Abstract
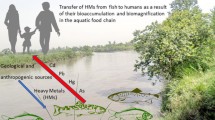
Since the Minamata incident in Japan, the public have become increasingly aware of the negative health effects caused by mercury pollution in the ocean. Consequently, there has been significant interest in the health of humans eating fish exposed to mercury (Hg). However, the toxicity of mercury to the marine fish themselves has received far less attention. In this review, we summarize mercury accumulation in marine fish and the toxicological effects of mercury exposure. Results showed that the bioaccumulation of mercury in marine fish was highly variable, and its concentration was affected by the specific physiological and ecological characteristics of different fish species. Mercury exposure can produce teratogenic, neurotoxic effects, and reproductive toxicity. These effects can then cause harm to cells, tissues, proteins and genes, and ultimately, the survival, growth, and behavior of marine fish. Future studies should afford more attention to the toxicological effect of mercury exposure upon marine fish.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mercury in water is mainly in the form of elemental mercury, divalent mercury and methylmercury. Elemental mercury (Hg0) is rarely found in seawater due to its high volatility and low bioavailability, so divalent inorganic mercury (Hg+ and Hg2+) and organic mercury (such as methyl, ethyl, and phenyl) are the major mercury species (Selin 2009; Gonzalez-Raymat et al. 2017). Mercury in the upper oceans has tripled compared to pre-anthropogenic conditions (Lamborg et al. 2014). The bioaccumulation of organic and divalent inorganic mercury in marine fish can directly and indirectly endanger population health; even low concentrations of mercury exposure are associated with developmental retardation and learning disabilities in fetuses, infants and children (Budtz-Jorgensen et al. 2002). The exposure of marine fish to mercury has not only been linked to population health, but could also act as a useful marker to reflect the ecological effects of mercury in the ocean.
The principal way by which humans are exposed to mercury is via ingestion, including the rice ingestion in the special Hg mining area (Li et al. 2012), and the consumption of seafood such as fish contaminated with mercury (Zaza et al. 2015). However, eating fish is not completely unprofitable. Nutrients in fish, such as docose hexaenoie acid (DHA), an n-3 long-chain polyunsaturated fatty acid, are important for the growth and development of the fetal brain, and fish are highly recommended for the prevention of cardiovascular disease (Liu et al. 2018; Anual et al. 2018). In marine fish, exposure to mercury can lead to reduced liver function and metabolism, altered behavior, impaired reproduction, deformity, damage to the gills and olfaction organs, and mortality (O’Bryhim et al. 2017; Huang et al. 2011).
Research studies have measured mercury levels in fish, assessed the health risks to humans of eating fish and have provided guidance for tolerable weekly intake (Anual et al. 2018; Jeevanaraj et al. 2016; Liu et al. 2018). However, far less is known about the abnormal development of marine fish due to mercury exposure. In fact, the reproductive toxicity and teratogenicity effects of mercury to embryos may also lead to a significant reduction in fishery production and quality. This article reviews the associated bioaccumulation characteristics of marine fishes, focusing particularly on the toxic effects of mercury on marine fish in laboratory studies.
Bioaccumulation of mercury in marine fish
Bacteria and phytoplankton are the primary entry-points of mercury into the food chain. Mercury can be actively absorbed and methylated by bacteria, some of which can be released back into the ocean (Schaefer et al. 2014). Phytoplankton can also actively accumulate methylmercury within their cells (Pickhardt and Fisher 2007); this cytoplasmic methylmercury can be more easily transferred to the trophic chain than inorganic mercury (Harding et al. 2018); this represents a key step in the bioaccumulation and biomagnification of mercury. In the ocean, trophic transfer is the main pathway for marine organisms to take in and accumulate mercury.
The methylation of inorganic mercury in the water column may represent an important source of methylmercury in pelagic marine food webs (Lehnherr et al. 2011), and studies have shown that the total mercury content of predatory pelagic fish, and their prey, is similar to that of dissolved organic mercury in seawater, which increases with water column depth, thus indicating that mercury content in marine predators was affected by their foraging depth (Choy et al. 2009). Horizontal habitats (i.e., distance from the coast), and trophic levels, also result in differences in mercury accumulation between species; moreover, coastal fish are more susceptible to contamination than offshore fish (Le Croizier et al. 2019).
Once mercury enters the body, it binds strongly with protein sulfhydryl groups (SH). As a consequence, the elimination and excretion of the bioaccumulated mercury is very slow and takes a long period of time (Chouvelon 2018; Maulvault et al. 2016). Body length and weight are the main factors responsible for intra-species differences in fish with respect to mercury concentrations. The bioaccumulation of mercury in marine fish is highly variable and its concentration is affected by species-specific differences in physiological and ecological characteristics (Table 1). Furthermore, mercury concentrations in fish tissues are known to be positively correlated with the iron, total organic carbon, ammonia, and nitrogen content within the water column but are negatively correlated with alkalinity, dissolved oxygen and pH (Glover et al. 2010). Consequently, there is a wide range of factors responsible for the affected by observed differences in the bioaccumulation of mercury in marine fish. All of these factors may exert influence over the toxicity of mercury in fish.
The highest total mercury levels in fish tissues were previously shown to be in the muscle, followed by liver or kidney. Fins showed the lowest levels, and there was no significant difference in the total mercury concentrations between different types of muscle (O’Bryhim et al. 2017; Harley et al. 2015). However, a study of mercury concentrations in southern California found that the level of mercury in the liver of the blue marlin was higher than that in the muscle (Vega-Sanchez et al. 2017). Therefore, it is still necessary to carefully examine the assumptions related to organ mercury concentrations and focus on methylmercury concentrations.
Toxic effects of mercury in marine fish
Even at low concentrations, low mercury concentrations in the ocean can still lead to genetic, biochemical, physiological, morphological and behavioral changes in fish (Huang et al. 2011). Mercury toxicity depends on a variety of factors, including speciation, bioavailability and the absorption and the transformation of mercury. These factors are also known to differ in different species, and the effects of mercury toxicity on different individuals, species and stages of life can vary greatly (Morcillo et al. 2016c).
Mercury toxicity in embryonic and larval stages of marine fish
When considering the entire life cycle, it is clear that the embryonic and larval stages are the most sensitive periods for mercury exposure in fish. Frequent mercury exposure can affect the development of several organs (such as abnormal eye development and cardiac malformation) and metabolic processes (Huang et al. 2011). Mercury toxicity can also affect the entire embryonic development process, can activate energy-consuming detoxification processes (Sfakianakis et al. 2015) and consumes a significant amount of energy, resulting in a reduction in energy that could otherwise be used for growth. Mercury toxicity can also cause slow development, morphological abnormality, dysfunction and even death (Sfakianakis et al. 2015). Sub-chronic toxicity tests on red sea bream (Pagrus major) indicated that mercury concentrations exceeding 20 µg/L can reduce hatching success, increase mortality and induce teratogenicity in both embryo and larvae (Huang et al. 2011). In a previous paper, the larvae of large yellow croaker (Pseudosciaena crocea) were exposed to 0–4 µg/L of methylmercury for 30 days; results showed that the expression of TCTP, GST3, Hsp70 and Hsp27 mRNA (related to immune response) were all up-regulated in the presence of methylmercury and that these changes occurred in a dose-dependent manner (Wu et al. 2018). Furthermore, mercury poisoning can also indirectly affect the survival skills (i.e. foraging and predator evasion) of fish. If fish are exposed to mercury, their feeding can be affected or even stopped; this may lead to a deficiency of essential nutrients for larval development (Liu et al. 2016).
Teratogenic effects of mercury on marine fish
Malformations due to mercury exposure exert a devastating effect on fish, since they not only affect the survival mode, growth rate and external morphology of the fish, but can also affect the most important behavioral characteristics in the life of fish, such as hunting, avoiding predators and long-distance migration (Mora-Zamorano et al. 2017, 2016; Webber and Haines 2003). Common malformations include those in the spine, bladder, head region and fins. The most common malformations observed in fish occur in the spine. The most obvious spinal deformities include lordosis (sacral doris), kyphosis (sacral curvature) and scoliosis (lateral curvature) (Morcillo et al. 2016b; Sfakianakis et al. 2015). Deformities (particularly skeletal deformities) can interfere with the ability of fish to interact with the environment and reduces their chances of survival (Huang et al. 2011). Poor nutrition can also result in bone deformities (Liu et al. 2016). Moreover, mercury metal ions can cause deformity by altering the integrity of the notochord during development or by inducing bone growth via neuromuscular effects (Avyle et al. 1989).
Hepatotoxicity of mercury on marine fish
Mercury exposure may cause hepatic histopathological damage (including vacuolization, parenchyma disorganization and pyknotic nucleus), and cause other syndromes. A previous study of the liver in marine medaka (Oryzias melastigma) exposed to different concentrations of mercuric chloride showed that mercury exposure increased the accumulation of mercury in the liver and subsequently impaired the ultrastructure of the liver (Wang et al. 2013; Chen et al. 2017). Quantitative proteomic analysis further suggested that Hg-induced hepatotoxicity may involve oxidative stress, cytoskeletal damage, immunotoxicity and changes in energy metabolism, indicating that mitochondria may be the primary target for mercury attack in cells (Wang et al. 2013; Chen et al. 2017). Olsvik et al. (2011) exposed Atlantic cod (Gadus morhua) larvae to mercury-rich sediments for 5 weeks and found that calreticulin, HSP70 and heme oxygenase mRNA were significantly up-regulated in fish gills. In the liver, calreticulin, heme oxygenase, transferrin and WAP65 were also upregulated but glutathione peroxidase 4B and zona pellucida 3 were significantly down-regulated.
Neurotoxic effects of mercury on marine fish
Although Hg can produce a variety of toxicity effects in organisms, the neurotoxicity of Hg has always been a significant concern. Mercury can accumulate in the brain and thereafter can cause significant damage. A previous study of marine medaka (O. melastigma) exposed to mercury chloride over long periods showed that inorganic mercury may cause neurotoxicity by inducing oxidative stress, cytoskeletal assembly dysfunction and metabolic disorders (Wang et al. 2015; Barboza et al. 2018a). Proteomic analysis of brain tissue from the Atlantic cod (Gadus morhua) also showed that after exposure to methylmercury, the levels of 71 proteins changed by 20% or more. These proteins were associated with major known molecular targets and mechanisms of methylmercury-induced neurotoxicity in mammals, such as mitochondrial dysfunction, oxidative stress (Berg et al. 2010). Changes of the proteome in the brain of juvenile beluga (Huso huso) further confirmed that methylmercury induces toxicity through oxidative stress and apoptosis, which suggested that chronic methylmercury exposure can cause an important metabolic defect in the brain (Keyvanshokooh et al. 2009). Mercury may also induce morphological changes in the brain, such as changes in the total number and volume of neurons and glial cells in specific areas of the brain, accompanied by changes in swimming behavior; it can also cause neurological damage over the long-term (Pereira et al. 2016). Studies have also shown that neurobehavioral deficits induced by mercury exposure can be passed down steadily to the next generation in white seabream (Diplodus sargus); these deficits were conveyed via the sperm and persisted in each generation. This startling discovery showed that such neurological diseases can persist in a fish population for generations, even after the source of the pollution has been removed (Senger et al. 2010).
Reproductive toxicity of mercury to marine fish
Mercury exposure is not entirely risk-free for adult fish. Mercury can accumulate in the gonads of fish and may affect the reproductive system and inhibit the growth and development of fish gonads (Liao et al. 2006). The hypothalamic–pituitary–gonadal (HPG) axis regulates reproductive activity by secreting different hormones and plays a key role during the normal development of the fish reproductive system (Dang et al. 2015). Mercury exposure can interfere with the expression of genes related to the HPG axis and alter sex hormone levels, potentially affecting fish reproduction. In addition, exposure to inorganic mercury is known to induce oxidative stress, leading to histological damage in the gonads of fish such as the thickening of tubule walls (Zhang et al. 2016). After exposure to mercury, testosterone levels in male testes are significantly reduced and spermatogenic degeneration and necrosis can also be detected, including Sertoli cell hypertrophy and interstitial inflammation (Liao et al. 2006; Zhang et al. 2016). Moreover, the connections between follicular cells and oocytes can be disrupted, which may lead to a delay in ovarian development (Zhang et al. 2016).
Other toxicological effect of mercury exposure to marine fish
In fish, the branchial epithelium is the site of gas exchange, acid-base balance, nitrogen waste excretion and ion regulation. Gills can interact directly and continuously with the surrounding environment, and represents the main target organ for environmental pollutants (Evans 1987; Brunelli et al. 2010; Wendelaar Bonga 1997; Barboza et al. 2018b). Inorganic mercury and methylmercury can exert toxic effects on gills by interfering with multiple metabolic pathways. Taurine is a sulfur-containing amino acid mainly involved in the maintenance of cellular homeostasis. Cell swelling caused by hypo-osmotic stress is followed by a regulatory volume decrease (RVD) that occurs via cellular extrusion of ions, mainly taurine (Fugelli and Thoroed 1990). Therefore, wild gold gray mullet (Liza aurata) exposure to mercury-containing sediments indicated that the accumulation of mercury in the gills of fish caused obstacles in the process of ionic regulation and osmotic regulation, due to the decrease of taurine and glycerophosphocholine, as well as the increase of creatine levels (Cappello et al. 2016).
In terms of histopathology, mercury can also affect the morphology of the gill epithelium, causing morphological and pathological changes in the sputum (such as cellular necrosis, shortening of the secondary lamellae, hyperplasia of the gill filaments and severe edema), thus resulting in impaired gill function. Rainbow trout (Oncorhynchus mykiss) exposed to 50 µg/L mercuric chloride or methylmercury showed a variety of morphological changes, including a reduction in the height of the lamellar cell ridges; other effects included vacuolated epithelial cells and the degeneration of chloride cells (Olson et al. 1973).
In addition to the toxicological effects of mercury exposure upon vital tissues in marine fish, as described above, there are also other effects which should be considered, for example the effects of such toxicity upon cellular proteins. For example, in a previous study, fibroblast SAF-1 cells, harvested from the marine teleost fish (Sparus aurata L.) were exposed to mercury for 24 h; this resulted in the increased production of reactive oxygen species and apoptotic cell death. The corresponding gene expression profile indicated that the potential mechanisms underlying the observed changes included induction of the metallothionein protective system, cellular oxidative stress and apoptosis (Morcillo et al. 2016a). In another study, erythrocytes from the gilthead seabream (Sparus aurata L.) and European sea bass (D. labrax L.) were exposed to methylmercury for 24 h in vitro; results showed that the gene expression profiles of heat shock protein 70 and 90, superoxide dismutase, metallothionein, glutathione reductase, anti-apoptosis Bcl2-related X protein, catalase, peroxidase, and calpain1, all changed. This showed that oxidative mechanisms were activated in order to protect cells but ultimately the cells could not cope and suffered apoptotic cell death (Morcillo et al. 2016b).
Conclusion
Even at low exposure levels, mercury can not only affect the genetic mutation, tissues, physiology, morphology and behavior of marine fish, but can also affect their survival, growth and development. Generally, the toxicity of pollutant exposure could be tested using an appropriate fish model, such as the zebrafish or medaka which both have a completely sequenced genome, a transparent body and for which laboratory conditions can be easily controlled. However, there are highly variable and significant differences among the teleosts due to a wide range of physiological and ecological characteristics. Carrying out more toxicity tests on marine fish species would help us to further elucidate the toxicological responses of marine fish to mercury.
Mercury accumulation in marine products has become an important global issue that must be addressed. Tracking the source of this mercury and understanding how mercury circulates in the marine ecosystem have become key research targets. It is clear that different publications have reported different orders of mercury pollution in fish tissues. Further experiments, which test the mercury concentration of specific organs, are now required to investigate the reasons for such differences. Moreover, the toxicity effect of mercury upon fish is also related to gender (Zhang et al. 2016); the reasons for this are also unknown and require further study.
References
Anual ZF, Maher W, Krikowa F, Hakim L, Ahmad NI, Foster S (2018) Mercury and risk assessment from consumption of crustaceans, cephalopods and fish from west peninsular Malaysia. Microchem J 140:214–221
Avyle MJ, Garvick SJ, Blazer VS, Hamilton SJ, Brumbaugh WG (1989) Skeletal deformities in smallmouth bass, Micropterus dolomieui, from Southern Appalachian Reservoirs. Arch Environ Contam Toxicol 18:688–696
Barboza LGA, Vieira LR, Branco V, Figueiredo N, Carvalho F, Carvalho C, Guilherminol L (2018a) Microplastics cause neurotoxicity, oxidative damage and energy-related changes and interact with the bioaccumulation of mercury in the European seabass, Dicentrarchus labrax (Linnaeus, 1758). Aquat Toxicol 195:49–57
Barboza LGA, Vieira LR, Branco V, Carvalho C, Guilherminol L (2018b) Microplastics increase mercury bioconcentration in gills and bioaccumulation in the liver, and cause oxidative stress and damage in Dicentrarchus labrax juveniles. Sci Rep 8:15655
Berg K, Puntervoll P, Valdersnes S, Goksøyr A (2010) Responses in the brain proteome of Atlantic cod (Gadus morhua) exposed to methylmercury. Aquat Toxicol 100:51–65
Brunelli E, Mauceri A, Fasulo S, Giannetto A, Maisano M, Tripepi S (2010) Localization of aquaporin 1 and 3 in the gills of the rainbow wrasse Coris julis. Acta Histochem 112:251–258
Budtz-Jorgensen E, Keiding N, Grandjean P, Weihe P (2002) Estimation of health effects of prenatal methylmercury exposure using structural equation models. Environ Health. https://doi.org/10.1186/1476-069X-1-2
Cabañero AI, Madrid Y, Cámara C (2004) Selenium and mercury bioaccessibility in fish samples: an in vitro digestion method. Anal Chim Acta 526(1):51–61
Cappello T, Pereira P, Maisano M, Mauceri A, Pacheco M, Fasulo S (2016) Advances in understanding the mechanisms of mercury toxicity in wild golden grey mullet (Liza aurata) by 1H NMR-based metabolomics. Environ Pollut 219:139–148
Chen QL, Sun YL, Liu ZH, Li YW (2017) Sex-dependent effects of subacute mercuric chloride exposure on histology, antioxidant status and immune-related gene expression in the liver of adult zebrafish (Danio rerio). Chemosphere 188:1–9
Chouvelon T, Cresson P, Bouchoucha M, Brach-Papa C, Bustamante P, Crochet S, Marco-Mirallea F, Thomas B, Knoery J (2018) Oligotrophy as a major driver of mercury bioaccumulation in medium-to high-trophic level consumers: a marine ecosystem-comparative study. Environ Pollut 233:844–854
Choy CA, Popp BN, Kaneko JJ, Drazen JC (2009) The influence of depth on mercury levels in pelagic fishes and their prey. Proc Natl Acad Sci USA 106(33):13865–13869
Dang Y, Giesy JP, Wang JH, Liu CS (2015) Dose-dependent compensation responses of the hypothalamic-pituitary-gonadal-liver axis of zebrafish exposed to the fungicide prochloraz. Aquat Toxicol 160:69–75
Djinovic-Stojanovic J, Nikolic D, Vranic D, Stefanovic S, Milijasevic M, Babic J, Jankovic S (2015) Distribution of mercury in three marine fish species. Procedia Food Sci 5:65–68
Evans DH (1987) The fish gill: site of action and model for toxic effects of environmental pollutants. Environ Health Perspect 71:47–58
Fugelli K, Thoroed SM (1990) Taurine and volume regulation in fish cells. Prog Clin Biol Res 351:481–488
Glover JB, Domino ME, Altman KC, Dillman JW, Castleberry WS, Eidson JP, Mattocks M (2010) Mercury in South Carolina Fishes, USA. Ecotoxicology 19(4):781–795
Gonzalez-Raymat H, Liu GL, Liriano C, Li YB, Yin YG, Shi JB, Jiang GB, Cai Y (2017) Elemental mercury: its unique properties affect its behavior and fate in the environment. Environ Pollut 229:69–86
Harding G, Dalziel J, Vass P (2018) Bioaccumulation of methylmercury within the marine food web of the outer Bay of Fundy, Gulf of Maine. PLoS One 13(7):e0197220
Harley J, Lieske C, Bhojwani S, Castellini JM, Lopez JA, O’Hara TM (2015) Mercury and methylmercury distribution in tissues of sculpins from the Bering Sea. Polar Bio 38(9):1535–1543
Huang W, Gao LA, Shan XJ, Lin LS, Dou SZ (2011) Toxicity testing of waterborne mercury with red sea bream (Pagrus major) embryos and larvae. Bull Environ Contam Toxicol 86(4):398–405
Jeevanaraj P, Hashim Z, Elias SM, Aris AZ (2016) Mercury accumulation in marine fish most favoured by Malaysian women, the predictors and the potential health risk. Environ Sci Pollut Res 23:23714–23729
Keyvanshokooh S, Vaziri B, Gharaei A, Mahboudi F, Esmaili-Sari A, Shahriari-Moghadam M (2009) Proteome modifications of juvenile beluga (Huso huso) brain as an effect of dietary methylmercury. Comp Biochem Physiol D-Genomics Proteomics 4(4):243–248
Lamborg CH, Hammerschmidt CR, Bowman KL, Swarr GJ, Munson KM, Ohnemus DC, Lam PJ, Heimburger LE, Rijkenberg MJA, Saito MA (2014) A global ocean inventory of anthropogenic mercury based on water column measurements. Nature 512(7512):65–69
Le Croizier G, Schaal G, Point D, Le Loc’h F, Machu E, Fall M, Munaron JM, Boye A, Walter P, Lae R (2019) Stable isotope analyses revealed the influence of foraging habitat on mercury accumulation in tropical coastal marine fish. Sci Total Environ 650:2129–2140
Lehnherr I, St Louis VL, Hintelmann H, Kirk JL (2011) Methylation of inorganic mercury in polar marine waters. Nat Geosci 4(5):298–302
Li P, Feng XB, Yuan XB, Chan HM, Qiu GL, Sun GX, Zhu YG (2012) Rice consumption contributes to low level methylmercury exposure in southern China. Environ Int 49:18–23
Liao CY, Fu JJ, Shi JB, Zhou QF, Yuan CG, Jiang GB (2006) Methylmercury accumulation, histopathology effects, and cholinesterase activity alterations in medaka (Oryzias latipes) following sublethal exposure to methylmercury chloride. Environ Toxicol Pharmacol 22(2):225–233
Liu Q, Kingler RH, Wimpee B, Dellinger M, King-Heiden T, Grzybowski J, Gerstenberger SL, Weber DN, Carvan MJ (2016) Maternal methylmercury from a wild-caught walleye diet induces developmental abnormalities in zebrafish. Reprod Toxicol 65:272–282
Liu YY, Buchanan S, Anderson HA, Xiao ZW, Persky V, Turyk ME (2018) Association of methylmercury intake from seafood consumption and blood mercury level among the Asian and Non-Asian populations in the United States. Environ Res 160:212–222
Matulik AG, Kerstetter DW, Hammerschlag N, Divoll T, Hammerschmidt CR, Evers DC (2017) Bioaccumulation and biomagnification of mercury and methylmercury in four sympatric coastal sharks in a protected subtropical lagoon. Mar Pollut Bull 116(1–2):357
Maulvault AL, Custodio A, Anacleto P, Reploho T, Pousao P, Nunes ML, Diniz M, Rosa R, Marques A (2016) Bioaccumulation and elimination of mercury in juvenile seabass (Dicentrarchus labrax) in a warmer environment. Environ Res 149:77–85
Meador JP, Robisch PA, Clark RC, Ernrst DW (1998) Elements in fish and sediment from the Pacific Coast of the United States: results from the national benthic surveillance project. Mar Pollut Bull 37(1–2):56–66
Mora-Zamorano FX, Klingler R, Murphy CA, Basu N, Head J, Carvan MJ (2016) Parental whole life cycle exposure to dietary methylmercury in Zebrafish (Danio rerio) affects the behavior of offspring. Environ Sci Technol 50(9):4808–4816
Mora-Zamorano FX, Klingler R, Basu N, Head J, Murphy CA, Binkowski FP, Larson JK, Carvan MJ (2017) Developmental methylmercury exposure affects swimming behavior and foraging efficiency of Yellow Perch (Perca flavescens) larvae. Acs Omega 2(8):4870–4877
Morcillo P, Esteban MA, Cuesta A (2016a) Heavy metals produce toxicity, oxidative stress and apoptosis in the marine teleost fish SAF-1 cell line. Chemosphere 144:225–233
Morcillo P, Romero D, Meseguer J, Esteban MA, Cuesta A (2016b) Cytotoxicity and alterations at transcriptional level caused by metals on fish erythrocytes in vitro. Environ Sci Pollut Res 23:12312–12322
Morcillo P, Meseguer J, Esteban MA, Cuesta A (2016c) In vitro effects of metals on isolated head-kidney and blood leucocytes of the teleost fish Sparus aurata L. and Dicentrarchus labrax L. Fish Shellfish Immunol 54:77–85
O’Bryhim JR, Adams DH, Spaet JLY, Mills G, Lance SL (2017) Relationships of mercury concentrations across tissue types, muscle regions and fins for two shark species. Environ Pollut 223:323–333
Olson KR, Fromm PO, Frantz WL (1973) Ultrastructural changes of rainbow trout gills exposed to methyl mercury or mercuric chloride. Fed Proc 32:261
Olsvik PA, Brattas M, Lie KK, Goksoyr A (2011) Transcriptional responses in juvenile Atlantic cod (Gadus morhua) after exposure to mercury-contaminated sediments obtained near the wreck of the German WW2 submarine U-864, and from Bergen Harbor, Western Norway. Chemosphere 83:552–563
Pereira P, Puga S, Cardoso V, Pinto-Ribeiro F, Raimundo J, Barata M, Pousão-Ferreira P, Pacheco M, Almeida A (2016) Inorganic mercury accumulation in brain following waterborne exposure elicits a deficit on the number of brain cells and impairs swimming behavior in fish (White seabream—Diplodus sargus). Aquat Toxicol 170:400–412
Pickhardt PC, Fisher NS (2007) Accumulation of inorganic and methylmercury by freshwater phytoplankton in two contrasting water bodies. Environ Sci Technol 41:125–131
Schaefer JK, Szczuka A, Morel FMM (2014) Effect of divalent metals on Hg(II) uptake and methylation by bacteria. Environ Sci Technol 48:3007–3013
Selin NE (2009) Global biogeochemical cycling of mercury: a review. Annu Rev Environ Resour 34:43–63
Senger MR, Rosemberg DB, Seibt KJ, Dias RD, Bogo MR, Bonan CD (2010) Influence of mercury chloride on adenosine deaminase activity and gene expression in zebrafish (Danio rerio) brain. Neurotoxicology 31(3):291–296
Sfakianakis DG, Renieri E, Kentouri M, Tsatsakis AM (2015) Effect of heavy metals on fish larvae deformities: a review. Environ Res 137:246–255
Sinkus W, Shervette V, Ballenger J, Reed LA, Plante C, White B (2016) Mercury bioaccumulation in offshore reef fishes from waters of the Southeastern USA. Environ Pollut 228:222–233
Taylor DL, Kutil NJ, Malek AJ, Collie JS (2014) Mercury bioaccumulation in cartilaginous fishes from Southern New England coastal waters: contamination from a trophic ecology and human health perspective. Mar Environ Res 99(4):20–33
Vega-Sanchez B, Ortega-Garcia S, Ruelas-Inzunza J, Frias-Espericueta M, Escobar-Sanchez O, Guzman-Rendon J (2017) Mercury in the Blue Marlin (Makaira nigricans) from the southern gulf of California: tissue distribution and inter-annual variation (2005–2012). Bull Environ Contam Toxicol 98(2):156–161
Wang MH, Wang YY, Zhang L, Wang J, Hong HS, Wang DZ (2013) Quantitative proteomic analysis reveals the mode-of-action for chronic mercury hepatotoxicity to marine medaka (Oryzias melastigma). Aquat Toxicol 130–131:123–131
Wang YY, Wang DZ, Lin L, Wang MH (2015) Quantitative proteomic analysis reveals proteins involved in the neurotoxicity of marine medaka Oryzias melastigma chronically exposed to inorganic mercury. Chemosphere 119:1126–1133
Webber HM, Haines TA (2003) Mercury effects on predator avoidance behavior of a forage fish, golden shiner (Notemigonus crysoleucas). Environ Toxicol Chem 22:1556–1561
Wendelaar Bonga SE (1997) The stress response in fish. Physiol Rev 77(3):591–625
Wu FZ, Huang W, Liu Q, Xu XQ, Zeng JN, Cao L, Hu J, Xu XD, Gao YX, Jia SH (2018) Responses of antioxidant defense and immune gene expression in early life stages of large yellow croaker (Pseudosciaena crocea) under methyl mercury exposure. Front Physiol 9:1436
Zaza S, de Balogh K, Palmery M, Alberto A, Stacchini P, Elena VR (2015) Human exposure in Italy to lead, cadmium and mercury through fish and seafood product consumption from Eastern Central Atlantic Fishing Area. J Food Compos Anal 40:148–153
Zhang QF, Li YW, Liu ZH, Chen QL (2016) Reproductive toxicity of inorganic mercury exposure in adult zebrafish: histological damage, oxidative stress, and alterations of sex hormone and gene expression in the hypothalamic-pituitary-gonadal axis. Aquat Toxicol 177:417–424
Acknowledgements
We would like to acknowledge the support of the National Natural Science Foundation of China (No. 41722110 and No. 41571474), the Jilin Province Natural Science Foundation of China (No. 20170101203JC).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zheng, N., Wang, S., Dong, W. et al. The Toxicological Effects of Mercury Exposure in Marine Fish. Bull Environ Contam Toxicol 102, 714–720 (2019). https://doi.org/10.1007/s00128-019-02593-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-019-02593-2