Abstract
The study explored (I) the concentration of heavy metals in water samples (II) their bioaccumulation in common carp Cyprinus carpio (III) and the subsequent genotoxicity in the selected organs of carp; from river Kabul, Khyber Pakhtunkhwa Pakistan. Except for Mercury (Hg) the water samples had all the heavy metals within permissible limits of recommended dietary allowance (RDA). Nonetheless a number of heavy metals (Zn, Ni, Cr, Cd, Pb and Hg) showed bioaccumulation at levels higher than permissible. Zinc (Zn) was the most while Cadmium (Cd) was the least accumulated metal in all tissue samples analyzed. The metal burden in different organs of C. carpio was in sequence of intestine > skin > liver > gills > muscle. The Comet assay established DNA damage in selected organs to be in accordance with metal burden; the most to least damage being in sequence of blood > intestine > skin > liver > gills > muscle. In conclusion assessment of DNA damage in the organs of C. carpio appears to be a useful bio-marker to evaluate genotoxic effects of heavy metal pollution.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
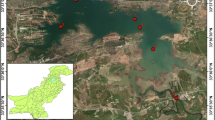
River Kabul originates from the base of Unai Pass in the Paghman Mountains of Afghanistan (Siraj et al. 2016). Before entering the valley of Peshawar the river is considered less polluted and safe because it is far away from dense industrial and human population and a dam called Warsak dam is constructed on it as water reservoir for electricity production (Yousafzai et al. 2008). In Pakistan during the course of its flow it receives the inputs of sewage, agricultural runoff and industrial effluents from various sources in densely populated towns and agricultural fields. Untreated wastes of more than 72 industries are discharged directly into the River Kabul, causing environmental contamination and affecting fish health and production also selected as polluted Sites 2 and 3 (Fig. 1).
To date fifty-four fish species have been identified from the Kabul River and its tributaries, of which thirty-five are described as common fish species (Rafique 2001). The common carp C. carpio is a prominent feature of fish fauna of the region. It is native to India, Bangladesh, Pakistan and Myanmar. The species has a very important place among the other fish species regarding its economic value, rapid growth and tasty meat. This fish prefers natural lakes, ponds and deep and slow flowing rivers. It can reach to a length of 1 m and weight of 40 kg (Geldiay and Balık 1988). It is a voracious omnivorous fish with zooplankton, phytoplankton, water plants and detritus being its major diet.
Heavy metals from both natural and anthropogenic sources are continuously released into aquatic ecosystems. These metals pose a serious threat to living organisms due to their toxicity, long persistence, bioaccumulation, and bio-magnification in food chains (Pandy et al. 2003). High concentrations of heavy metals could damage DNA in fish (Serpi et al. 2006). For instance genotoxic effects of cadmium (Cd) and mercury (Hg) are well known for some fish species (Risso-De Faverney et al. 2001).
In this scenario fish can act as model animals for pollution monitoring studies in aquatic ecosystems (Kumar et al. 2009). The current study which is an effort in this direction first demonstrates the concentrations of heavy metals (Zn, Ni, Cr, Cu, Cd, Pb, Fe, Mn and Hg) in river water. Next it highlights bioaccumulation in selected organs of common carp (muscle, gills, intestine, skin and liver). Finally it investigates genotoxicity in various organs. The study verifies that even when heavy metals are under permissible values of aquatic environment they can still cause bioaccumulation and DNA damage.
Materials and Methods
This study involved three sites along the stretch of the River Kabul in Khyber Pakhtunkhwa, Pakistan. One site “Warsak dam” was designated as reference site (Site 1) due to relatively less human activities in its surrounding, while the other two sites i.e. Aman Garh (Site 2) and Nowshera (Site 3) were designated as polluted sites. These two sites were receiving pollutants from the industries, domestic, municipal and agricultural sources from Aman Garh industrial zone, Mardan, Nowshera and Risalpur cities and the adjacent areas (Fig. 1).
Water samples of about 1 L were collected from the sampling sites in cleaned polypropylene bottles, then filtered with Whatmann filter paper and preserved in 5 mL of 55% HNO3 per liter of water to prevent metal adsorption on the inner surface of the container and stored at 4°C before their analyses.
Twenty fresh samples each of the C. carpio were collected through professional local fishermen from selected sites. Live fish samples were handed over to the researcher, immediately anesthetized and the blood was collected by cardiac punctures through sterilized syringes, immediately transferred to EDTA containing glass tubes to prevent blood clotting. The collected fishes were dissected for obtaining organs like an intestine, gills, skin, liver and muscle, then washed with distilled water and shifted to marked sterilized polythene bags. One gram tissue from each organ was grinded on grinder machine and 5 mL PBS was added to the grinded tissue to obtain cell suspension. The freshly made cell suspension and blood was used in comet assay. For metal analysis the remaining portions of each organ were placed in properly marked polythene bags and were stored in the freezer at − 20°C until further analysis.
For the metal analysis each 100 mL of acidified water samples were evaporated in a volumetric flask on a hot plate to about 20 mL within a fume cupboard. After cool, 5 mL of HNO3 (55%) and 10 mL of percholoric acid (70%) were added. The mixture was evaporated on a hot plate until the brown fumes converted into dense white fumes of perchloric acid. The samples were allowed to cool and diluted to 100 mL with distilled water. The solutions were then aspirated into an atomic absorption spectrophotometer (Perkin Elmer, Analyst A 800) by using an air acetylene flame for the determination of these metals. Standard curves were prepared and the optical densities (ODs) obtained were calibrated against the standard curves to know the concentration of metals like Zn, Ni, Cr, Cu, Cd, Pb, Fe, Mn and Hg present in the water samples (Table 1).
The tissue samples like gills, skin, liver, intestine and muscle were thawed, rinsed in distilled water and blotted with blotting paper. Fifty grams of each tissue were shifted to 100 mL volumetric flasks. Samples were digested according to the methods described in our previous work (Siraj et al. 2016). To each flask, 5 mL nitric acid (55%) and 1 mL perchloric acid (70%) were added, and the flasks were kept overnight. The next day, a second dose of 5 mL nitric acid (55%) and 4 mL perchloric acid (70%) were added to each flask. The flasks were then placed on a hot plate and allowed to digest at 200 to 250°C until a transparent and clear solution was obtained. After digestion, samples were cooled and were diluted to 100 mL with de-ionized water. Atomic absorption spectrophotometer (Perkin Elmer, Analyst A 800) was used to determine the concentration of heavy metals (Zn, Ni, Cr, Cu, Cd, Pb, Fe, Mn and Hg) in the tissue samples. Each sample was analyzed in triplicate, and the results were given as (μg/g wet weight).
Assessment of genotoxicity was conducted through comet assay according to the method mentioned in our previous publication (Khisroon et al. 2015), with slight modifications. Pre-coated slides were prepared with 1% normal melting agarose (NMA). 75 μL of low melting point agarose (LMPA) (1%) mixed with 10 μL of blood or cell suspension of grinded tissues and added to pre-coated slides and kept in 4°C for 5 min to solidify. 85 μL of LMPA was added to the slide and was returned to 4°C for 5 min.The slides were then transferred to lysing solution [(2.5 M NaCl, 100 mM Na2EDTA, 10 mM Tris, pH 10) with 1% Triton X-100 and 10% DMSO added just before use] and placed in the refrigerator for at least 2 h at 4°C.After lysing the slides were placed in the electrophoresis buffer (300 mM NaOH and 1 mM EDTA, pH 13) and left for 20 min and then subjected to electrophoresis for 25 min at 300 mA and 25 V. After electrophoresis, the slides were washed with neutralization buffer (400 mM Tris, pH 7.5).The slides were stained with 70 μL DNA specific fluorescent dye acridine orange (20 μg/mL), cover slips were placed on it, and viewed at 200× of fluorescent microscope (Nikon Eclipse 80 i) equipped with 450–490 nm excitation filter. In order to calculate DNA damage, 100 cells per sample were chosen randomly and analyzed visually according to comet appearance. Five classes, i.e. from class 0 (no DNA damage) to class 4 (maximum DNA damage) gave sufficient declaration (Collins 2004). Total comet score (TCS) was then calculated according to the formula, TCS = 0(n) + 1(n) + 2(n) + 3(n) + 4(n), where “n” indicates number of cells in each class (Collins 2004).
Mean and standard error (SE) of the data was taken. Statistical analysis of the results was conducted by using SPSS version 20.0 (IBM, Armonk, NY, USA) for windows software. Differences in the means of fishes of Site 1 (reference site), Site 2, and Site 3 were calculated using one-way analysis of variance (ANOVA) and Tukey test (p ≤ 0.05).
Results and Discussion
pH values of water samples from Sites 1, 2 and 3 were 7.1, 7.8 and 7.9, respectively. Mean values of metals in water at selected sites of the River Kabul and the permissible guide lines of these metals by the National Environmental Quality Standards (NEQS) are given in Table 2. All metals except Hg were within the permissible limits at all the three sites of River Kabul. The general pattern of metal concentrations in water sample at reference site (Site 1) was Zn > Ni > Cr > Mn > Cd > Fe > Hg > Cu > Pb, at polluted Site 2 was Zn > Pb > Cd > Ni > Fe > Mn > Cu > Hg > Cr and at polluted Site 3 was Zn > Pb > Cd > Fe > Ni > Mn > Hg > Cu > Cr, respectively. Different sites had an order of Site 3 > Site 2 > Site 1, respectively. Mean concentration (µg/g wet weight) of metals in gills, muscle, intestine, skin and liver of C. carpio has been given in Table 3. The metal concentration in different organs of C. carpio were in a sequence of Intestine > skin > liver > gills > muscle.
The general pattern of metal concentrations in the skin at the polluted Sites (2 and 3) were in the order of Zn > Cr > Pb > Cu > Ni > Fe > Mn > Hg > Cd, in gills were Zn > Cr > Cu > Pb > Ni > Fe > Mn > Hg > Cd, in intestine were Zn > Pb > Cr > Cu > Fe > Ni > Mn > Hg > Cd, in liver were Zn > Cr > Cu > Pb > Fe > Mn > Hg > Ni > Cd and in muscle were Zn > Cr > Cu > Pb > Fe > Mn > Hg > Fe > Mn > Ni > Cd.
Heavy metals such as Zn, Ni, Cr, Cu and Pb appeared to be accumulating more in the gills and liver than intestine, skin and muscles. Heavy metals like Cu and Zn are essential elements as these are carefully regulated by physiological mechanisms in most organisms (Eisler 1993). However, they are regarded as potential hazards that can endanger both animals and human health. Knowledge of their concentrations in the fish is therefore important both with respect to nature management and human consumption of fish (Jabeen and Chaudhry 2013).
High levels of Zn in the intestine, skin, liver, gills and muscle in present findings may pose adverse effects on the fish health. Mercury is a highly toxic and the most closely monitored contaminant in fish. More Hg concentration in the intestine and gills indicated the contamination of river water with mercury (Chaudhry and Jabeen 2011). Heavy metal like mercury affects behavior of vertebrates, inhibits enzyme activity and increases abnormal cell division, it is important to investigate mercury contamination in freshwater fish for the sake of fish as well as the consumer’s health. Cr is also known as a carcinogen and its high levels in fish tissue are of prime concern (Holmes et al. 2008).
Genotoxicity is a deleterious action, which affects a cell’s genetic material (Smith 1996). Heavy metals may be genotoxic that can damage DNA of a living cell (Igwilo et al. 2006). DNA damage/total comet score (TCS) in gills, muscle, intestine, skin and liver of C. carpio at three sites of the River Kabul has been shown in Table 4. All the studied tissues of fish from polluted sites showed significantly higher (p < 0.05) degree of DNA damage (TCS) as compared to those from reference site. Our results also agree with other previous data which shows increase in DNA damage in fish from polluted areas as compared to control sites (Javed et al. 2016; Hussain et al. 2016). Fish is a useful genetic model to find out the genotoxicological impacts of aquatic pollution (Kumar et al. 2009). Similarly in a previous finding it has been investigated that fish is used in testing of substances such as heavy metals, biocides, agrochemical, food additives and pharmaceuticals for genotoxicity (Brendler-Schwaab et al. 2005). The significantly higher levels of studied metals in blood and intestine could be linked to greater degree of DNA damage in C. carpio from polluted waters of River Kabul.
In conclusion, the results of the present study showed that water samples had all the heavy metals with in permissible limits of RDA except Hg. Furthermore Zn, Ni, Cr, Cd, Pb and Hg in muscle and skin of fish had crossed the permissible RDA limits. Zn was the highly and Cd was the least accumulated metals in all tissues of the fish inhabiting in polluted and control waters. Overall level of DNA damage in different organs was in sequence of blood > intestine > skin > liver > gills > muscle.
References
Brendler-Schwaab S, Hartman A, Pfuhler S, Speit G (2005) The in vivo comet assay: use and status in genotoxicity testing. Mutagenesis 20:245–254
Chaudhry AS, Jabeen F (2011) Assessing metal, protein, and DNA profiles in Labeo rohita from the Indus River in Mianwali, Pakistan. Environ Monit Assess 174:665–679
Collins AR (2004) The comet assay for DNA damage and repair: principles, applications, and limitations. Mol Biotechnol 26:249–261
Eisler R (1993) Zinc hazards to fish, wildlife and invertebrates: A synoptic review. U.S. Department of the Interior, Fish Wildlife Service. Biol Rep 10:106
Geldiay R, Balık S (1988) Turkiye Tatlısu Balıkları, Ege Universitesi Fen Fakultesi Kitaplar Serisi No: 97: 519, Bornova, İzmir
Holmes AL, Wise SS, Wise JP (2008) Carcinogenicity of hexavalent chromium. Ind J Med Res 128:353–372
Hussain B, Sultana T, Sultana S et al (2016) Variation in genotoxic susceptibility and biomarker responses in Cirrhinus mrigala and Catla catla from different ecological niches of the Chenab River. Environ SciPollut Res 23:14589–14599
Igwilo IO, Afonne OJ, Maduabuchi UJ, Orisakwe OE (2006) Toxicological study of the Anambra River in Otuocha, Anambra State, Nigeria. Arch Environ Occup Health 61:205–208
Jabeen F, Chaudhry AS (2013) Metal uptake and histological changes in gills and liver of Oreochromis mossambicus inhabiting Indus River. Pakistan J Zool 45:9–18
Javed M, Ahmad I, Usmani N, Ahmad M (2016) Studies on biomarkers of oxidative stress and associated genotoxicity and histopathology in Channa punctatus from heavy metal polluted canal. Chemosphere 151:210–219
Khisroon M, Khan A, Naseem M, Ali N, Khan S, Rasheed SB (2015) Evaluation of DNA damage in lymphocytes of radiology personnel by comet assay. J Occup Health 57:268–274
Kumar R, Nagpure NS, Kushwaha B et al (2009) Investigation of the genotoxicity of malathion to freshwater teleost fish Channa punctatus (Bloch) using the micronucleus test and comet assay. Arch Environ ContamToxicol 58:123–130
Pandy S, Parvez S, Sayeed I et al (2003) Biomarkers of oxidative stress: a comparative study of river Yamuna fish Wallago attu (Bl. & Schn). Sci Total Environ 309:105˗115
Rafique (2001) Fish fauna of the Hamalayas in Pakistan with comments on the origin and dispersal of its high Asian elements. Pakistan J Zool 33:279˗288
Risso-De Faverney C, Devaux A et al (2001) Cadmium induces apoptosis and genotoxicity in rainbow trout hepatocytes through generation of reactive oxygene species. Aquat Toxicol 53:65–76
Serpi U, Ayla C, Ozlem FC, Aysin G (2006) Cadmium-induced genotoxicity, cytotoxicity and lipid peroxidation in Allium sativum and Vicia faba. Oxford J Life Sci Med Mutagen 21:77˗81
Siraj M, Khisroon M, Khan A (2016) Bioaccumulation of heavy metals in different organs of Wallago attu from River Kabul Khyber Pakhtunkhwa, Pakistan. Biol Trace Elem Res 172:242–250
Smith TM (1996) The mechanism of benzene-induced leukemia: a hypothesis and speculations on the causes of leukemia. Environ Health Perspect 104:1219–1225
Yousafzai AM, Khan A, Shakoori AR (2008) Heavy metal pollution in River Kabul affecting the inhabitant fish population. Pak J Zool 40:331–339
Acknowledgements
The authors acknowledge the University of Peshawar for providing the research facilities. We are also thankful to those fishermen who helped us in collecting the fish samples.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Siraj, M., Khisroon, M., Khan, A. et al. Bio-monitoring of Tissue Accumulation and Genotoxic Effect of Heavy Metals in Cyprinus carpio from River Kabul Khyber Pakhtunkhwa Pakistan. Bull Environ Contam Toxicol 100, 344–349 (2018). https://doi.org/10.1007/s00128-017-2265-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-017-2265-5