Abstract
Objective
Interest has recently focused on the use of neurohormonal markers such as atrial natriuretic peptide (ANP) and brain natriuretic peptide (BNP) as indices of left ventricular systolic dysfunction and prognosis in heart failure. Also, peptides belonging to the interleukin-6 (IL-6) family have been shown to induce ANP and BNP secretion. We hypothesized that BNP and ANP spillover in the peripheral circulation reflects left ventricular dysfunction and IL-6 production in septic shock.
Design and setting
Retrospective, clinical study in the medical intensive care unit of a university hospital.
Patients and participants
17 patients with septic shock and 19 control subjects.
Interventions
Collection of clinical and demographic data in relation to ANP, BNP, IL-6, and soluble TNF receptors (sTNF-R-p55, sTNF-R-p75) in plasma over a period of 4 days.
Measurements and results
In septic shock we found a significant increase in ANP (82.7±9.9 vs. 14.9±1.2 pg/ml) and BNP (12.4±3.6 vs. 5.5±0.7 pg/ml). Plasma ANP peaked together with IL-6. Peaks of ANP and IL-6 were significantly correlated (r=0.73; p<0.01). BNP was inversely correlated to cardiac index (r=–0.56; p<0.05).
Conclusions
ANP and BNP increase significantly in patients with septic shock. BNP reflects left ventricular dysfunction. ANP is related to IL-6 production rather than to cardiovascular dysfunction.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Atrial natriuretic peptide (ANP) is a hormone with a wide range of potent biological effects, including natriuresis, diuresis, vasodilation, and inhibition of the renin-angiotensin-aldosterone axis and the sympathetic nervous system [1]. In adult mammals ANP is secreted mainly from the atria [2]. Brain natriuretic peptide (BNP), first isolated from the porcine brain [3] and subsequently from mammalian organs including human hearts, forms together with ANP a peptide family involved in the regulation of blood pressure and fluid volume. In healthy subjects this novel natriuretic peptide is mainly secreted from the ventricles of the heart [4]. Increased plasma levels of ANP and BNP have been reported in patients with congestive heart failure [5, 6]. In these patients both natriuretic peptides increase with the severity of disease [5, 6]. Enhanced plasma concentrations of ANP and BNP in ischemic and congestive heart failure predict poor prognosis [6, 7, 8]. Plasma levels of natriuretic peptides in patients with septic shock in relation to cardiovascular function has not yet been evaluated.
ANP and BNP are secreted in response to atrial or ventricular volume expansion and pressure overload, respectively. Proinflammatory cytokines stimulated in heart failure might contribute to ANP and BNP secretion from the heart. In vitro studies show an enhanced gene expression of BNP and prepro-ANP, the precursor form of circulating ANP following stimulation of cultured cardiomyocytes with interleukin (IL)-1β [9, 10]. Increased secretion of both natriuretic peptides following stimulation with peptides belonging to the IL-6 family has recently been reported in cultured cardiomyocytes [11]. In vivo cardiotrophin-1 and IL-6, both members of the IL-6 related family, increase in heart failure [12, 13], and cosecretion of IL-6 and ANP as well as cardiotrophin-1 and BNP has been reported [12, 14, 15]. Tumor necrosis factor-α (TNF-α) comparable to IL-6, increases in a broad spectrum of cardiovascular diseases ranging from severe heart failure of various causes to asymptomatic ischemic left ventricular impairment [16]. In experimental settings TNF-α induces left ventricular dysfunction, pulmonary edema, and cardiomyopathy [17]. Soluble TNF receptors have a longer half-life in plasma than TNF itself, which make them particularly suitable for detecting TNF production in plasma.
Changes in cardiovascular functional parameters in sepsis are associated by a several-fold increase in IL-6. Although pleiotropic actions of IL-6 have been described in humans (for review see [18]), the relationship between IL-6 and natriuretic peptides has not yet been investigated in septic patients. Patients with septic shock show a reversible left ventricular systolic dysfunction commonly masked by a concomitant elevation in cardiac index (CI) [19]. Survivors of septic shock were found to have decreased systolic function and an increase in left ventricular end-diastolic diameter. These changes in left ventricular function were of rapid onset and reversible in survivors within several days [20]. Paradoxically the changes seen were less profound in those who died [21]. At present it is not known whether natriuretic peptides reflect left ventricular function in septic shock. However, this may be a question of considerable interest because left ventricular systolic dysfunction occurs in septic patients and has been related to prognosis [19].
The present study assessed ANP and BNP spillover in the peripheral circulation in relation to cardiovascular function and systemic inflammation i.e., IL-6 and soluble TNF-α receptor release in patients with septic shock.
Patients and methods
Patient population
Patients with septic shock and without renal or hepatic failure (5 women, 12 men; mean age 61±2.7 years) were selected from the placebo group of the "Score-Based Immunoglobulin G Therapy in Sepsis" study [22, 23] (Table 1). Renal failure was defined as increased retention above normal on the day of diagnosis. Hepatic failure was defined by transaminases being more than twofold above normal or by increased bilirubin values. On the basis of the underlying disease 4 patients were classified as medical and 13 as surgical patients. Catecholamines were administered intravenously in all patients. All patients needed mechanical ventilation between 4 and 32 days (mean 15.4±2.2 days). Septic shock was defined according to international criteria [24, 25]. Severity of sepsis was determined by the sepsis score according to Elebute and Stoner [26]. Severity of disease was quantified by the Acute Physiology and Chronic Health Evaluation II (APACHE II ) score [27]. Laboratory, biochemical, hemodynamic, and physical parameters were determined on the day of diagnosis (day 0) and on the first and fourth day after diagnosis. All patients with septic shock were followed for 28 days. The date of death was recorded and duration of follow-up calculated.
The study was approved by the local ethics committee, with informed consent of all participants or, in the case of unconscious patients, of the closest relatives.
Control group
Nineteen subjects diagnosed for suspected cardiac disease without sepsis or renal failure (4 women, 15 men; mean age 61±2.1 years) served as the control group. After elective admission to hospital for suspected coronary artery disease these patients were prospectively enrolled after intracardiac catheter diagnosis confirmed normal left ventricular function by levocardiography in standard projections, and coronary artery stenosis was excluded. Furthermore, two-dimensional echocardiography with continuous and pulse-wave Doppler was performed to exclude underlying valvular disease which might affect plasma levels of natriuretic peptides. A single measurement of natriuretic peptide and proinflammatory cytokine plasma levels was performed.
Blood sampling and determination of plasma levels of ANP, BNP, IL-6, sTNF-R-p55, and sTNF-R-p75
Blood samples were drawn by venipuncture in supine position, immediately placed on ice, and centrifuged within 30 min. Plasma was aspirated and transferred into polystyrol test tubes stored at −70°C until analysis. Plasma ANP and BNP concentrations were measured as described previously [28]. Briefly, for extraction plasma samples were thawed and adsorbed as 1-ml aliquots to activated Amberlite XAD2 resin chromatography columns (Bio-Rad Laboratories, Richmond, Calif., USA) followed by washing with trifluoroacetic acid (1 mmol/l). Elution was performed using a mobile phase consisting of 1 mol/l acetonitrile (80% vol.) and 1 mmol/l trifluoroacetic acid (20% vol.). The lyophilized samples were reconstituted in ANP or BNP assay buffer. Plasma ANP was measured by radioimmunoassay (Amersham, Buckinghamshire, UK). Plasma BNP was measured by radioimmunoassay (Peninsula Laboratories, Belmont, Calif., USA). Serum levels of IL-6 were measured by commercially available enzyme linked immunosorbent assay (ELISA, Medgenix, Ratingen, Germany). Measurements for both soluble TNF receptors, sTNF-R-p55 and sTNF-R-p75, the more stable form reflecting TNF production, were obtained by enzyme-linked immunological assays as described previously [29].
Measurement of cardiovascular functional parameters
Cardiac catheterization in septic patients was performed using a pulmonary artery catheter inserted into the cephalic vein, and right atrial pressure was measured. Cardiac output was determined by thermodilution technique in triplicate. Arterial pressure was measured by catheters inserted into the radial or femoral artery using Statham pressure transducers. Cardiac output, systemic vascular resistance (SVR), and left ventricular stroke work index (LVSWI) were calculated using standard formulas [30].
Statistical analysis
All data are expressed as mean ±standard error. Hemodynamic data, cytokine levels, and plasma levels of ANP and BNP were compared between groups using the Mann-Whitney nonparametric statistical test. Pearson's correlation coefficient was computed between plasma ANP and BNP, hemodynamic parameters, and plasma concentrations of IL-6, sTNF-R-p55, and sTNF-R-p75. A p value less than 0.05 was considered as statistically significant.
Results
Plasma levels of ANP and BNP
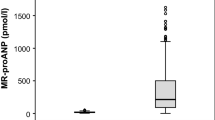
Plasma levels of ANP in patients with septic shock were 82.7±9.9 pg/ml, significantly higher than in control subjects (14.9±1.2 pg/ml, p<0.01; Fig. 1a). Plasma BNP were significantly higher than in controls (12.4±3.6 vs. 5.5±0.7 pg/ml, p<0.05; Fig. 1b). From day 0 to day 1 plasma ANP decreased significantly (64.4±7.2 pg/ml, p<0.05 vs. day 0) in patients with septic shock. On day 4 plasma levels of ANP in patients with septic shock were still higher than baseline levels of control subjects (71.5±6.9 pg/ml, p<0.05). BNP showed the highest levels in patients with septic shock on day 0 (12.4±3.6 pg/ml) without significant differences up to day 4 (9.8±2.1 pg/ml).
Plasma levels of atrial natriuretic peptide (ANP) in control subjects and patients with septic shock. b Plasma levels of brain natriuretic peptide (BNP) in control subjects and patients with septic shock. Plasma levels were determined on the day of diagnosis in duplicate. Horizontal bars Statistical mean. *p<0.05 vs. control **p<0.01
Interleukin-6 and soluble tumor necrosis factor receptors (sTNF-R-p55, TNF-R-p75)
IL-6 values in septic shock were significantly higher than in controls (3035±949 vs. 18.3±7.2 pg/ml, p<0.01). In patients with septic shock plasma levels of IL-6 significantly dropped from day 0 to day 1 (852±244 pg/ml, p<0.05 vs. day 0). On day 4 plasma IL-6 was still significantly higher in patients with septic shock than in controls (423±156 pg/ml, p<0.05). The concentrations of neither sTNF-R-p55 nor sTNF-R-p75 changed significantly over 4 days, with initial values of 7.6±0.8 ng/ml and 11.0±1.1 ng/ml, respectively.
Cardiovascular functional parameters
Cardiovascular functional parameters are given in Table 2. All septic patients showed hypotensive blood pressure values and tachycardia on the day of diagnosis of septic shock (Table 2). Stroke volume was in the normal range (Table 2). SVR in patients with septic shock was lowered (Table 2). CI was not increased in terms of absolute values; however, there was a tendency to higher values in septic shock (Table 2). LVSWI was lower in septic patients than in controls (Table 2), and right atrial pressure was elevated (Table 2).
Relationship between plasma levels of natriuretic peptides, cardiovascular functional parameters, IL-6, and soluble TNF receptors
On the day of diagnosis a highly significant correlation was found between IL-6 and ANP (Fig. 2) and between IL-6 and sTNF-R-p55 (r=0.66, p<0.01) but not between ANP and sTNF-R-p55 (r=0.20, p=0.48) or sTNF-R-p75 (r=0.02, p=0.94). A weak, statistically nonsignificant correlation was observed between ANP and right atrial pressure (r=0.56, p=0.09) but not between plasma ANP and CI (r=0.27, p=0.42) or SVR (r=−0.44, p=0.13). IL-6 was significantly correlated with right atrial pressure (r=0.58, p<0.05) but not with other cardiovascular parameters. BNP was inversely correlated with CI (Fig. 3). BNP was correlated neither with further cardiovascular parameters nor with IL-6, sTNF-R-p55, or sTNF-R-p75. Severity of disease (APACHE II) and sepsis score were not correlated with plasma ANP or BNP at any time point.
28 day follow-up in septic shock
Five patients died within 28 days in multiple organ failure, resulting in a 28-day mortality of 29.4%. Patients with septic shock were divided into two groups based on outcome (survivors and nonsurvivors) 28 days after diagnosis. These groups did not differ in baseline characteristics or in natriuretic peptide or proinflammatory cytokine levels. None of the hemodynamic parameters initially determined except SVR (465.7±37.9 vs. 655.8±34.8 dyn × s−1 × cm−5, p<0.01) differentiated survivors from nonsurvivors.
Discussion
We demonstrated a several-fold, significant increase in circulating ANP in plasma of patients with septic shock (Fig. 1). ANP was clearly correlated with excessively elevated IL-6 plasma levels (Fig. 2). The concentration of plasma ANP in our septic patients is comparable to levels reported in patients with severe congestive heart failure NYHA III–IV [31]. BNP was inversely correlated with CI (Fig. 3). The increase in both ANP and BNP in plasma of patients with septic shock lasted more than 1 day and was still detectable 4 days after diagnosis.
Myocardial dysfunction in septic shock
Patients with septic shock showed a CI in the normal range despite an extremely lowered SVR. A reversible left ventricular systolic dysfunction occurring in septic shock, however, is commonly masked by a concomitant normal or elevated CI due to the lowering in afterload reduction as indicated by a decreased SVR [19]. We found only a weak correlation between CI and SVR. This finding might be interpreted as an inadequate increase in CI in the presence of an extremely lowered SVR. Therefore to further elucidate left ventricular systolic function we calculated LVSWI. As a measure of external left ventricular work this is frequently used as a reliable parameter for quantifying cardiac function in sepsis studies [32, 33]. Our data clearly show lowered values of LVSWI in septic shock, confirming left ventricular systolic dysfunction in our patients.
Cardiovascular function and natriuretic peptides
ANP and BNP plasma levels were increased acutely on the day of diagnosis in septic shock. The plasma level of ANP increased more markedly than that of BNP in the early phase of the disease and dropped until day 4, with plasma levels still significantly higher than control subjects. BNP increased inversely in proportion to CI without significant changes up to day 4. The different behavior of both natriuretic peptides may be due to different sites and mechanisms of release. It has been shown that BNP is predominantly synthesized in and secreted from the ventricles, in contrast to ANP, the major producer of which are the atria [34, 35]. Acute stimuli such as acute volume expansion in vivo or atrial muscle stretch in vitro are met by the endocrine heart with an increase in the rate of release of ANP from stores without an apparent effect on synthesis and with a significant increase in plasma levels of ANP [36]. Atrial stretch has been identified as a major determinant of ANP release [37]. However, despite a statistical trend there was no significant correlation in our septic patients between plasma ANP and right atrial filling pressure (r=0.56. p=0.09). The lack of a correlation between plasma ANP and right atrial pressure argues against atrial stretch as the only factor responsible for the significant increase in plasma ANP.
In contrast, in ventricular overload a rapid induction of ventricular BNP gene expression and secretion has been shown [35]. In septic patients an increased end-diastolic volume might compensate for a decreased ejection fraction and result in a cardiac output in the normal range [38]. Although we did not measure left ventricular ejection fraction directly, the lowered LVSWI in our patients with septic shock indicates a lower than normal systolic left ventricular function. An increase in ventricular distension due to an increased enddiastolic volume may result in enhanced ventricular wall stress, a sufficient stimulus for BNP. In the presence of a lowered ejection fraction, an increased filling volume of the left ventricle could maintain the measured CI in the normal range. This assumption of increased filling volumes is in good agreement with the elevated pulmonary capillary wedge pressure and central venous pressure found in our patients (Table 2). A resulting rise in ventricular distension may explain the raised plasma levels of BNP and its rather weak correlation to CI in the presence of a lowered ejection fraction. Thus, elevated BNP levels might be interpreted as an indicator of a reduced systolic left ventricular function comparable to findings in congestive heart failure also in septic shock.
Interleukin-6 and atrial natriuretic peptide
On the day of diagnosis of septic shock we found higher plasma concentrations of IL-6 and ANP than in control subjects and a strong correlation between IL-6 and ANP. The precise mechanism of IL-6 correlated ANP production in septic shock has not been fully elucidated. In human septic shock subendocardial hypoperfusion, with redistribution of intramyocardial blood flow to the epicardial vessels, i.e., arteriole-venule shunting or a defect in oxygen utilization at the cellular level, may increase regional ventricular wall stretch owing to depression of myocardial contraction at the atrial and ventricular level (for review see [19]). Wall stretch can activate the Janus kinase/signal transducer and activator of transcription (JAK/STAT) pathway in (neonatal) cardiomyocytes, and this activation augments IL-6 mRNA expression and depends mainly on the IL-6 release [39]. ANP secretion and protein synthesis have been reported to be stimulated by members of the IL-6 family by a JAK/STAT dependent pathway [11]. Since IL-6 related peptides are also produced in the atrium and ventricles of the human heart [40] and gp130, the common signal transducer of IL-6 related cytokines, is ubiquitously expressed including in the heart [11], our data are consistent with the concept of an IL-6 promoted transcription and secretion of ANP also in septic shock in addition to increased atrial pressures.
In agreement with our findings, IL-6 has been reported by others to be increased in patients with left ventricular dysfunction associated with septic shock (for review see [20]). Data from patients with congestive and ischemic heart failure showed a TNF-α dependent IL-6 release. IL-6 showed a good correlation to sTNF-R-p55 and ANP in our study. In support of a specific, IL-6 dependent mechanism of ANP release, however, IL-6 but not soluble TNF receptors, the more stable product of TNF production, have been found to be highly significantly correlated with ANP. Furthermore, IL-6 in contrast to soluble TNF receptors decreased on the following day, accompanied by a fall in plasma ANP. ANP and IL-6 maintained plasma levels still significantly higher than controls up to day 4, while sTNF receptors did not change between day 0 and day 4.
In conclusion, we found significant elevated plasma levels of ANP and BNP in patients with septic shock. To our knowledge, this is the first study showing a correlation between ANP and excessively increased IL-6 and BNP and CI in patients with septic shock. Our data suggest that excessively increased proinflammatory cytokines, in example, IL-6, contributes more than hemodynamic changes to the modulation of ANP in these patients. Furthermore, BNP may offer a valuable parameter for determining reduced left ventricular function in patients with septic shock. Future studies evaluating the precise mechanism underlying an enhanced BNP secretion in septic shock should combine hemodynamic measurements and echocardiographic determination of left ventricular function. Studies comprising larger study populations are needed to evaluate a potential prognostic value of certain hemodynamic parameters, such as SVR and natriuretic peptides in those patients.
References
Nakao K, Ogawa Y, Suga S, Imura H (1992) Molecular biology and biochemistry of the natriuretic system. I. Natriuretic peptides. J Hypertens 10:907–912
Bold AJ de (1985) Atrial natriuretic factor: a hormone produced by the heart. Science 230:767–770
Sudoh T, Kangawa K, Minamino N, Matsuo H (1988) A new natriuretic peptide in porcine brain. Nature 332:78–81
Yasue H, Yoshimura M, Sumida H, Kikuta K, Kugiyama K, Jougasaki M, Ogawa H, Okumura K, Mukoyama M, Nakao K (1994) Localization and mechanism of secretion of B-type natriuretic peptide in comparison with those of A-type natriuretic peptide in normal subjects and patients with heart failure. Circulation 90:195–203
Mukoyama M, Nakao K, Saito Y, Ogawa Y, Hosoda K, Suga S, Shirakami G, Jougasaki M, Imura H (1990) Increased human brain natriuretic peptide in congestive heart failure. N Engl J Med 313:757–758
Gottlieb S, Kukin ML, Ahern D, Packer M (1989) Prognostic importance of atrial natriuretic peptide in patients with chronic heart failure. J Am Coll Cardiol 13:153–159
Arakawa N, Nakamura M, Aoki H, Hiramori K (1996) Plasma brain natriuretic peptide concentrations predict survival after acute myocardial infarction. J Am Coll Cardiol 27:1656–1661
Hall C, Rouleau JL, Moye L, de Champlain J, Bichet D, Klein M, Sussex B, Packer M, Rouleau J, Arnold MO, Lamas GA, Sestier F, Gottlieb S, Wun CC, Pfeffer MA (1994) N terminal proatrial natriuretic factor. An independent predictor of long term prognosis after myocardial infarction. Circulation 89:1934–1942
Thaik CM, Calderone A, Takahashi N, Colucci WS (1995) Interleukin-1β modulates the growth and phenotype of neonatal rat cardiac myocytes. J Clin Invest 96:1093–1099
He Q, LaPointe MC (1999) Interleukin-1β regulation of the human brain natriuretic peptide promotor involves Ras-, Rac-, and p38 kinase-dependent pathways in cardiac myocytes. Hypertension 33:283–289
Kuwahara K, Saito Y, Harada M, Ishikawa M, Ogawa E, Miyamoto Y, Hamanaka I, Kamitani S, Kajiyama N, Takahashi N, Nakagawa O, Masuda I, Nakao K (1999) Involvement of cardiotrophin-1 in cardiac myocyte-nonmyocyte interactions during hypertrophy of rat cardiac myocytes in vitro. Circulation 100:1116–1124
MacGowan GA, Mann DL, Kormos RL, Feldman AM, Murali S (1997) Circulating interleukin-6 in severe heart failure. Am J Cardiol 79:1128–1131
Talwar S, Downie PF, Squire IB (1999) An immunolumino-metric assay for cardiotrophin-1: a newly identified cytokine is present in normal human plasma and is increased in heart failure. Biochem Biophys Res Commun 261:567–571
Tsutamoto T, Hisanga T, Wada A, Maeda K, Ohnishi M, Fukai D, Mabushi N, Sawaki M, Kinoshita M (1998) Interleukin-6 spillover in the peripheral circulation increase with the severity of heart failure, and the high plasma level of interleukin-6 is an important prognostic predictor in patients with congestive heart failure. J Am Coll Cardiol 31:391–398
Talwar S, Squire IB, Downie PF, Davies JE, Ng LL (2000) Plasma N terminal pro-brain natriuretic peptide and cardiotrophin 1 are raised in unstable angina. Heart 84:421–424
Torre-Amione G, Kapadia S, Benedct C, Oral H, Young JB, Mann DL (1996) Proinflammatory cytokine levels in patients with depressed left ventricular dysfunction. J Am Coll Cardiol 27:1201–1206
Yokoyama T, Vaca L, Rossen RD, Durante W, Hazarika P, Mann DL (1993) Cellular basis for the negative inotropic effects of tumor necrosis factor-alpha in the adult mammalian heart. J Clin Invest 92:2303–2312
Werdan K (1998) The activated immune system in congestive heart failure-from dropsy to the cytokine paradigm. J Intern Med 243:87–92
Price S, Anning PB, Mitchell JA, Evans TW (1999) Myocardial dysfunction in sepsis: mechanisms and therapeutic implications. Eur Heart J 20:715–724
Müller-Werdan U, Werdan K (1999) Septic cardiomyopathy. Curr Opin Crit Care 5:415–421
Parker MM, Shelhamer JH, Natanson C, Alling DW, Parillo JE (1989) Serial cardiovascular variables in survivors and nonsurvivors of human septic shock: heart rate as an early predictor of prognosis. Crit Care Med 15:923–929
Pilz G, Fateh-Moghadam S, Viell B, Bujdoso O, Döring G, Marget W, Werdan K (1993) Supplemental immunoglobulin therapy in sepsis and septic shock-comparison of mortality under treatment with polyvalent i.v. immunoglobulin versus placebo: protocol of a multicenter, randomized, prospective, double-blind trial. Theor Surg 8:61–83
Werdan K, Pilz G, and the SBITS Study Group (1998). Polyvalent immune globulins. Shock [Suppl] 7:1918
Bone R (1996) Sir Isaac Newton, sepsis, SIRS and CARS. Crit Care Med 24:1125–1128
American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference Committee (1992) Definitions for sepsis and multiple organ failure and guidelines for the use of innovative therapies in sepsis. Crit Care Med 20:864–874
Elebute EA, Stoner HB (1983) The grading of sepsis. Br J Surg 70:29–31
Knaus WA, Draper EA, Wagner DP, Zimmermann JE (1985) APACHE II: a severity of disease classification system. Crit Care Med 13:818–829
Gerbes AL, Witthaut R, Gülberg V, Thibault G, Bilzer M, Jüngst D (1992) Role of the liver in splanchnic extraction of atrial natriuretic factor in the rat. Hepatology 16:790–793
Fraunberger P, Pfeiffer M, Cremer P, Holler E, Nagel D, Dehart I, Thein M, Walli AK, Seidel D (1992) Validation of an automated enzyme immunoassay for interleukin-6 for routine clinical use. Clin Chem Lab Med 36:797–801
Baim DS, Grossman W (1996) Cardiac catheterization, angiography, and intervention, 5th edn. Williams & Wilkins, Baltimore
Muders F, Kromer EP, Griese DP, Pfeifer M, Hense HW, Riegger GAJ, Elsner D (1997) Evaluation of plasma natriuretic peptides as markers for left ventricular dysfunction. Am Heart J 134:442–449
Bakker J, Vincent JL (1993) Effects of norepinephrine and dobutamine on oxygen transport and consumption in a dog model of endotoxic shock. Crit Care Med 21:1248–1249
Pilz G, R Appel, P McGinn (1993) Calculation of the left ventricular stroke work index. Crit Care Med 21:1248–1249
Raine AEG, Erne P, Bürgisser E, Müller FB, Bolli P, Burkart F, Bühler FR (1986) Atrial natriuretic peptide and atrial pressure in patients with congestive heart failure. N Engl J Med 315:533–537
Hama N, Ito H, Shirakami G, Nakagawa O, Suga S, Ogawa Y, Masuda I (1995) Rapid ventricular induction of brain natriuretic peptide gene expression in experimental acute myocardial infarction. Circulation 92:1558–1564
Ogawa T, Linz W, Stevenson M, Bruneau BG, de Bold MLK, Chen JH, Eid H, Schölkens BA, de Bold AJ (1996) Evidence for load dependent and load independent determinants of cardiac natriuretic peptide production. Circulation 93:2059–2067
Edwards BS, Zimmermann RS, Schwab TR, Heublin DM, Burnett JC (1988) Atrial stretch, not pressure, is the principal determinant controlling the acute release of atrial natriuretic factor. Circ Res 62:191–195
Ognibene FP, Parker MM, Natanson C, Shelhamer JH, Parrillo JE (1988) Depressed left ventricular performance. Response to volume infusion in patients with sepsis and septic shock. Chest 93:903–910
Pan J, Fukuda K, Saito M, Matsuzaki J, Kodama H, Sano M, Takahashi T, Kato T, and Ogawa S (1999) Mechanical stretch activates the JAK/STAT pathway in rat cardiomyocytes. Circ Res 84:1127–1136
Tsuruda T, Jougaschi M, Boerrigter G, Huntley BK, Chen HH, Dàssoro AB, Lu SC, Sarson AM, Cataliotti A, Burnett JC Jr (2002) Cardiotrophin-1 stimulation of cardiac fibroblast growth. Roles for glycoprotein 130/ leukemia inhibitory factor receptor and the endothelin type A receptor. Circ Res 90:128–134
Acknowledgements
This work was supported by a grant from the Deutsche Forschungsgemeinschaft to R.W. (Wi 1616/1). Patients with septic shock were enrolled in the "Score-Based Immunoglobulin Therapy of Sepsis" study [22, 23]. Mrs. Anke Rasch is gratefully thanked for the preparation of illustrations in the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Witthaut, R., Busch, C., Fraunberger, P. et al. Plasma atrial natriuretic peptide and brain natriuretic peptide are increased in septic shock: impact of interleukin-6 and sepsis-associated left ventricular dysfunction. Intensive Care Med 29, 1696–1702 (2003). https://doi.org/10.1007/s00134-003-1910-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-003-1910-0