Abstract
Environmental pollution of fish with organic contaminants is a topic of rising attention in Morocco. Polycyclic aromatic hydrocarbons (PAH) are prominent organic contaminants which are rapidly metabolized in fish. Their metabolites are accumulated in the bile fluid and can be used to assess PAH exposure. The two PAH metabolites 1-hydroxypyrene and 1-hydroxyphenanthrene were quantified in European eels (Anguilla anguilla) from two Moroccan river systems by high-performance liquid chromatography with fluorescence detection. Mean values ranged from 52 to 210 ng/mL 1-hydroxypyrene and from 61 to 73 ng/mL 1-hydroxyphenanthrene. The overall concentrations of PAH metabolites in eel from Morocco appeared moderate compared to eel from European rivers and coastal sites. The present study provides first information on concentrations of PAH metabolites in fish from Morocco.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Environmental pollution of fish is a topic of rising attention in Morocco (Bouachrine et al. 1998; El Morhit et al. 2009; Wariaghli et al. 2013). However, only few studies have been carried out so far, describing the potential threat of organic pollution to Moroccan aquatic ecosystem (Azdi et al. 2006; Chafik 2009; Er-Raioui et al. 2009; Hajjaj Hassani et al. 2006). The contamination with polycyclic aromatic hydrocarbons (PAH) has not yet been studied in fish from Moroccan waters. PAH are ubiquitous environmental contaminants found in marine sediments and waters associated with urbanized estuarine and coastal pollution as well as in rivers (Blahová et al. 2010; Meador et al. 1995). PAH are derived from both natural and anthropogenic sources. The latter can be related to pyrolysis and incomplete combustion of organic matter (Eisler 2007). Natural sources for PAH are e.g. forest fires and degradation of biological materials, which has led to the presence of these compounds in sediments and to the formation of fossil fuels (Eisler 2007). For the aquatic environment wastewater, atmospheric deposition and petroleum spillage are further prominent sources. PAHs and their intermediate degradation products have the potential to induce toxic or mutagenic effects in fish (Brinkmann et al. 2010, 2014; Monteiro et al. 2000) and in humans (Chen and Liao 2006).
PAH metabolites in the bile fluid are widely accepted as measures for PAH exposure in fish because of the rapid metabolization of PAH in vertebrates (Meador et al. 1995). As a consequence, PAH metabolites in fish are recommended as core monitoring parameters in European Seas (HELCOM 2012; OSPAR 2008). High performance liquid chromatography (HPLC) is widely used for the determination of PAH metabolites in different fish species (Harman et al. 2009; Kammann 2007; Pikkarainen 2006; Tairova et al. 2009; Vuorinen et al. 2006) and has been covered in a recent intercalibration exercise (Kammann et al. 2013).
The European eel (Anguilla anguilla) is probably one of the most vulnerable fish species to chemical pollution during its feeding and growth phase in freshwater as it is long-living, bottom dwelling, carnivore and has a high body fat content. Due to this, adverse effects of different contaminants have been speculated as a possible cause for the decline of the European eel stock, which is still considered as endangered and outside safe biological limits (ICES 2013). Since eels are quite heavily exploited in Moroccan inland waters (ICES 2011), a pollution assessment is needed, not only with regard to thresholds for human consumption but also to detect possible biological effects of contaminants on this species. Although some studies exist on PAH metabolites in eel (Kammann et al. 2014; Nagel et al. 2012a; Ruddock et al. 2003; Szlinder-Richert et al. 2014) up to now no data are available for PAH metabolites in fish from Moroccan waters. The aim of this study was to bridge this gap and to provide first information on concentrations of PAH metabolites of eels in Morocco; to evaluate spatial differences in PAH contamination and to discuss the possible threat for eel due to PAH contamination.
Materials and Methods
PAH metabolites in bile were determined by the method described by Kammann et al. (2014) with modified HPLC conditions. In brief, 25 µL fish bile were mixed with 95 µL water and 5 µL β-glucuronidase/arylsulfatase solution (30–60 U/mL). The mixture was subsequently incubated for 2 h at 37°C on a heated shaker. The enzymatic reaction was stopped by the addition of 125 µL of ethanol. After centrifugation, the supernatants were used for HPLC analysis immediately. PAH metabolites 1-hydroxypyrene (1-OH-Pyr) and 1-hydroxphenantrene (1-OH-Phen) were separated by HPLC (Lachrom System; Merck Hitachi). Samples were chromatographed on a Nucleosil 100-3 C18 (3 × 125 mm) reversed phase column at a flow of 0.55 mL/min. The initial mobile phase was acetonitrile/0.1 % trifluoroacetic acid 50/50 (v/v) changing progressively after 10 min to 60 % actonitrile over 4 min and afterwards to 100 % acetonitrile within 2 min. A fluorimetric detector was attached to the HPLC system. Standard solutions were diluted in acetonitrile with 5 mg/mL ascorbic acid. The excitation/emission wavelength pairs for 1-OH-Pyr and 1-OH-Phen were 346/384 and 256/380 nm, respectively. For the quantification of bile pigments 25 µL of bile was added to 475 µL of water and the absorbance was recorded at 380 nm in microplates (Fluostar Optima, BMG Labtech, Offenburg, Germany).
Quantification was performed via external standards using a five point calibration. The limit of detection (LOD) and the limit of quantification (LOQ) were calculated according to DIN 32645 (DIN 1994). The LOD and the LOQ for 1-OH-Pyr (1-OH-Phen) was 3.4 (0.5) and 22.5 (1.7) ng/mL bile, respectively. For internal quality assurance a fish bile sample as laboratory reference material was included in every sample batch to monitor the stability of the method (variation coefficient 15 % for 1-OH-Pyr). Participation in an intercalibration exercise leads to acceptable z-scores below ±2 for the laboratory performing the chemical analysis (Kammann et al. 2013). Every bile sample was analyzed twice. Blanks, five standard concentrations and the laboratory reference material were included in every sample batch. The recovery of 1-OH-Pyr was 98 %. Certified Standard solutions of 1-OH-Pyr and 1-OH-Phen were purchased by LGC (Dr. Ehrenstorfer Standards distributed by LGC, Middlesex, UK). All other chemicals were obtained from Merck (Darmstadt, Germany).
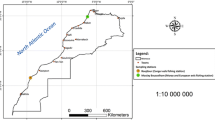
45 European eels (A. anguilla) in their yellow stage were collected from Sebou and Loukkos rivers/estuaries in Morocco during October/November 2009 by local fishermen using fyke nets. Yellow eels were chosen as this developmental stage is regarded as primarily sedentary (Belpaire and Goemans 2007). Sampling locations are shown in Fig. 1. Body length and weight were recorded for each fish (Table 1). After opening the body cavity, bile fluid was collected by a disposable syringe. Bile samples of approximately 0.1–0.5 mL were immediately frozen and stored at −20°C until analysis.
The Sebou estuary (34°16′N, 6°34′W) is located in the northwest of Morocco, at the Atlantic coast (Fig. 1), draining a surface area of 40,000 km2. The Sebou is the main purveyor of water to the Gharb plain, a major agricultural region, where fertilizers and pesticides are widely used. Fish for the present study were sampled at two sites in Sebou river: “Sebou up” receive its pollution mainly by discharges from the cities Fes and Meknes whereas “Sebou down” represent an area with highly developed industrial activity (paper mills, sugar plants, tanneries, olive oil mills, oil refineries, alcohol industries etc.). The Loukkos river (35°09′N, 6°05′W) drains a catchment of 3730 km2 (El Morhit et al. 2012). The landscape is composed of wetlands (coastal lagoons, swamps and rice fields), forests, and agricultural zones. The Loukkos watershed is less industrialized than “Sebou down”.
One-way analysis of variance (ANOVA) was applied to test for differences between sampling stations using Scheffe’s test (p ≤ 0.05).
Results and Discussion
With the present study we addressed the question if a possible exposure of eel to PAH in Moroccan rivers is reflected in spatially different concentrations of PAH metabolites and if this contamination might be a potential threat for the Moroccan eels from Sebou and Loukkos, the most important eel catchments in Morocco (ICES 2011). The PAH metabolites 1-OH-Pyr and 1-OH-Phen were detected in all samples (Table 1). The level of 1-OH-Pyr in eel from “Loukkos” were significantly higher when compared with “Sebou up” (p < 0.05; Fig. 2). The lower concentrations of 1-OH-Pyr observed at “Sebou up” reflect the less contaminated the river before entering the highly industrialized area around the Sebou basin at “Sebou down”. However, highest concentrations of PAH metabolites with statistical significant difference to “Sebou up” were found in eel from Loukkos, which is a less industrialized area. Although the Loukkos estuary is known for its intense agricultural activity (El Bakouri et al. 2008) and the sampling site is in an area directly affected by discharged water especially from rice fields, this does not explain elevated PAH levels. Due to the size of the area, unknown sources of contamination cannot be fully excluded. At the same time, a movement of eels even in the yellow eel stage is another possible explanation for the detected results.
In accordance with Nagel et al. (2012a) and Ruddock et al. (2003) 1-OH-Pyr was found to be the dominant compound in eel bile, except in samples from “Sebou up” where both metabolites are present in nearly equal concentrations (Table 1). Comparing the results of the present study to similar investigations from several European countries (Table 2), it is shown that the means of both PAH metabolite concentrations, especially in eel from the United Kingdome and Germany are higher compared to concentrations found in eel from Morocco. However, eel caught in Poland contained PAH metabolites in similar concentrations compared to the eels investigated in the present study. The studies cited in Table 2 cover eels caught in rivers, lakes or lagoons as well as in estuaries and can therefore be considered as directly comparable. Since PAH metabolite concentrations in eel bile are influenced by the developmental stage (Nagel et al. 2012b), such a comparison is only valid by looking at the same life history stage, in this case yellow eels.
To evaluate the measured PAH metabolite concentrations with respect to a possible adverse effect of PAH contamination for aquatic organisms, the results were compared to an internationally agreed threshold value for fish. Since no such thresholds exist for eel, the respective concentration for Atlantic cod (Gadus morhua) of 483 ng/mL bile was used (ICES 2012). Only few fish in the present study are close to this threshold value for 1-OH-Pyr. Even if this threshold has been calculated for another fish species, a comparison indicates that the PAH contamination in Moroccan eels might be of biological significance. However, the cited threshold value can only provide a rough guidance since it has been calculated for a marine teleost species with a different life history compared to the diadromous European eel. Therefore, it cannot be excluded, that PAH, especially together with other organic or inorganic contaminants or environmental impacts, may have some adverse effect for the eel.
With this study on PAH contamination in fish from Morocco information is provided on PAH metabolite amplitudes and spatial differences. Concentrations of PAH metabolites in eel appear moderate compared to fish caught in European waters and to internationally accepted threshold levels in other teleost species.
References
Azdi M, Moukrim A, Burgeot T, Budzinski H, Chiffoleau JF, Kaaya A, Zekhnini A, Narbonne JF, Guarrigues P (2006) Hydrocarbon pollution along Moroccan coasts and BPH activity in the mussel Perna perna. Polycycl Aromat Compd 26:265–282
Belpaire C, Goemans G (2007) Eels: contaminant cocktails pinpointing environmental contaminant. ICES J 64:1423–1436
Blahová J, Havelková M, Kružíková K, Hilscherová K, Halouzka R, Modrá H, Grabic R, Halířová J, Jurčíková J, Ocelka T, Haruštiaková D, Svobodová Z (2010) Assessment of contamination of the Svitava and Svratka rivers in the Czech Republic using selected biochemical markers. Environ Toxicol Chem 29:541–549
Bouachrine M, Fekhaoui M, Bennasser L, Idrissi L (1998) Distribution of selected metals in tissue samples of fish from an industrially contaminated stream (The River Sebou, Morocco). Acta Hydrobiol 40:173–179
Brinkmann M, Hudjetz S, Cofalla C, Roger S, Kammann U, Zhang X, Wiseman S, Giesy J, Hecker M, Schüttrumpf H, Wölz J, Hollert H (2010) A combined hydraulic and toxicological approach to assess re-suspended sediments during simulated flood events. Part I—multiple biomarkers in rainbow trout. J Soils Sediments 10:1347–1361
Brinkmann M, Eichbaum K, Kammann U, Hudjetz S, Cofalla C, Buchinger S, Reifferscheid G, Schüttrumpf H, Preuss T, Hollert H (2014) Physiologically-based toxicokinetic models help indentifying the key factors affecting contaminant uptake during flood events. Aquat Toxicol 152:38–42
Chafik T (2009) Evaluation of the soil contamination of Tangier (Morocco) by the determination of BTEX, PCBs, and PAHs. Soil Sediment Contam 18:766
Chen SC, Liao CM (2006) Health risk assessment on human exposed to environmental polycyclic aromatic hydrocarbons pollution sources. Sci Total Environ 366:112–123
DIN, Deutsches Institut für Normung e.V. (1994) 32645, Nachweis-, Erfassungs- und Bestimmungsgrenze. Berlin Beuth Verlag, Berlin
Durif CMF, Dufour S, Elie P (2005) The silvering process of Anguilla anguilla: a new classification from yellow resident to silver migrating stage. J Fish Biol 66:1025–1043
Eisler R (2007) Eisler’s encyclopedia of environmentally hazardous priority chemicals, 1st edn. Elsevier, Amsterdam
El Bakouri H, Ouassini A, Morillo J, Usero J (2008) Pesticides in ground water beneath Loukkos perimeter, Northwest Morocco. J Hydrol 348:270–278
El Morhit M, Fekhaoui M, Élie P, Girard P, Yahyaoui A, El Abidi A, Jbilou M (2009) Heavy metals in sediment, water and the European glass eel, Anguilla Anguilla (Osteichthyes: Anguillidae), from Loukkos River estuary (Morocco, eastern Atlantic). Cybium 33:219–222
El Morhit M, Fekhaoui M, Serghini A, El Blidi S, El Abidi A, Yahyaoui A, Hachimi M (2012) Étude de l’évolution spatio-temporelle des paramètres hydrologiques caractérisant la qualité des eaux de l’estuaire du Loukkos (Maroc). Bulletin de l’Institut Scientifique, Rabat, section Sciences de la Vie. 34(2):151–162
Er-Raioui H, Bouzid S, Marhraoui M, Saliot A (2009) Hydrocarbon pollution of the Mediterranean coastline of Morocco. Ocean Coast Manag 52:124–129
Hajjaj Hassani L, Garrido Frenich A, Benajiba MH, González Rodríguez MJ, Martínez Vidal JL, Debdoubi A (2006) Assessment of butyltin and phenyltin pollution in the sea mullet, Mugil cephalus, along the Moroccan and Spanish Coasts (Mediterranean Sea). Arch Environ Contam Toxicol 51:608–614
Harman C, Holth TF, Hylland K, Thomas K, Grung M (2009) Relationship between polycyclic aromatic hydrocarbon (PAH) accumulation in semipermeable membrane devices and PAH bile metabolite levels in Atlantic cod (Gadus morhua). J Toxicol Environ Health A 72:234–243
HELCOM (2012) HELCOM Core indicators. http://www.helcom.fi/Lists/Publications/BSEP136.pdf#search=core%20set. Accessed 2 April 2015
ICES (2011) International council for the exploration of the sea, report of the 2011 session of the joint EIFAAC/ICES working group on eels. Lisbon, Portugal. ICES CM 2011/ACOM: 18, 223 pp
ICES (2012) International council for the exploration of the sea, cooperative research report 315: integrated marine environmental monitoring of chemicals and their effects. In: Davies IM, Vethaak D (eds) Copenhagen, Denmark, 277 pp
ICES (2013) International council for the exploration of the sea, report of the workshop on evaluation progress eel management plans (WKEPEMP), 13–15 May 2013, Copenhagen, Denmark. ICES CM 2013/ACOM:32. 757 pp
Kammann U (2007) PAH metabolites in bile fluide of dab (Limanda limanda) and flounder (Platichthys flesus): spatial distribution and seasonal changes. Environ Sci Pollut Res 14:102–108
Kammann U, Askem C, Dabrowska H, Grung M, Kirby MF, Koivisto P, Lucas C, McKenzie M, Meier S, Robinson C, Tairova ZM, Tuvikene A, Vuorinen PJ, Strand J (2013) Interlaboratory proficiency testing for measurement of the PAH metabolite 1-hydroxypyrene in fish bile for marine environmental monitoring. J AOAC Int 96:635–641
Kammann U, Brinkmann M, Freese M, Pohlmann J-D, Stoffels S, Hollert H, Hanel R (2014) PAH metabolites, GST and EROD in European eel (Anguilla anguilla) as possible indicators for eel habitat quality in German rivers. Environ Sci Pollut Res 1:2519–2530
Meador J, Stein J, Reichert W, Varanasi U (1995) Bioaccumulation of polycyclic aromatic hydrocarbons by marine organisms. Rev Environ Contam Toxicol 143:79–165
Monteiro PR, Reis-Henriques MA, Coimbra J (2000) Polycyclic aromatic hydrocarbons inhibit in vitro ovarian steroidogenesis in the flounder (Platichthys flesus L.). Aquat Toxicol 48:549–559
Nagel F, Kammann U, Wagner C, Hanel R (2012a) Metabolites of polycyclic aromatic hydrocarbons (PAHs) in bile as biomarkers of pollution in European eel (Anguilla anguilla) from German rivers. Arch Environ Contam Toxicol 62:254–263
Nagel F, Wagner C, Hanel R, Kammann U (2012b) The silvering process in European eel (Anguilla anguilla) influences PAH metabolite concentration in bile fluid—consequences for monitoring. Chemosphere 87:91–96
OSPAR Commission (2008) Coordinated environmental monitoring programme (CEMP). http://www.ospar.org/content/content.asp?menu=00900301400000. Accessed 2 April 2015
Pikkarainen AL (2006) Ethoxyresorufin-O-deethylase (EROD) activity and bile metabolites as contamination indicators in Baltic Sea perch: determination by HPLC. Chemosphere 65:1888–1897
Ruddock PJ, Bird DJ, McEvoy J, Peters LD (2003) Bile metabolites of polycyclic aromatic hydrocarbons (PAHs) in European eels Anguilla anguilla from United Kingdom estuaries. Sci Total Environ 301:105–117
Szlinder-Richert J, Nermer T, Szatkowska U (2014) PAH metabolites in European eels (Anguilla anguilla) as indicators of PAH exposure: different methodological approaches. Sci Total Environ 496:84–91
Tairova ZM, Giessing AMB, Hansen R, Andersen O (2009) 1-Hydroxypyrene as a biomarker of PAH exposure in the marine polychaete Nereis diversicolor. Mar Environ Res 67:38–46
Vuorinen PJ, Keinänen M, Vuontisjärvi H, Barsiene J, Broeg K, Förlin L, Gercken J, Kopecka J, Köhler A, Parkkonen J, Pempkowiak J, Schiedek D (2006) Use of biliary PAH metabolites as a biomarker of pollution in fish from the Baltic Sea. Mar Pollut Bull 53:479–487
Wariaghli F, Tilghman-Sibille A, El Abidi A, El Hamri H, Fekhaoui M, Yahyaoui A (2013) Anguilla anguilla L.: evaluation of the degree of heavy metal contamination in the Sebou estuary and in Moulay Bousselham lagoon reserve (Morocco). Int J Aquatic Sci 4:69–82
Acknowledgments
This work was supported by a Grant from the German Academic Exchange Service (DAAD) (A/08/97890) as well as the Moroccan-German Programme of Scientific Research (PMARS) Project 01DH13008 (“Traceability of Fish and Fish Products”). We thank Alexander Schulz for his skillful technical assistance. Any experiments comply with the current laws of the country in which they were performed.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wariaghli, F., Kammann, U., Hanel, R. et al. PAH Metabolites in Bile of European Eel (Anguilla anguilla) from Morocco. Bull Environ Contam Toxicol 95, 740–744 (2015). https://doi.org/10.1007/s00128-015-1586-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-015-1586-5