiology of severe sepsis in the United States: analysis of incidence, outcome and associated costs of care. Crit Care Med 29:1303–1310
Iregui M, Ward S, Sherman G, Fraser VJ, Kollef MH (2002) Clinical importance of delays in the initiation of appropriate antibiotic treatment for ventilator-associated pneumonia. Chest 122:262–268
Garnacho-Montero J, Garcia-Garmendia JL, Barrero-Almodovar AE, Jimenez-Jimenez FJ, Perez-Paredes C, Ortiz-Leyba C (2003) Impact of adequate empirical antibiotic therapy on the outcome of patients admitted to the intensive care unit with sepsis. Crit Care Med 31:2742–2751
Dellinger RP, Carlet JM, Masur H, Gerlach H, Calandra T, Cohen J, Gea-Banacloche J, Keh D, Marshall JC, Parker MM, Ramsay G, Zimmerman JL, Vincent JL, Levy MM; Surviving Sepsis Campaign Management Guidelines Committee (2004) Surviving sepsis campaign guidelines for management of severe sepsis and septic shock. Crit Care Med 32:858–873
Pepys MB, Baltz ML (1983) Acute phase proteins with special reference to C-reactive protein and related proteins (pentaxins) and serum amyloid A protein. Adv Immunol 34:141-212
Gabay C, Kushner I (1999) Acute-phase proteins and other systemic responses to inflammation. N Engl J Med 340:448–454
Mackowiak PA, Bartlett JG, Borden EC, Goldblum SE, Hasday JD, Munford RS, Nasraway SA, Stolley PD, Woodward TE (1997) Concepts of fever: recent advances and lingering dogma. Clin Infect Dis 25:119–138
Johanson WG Jr, Pierce AK, Sanford JP, Thomas GD (1972) Nosocomial respiratory infections with gram-negative bacilli. The significance of colonization of the respiratory tract. Ann Intern Med 77:701–706
Marquette CH, Georges H, Wallet F, Ramon P, Saulnier F, Neviere R, Mathieu D, Rime A, Tonnel AB (1993) Diagnostic efficiency of endotracheal aspirates with quantitative bacterial cultures in intubated patients with suspected pneumonia. Comparison with the protected specimen brush. Am Rev Respir Dis 148:138–144
Ferreira FL, Bota DP, Bross A, Melot C, Vincent JL (2001) Serial evaluation of the SOFA score to predict outcome in critically ill patients. JAMA 286:1754–1758
Povoa P, Almeida E, Moreira P, Fernandes A, Mealha R, Aragao A, Sabino H (1998) C-reactive protein as an indicator of sepsis Intensive Care Med 24:1052–1056
Ugarte H, Silva E, Mercan D, De Mendonça A, Vincent JL (1999) Procalcitonin used as a marker of infection in the intensive care unit. Crit Care Med 27:498–504
Suprin E, Camus C, Gacouin A, Le Tulzo Y, Lavoue S, Feuillu A, Thomas R (2000) Procalcitonin: a valuable indicator of infection in a medical ICU. Intensive Care Med 26:1232–1238
Miller PR, Munn DD, Meredith JW, Chang MC (1999) Systemic inflammatory response syndrome in the trauma intensive care unit: who is infected? J Trauma 47:1004–1008
Hambach L, Eder M, Damman E, Schrauder A, Sykora KW, Dieterich C, Kirschner P, Novotny J, Ganser A, Hertenstein B (2002) Diagnostic value of procalcitonin serum levels in comparison with C-reactive protein in allogeneic stem cell transplantation. Haematologica 87:643–651
Reny JL, Vuagnat A, Ract C, Benoit MO, Safar M, Fagon JY (2002) Diagnosis and follow-up of infections in intensive care patients: value of C-reactive protein compared with other clinical and biological variables. Crit Care Med 30:529–535
Povoa P (2002) C-reactive protein: a valuable marker of sepsis. Intensive Care Med 28:235–243
Guven H, Altintop L, Baydin A, Esen S, Aygun D, Hokelek M, Doganay Z, Bek Y (2002) Diagnostic value of procalcitonin levels as an early indicator of sepsis. Am J Emerg Med 20:202–206
BalcI C, Sungurtekin H, Gurses E, Sungurtekin U, Kaptanoglu B (2003) Usefulness of procalcitonin for diagnosis of sepsis in the intensive care unit. Crit Care 7:85–90
Chirouze C, Schuhmacher H, Rabaud C, Gil H, Khayat N, Estavoyer JM, May T, Hoen B (2002) Low serum procalcitonin level accurately predicts the absence of bacteremia in adult patients with acute fever. Clin Infect Dis 35:156–161
Luzzani A, Polati E, Dorizzi R, Rungatscher A, Pavan R, Merlini A (2003) Comparison of procalcitonin and C-reactive protein as markers of sepsis. Crit Care Med 31:1737–1741
Tugrul S, Esen F, Celebi S, Ozcan PE, Akinci O, Cakar N, Telci L (2002) Reliability of procalcitonin as a severity marker in critically ill patients with inflammatory response. Anaesth Intensive Care 30:747–754
Steel DM, Whitehead AS (1994) The major acute phase reactants: C-reactive protein, serum amyloid P component and serum amyloid A protein. Immunol Today 15:81–88
Oberhoffer M, Karzai W, Meier-Hellmann A, Bogel D, Fassbinder J, Reinhart K (1999) Sensitivity and specificity of various markers of inflammation for the prediction of tumor necrosis factor-alpha and interleukin-6 in patients with sepsis. Crit Care Med 27:1814–1818
Lobo SM, Lobo FR, Bota DP, Lopes-Ferreira F, Soliman HM, Melot C, Vincent JL (2003) C-reactive protein levels correlate with mortality and organ failure in critically ill patients. Chest 123:2043–2049
Krabbe KS, Bruunsgaard H, Hansen CM, Moller K, Fonsmark L, Qvist J, Madsen PL, Kronborg G, Andersen HO, Skinhoj P, Pedersen BK (2001) Ageing is associated with a prolonged fever response in human endotoxemia. Clin Diagn Lab Immunol 8:333–338
Opal SM, Cohen J (1999) Clinical gram-positive sepsis: does it fundamentally differ from gram-negative bacterial sepsis? Crit Care Med 27:1608–1616
Byl B, Deviere J, Saint-Hubert F, Zech F, Gulbis B, Thys JP (1997) Evaluation of tumor necrosis factor-alpha, interleukin-6 and C-reactive protein plasma levels as predictors of bacteremia in patients presenting signs of sepsis without shock. Clin Microbiol Infect 3:306–316
Fassbender K, Pargger H, Muller W, Zimmerli W (1993) Interleukin-6 and acute-phase protein concentrations in surgical intensive care unit patients: diagnostic signs in nosocomial infection. Crit Care Med 21:1175–1180
Rothenburger M, Markewitz A, Lenz T, Kaulbach HG, Marohl K, Kuhlmann WD, Weinhold C (1999) Detection of acute phase response and infection. The role of procalcitonin and C-reactive protein. Clin Chem Lab Med 37:275–279
Enguix A, Rey C, Concha A, Medina A, Coto D, Dieguez MA (2001) Comparison of procalcitonin with C-reactive protein and serum amyloid for the early diagnosis of bacterial sepsis in critically ill neonates and children. Intensive Care Med 27:211–215
Vincent JL (2000) Procalcitonin: THE marker of sepsis? Crit Care Med 28:1226–1228
Meisner M, Tschaikowsky K, Palmaers T, Schmidt J (1999) Comparison of procalcitonin (PCT) and C-reactive protein (CRP) plasma concentrations at different SOFA scores during the course of sepsis and MODS. Crit Care 3:45–50
Bonten MJ, Froon AH, Gaillard CA, Greve JW, de Leeuw PW, Drent M, Stobberingh EE, Buurman WA (1997) The systemic inflammatory response in the development of ventilator-associated pneumonia. Am J Respir Crit Care Med 156:1105–1113
Pepys MB, Berger A (2001) The renaissance of C reactive protein. BMJ 322:4–5
Rello J, Diaz E, Roque M, Mariscal D, Valles J (1999) Risk factors for developing pneumonia within 48 hours of intubation. Am J Respir Crit Care Med 159:1742–1746
Woiciechowsky C, Schoning B, Cobanov J, Lanksch WR, Volk HD, Docke WD (2002) Early IL-6 plasma concentrations correlate with severity of brain injury and pneumonia in brain-injured patients. J Trauma 52:339–345
Luna CM (2004) C-reactive protein in pneumonia: let me try again. Chest 125:1192–1195
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported in part by Red Respira (isciii-RTIC C03/11), CIRIT SGR 2001/414 and Distinció a la Recerca Universitaria (JR).
Rights and permissions
About this article
Cite this article
Sierra, R., Rello, J., Bailén, M.A. et al. C-reactive protein used as an early indicator of infection in patients with systemic inflammatory response syndrome. Intensive Care Med 30, 2038–2045 (2004). https://doi.org/10.1007/s00134-004-2434-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-004-2434-y