Abstract
The aim of this work was to evaluate contamination by polychlorinated biphenyls and mercury in soil, and to assess exposure level to both contaminants in children living in San Felipe, Nuevo Mercurio, Zacatecas, Mexico. We found soil levels of total polychlorinated biphenyls ranging from non detectable (nd) to 190 μg/kg. Mercury soil levels ranged from 8.9 to 10215.0 mg/kg. Exposure levels of total polychlorinated biphenyls assessed in blood and urinary mercury in children living in the studied community were 1,600 ± 8,800 ng/g lipid and 4.2 ± 7.1 μg/g creatinine, respectively.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
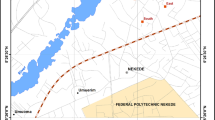
In Mexico, sources of persistent organic pollutants (POPs) and metals exist in mining, agricultural, industrial, small industry and petroleum sites. Moreover, uncontrolled areas, such as waste disposal and inclusive natural sources generate contaminated areas in several regions of the country (Trejo-Acevedo et al. 2009). In this context, we studied a mining site (San Felipe, Nuevo Mercurio, Zacatecas), where mercury (Hg) was the principal extracted mineral. In addition, the mine was an area formerly used to store wastes and several hazardous substances, including hundreds of storage drums containing polychlorinated biphenyls (PCBs). San Felipe, Nuevo Mercurio (SFNM; 600 inhabitants) is a mining site located 200 km north of Zacatecas city in Mexico (Fig. 1). The mining facility containing a raw mineral crusher and a mining waste disposal site (mining tailings) is located within the town (Fig. 2). Moreover, the mine was used for clandestine PCBs residues disposal (Fig. 2).
PCBs are a family of lipophilic compounds once used extensively in industry, particularly as heat transfer chemicals in electric transformers and capacitors as well as hydraulic fluids and lubricants in heavy electrical equipment (ATSDR 2008). The toxicity of PCBs and related compounds was first recognized following two industrial accidents–one in Japan in the late 1960s, the other in Taiwan in the late 1970s—when PCBs used as a heat transfer agent leaked into rice oil during its manufacture. Adults who ate the contaminated oil developed skin rashes and peripheral nervous system numbness and tingling (ATSDR 2008). Infants born to women who had eaten the contaminated oil also had skin rashes and exhibited poorer intellectual functioning during infancy and childhood (ATSDR 2008).The other contaminant assessed in this study is Hg, which is a heavy metal that enters the environment from both natural and anthropogenic sources (ATSDR 2008). Hg exists in three forms, the metallic element (Hg0), inorganic salts and organic compounds, each of which possesses different bioavailability and toxicity. The neurotoxicity of Hg was first recognized in the 1950s when residents of southern Japan ate fish from Minamata Bay which were highly contaminated by industrial pollution. This exposure led to mental retardation, motor damage, ataxia, and seizures in children born to women who ate the contaminated fish during pregnancy (ATSDR 2008). Moreover, epidemiological and laboratory studies indicate that PCBs and Hg (principally organic Hg) may have additive or interactive adverse effects on nervous system function (Powers et al. 2009). Because of this historical background, the aim of this work was to evaluate the exposure level of PCBs and Hg in children living in San Felipe, Nuevo Mercurio.
Materials and Methods
In order to confirm the presence of PCBs and Hg in soil in SFMN, surface soil samples (1–5 cm in depth) were obtained from different areas of the town (Fig. 2). We used systematic sampling within a 400-m grid. Areas repeatedly used by children either at school or at home (backyards) were included in the sampling area. We divided the study site into four zones: Zone 1, the downtown area of SFNM and recreational areas for children; Zone 2, high mines and Hg extraction ovens; Zone 3, small mines and sites where the PCBs drums were stored; Zone 4, where the wastes contaminated with PCBs were stored.
All children attending primary school located in this community were screened for study eligibility through in-person interviews. Children who had lived in the selected area since birth and who were 6–14 years old at the time of the study were eligible to participate. After informed consent was obtained, a questionnaire was used to help document the history of each child and blood and urine samples were taken.
For PCBs in soil, we determined the concentrations of 14 PCB congeners (International Union for Pure and Applied Chemistry numbers 28, 52, 99, 101, 105, 118, 128, 138, 153, 156, 170, 180, 183, and 187). Briefly, one gram of soil was accurately weighed and placed in Green-Chem LEV glasses and 14 mL of dichloromethane were added. The extraction was performed in a microwave oven (CEM), and allowed to cool at 4°C in a cold-room. The extract was filtered (0.45 μm) and diluted with dichloromethane to 20 mL then evaporated to 4 mL and hexane was added three times (4 mL). The extract was cleaned with Florisil columns (1,000 mg/6 mL, Baker), and the PCBs were eluted with 12 mL of diethyl-ether:hexane 6%. The extract was then evaporated with nitrogen gas flow to approximately 0.5 mL and diluted with hexane to 1 mL. For PCBs in soil, Analytical Reference Material EC-2 (Environmental Canada, National Water Research Institute) was used. Our extraction efficiency was 90–110% for all tested analytes. We also determined the concentrations of 14 PCB congeners (the same congeners that were in the soil analysis) in plasma samples as described by Trejo-Acevedo et al. (2009). Briefly, a 2 mL aliquot of plasma was first extracted with a mixture of ammonium sulfate/ethanol/hexane (1:1:3), and the extract was then concentrated and cleaned on Florisil columns. The quantification of soil and plasma samples was performed using a HP 6890 gas chromatograph coupled with a HP 5973 mass spectrometer. A HP5-MS column, 60 m × 0.25 mm ID, 0.25-μm film thickness was used (J&W Scientific, Bellefonte, PA, USA). Column temperatures were: initial, 100°C (2 min), final, 310°C (rates: 20°C/min up to 200°C, 10.0°C/min up to 245°C, 4.0°C/min up to 280°C and 30°C/min up to 310°C). Injector temperature was 270°C operated in pulsed splitless mode. Helium was used as the carrier gas at a linear velocity of 1.0 mL/min. α-Hexachlorocyclohexane-C13, endrin-C13 and PCB-141-C13 were added as internal standards to all samples. Under these conditions and using the data of nine replicates near the lowest concentration attainable on the calibration curve, the method detection limits for PCBs were approximately 0.30 μg/L. Quantification of PCBs in plasma was part of the Interlaboratory Comparison Program organized by the Institute National de Santé Publique du Quebec (Quebec, Canada) with results within the limits of tolerance. Our accuracy in this program was 80–120% for all tested analytes.
For total Hg in the soil analysis, one gram of soil was accurately weighed and placed in a 100 mL volumetric flask and 20 mL of HNO3 were added. The digestion was performed in a microwave oven (CEM), and allowed to cool to room temperature. The digest was filtered (0.45 μm) and diluted with deionized water to 20 mL. Total Hg was measured by atomic absorption spectrometry (AAnalyst 100-FIAS; Norwalk, USA). The detection limit for Hg was 0.1 μg/L. The digestion procedure for total Hg analysis was verified using the certified reference material of the National Institute of Standards and Technology (NIST, mountain soil 2711). The recovery percentage was 92%. For Hg in the urine analysis, urine samples of approximately 100 mL were collected in polyethylene bottles. Then 5 mL of the sample were digested with 10 mL of HNO3 in a microwave oven (CEM), subsequently the Hg was reduced by adding K2MnO4 5%. Total Hg was measured by atomic absorption spectrometry (AAnalyst 100-FIAS; Norwalk, USA). The detection limit for total Hg analysis was 2 μg/mL. The digestion procedure for total Hg analysis was verified using the certified reference material, Clincheck urine control lyophil (D-80335 Munich/Germany). The recovery percentage was 95%. Finally, the levels of Hg in urine were adjusted by creatinine (crt).
To satisfy normality criteria, the levels of PCBs and Hg in soil, PCBs in plasma and urinary Hg were logarithm-transformed. Therefore, all of the results are shown as geometric means. Mean levels of PCBs and Hg in soil between zones were compared, using one way analysis of variance (ANOVA), followed by Tukey’s test. For all statistical analyses we used JMP IN 7.0 software (SAS Institute, Inc., Cary, NC, USA).
Results and Discussion
Table 1 shows the mean levels of total PCBs in the four zones sampled. The highest levels of total PCBs were in Zones 2 and 3 (35.5 μg/kg and 33.5 μg/kg, respectively), and in Zones 1 and 2 mean levels of 14.2 μg/kg and 15.0 μg/kg were found. Levels of total Hg in soil are shown in Table 2. For Hg, similar results to those for PCBs were found, the highest levels of total Hg were found in Zones 2 and 3 with a mean level of 687.0 mg/kg and 1569.0 mg/kg, respectively. While, in Zones 1 and 4, the mean levels of Hg in soil were 34.7 mg/kg and 118.0 mg/kg, respectively.
For total PCBs, the guidelines in Mexico for agricultural soil are 500 μg/kg, for residential soil 5,000 μg/kg and for industrial soil 25,000 μg/kg (Norma Oficial Mexicana NOM-133-ECOL- 2000). All zones were found to have soil levels lower than those in the guidelines for Mexico. When comparing the results with the guidelines for total PCBs in Canada (Canadian Soil Quality Guidelines 1999) of 0.5 mg/kg for agricultural, 1.3 mg/kg for residential and 33 mg/kg for industrial soil, similar conclusions can be made. All zones had soil levels lower than the Canadian guidelines.
The guidelines for Hg0 in agricultural/residential and industrial soils in Mexico are 25 and 310 mg/kg, respectively (NOM-147-SEMARNAT/SSA1-Norma Oficial Mexicana NOM-147-SEMARNAT/SSA1-2004). It is important to mention that this guide is for remediation of soil in agricultural/residential and industrial zones. We note that the four zones sampled had levels higher than the guideline for agricultural/residential soil, and only Zones 2 and 3 had levels higher than the industrial guideline for soil in Mexico. When compared with guidelines in Canada (Canadian Soil Quality Guidelines for the Protection of Environmental and Human Health 1999), soil in the four zones studied in Mexico had levels higher than the Canadian agricultural/residential guidelines (6.6 mg/kg) and only Zone 1 had levels lower than industrial guidelines for Canada (50.0 mg/kg). However, when compared with guidelines in the United Kingdom for inorganic Hg (3,600 mg/kg; Environmental Agency 2009), only 11% and 13% of soil samples in Zone 2 and zone 3, respectively, had levels above these guidelines (Table 2). We should remember that we assessed total mercury in soil, and the guidelines in Mexico and Canada are for Hg0 and in the United Kingdom inorganic mercury was assessed. In addition, at this point it is important to note that Zone 1 is the zone where the children are living (home, school and recreational area for children).
Considering that soil is an important pathway of exposure for children, specific biomarkers of exposure to these agents were assessed in both communities. The exposure levels to PCBs and Hg in children are shown in Table 3. The mean level for total PCBs in blood was 1,600 ng/g lipid and for urinary Hg was 13.5 mg/g crt. When PCBs levels in blood found in this study were compared with levels reported in others studies around the world, we noted that children living in SFMN had higher levels (1,600 ng/g lipid) than children living in China (40.6 ng/g lipid; Shen et al. 2010) and children living in the Slovak Republic (352.8 ng/g lipid; Trnovec et al. 2010). Moreover, when compared with a reference value for total PCBs in Germany in the whole blood of children aged 7–14 (0.9–1.0 μg/L; Schulz et al. 2009), the levels in children living in SFNM were higher (mean levels of 11.4 μg/L). The mean level of Hg in children in this study was higher than the level observed in persons who are not occupationally exposed to Hg (5 mg/g crt; United Nations Industrial Development Organization (UNIDO 2003) and the level found in NHANES III for females aged 16–49 years (NHANES III. Third National Report on Human Exposure to Environmental Chemicals 2005). This level was also higher than the levels found in children in the Czech Republic (0.16 μg/g crt; Puklová et al. 2010).
Our study has some limitations, including the lack of information regarding other environmental media and dietary sources. However, our data indicates high exposure levels in children living in the community studied in this work. Children appear to be particularly suitable for a monitoring program, as they are not directly exposed to occupational pollution. Thus, children normally reflect present trends of environmental exposure more accurately than adults (IPCS 2006). Moreover, it is well established that children are potentially at higher risk than adults for adverse health effects resulting from exposure to many environmental chemicals (IPCS 2006). Finally, regarding health risks, it is difficult to associate a specific health effect with the levels found in the children studied. It is also very difficult to predict a specific effect when the exposure is to both toxicants (PCBs and Hg). Therefore, more studies are needed in order to evaluate the health effects of exposure to multiple toxins. Children are a particularly vulnerable population, and children living in developing countries deserve special attention. Moreover, an international environmental health program for children is clearly needed, and biomonitoring of toxicants on a global scale should be the first step towards the prevention of toxin-induced illnesses in this vulnerable population.
References.
ATSDR (2008) Toxicological profile for Polychlorinated Biphenyls and Mercury. Agency for toxic substances and diseases registry. Atlanta, GA. USA
Canadian Soil Quality Guidelines for the Protection of Environmentaland Human Health (1999) Mercury (inorganic) and Polychlorinated biphenyls (total). http://ceqg-rcqe.ccme.ca/download/en/270/. http://ceqg-rcqe.ccme.ca/download/en/274/. Accessed 03 Nov 2010
Environmental agency (2009) Soil Guideline Values for mercury in soil. Science. ReportSC050021/MercurySGV. http://www.environmentagency.gov.uk/static/documents/Research/SCHO0309BPQG-e-e.pdf. Accessed 04 Apr 2010
IPCS (2006) Principles for Evaluating Health Risk in Children Associated with Exposure to Chemical, Environmental Health Criteria 237
NHANES III Third National Report on Human Exposure to Environmental Chemicals (2005) Department of Health and Human Services (CDC). Atlanta, Georgia
Norma Oficial Mexicana NOM-133-ECOL-2000. http://siscop.ine.gob.mx/descargas/legislacion/nom_bpcs.pdf. Accessed 03 Nov 2010
Norma Oficial Mexicana NOM-147-SEMARNAT/SSA1-2004. http://www.inforural.com.mx/IMG/pdf/DOF-SEMARNAT-020307.pdf Accessed 03 Nov 2010
Powers BE, Poon E, Sable H, Schantz SL (2009) Developmental exposure to pcbs, mehg, or both: long-term effects on auditory function. Environ Health Perspect 117:1101–1107
Puklová V, Krsková A, Cerná M, Cejchanová M, Rehůřková I, Ruprich J, Kratzer K, Kubínová R, Zimová M (2010) The mercury burden of the Czech population: an integrated approach. Int J Hyg Environ Health 213:243–251
Schulz C, Angerer J, Ewers U, Heudorf U, Wilhelm M (2009) Revised and new reference values for environmental pollutants in urine or blood of children in Germany derived from the German environmental survey on children 2003–2006 (GerES IV). Int J Hyg Environ Health 212:637–647
Shen H, Ding G, Han G, Wang X, Xu X, Han J, Lou X, Xu C, Cai D, Song Y, Lu W (2010) Distribution of PCDD/Fs, PCBs, PBDEs and organochlorine residues in children’s blood from Zhejiang. China. Chemosphere. 80:170–175
Trejo-Acevedo A, Díaz-Barriga F, Carrizales L, Domínguez G, Costilla R, Ize-Lema I, Yarto-Ramírez M, Gavilán-García A, JesúsMejía-Saavedra J, Pérez-Maldonado IN (2009) Exposure assessment of persistent organic pollutants and metals in Mexican children. Chemosphere 74:974–980
Trnovec T, Sovcíková E, Pavlovcinová G, Jakubíková J, Jusko TA, Husták M, Jurecková D, Palkovicová L, Kocan A, Drobná B, Lancz K, Wimmerová S (2010) Serum PCB concentrations and cochlear function in 12-year-old children. Environ Sci Techno l44:2884–2889
United Nations Industrial Development Organization (UNIDO; 2003). Removal of barriers to introduction of cleaner artisanal gold mining and extraction technologies. http://www.cetem.gov.br/gmp/GMP_News/GMP_News_January_2003.pdf. Accessed 03 Nov 2010
Acknowledgments
This work was supported by grant from the Consejo Nacional de Ciencia y Tecnología, Mexico, CONACYT-SALUD-13749.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Costilla-Salazar, R., Trejo-Acevedo, A., Rocha-Amador, D. et al. Assessment of Polychlorinated Biphenyls and Mercury Levels in Soil and Biological Samples from San Felipe, Nuevo Mercurio, Zacatecas, Mexico. Bull Environ Contam Toxicol 86, 212–216 (2011). https://doi.org/10.1007/s00128-010-0165-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-010-0165-z