Abstract
Aims/hypothesis
Patient-reported outcome measures (PROMs) are increasingly used as key performance indicators in chronic illness. We sought to review the value of these tools in assessing health-related quality of life (HRQOL) in patients with diabetes-related foot disease and identify the impact of each foot problem on life quality.
Methods
A systematic review of literature on HRQOL PROMs in diabetes-related foot disease was performed according to Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines. The quality of eligible studies was evaluated within pre-existing criteria.
Results
53 studies written between 1995 and 2010 met the inclusion criteria. A variety of HRQOL PROMs were used. Disease-specific tools were better than generic at quantifying temporal changes in life quality and showed greater sensitivity to ulcer/neuropathic severity. No studies have simultaneously evaluated disease-specific tools. Generic and utility HRQOL PROMs are frequently used as secondary outcome measures in randomised trials and cost–utility analysis.
HRQOL is depressed in diabetes, further impaired by the presence of foot disease. Ulcer healing is associated with improvements in HRQOL. Patients with active ulceration report poorer HRQOL than those whom have undergone successful minor lower extremity amputation (LEA) but there is a paucity of quality data on HRQOL outcomes for diabetes-related LEA.
Conclusions/interpretation
No one PROM was identified as a ‘gold standard’ for assessing HRQOL in diabetes-related foot disease. Specific areas for further development include the most valid HRQOL PROM with disease-specific content; HRQOL outcomes in minor and major amputations and the role of HRQOL tools in routine clinical care.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Management of diabetes-related foot disease demands a multidisciplinary approach with treatments that are often intensive and prolonged. As a result, hospitalisations are frequent and costs are high. Evidence to support any one approach over another, in terms of conventional endpoints such as wound healing or major amputation, is thin [1]. Given the lack of clear benefit in terms of conventional outcome measures, the use of other markers of treatment success may be more appropriate.
One such marker is health-related quality of life (HRQOL). HRQOL is a subjective measure of a person’s physical and psychological well-being and represents a patient’s assessment of how a particular disease or intervention has affected their life. Information about a patient’s HRQOL is commonly gathered using ‘Patient-Reported Outcome Measures’ (PROMs). These involve the patient responding to a number of questions on themes such as physical functioning, social functioning and mental well-being, and may include both generic and disease-specific questions. Responses are analysed to produce a multi-dimensional quality of life ‘score’ that can be measured repeatedly to help clinicians identify an improvement or deterioration in a patient’s health status. The use of PROMs has been recommended in the evaluation of healthcare technologies/services and in regulatory decision-making in the UK National Health Service (NHS) [2].
The purpose of this review is to summarise and analyse the literature pertaining to HRQOL PROMs used in the spectrum of diabetes-related foot disease. Specifically, we aim to assess the validity of HRQOL PROMs and examine specific factors affecting HRQOL in patients with diabetes and foot disease.
Methods
Systematic searches using the keywords ‘diabetes mellitus and foot’ OR ‘diabetic foot’ AND ‘neuropathy/ulcer/Charcot arthropathy/amputation/lower extremity amputation’ AND ‘health related quality of life/quality of life/QALY/patient reported outcome measure’ were performed using the ‘explosion technique’ where possible in Medline, PubMed, EMBASE and the Cochrane Library. Articles were limited to English language, human studies and peer review publications, and included all papers identified from 1966 to 7 February 2011. Bibliographies of relevant citations were screened for further articles of relevance.
Inclusion criteria specified that studies had to assess HRQOL specifically pertaining to foot health in patients with diabetes using structured PROMs. Studies using PROMs to look at general aspects of HRQOL in diabetes were excluded, as were studies that used ‘self-evaluation’ (i.e. free texting/interviews without formulaic format) as a means of recording HRQOL.
HRQOL PROMs were rated according to pre-defined criteria (assessing study quality, quality of tool employed and the generalisability of outcomes) [3]. The score was adapted to ensure its relevance to diabetes-related foot disease [4] and an overall quality ‘score’ (range, 0–15) was assigned as a gross marker of fulfilment of the criteria—where 0 represented poorest quality and 15 best quality.
The literature review conformed to Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement standards [5, 6]. Owing to heterogeneity of study design, varying methodology and PROMs used, a quantitative pooled meta-analysis was not performed.
Results
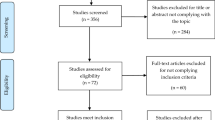
Initial search strategies revealed 203 papers. When abstracts and reference lists were scrutinised and exclusion/inclusion criteria applied, 53 studies were eligible and included (Fig. 1). The median quality ‘score’ [3] of the 53 included studies assessed was 10 (range 4–15) (Table 1).
PROMs used to assess HRQOL in diabetes-related foot disease
A variety of generic, summary, utility, dimension specific, disease-specific and site-specific PROMs were used to assess HRQOL in diabetes-related foot disease (Tables 1 and 2).
Generic PROMs
The most frequently encountered tool, used in 27 studies, was the generic HRQOL tool: the Medical Outcomes Study 36-item Short-Form Health Survey (SF-36) (http://www.qualitymetric.com). The SF-36 is commonly used as a ‘gold standard’ PROM to verify construct validity when developing disease-/site-/dimension-specific PROMs. It has shown efficacy in assessing HRQOL in diabetes-related foot disease [7–18] (Fig. 2). When evaluated alongside site-/disease-specific PROMs, the Diabetic Foot Ulcer Scale (DFS) [10–12] and the Cardiff Wound Index Scale (CWIS) [8, 19], statistically significant correlations between the assessed ‘life domains’ (e.g. locomotor abilities, mental wellness, personal care) make it an appropriate measure (Fig. 3).
Comparison of SF-36 physical component scores (PCS, dark grey bars) and mental component scores (MCS, light grey bars) from studies using the SF-36 to record HRQOL in diabetes-related foot disease. Scores compared to those from patients with diabetes without foot disease and the general US population [50]. Scores as stated in respective studies. aStudies in which component scores were not stated in text. PCS and MCS calculated from subscores at SF-36, PCS, MCS and NBS Calculator, available at www.sf-36.org/nbscalc/index.shtml, accessed 1 November 2010 NTSS-6-SA, Neuropathic total symptom score—self-administered
Comparison of mean SF-36 domain scores in patients with type 2 diabetes and DFU undergoing amputation vs medical management. Scores compared with patients with diabetes without foot disease [41]. Scores range from 0 to 100 with a score of 0 reflecting poorest life HRQOL. Mid-grey bars, General Perception of Health; light grey bars, Pain Score; dark grey bars, Physical Functioning. AKA, above-knee amputation; BKA, below-knee amputation; TMA, transmetatarsal amputation; TA, toe amputation
The SF-36 has shown sensitivity when correlating HRQOL scores with diabetic foot ulcers (DFUs) [13] and neuropathy severity [20]. Furthermore it can show temporal changes in HRQOL [21] and has been used in a number of randomised controlled trials assessing the treatment of diabetes-related foot disease [9, 12, 22–26] in which improvements in primary outcomes (i.e. ulcer healing and improved pain scores) were associated with appropriate changes in HRQOL scores.
In contrast, a Nottingham-based randomised controlled trial assessing the use of different dressing types in the treatment of DFUs used the SF-36 alongside the CWIS to document HRQOL as a secondary outcome measure. Whereas the CWIS showed statistically significant improvements in HRQOL between healed and non-healed ulcers, the SF-36 failed to show any difference [27]. This poor sensitivity to ulcer healing was presumed by the study group to relate to the large volume of incomplete SF-36 questionnaires at follow-up, suggesting that it is a poorly accepted tool. Most observational studies assessed by this review have, however, reported good response rates of >70% when using the SF-36.
The SF-36 is well equipped to generate a generic measure of HRQOL and can be truncated into the Medical Outcomes Study 6D Short-Form Health Utility Index (SF-6D) for health utility analysis. However, the SF-36 lacks specificity and may be confounded by other (non-foot) complications of diabetes [13, 14].
Other generic measures of HRQOL (the Medical Outcomes Study 12-item Short-Form Health Survey [SF-12], the Research and Development 36-item Form [RAND-36], the Nottingham Health Profile, the Sickness Index Profile) show appropriate changes in HRQOL according to severity of foot disease [28–31] but remain subject to confounding factors as a result of poor specificity.
Health utility PROMs
Health utility PROMS have been used for the economic evaluation of foot care/treatment in diabetes with variable outcomes in terms of calculated health states (Table 2). To ensure comparability between cost–utility analyses and to guide healthcare resource allocation, the National Institute for Health and Clinical Excellence (NICE) have specified the EuroQOL 5D Health Utility Index (EQ-5D) as the preferred PROM for use in the UK [32].
Diabetic foot-specific PROMs
PROMs specifically designed to assess diabetes-related foot problems include the DFS [12], NeuroQoL [31] and the Carrington foot questionnaire [33].
The DFS was developed using semi-structured interviews and focus groups of patients with DFUs and their care givers [12]. It has shown internal consistency, reliability, validity [12] and sensitivity to wound severity [11, 15] and healing [15]. A shortened version, the DFS Short Form (DFS-SF) [34] showed similar robustness and sensitivity. The DFS-SF has statistically significant regression correlations to the DFS and SF-36 and, at only 29 questions, is a more ‘user-friendly’ tool for everyday clinical practice.
NeuroQoL was designed to assess HRQOL in patients with diabetes complicated by peripheral neuropathy and DFUs. It has been validated against the SF-12 to show construct validity and sensitivity to neuropathic symptoms, which the SF-12 (as a generic instrument) is unable to detect. Factor analyses have demonstrated internal consistency and its brevity makes it feasible and acceptable.
NeuroQoL has proven proficient in assessing the impact of advancing neuropathy on HRQOL. It is less useful in showing the relationship between DFU severity and HRQOL. This may reflect the tool’s focus on neuropathic (as opposed to ulcerative) symptoms. It could also be a consequence of diminished nociceptive responses in patients with ulcers, reducing pain scores (and thus indicating better HRQOL). However, a well-designed UK cross-sectional population study (using NeuroQoL) disputes this theory; showing more severe neuropathy to be associated with a higher prevalence of pain and a poorer HRQOL [7]. NeuroQoL’s poor sensitivity to DFUs may therefore be related to its failure to assess ulcer-related therapies (e.g. non-weight-bearing regimens, dressing changes and antibiotic therapy).
The Norfolk Quality of Life in Diabetic Neuropathy Questionnaire (Norfolk QoL-DN) was designed to assess all aspects of diabetes-related neuropathic disease. It is intended for use as a diagnostic aide as well as for disease monitoring and treatment evaluation [20, 35]. Despite robust validation, the Norfolk QoL-DN [35, 36] lacks specificity for peripheral neuropathy, limiting its use in assessing the impact of diabetes-related foot disease.
Disease-specific tools for diabetes-related peripheral neuropathy (DPN) have shown sensitivity to symptom severity but no attempts have been made to evaluate their efficacy in assessing temporal changes in HRQOL in individuals with DPN. As a result their use in disease monitoring is currently limited.
Wound-specific PROMs
The CWIS has validity in assessing chronic wounds but is not specific to DFUs [8, 19]. Evaluation alongside the SF-36 showed significant correlations in all domains [8, 19] but sensitivity to severity of DFUs was lacking when CWIS HRQOL values were compared against ulcer severity (as determined by the University of Texas Wound Classification system) [19]. The CWIS is able to discriminate between healed and unhealed ulcers [8, 27]
Combined generic and disease-specific PROMs
Combining generic and disease-specific PROMs produces more useful information on outcomes [4]. Different measures provide complementary evidence, with disease-specific tools offering specific clinical information and reflecting treatment effects and generic measures collecting information more transferrable to the service provider and highlighting unforeseen intervention effects. There are no ‘combined PROMs’ in use in diabetes-related foot disease.
The impact of foot complications on HRQOL in patients with diabetes
Foot monitoring and podiatry
Individuals with diabetes without foot disease have a lower HRQOL compared with the general population (Fig. 2) [7, 8, 11, 12, 14, 28, 29, 33, 35, 36]. This may relate to the general lifestyle/complications of diabetes but may also reflect the commitment needed to preserve foot health. Patient focus groups have shown that preventative foot care practices (regular visits to the podiatrist, having to wear restrictive footwear, daily foot care regimens and restrictions on activities, etc) negatively impacts HRQOL [8, 37].
DPN
DPN may be asymptomatic but when painful impacts on HRQOL. Functional problems (disturbed balance; reduced foot sensation; disturbed sleep; limitations in footwear) and psychological fears of advancing disease, ulceration and amputation often exist together. DPN is difficult to study in terms of impact on HRQOL as the classical view is that less advanced neuropathy is associated with more pain [38]. In advanced foot disease (e.g. ulceration), it is difficult to determine the exact impact of neuropathy in isolation.
Ulceration
Ulcer healing has a positive impact on HRQOL scores whilst persisting ulcers have a progressively negative effect on life quality [8–10, 12]. HRQOL scores in patients who have undergone successful minor amputation for DFUs are significantly superior to those with persisting ulcers [30, 33] (although definitions of successful amputation are vague). Those who undergo major lower extremity amputation (LEA) have worse HRQOL scores than those with active DFUs [30, 33].
One study assessing variables that determine HRQOL in DFUs showed: presence of infection, co-existing peripheral arterial disease and ulcer size to be the most important [14]. When evaluating the influence of pain on HRQOL in DFUs, the same group reported that larger persisting ulcers (>5 cm) were less painful than smaller ulcers of shorter existence [15], but were associated with poorer HRQOL due to ‘non pain related’ factors. Indeed focus groups have found that HRQOL in DFUs most closely relate to the impact of treatments (regular dressing changes, poorer mobility caused by non weight bearing regimens, prolonged antibiotic therapy), anxieties around possibilities of future amputation, financial losses secondary to medical incapacity and the day to day disruption on social and family life rather than the ulcer size or duration [39].
Charcot arthropathy
Few studies formally assessed HRQOL in patients with diabetes and Charcot disease [17, 18, 40]. Of those studies that identified Charcot arthropathy as a separate subgroup, it was suggested (using the American Academy of Orthopaedic Surgeons – Diabetic Foot Questionnaire [AAOS –DFQ] and SF-36) that the health status in these patients was comparable to that following minor LEA [40]. However these studies were small and a more detailed analysis of a broader spectrum of patients at different stages of Charcot arthropathy is required.
Amputation
Outcomes in amputation have focused on functional status and mobility rather than HRQOL. A large cost–utility analysis performed in USA used three components of the SF-36 to assess HRQOL in patients with diabetes [41]. Those with active ulcers had significantly lower HRQOL scores for ‘physical functioning’ than those who had undergone successful toe or transmetatarsal amputations. Physical functioning in below knee amputation was no different to that in DFUs, but above knee amputation scores were worse (Fig. 4). Despite the large overall size of this cross-sectional analysis, the subgroups of interest were small and amputation groups poorly representative. Only patients who had undergone successful operations were included in this study (although ‘success’ was poorly defined), with no account for amputation related morbidity and mortality.
Mean EQ-5D scores for patients with active ulceration/healed ulcers/minor amputation/major amputation. Possible scores range from –0.594 to 1. A score of 0 represents no life quality; scores <0 reflect a perceived life quality worse than death; a score of 1 indicates best possible life quality [43, 44]
In the UK, a prospective cohort study used the SF-36 to chart HRQOL outcomes in patients with their first diagnosis of DFUs. No significant deterioration in HRQOL scores was found (when adjusted for confounders) in those who went on to amputation [42]. Similarly, observational studies in Sweden [43, 44] and France [45], using the EQ-5D and SF-36, respectively, found (with the exception of major amputation, where scores were considerably lower) poorer HRQOL scores in those with active DFUs compared with patients who had undergone successful minor amputations. The Swedish group showed no difference in scores for minor amputation and primary ulcer healing (Fig. 4). Two other small observational studies [30, 33] have shown similar patterns.
Of key importance, all the studies identified that evaluate HRQOL in patients post amputation were of poor quality with small, highly selected patient groups (i.e. well healed rather than amputation stumps with complications).
Environment, delivery and modality of care
A supportive multi-disciplinary team (MDT) for diabetes care is associated with improved self-management and health-related outcomes [46–48]. Using generic and disease-specific PROMs (SF-36 and DFS), a French observational study [11] found better HRQOL outcomes for patients managed in tertiary centres, with less deterioration in physical health scores, less irritation due to ulcer appearance, shorter durations of foot ulcer care, greater closeness with partners and friends and greater satisfaction with overall medical care. These results were echoed by patients cared for by a devoted MDT in Thailand [49].
Discussion
Generic, diabetes foot-specific, utility, complication-specific, summary, site-specific and combination PROMs all have validity in measuring HRQOL in patients with diabetes-related foot disease. However, each has its limitations and none can be considered a suitable tool in every study or complication setting. Of the generic tools, the SF-36 shows sensitivity to foot disease and has been used most frequently but lacks NICE approval for use in utility studies (who favour the EQ-5D). Of the disease-specific tools, the DFS and NeuroQoL are the most validated, but no studies have compared them simultaneously and, as their titles suggest, they fail to encompass the full spectrum of diabetes-related foot disease. The CWIS shows promise in assessing HRQOL in active ulceration, but is non-specific for DFUs and may fail to capture aspects of this disease. Using a combination of generic and disease-specific PROMs should produce the most meaningful outcomes, but as indicated by the poor response rate in one randomised trial, the use of two instruments (e.g. CWIS and SF-36) may be cumbersome and unfeasible in clinical practice [27].
Generic HRQOL tools are limited in their abilities in identifying the factors reducing HRQOL (for example, a low score on ‘role performance’ may result from retinopathy rather than foot disease). Their advantage is that they can be compared against pre-existing figures from the general population and translated into cost–utility analysis making these tools more amenable to the research setting.
Disease-specific tools (DFS, NeuroQoL) offer greater insight into specific issues impairing health status. They show greater sensitivity to changing foot health/disease severity and are subject to fewer confounders, offering greater insights into lifestyle factors that may be improved by the MDT. Limitations include validation in smaller numbers of studies, exclusivity to disease state (cannot be given to a disease free control group) and narrow focus (they may miss unexpected aspects of impaired HRQOL, e.g. gastrointestinal side effects from antibiotics used to treat osteomyelitis that would not exist with surgical management). In contrast, these tools offer more use in the clinical setting for disease monitoring.
The quality of studies available was variable. The assessment of test-retest reliability and use of comparable HRQOL tools to assess construct validity of PROMs was often lacking and should be encouraged when validating any PROM. The ‘quality score’ used in this review is a gross estimate of ‘quality’ pertaining to the correct use of a PROM [3]. However, a well designed RCT that uses a single PROM as a secondary outcome measure may have a poor quality score due to a number of issues such as an ungeneralisable patient group, lack of re-test validation and lack of discussion of HRQOL domains, etc. The score is therefore more useful in observational studies designed to evaluate PROMs.
HRQOL PROMs have shown the negative impact of advancing foot disease on quality of life. Where uncertainty lies is in outcomes post LEA, where only a small number of poorly generalisable studies have assessed patient reported HRQOL using PROMs [7, 30, 33, 41–43, 45]. The suggestions from these studies (that HRQOL after successful minor amputation is superior to that of active ulceration) could form a large paradigm shift in clinical approach and management of DFUs. However, patient HRQOL after unsuccessful or complicated LEA is a neglected area of study and given that amputations are associated with significant peri-operative and late morbidity, more work in this area is required before any direct conclusions can be made.
Studies indicate that patient-reported HRQOL in DFUs is superior in patients managed by an MDT [11, 49]. There is no data to suggest whether MDT management would improve HRQOL in patients with less severe foot disease.
Preserving HRQOL in patients with diabetes-related foot disease is of high importance and should be measured accurately in order to guide appropriate management of these patients. Using HRQOL PROMs can offer valid insights into this complex and ever increasing disease; shaping service provision and ensuring cost effective patient care.
This review has demonstrated that a number of tools exist to assess HRQOL in patients with foot-related complications of diabetes. There is no one ideal PROM from which to assess HRQOL and each tool has its limitations. Clinicians and researchers should be aware of these limitations before implementation in individual settings.
Abbreviations
- CWIS:
-
Cardiff Wound Index Scale
- DFS:
-
Diabetic Foot Ulcer Scale
- DFS-SF:
-
Diabetic Foot Ulcer Scale Short Form
- DFU:
-
Diabetic foot ulcer
- DPN:
-
Diabetes-related peripheral neuropathy
- EQ-5D:
-
EuroQOL 5D Health Utility Index
- HRQOL:
-
Health-related quality of life
- LEA:
-
Lower extremity amputation
- NICE:
-
National Institute for Health and Clinical Excellence
- Norfolk QoL-DN:
-
Norfolk Quality of Life in Diabetic Neuropathy Questionnaire
- PROM:
-
Patient-recorded outcome measure
- RAND-36:
-
Research and Development 36-item Form
- SF-12:
-
Medical Outcomes Study 12-item Short-Form Health Survey
- SF-36:
-
Medical Outcomes Study 36-item Short-Form Health Survey
References
Hinchliffe RJ, Valk GD, Apelqvist J et al (2008) A systematic review of the effectiveness of interventions to enhance the healing of chronic ulcers of the foot in diabetes. Diabetes Metab Res Rev 24(Suppl 1):S119–S144
Darzi A (2008) Quality and the NHS next stage review. Lancet 371:1563–1564
Cornish D, Holterhues C, van de Poll-Franse LV, Coebergh JW, Nijsten T (2009) A systematic review of health-related quality of life in cutaneous melanoma. Ann Oncol 20(Suppl 6):vi51–vi58
Fitzpatrick R, Davey C, Buxton MJ, Jones DR (1998) Evaluating patient-based outcome measures for use in clinical trials. Health Technol Assess 2:i–iv, 1–74
Liberati A, Altman DG, Tetzlaff J et al (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol 62:e1–e34
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol 62:1006–1012
Davies M, Brophy S, Williams R, Taylor A (2006) The prevalence, severity, and impact of painful diabetic peripheral neuropathy in type 2 diabetes. Diabetes Care 29:1518–1522
Price P, Harding K (2004) Cardiff Wound Impact Schedule: the development of a condition-specific questionnaire to assess health-related quality of life in patients with chronic wounds of the lower limb. Int Wound J 1:10–17
Nabuurs-Franssen MH, Huijberts MS, Nieuwenhuijzen Kruseman AC, Willems J, Schaper NC (2005) Health-related quality of life of diabetic foot ulcer patients and their caregivers. Diabetologia 48:1906–1910
Ribu L, Birkeland K, Hanestad BR, Moum T, Rustoen T (2008) A longitudinal study of patients with diabetes and foot ulcers and their health-related quality of life: wound healing and quality-of-life changes. J Diabetes Complications 22:400–407
Valensi P, Girod I, Baron F, Moreau-Defarges T, Guillon P (2005) Quality of life and clinical correlates in patients with diabetic foot ulcers. Diabetes Metab 31:263–271
Abetz L, Sutton M, Brady L, McNulty P, Gagnon D (2002) The Diabetic Foot Ulcer Scale (DFS): a quality of life instrument for use in clinical trials. Pract Diabetes Int 19:167–175
Ribu L, Hanestad BR, Moum T, Birkeland K, Rustoen T (2007) Health-related quality of life among patients with diabetes and foot ulcers: association with demographic and clinical characteristics. J Diabetes Complications 21:227–236
Ribu L, Hanestad BR, Moum T, Birkeland K, Rustoen T (2007) A comparison of the health-related quality of life in patients with diabetic foot ulcers, with a diabetes group and a nondiabetes group from the general population. Qual Life Res 16:179–189
Ribu L, Rustoen T, Birkeland K, Hanestad BR, Paul SM, Miaskowski C (2006) The prevalence and occurrence of diabetic foot ulcer pain and its impact on health-related quality of life. J Pain 7:290–299
Huang IC, Hwang CC, Wu MY, Lin W, Leite W, Wu AW (2008) Diabetes-specific or generic measures for health-related quality of life? Evidence from psychometric validation of the D-39 and SF-36. Value Health 11:450–461
Pakarinen TK, Laine HJ, Maenpaa H, Mattila P, Lahtela J (2009) Long-term outcome and quality of life in patients with Charcot foot. Foot Ankle Surg 15:187–191
Sochocki MP, Verity S, Atherton PJ et al (2008) Health related quality of life in patients with Charcot arthropathy of the foot and ankle. Foot Ankle Surg 14:11–15
Jaksa PJ, Mahoney JL (2010) Quality of life in patients with diabetic foot ulcers: validation of the Cardiff Wound Impact Schedule in a Canadian population. Int Wound J 7(6):502–507
Currie CJ, Poole CD, Woehl A et al (2006) The health-related utility and health-related quality of life of hospital-treated subjects with type 1 or type 2 diabetes with particular reference to differing severity of peripheral neuropathy. Diabetologia 49:2272–2280
Ware JE Jr, Kosinski M, Bjorner JB, Turner-Bowker DM, Gandek B, Maruish ME (2007) User’s manual for the SF-36v2 health survey, 2nd edn. QualityMetric, Inc, Lincoln
Selvarajah D, Gandhi R, Emery CJ, Tesfaye S (2010) Randomized placebo-controlled double-blind clinical trial of cannabis-based medicinal product (Sativex) in painful diabetic neuropathy: depression is a major confounding factor. Diabetes Care 33:128–130
Swislocki A, Orth M, Bales M et al (2010) A randomized clinical trial of the effectiveness of photon stimulation on pain, sensation, and quality of life in patients with diabetic peripheral neuropathy. J Pain Symptom Manage 39:88–99
Armstrong DG, Lavery LA, Wrobel JS, Vileikyte L (2008) Quality of life in healing diabetic wounds: does the end justify the means? J Foot Ankle Surg 47:278–282
Rauck RL, Shaibani A, Biton V, Simpson J, Koch B (2007) Lacosamide in painful diabetic peripheral neuropathy: a phase 2 double-blind placebo-controlled study. Clin J Pain 23:150–158
Rosenstock J, Tuchman M, LaMoreaux L, Sharma U (2004) Pregabalin for the treatment of painful diabetic peripheral neuropathy: a double-blind, placebo-controlled trial. Pain 110:628–638
Jeffcoate WJ, Price PE, Phillips CJ et al (2009) Randomised controlled trial of the use of three dressing preparations in the management of chronic ulceration of the foot in diabetes. Health Technol Assess 13:1–86, iii–iv
Benbow SJ, Wallymahmed ME, MacFarlane IA (1998) Diabetic peripheral neuropathy and quality of life. QJM 91:733–737
Meijer JW, Trip J, Jaegers SM et al (2001) Quality of life in patients with diabetic foot ulcers. Disabil Rehabil 23:336–340
Peters EJ, Childs MR, Wunderlich RP, Harkless LB, Armstrong DG, Lavery LA (2001) Functional status of persons with diabetes-related lower-extremity amputations. Diabetes Care 24:1799–1804
Vileikyte L, Peyrot M, Bundy C et al (2003) The development and validation of a neuropathy- and foot ulcer-specific quality of life instrument. Diabetes Care 26:2549–2555
National Institute for Health and Clinical Excellence (2008) Guide to the methods of technology appraisal, 2008. NICE, London
Carrington AL, Mawdsley SK, Morley M, Kincey J, Boulton AJ (1996) Psychological status of diabetic people with or without lower limb disability. Diabetes Res Clin Pract 32:19–25
Belury MA, Mahon A, Banni S (2003) The conjugated linoleic acid (CLA) isomer, t10c12-CLA, is inversely associated with changes in body weight and serum leptin in subjects with type 2 diabetes mellitus. J Nutr 133:257S–260S
Vinik EJ, Hayes RP, Oglesby A et al (2005) The development and validation of the Norfolk QOL-DN, a new measure of patients' perception of the effects of diabetes and diabetic neuropathy. Diabetes Technol Ther 7:497–508
Vinik EJ, Paulson JF, Ford-Molvik SL, Vinik AI (2008) German-translated Norfolk quality of life (QOL-DN) identifies the same factors as the English version of the tool and discriminates different levels of neuropathy severity. J Diabetes Sci Technol 2:1075–1086
Hjelm K, Nyberg P, Apelqvist J (2002) Gender influences beliefs about health and illness in diabetic subjects with severe foot lesions. J Adv Nurs 40:673–684
Duby JJ, Campbell RK, Setter SM, White JR, Rasmussen KA (2004) Diabetic neuropathy: an intensive review. Am J Health Syst Pharm 61:160–173
Brod M (1998) Quality of life issues in patients with diabetes and lower extremity ulcers: patients and care givers. Qual Life Res 7:365–372
Pinzur MS, Evans A (2003) Health-related quality of life in patients with Charcot foot. Am J Orthop (Belle Mead NJ) 32:492–496
Eckman MH, Greenfield S, Mackey WC et al (1995) Foot infections in diabetic patients. Decision and cost-effectiveness analyses. JAMA 273:712–720
Winkley K, Stahl D, Chalder T, Edmonds ME, Ismail K (2009) Quality of life in people with their first diabetic foot ulcer: a prospective cohort study. J Am Podiatr Med Assoc 99:406–414
Ragnarson Tennvall G, Apelqvist J (2000) Health-related quality of life in patients with diabetes mellitus and foot ulcers. J Diabetes Complicat 14:235–241
Tennvall GR, Apelqvist J, Eneroth M (2000) Costs of deep foot infections in patients with diabetes mellitus. PharmacoEconomics 18:225–238
Boutoille D, Feraille A, Maulaz D, Krempf M (2008) Quality of life with diabetes-associated foot complications: comparison between lower-limb amputation and chronic foot ulceration. Foot Ankle Int 29:1074–1078
Tang TS, Brown MB, Funnell MM, Anderson RM (2008) Social support, quality of life, and self-care behaviors among African Americans with type 2 diabetes. Diabetes Educ 34:266–276
Garay-Sevilla ME, Nava LE, Malacara JM et al (1995) Adherence to treatment and social support in patients with non-insulin dependent diabetes mellitus. J Diabetes Complications 9:81–86
Davies S, Gibby O, Phillips C, Price P, Tyrrell W (2000) The health status of diabetic patients receiving orthotic therapy. Qual Life Res 9:233–240
Rerkasem K, Kosachunhanun N, Tongprasert S, Guntawongwan K (2009) A multidisciplinary diabetic foot protocol at Chiang Mai University Hospital: cost and quality of life. Int J Low Extrem Wounds 8:153–156
McHorney CA, Ware JE Jr, Rogers W, Raczek AE, Lu JF (1992) The validity and relative precision of MOS short- and long-form health status scales and Dartmouth COOP charts. Results from the Medical Outcomes Study. Med Care 30(5 Suppl):MS253–MS265
Armstrong DG, Lavery LA, Fleischli JG, Gilham KA (1997) Is electrical stimulation effective in reducing neuropathic pain in patients with diabetes? J Foot Ankle Surg 36:260–263
Backonja M, Beydoun A, Edwards KR et al (1998) Gabapentin for the symptomatic treatment of painful neuropathy in patients with diabetes mellitus: a randomized controlled trial. JAMA 280:1831–1836
Piaggesi A, Schipani E, Campi F et al (1998) Conservative surgical approach versus non-surgical management for diabetic neuropathic foot ulcers: a randomized trial. Diabet Med 15:412–417
Coffey JT, Brandle M, Zhou H et al (2002) Valuing health-related quality of life in diabetes. Diabetes Care 25:2238–2243
Dhawan V, Spratt KF, Pinzur MS, Baumhauer J, Rudicel S, Saltzman CL (2005) Reliability of AOFAS diabetic foot questionnaire in Charcot arthropathy: stability, internal consistency, and measurable difference. Foot Ankle Int 26:717–731
Evans AR, Pinzur MS (2005) Health-related quality of life of patients with diabetes and foot ulcers. Foot Ankle Int 26:32–37
Willrich A, Pinzur M, McNeil M, Juknelis D, Lavery L (2005) Health related quality of life, cognitive function, and depression in diabetic patients with foot ulcer or amputation. A preliminary study. Foot Ankle Int 26:128–134
Goodridge D, Trepman E, Sloan J et al (2006) Quality of life of adults with unhealed and healed diabetic foot ulcers. Foot Ankle Int 27:274–280
Tarride JE, Gordon A, Vera-Llonch M, Dukes E, Rousseau C (2006) Cost-effectiveness of pregabalin for the management of neuropathic pain associated with diabetic peripheral neuropathy and postherpetic neuralgia: a Canadian perspective. Clin Ther 28:1922–1934
Tolle T, Xu X, Sadosky AB (2006) Painful diabetic neuropathy: a cross-sectional survey of health state impairment and treatment patterns. J Diabetes Complications 20:26–33
Casellini CM, Barlow PM, Rice AL et al (2007) A 6-month, randomized, double-masked, placebo-controlled study evaluating the effects of the protein kinase C-β inhibitor ruboxistaurin on skin microvascular blood flow and other measures of diabetic peripheral neuropathy. Diabetes Care 30:896–902
Lewko J, Politynska B, Kochanowicz J et al (2007) Quality of life and its relationship to the degree of illness acceptance in patients with diabetes and peripheral diabetic neuropathy. Adv Med Sci 52(Suppl 1):144–146
Nelson SC, Little ER Jr (2007) The 36-item Short-Form Health Survey outcome evaluation for multiple lower-extremity nerve decompressions in diabetic peripheral neuropathy: a pilot study. J Am Podiatr Med Assoc 97:121–125
Happich M, John J, Stamenitis S, Clouth J, Polnau D (2008) The quality of life and economic burden of neuropathy in diabetic patients in Germany in 2002—results from the Diabetic Microvascular Complications (DIMICO) study. Diabetes Res Clin Pract 81:223–230
Lavery LA, Murdoch DP, Williams J, Lavery DC (2008) Does anodyne light therapy improve peripheral neuropathy in diabetes? A double-blind, sham-controlled, randomized trial to evaluate monochromatic infrared photoenergy. Diabetes Care 31:316–321
Lincoln NB, Radford KA, Game FL, Jeffcoate WJ (2008) Education for secondary prevention of foot ulcers in people with diabetes: a randomised controlled trial. Diabetologia 51:1954–1961
Mittlmeier T, Klaue K, Haar P, Beck M (2010) Should one consider primary surgical reconstruction in charcot arthropathy of the feet? Clin Orthop Relat Res 468:1002–1011
Spiegelhalter DJ, Gore SM, Fitzpatrick R, Fletcher AE, Jones DR, Cox DR (1992) Quality of life measures in health care. III: Resource allocation. BMJ 305:1205–1209
Bann CM, Fehnel SE, Gagnon DD (2003) Development and validation of the Diabetic Foot Ulcer Scale-short form (DFS-SF). PharmacoEconomics 21:1277–1290
Contribution statement
All authors were involved in the conception of the paper or analysis and interpretation of data; the drafting or revision of the article; and the review of the final version for publication.
Duality of interest
The authors declare that there is no duality of interest associated with this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hogg, F.R.A., Peach, G., Price, P. et al. Measures of health-related quality of life in diabetes-related foot disease: a systematic review. Diabetologia 55, 552–565 (2012). https://doi.org/10.1007/s00125-011-2372-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-011-2372-5