Abstract
Purpose
To analyze the effects of genetic and environmental factors on sense of coherence (SOC) in young adulthood and whether family environment measured in childhood modifies these effects.
Methods
SOC was measured at 20–27 years of age in 3,193 Finnish twins using the Antonovsky’s 13-item short scale. The twins and their parents had rated their emotional family environment independently when the twins were 12 years of age. The data were analyzed using applications of structural linear equation modeling to twin data.
Results
Females rated SOC 2.42 points lower than males. Additive genetic factors explained 39 % of the variation of SOC in males and 49 % in females, whereas the rest of the variation was explained by environmental factors unique to each twin individual. For the dimensions of SOC, the highest genetic correlation was found between comprehensibility and manageability (0.90 in males and 0.97 in females). SOC was strongest in the participants who had reported supportive family atmosphere and low relational tensions to parents in childhood. These participants also had higher genetic variance and lower unique environmental variance of SOC when compared to those who reported emotionally more stressful family environment. The results were similar when we used parental rating of family environment.
Conclusion
Genetic factors are important for SOC, but genetic influences are much greater in supportive family environments. This emphasizes the importance of childhood home for the development of strong SOC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Health psychology has traditionally emphasized psychological risk factors of health, such as temperament, personality traits and stress. The theory of sense of coherence (SOC), originally developed by Aaron Antonovsky [1], in contrast focuses on personality factors maintaining good health rather than compromising health. According to this theory, those who regard the world as comprehensible, manageable and meaningful, i.e., have strong SOC, can cope better with stressful life situations. Thus, negative life experiences have a less adverse effect on their health compared to those with weak SOC [2]. In previous follow-up studies, strong SOC has been found to predict better self-rated health [3], lower risk of psychiatric disorders [4] and lower mortality [5], suggesting that strong SOC is a predictor rather than a consequence of good health.
According to Antonovsky, especially childhood is an important period for the development of strong SOC. Experiences in adolescence can affect the development of SOC as well, and finally it is stabilized between 20 and 30 years of age, with greatest stabilization, however, taking place in individuals who have developed a strong SOC [2]. Empirical studies based on two Finnish cohorts found that SOC in adulthood was very stable over five [6] and more than 10 years of follow-up [7] supporting the original theory on the stability of SOC. An issue receiving but little attention so far is, however, the role of genetic factors. There is extensive evidence showing a strong genetic component behind most, if not all, psychological traits, such as personality and social attitudes, explaining typically about half of the inter-individual variation in these traits [8]. The only study on the heritability of SOC we are aware of was based on Swedish twin data from 326 pairs. This study found that 35 % of the variation of SOC was accounted for by genetic factors whereas environmental factors shared by co-twins were statistically non-significant and accounted for only 7 % of the variation [9].
Genetic and environmental factors are, however, unlikely to act independently but rather interact with each other. In Dutch studies, religious upbringing was found to decrease genetic variance of disinhibition [10], the experience of sexual assault to increase genetic variance of borderline personality features [11] and parental divorce to increase environmental variation in internalizing and externalizing problem behavior in children [12]. Earlier analyses of data from the Finnish twins used also in the present study have also illustrated effects of parental monitoring and home atmosphere on the heritability of adolescent substance use and externalizing behaviors associated with risk for early substance abuse [13]. These studies suggest that environmental factors may suppress or enhance the effects of genes affecting psychosocial development, or trigger personality disorders in genetically susceptible persons.
Interactions between genetic and environmental factors may also be an explanation behind the somewhat inconsistent results on effects of childhood environments on many psychological traits. Studies associating childhood environment with later psychological outcomes have given clear evidence of the importance of parent–child relationships on later mental health [14, 15], whereas previous twin studies have found only modest evidence on the role of environmental factors shared by co-twins behind the variation of many psychological traits [16]. It is possible that childhood environment has effects on the development of many psychological traits, including SOC, but because such effects interact with genetic predispositions of children, they cannot be disentangled from genetic factors without direct measures of childhood environment rarely available in twin studies [17].
In this study, we aim to analyze genetic and environmental factors and their interactions affecting SOC in a large longitudinal set of Finnish twins with the measurements of SOC in young adulthood and home environment in childhood. We will first analyze the heritability of SOC and how genetic and environmental factors explain the mutual correlations between the dimensions of SOC. Secondly, we will analyze the modulating effects on heritability of adult SOC by family environment reported in childhood by twins themselves and independently by their parents.
Method
Participants
The data were derived from the longitudinal FinnTwin12 study, which comprises all Finnish twins born in 1983–1987 [18]. The study protocol was approved by the Ethical Committee of the University of Helsinki and the IRB of Indiana University. The names and postal addresses of the twins and their parents were received from the Finnish population registry covering the entire population by identifying children born at the same day to the same mother. The baseline questionnaire was posted to twins in the autumn of the year when they reached the age of 11 years. Valid responses were received from 5,184 twins (the response rate 94 %). At the same time, a questionnaire about the twins’ childhood and rearing was posted to the parents of the twins (response rate 87 %). In the majority of families, the twins’ mother (60 %) or mother and father jointly (35 %) completed this family questionnaire. A follow-up study was conducted during the years 2006–2011 for all twins who participated in the baseline study at their average age of 24 years (range 20–27 years). However, because of incomplete contact information, death or other reasons, this invitation could not be sent to 359 twins. Most twins replied by mail, while a minority who also participated in a clinical study filled in the questionnaire during the study visit (n = 818). Together, we received information from 3,193 twins yielding a response rate of 66 % (58 % of all twins belonging to these birth cohorts).
After removing 155 twins with uncertain information on zygosity, our realized sample comprised 478 monozygotic (MZ), 395 same-sex dizygotic (SSDZ) and 382 opposite-sex dizygotic (OSDZ) complete twin pairs. Zygosity was determined at baseline using questionnaire items on physical similarity and confusability of appearance at school age and was supplemented by parental response to items developed for zygosity classification of twin children. School photographs and additional information from twins’ mothers were obtained if classification was unclear. We validated the questionnaire-based zygosity classification in these data using 395 same-sex pairs as young adults whose DNA was tested and found that the zygosity was confirmed among 97 % of the pairs [19]. This suggests good reliability of this method.
Measures
Sense of coherence was assessed during the follow-up study using the Antonovsky’s 13-item short scale derived from the original 29-item Orientation to Life Questionnaire [2]. This scale measures the three dimensions of SOC, i.e., comprehensibility, manageability and meaningfulness, reflecting the cognitive, instrumental/behavioral and motivational aspects of SOC. The Cronbach’s alpha was satisfactory for SOC (α = 0.85), but, expectedly because of fewer items, somewhat lower for its components: α = 0.69 for 5-item comprehensibility, 0.64 for 4-item manageability, and 0.72 for 4-item meaningfulness. We removed data from 14 twins because of incomplete information on SOC (more than 1 missing item). For 45 additional twins, with a single missing item, we replaced that item with the mean of all other items. SOC scores showed a positive correlation with age in both men (r = 0.13, p < 0.0001) and women (r = 0.13, p < 0.0001), and accordingly, we adjusted SOC and its components for age in all genetic analyses.
Home environment was assessed during the baseline study using an 8-item scale. Both co-twins and both of their parents independently rated on 5-point scales the degree to which their home is (1) warm, caring; (2) creative, supportive; (3) trusting, understanding; (4) open; (5) authoritarian; (6) unjust; (7) argumentative; and (8) indifferent. The response alternatives ranged from ‘does not hold true at all’ to ‘holds completely true’. Information on all these ratings was available for 2,811 twins. As discussed earlier [20], the items 1–3 describe home atmosphere and items 6–8 relational tensions to parents. In un-rotated factor analysis, the first factors explained the major part of the variation for home atmosphere (52 % for self-rating and 61 % for parental rating) and relational tensions (52 % for self-rating and 57 % for parental rating), whereas eigenvalues for the second factors were low (<0.83) suggesting one factor solution. We thus used these factors in the modeling. The item 5 (authoritarian) did not fit well on either of the factors and was thus excluded from these analyses. The factor modeling was conducted using SPSS statistical software, version 15.0. The factors were scaled in a way that high scores indicate good family atmosphere and low relational tensions to parents.
We used the information on home environment to analyze selective participation during the follow-up because it was lower (66 %) than at baseline survey (94 %). When we analyzed this using regression analyses, we found that there was no difference between respondents and non-respondents in home environment when rated by twins (p = 0.68 for home atmosphere and p = 0.19 for parental relationship) or parental relations reported by their parents (p = 0.11). Only for home atmosphere rated by parents, we found that non-respondents had somewhat poorer home atmosphere than respondents (difference 0.08, p = 0.03).
Statistical methods
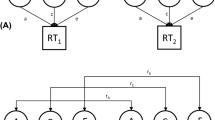
Estimation of the genetic contribution to interindividual variability in SOC was based on the applications of linear structural equation modeling to twin data [21]. The basis of this comes from comparisons of similarity between MZ and DZ twins: MZ twins have the same genomic sequence, while DZ pairs shared, on average, 50 % of their segregating genes. In genetic twin modeling, trait variation is assumed to arise from four possible sources and hence can be decomposed into those four components: (1) additive genetic variation (A) including all main effects of the alleles affecting the trait (correlation 1 within MZ and 0.5 within DZ twins); (2) dominance genetic variation (D) caused by interactions between alleles in the same locus (correlation 1 within MZ and 0.25 within DZ twins); (3) common environment (C) including environmental factors shared by co-twins (correlation 1 within both MZ and DZ twins), and (4) specific environment (E) including environmental factors unique to each twin individual and any measurement error (0 both within MZ and DZ twins). However, because we had only twins reared together available, we were not able to estimate dominance genetic and common environmental effects simultaneously. This is because common environmental factors make DZ twins more similar and dominance genetic factors less similar than MZ twins as compared to the situation when only additive genetic and specific environmental factors would affect the trait [22].
Twin modeling makes the assumptions of equality of means and variances between MZ and DZ twins and first and second co-twin within a pair. Those assumptions were tested by comparing the fit of genetic models to saturated models, which do not make these assumptions. Further we tested in a series of univariate models whether the size of the variance components was equal in males and females and whether there existed any sex-specific genetic effect seen as lower correlations within OSDZ pairs compared to SSDZ pairs. The comparisons of the model fit between nested models were based on χ 2-goodness-of-fit statistics and degrees of freedom (df). The modeling was continued by analyzing the correlations between the dimensions of SOC using multivariate Cholesky decomposition. This method decomposes the variation and co-variation in the data into a series of uncorrelated genetic and environmental factors. Using this method, we calculated genetic and environmental correlations between the dimensions of SOC and the proportions to which much these correlations explain the trait correlations between these dimensions.
Next, we addressed the hypothesis that childhood home environment modifies the genetic and environmental variances of SOC. This was done using gene–environment interaction models among those participants for whom we had the information on home environment available. In this model, the factors of home environment can increase or decrease genetic and environmental variation of SOC [17]. Further, variation in home environment can affect the mean of SOC modeled as a mean modification effect. This effect takes into account gene–environment correlations as well as a causal effect of home environment on SOC. All genetic modelings were carried out using the Mx statistical package, version 1.7.03 [23]. The raw data analysis option was used, which allows including also data from twins without their co-twins. In all analyses, the effect on confidence intervals of clustered data, i.e., sampling twin pairs instead of unrelated individuals, was taken into account using the clustered sample option of the Stata statistical package, version 10.1 [24].
Results
Table 1 presents the means and standard deviations (SD) for SOC and its components measured in adulthood and the factors of home environment measured in childhood by sex and zygosity. Women had lower scores for SOC [difference in means 2.42, 95 % confidence interval (CI) 1.59–3.25] and for its dimensions, i.e., comprehensibility (1.63, 95 % CI 1.28–1.99), manageability (1.34, 95 % CI 1.05–1.63) and meaningfulness (0.56, 95 % CI 0.25–0.87) in the whole data set, when adjusted for age. In the factors of home environment, the only sex difference was that females rated less tension with their parents than males (difference in means 0.10, 95 % CI 0.02–0.18).
In univariate genetic modeling, we selected the additive genetic/common environment/specific environment (ACE) model as the starting point, because the observed twin correlations suggested the presence of common environmental component (data not shown). No sex-specific genetic effect was found for total SOC score (Δχ 21 = 0.05, p = 0.82), but it was present for all of its components when analyzed separately (Δχ 21 = 30.2-47.8, p < 0.0001). This suggests that the genes underlying the SOC components may differ between men and women. Accordingly, we took it into account in all further modeling.
In the ACE model, the common environmental components were modest in size and statistically non-significant (Table 2). However in some models, additive genetic components were also statistically non-significant, and thus we pooled men and women to increase statistical power. The results in the pooled data showed strong and statistically significant additive genetic effects (a 2 = 0.36 for SOC and 0.25–0.42 for the components of SOC) whereas common environmental components were small and statistically non-significant (c 2 = 0.09 and 0.00–0.10, respectively). Thus, we used the reduced additive genetic/specific environment (AE) model in the further analyses. The fit of AE model differed from the saturated models for some of these traits (Δχ 215 = 24.0–35.0, p = 0.07–0.002). However, no systematic differences were found in means and variances between MZ and DZ twins (Table 1), suggesting that these violations were rather because of random variation and did not reflect the need of a more complex statistical model.
We then analyzed the trait correlations between these dimensions (Table 3). The highest correlation was found between comprehensibility and manageability (r = 0.69 in males and 0.72 in females) and the lowest between manageability and meaningfulness (r = 0.52 and 0.53, respectively). When these correlations were decomposed using Cholesky decomposition, this difference in the size of the trait correlations was found to be mainly because of the high genetic correlation between the genetic components of comprehensibility and manageability (r A = 0.90 and 0.97, respectively), whereas unique environmental correlations were roughly similar (r E = 0.42–0.58). Additive genetic factors explained from 40 to 63 % and unique environmental factors from 37 to 60 % of the trait correlations.
Finally, we analyzed how the factors of home environment self-reported by twins and reported by their parents modified the effects of additive genetic and specific environmental influences on SOC and its components. Because detection of significant gene–environment interactions requires greater sample sizes due to statistical power requirements than univariate models or Cholesky decomposition and because we were able to equate the parameters estimates for males and females in all models (Δχ 25 = 0.07–10.12, p = 1–0.07), we analyzed data from men and women together. When analyzing SOC, good family atmosphere and low relational tensions to parents were associated with higher SOC observed as positive mean moderator effects (regression coefficients 0.51–1.16). These family environmental measures increased the additive genetic variation and decreased specific environmental variation of SOC; the variance modification effects were statistically significant except for self-rated parental relationship (Fig. 1, full results of mean and variance modifications effects with 95 % CIs are presented in “Appendix”). When we repeated these analyses for the components of SOC, we found that the associations were generally similar to the results for total SOC, but some of the estimates were not statistically significant (see “Appendix”).
Discussion
In this longitudinal study of young adult Finnish twins, we found that genetic differences between individuals explained from a third to one-half of the variation of SOC and its components, which is very close to the heritability estimate for SOC found in a previous Swedish twin study [9]. In the light of the previous studies in behavior genetics, these results are not surprising, and very similar heritability estimates have been found for many other behavioral traits, such as personality, which probably originated in neuro-physiological differences between individuals [8]. Especially the role of dopamine and serotonin systems in the brain laying behind temperamental differences has been discussed [25], and the previous efforts to find candidate genes affecting temperament and personality differences have mainly focused on genes associated with these neuro-regulatory systems [26, 27]. Previous genome-wide association studies of personality have not yielded many genes having significant effect on these traits, suggesting that each of the individual genes plays only a minor role in accounting for inter-individual differences, and that hundreds if not thousands of genes may be involved as has been found for intelligence [28]. However, the previous studies have been relatively small and larger sample sizes may be needed to detect individual genes underlying personality traits. We also found that correlations between the dimensions of SOC could be attributed to genetic factors. Especially the high correlation between comprehensibility and manageability also found in two previous studies being higher than between the other dimensions of SOC [3, 29], was explained by largely the same set of genes contributing to these dimensions of SOC. However, it is noteworthy that about half of the correlation between the dimensions of SOC was explained by environmental correlations, suggesting that partly a common subset of environmental factors underlies all three dimensions of SOC.
Our more novel and major results concerned how family environment modifies the genetic architecture of SOC. We found that children who grew-up in supportive family atmosphere and experienced few emotional tensions with their parents showed more genetic and less environmental variation in SOC in adulthood than children who grew-up in emotionally less favorable family environments. It is highly unlikely that these results would be because of the effect of SOC on reporting of family environment, first, because the participants reported their family environment more than one decade earlier than the SOC test was conducted and, second, because we obtained very similar results when family environment was reported by the twins’ parents. These results suggest that a supportive family environment enhances a child’s genetic potential for the development of strong SOC and the emotional stress related to less optimal family environment increases environmental variation in the development of SOC. Very similar results have been found for other behavioral traits including disinhibition [10], substance use [13] and borderline personality [11]. There has been extensive research activity seeking to identify the molecular genetic background of these interactions focusing especially on genes associated with the serotonin neuroregulatory system [25], but the results are not conclusive so far. For example, Caspi et al. [30] reported interaction between stressful life events and the polymorphisms of serotonin transporter gene 5-HTTLPR when predicting major depression in adulthood, but these findings have later been disputed [31]. Our results suggest that gene–environment interactions are not limited to mental or psychosocial disorders in which environmental stress may trigger the disorder in genetically susceptible persons, but are also present when analyzing general measures of psychological well-being in the population at large.
We found, too, that a favorable family environment not only modified the genetic and environmental variation of SOC but also correlated with SOC. The effect of good family environment on strong SOC has been previously reported in many studies [32–35], and already Antonovsky emphasized the importance of childhood environment for the development of SOC [2]. It is, however, possible that these associations may also reflect gene–environment correlations when parents with strong SOC create supportive family environment but concurrently transmit trait-relevant genetic dispositions to their offspring. Because of methodological limitations, we could not analyze this issue directly since family environment is modeled as part of shared environment even in the case that it would correlate with genetic factors. Thus, in our models, the mean modification effects include both gene–environment correlations and the direct effects of family environment on SOC. However, our results showing strong genetic variation and lack of common environmental variation in SOC suggest that gene–environment correlations can well explain at least partly the correlation between family environment and SOC.
Our study enhances understanding of the role of common environmental factors for SOC and more generally, for other psychological factors, as well. Previous twin studies have given very little evidence on the importance of common environmental factors [16] even when there is clear evidence, for example, of the importance of parent–child relationship on later mental health [14, 15]. One possible explanation is the lack of power to detect both additive genetic and common environmental effect simultaneously. However, we found that the point estimate for the proportion of variation of SOC explained by common environmental effects was only 9 % (95 % CI 0–27 %), showing that these effects explain much less variation in SOC than do additive genetic factors. Even when the confidence intervals are wide, this point estimate is very close to the point estimate of common environmental factors found in the previous Swedish twin study on SOC, i.e., 7 % [9]. Despite such modest direct effects, our results suggest that family environment is of much importance in children’s developmental outcomes. It affects developmental outcomes by shaping genetic and specific environmental influences to make children more prone to external environmental exposures in less supportive family environments, while enabling children to realize their genetic potential to develop strong SOC in supportive family environments. The modest effects of common environmental influences found for many psychological traits should not be interpreted to mean that family environment is unimportant, but rather that its importance is to be found in interactions, not in direct effects.
In this study, we also found that the mean levels of SOC and its dimensions were somewhat lower in females compared to males: a finding consistent with previous studies [36, 37]. In the light of these results, it is interesting that the relative sizes of genetic and environmental effects were very similar in men and women. This suggests that the lower SOC of women is not, for example, due to higher environmental pressure in females, which would increase environmental variance, but rather reflects more qualitative differences, for example, in hormonal levels or different role expectations in men and women. We also found that for the components of SOC, sex-specific genetic effect was statistically significant because of lower correlations in OSDZ pairs as compared to SSDZ pairs. This would suggest that partly different sets of genes affect the development of these dimensions in males and females. However, these results should be considered with caution, because for total SOC we found no evidence of sex-specific genetic effects.
Our data have both strength and limitations. Our main strength is that we have time-lagged measures of SOC and family environment in a large population-based sample of twins allowing us to analyze both the effects of genes and environment and their interaction effects. It is also an advantage that SOC is measured in young adulthood when, according to Antonovsky, SOC is already stabilizing, whereas the measurements of home environment have been conducted in childhood which is important phase of life in the development of SOC [2]. This also decreases effects of recall bias that might well be associated with SOC and, further, we had independent ratings of home environment by parents in addition to self-ratings of twins. A limitation of our data is that we did not measure SOC of parents, which would be necessary to analyze whether the correlation between family environment and SOC is due to gene–environment correlations. Further, because information on parental SOC was not available, we necessarily assumed random mating for level of SOC. However, it is unlikely that assortative mating by SOC would have affected our results because it should increase DZ correlations and thus lead to overestimation of shared environmental effects not found in this study.
In conclusion, genetic factors are important for SOC but genetic influences are much greater in supportive family environments. This result emphasizes the long-lasting consequences of childhood family environment for further mental health.
References
Antonovsky A (1979) Health, stress and coping. Jossey-Bass, San Francisco
Antonovsky A (1987) Unraveling the mystery of health. How people manage stress and stay well. Jossey-Bass, San Francisco
Suominen S, Helenius H, Blomberg H, Uutela A, Koskenvuo M (2001) Sense of coherence as a predictor of subjective state of health: results of 4 years of follow-up of adults. J Psychosom Res 50:77–86
Kouvonen AM, Väänänen A, Vahtera J, Heponiemi T, Koskinen A, Cox SJ et al (2010) Sense of coherence and psychiatric morbidity: a 19-year register-based prospective study. J Epidemiol Community Health 64:255–261
Surtees P, Wainwright N, Luben R, Khaw KT, Day N (2003) Sense of coherence and mortality in men and women in the EPIC-Norfolk United Kingdom prospective cohort study. Am J Epidemiol 158:1202–1209
Feldt T, Lintula H, Suominen S, Koskenvuo M, Vahtera J, Kivimäki M (2007) Structural validity and temporal stability of the 13-item sense of coherence scale: prospective evidence from the population-based HeSSup study. Qual Life Res 16:483–493
Hakanen JJ, Feldt T, Leskinen E (2007) Change and stability of sense of coherence in adulthood: longitudinal evidence from the Health Child Study. J Res Personal 41:602–617
Bouchard TJ Jr, McGue M (2003) Genetic and environmental influences on human psychological differences. J Neurobiol 54:4–45
Hansson K, Cederblad M, Lichtenstein P, Reiss D, Pedersen N, Belderhiser J et al (2008) Individual resiliency factors from a genetic perspective: results from a twin study. Fam Proc 47:537–551
Boomsma DI, de Geus EJ, van Baal GC, Koopmans JR (1999) A religious upbringing reduces the influence of genetic factors on disinhibition: evidence for interaction between genotype and environment on personality. Twin Res 2:115–125
Distel MA, Middeldorp CM, Trull TJ, Derom CA, Willemsen G, Boomsma DI (2011) Life events and borderline personality features: the influence of gene–environment interaction and gene–environment correlation. Psychol Med 41:849–860
Robbers S, van Oort F, Huizink A, Verhulst F, van Beijsterveldt C, Boomsma D et al (2012) Childhood problem behavior and parental divorce: evidence for gene–environment interactions. Soc Psychiatry Psychiatr Epidemiol 47:1539–1548
Dick DM, Viken R, Purcell S, Kaprio J, Pulkkinen L, Rose RJ (2007) Parental monitoring moderates the importance of genetic and environmental influences on adolescent smoking. J Abnorm Psychol 116:213–218
Lukaschek K, Kruse J, Emeny RT, Lacruz ME, von EisenhartRothe A, Ladwig KH (2013) Lifetime traumatic experiences and their impact on PTSD: a general population study. Soc Psychiatry Psychiatr Epidemiol 48:525–532
Morgan Z, Brugha T, Fryers T, Stewart-Brown S (2012) The effects of parent–child relationships on later life mental health status in two national birth cohort. Soc Psychiatry Psychiatr Epidemiol 47:1707–1715
DaveySmith G (2011) Epidemiology, epigenetics and the ‘Gloomy Prospect’: embracing randomness in population health research and practice. Int J Epidemiol 40:537–562
Purcell S (2002) Variance components models for gene–environment interaction in twin analysis. Twin Res 5:554–571
Kaprio J, Pulkkinen L, Rose RJ (2002) Genetic and environmental factors in health-related behaviors: studies on Finnish twins and twin families. Twin Res 5:366–371
Jelenkovic A, Ortega-Alonso A, Rose RJ, Kaprio J, Rebato E, Silventoinen K (2011) Genetic and environmental influences on growth from late childhood to adulthood: a longitudinal study of two Finnish twin cohorts. Am J Hum Biol 23:764–773
Latendresse SJ, Rose RJ, Viken RJ, Pulkkinen L, Kaprio J, Dick DM (2010) Examining the etiology of associations between perceived parenting and adolescents’ alcohol use: common genetic and/or environmental liabilities? J Stud Alcohol Drugs 71:313–325
Posthuma D, Beem AL, de Geus EJ, van Baal GC, von Hjelmborg JB, Iachine I et al (2003) Theory and practice in quantitative genetics. Twin Res 6:361–376
Neale MC, Cardon LR (2003) Methodology for genetic studies of twins and families. Kluver Academic Publisher, Dordrecht
Neale MC (2003) Mx: statistical modeling: Box 710 MCV, Richmond, VA 23298. Department of Psychiatry
Williams R (2000) A note on robust variance estimation for cluster-correlated data. Biometrics 56:645–646
Keltikangas-Järvinen L, Salo J (2009) Dopamine and serotonin systems modify environmental effects on human behavior: a review. Scand J Psychol 50:574–582
Frustaci A, Pozzi G, Gianfagna F, Manzoli L, Boccia S (2008) Meta-analysis of the brain-derived neurotrophic factor gene (BDNF) Val66Met polymorphism in anxiety disorders and anxiety-related personality traits. Neuropsychobiology 58:163–170
Calati R, Porcelli S, Giegling I, Hartmann AM, Moller HJ, De Ronchi D et al (2011) Catechol-o-methyltransferase gene modulation on suicidal behavior and personality traits: review, meta-analysis and association study. J Psychiatr Res 45:309–321
Davies G, Tenesa A, Payton A, Yang J, Harris SE, Liewald D, et al (2011) Genome-wide association studies establish that human intelligence is highly heritable and polygenic. Mol Psychiatry 16:996–1005
Honkinen PL, Suominen S, Rautava P, Hakanen J, Kalimo R (2006) The adult sense of coherence scale is applicable to 12-year-old schoolchildren–an additional tool in health promotion. Acta Paediatr 95:952–955
Caspi A, Sugden K, Moffitt TE, Taylor A, Craig IW, Harrington H et al (2003) Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene. Science 301:386–389
Risch N, Herrell R, Lehner T, Liang KY, Eaves L, Hoh J et al (2009) Interaction between the serotonin transporter gene (5-HTTLPR), stressful life events, and risk of depression: a meta-analysis. JAMA 301:2462–2471
Volanen SM, Lahelma E, Silventoinen K, Suominen S (2004) Factors contributing to sense of coherence among men and women. Eur J Public Health 14:322–330
Lundberg O (1997) Childhood conditions, sense of coherence, social class and adult ill health: exploring their theoretical and empirical relations. Soc Sci Med 44:821–831
Feldt T, Kokko K, Kinnunen U, Pulkkinen L (2005) The role of family background, school success, and career orientation in the development of sense of coherence. Europ Psychologist 10:298–308
Volanen SM, Suominen S, Lahelma E, Koskenvuo M, Silventoinen K (2006) Sense of coherence and its determinants: a comparative study of the Finnish-speaking majority and the Swedish-speaking minority in Finland. Scand J Public Health 34:515–525
Anson O, Paran E, Neumann L, Chernichovsky D (1993) Gender differences in health perceptions and their predictors. Soc Sci Med 36:419–427
Larsson G, Kallenberg K (1996) Sense of coherence, socio-economic conditions and health. Eur J Public Health 6:175–180
Acknowledgments
Data collection and analysis have been supported by National Institute of Alcohol Abuse and Alcoholism (Grants AA-12502, AA-00145, and AA-09203 to RJR), the Academy of Finland (Grants 100499, 205585, 118555, 141054 and 264146 to JK and 266592 for KS) and the Academy of Finland Center of Excellence in Complex Disease Genetics (Grant Numbers: 213506, 129680).
Conflict of interest
None to declare.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
See Table 4
Rights and permissions
About this article
Cite this article
Silventoinen, K., Volanen, SM., Vuoksimaa, E. et al. A supportive family environment in childhood enhances the level and heritability of sense of coherence in early adulthood. Soc Psychiatry Psychiatr Epidemiol 49, 1951–1960 (2014). https://doi.org/10.1007/s00127-014-0851-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00127-014-0851-y