Abstract
Aims/hypothesis
Microbial factors influence the development of diabetes in NOD mice. Studies in germ-free animals have revealed important roles of microbiota in the regulation of Th17 and forkhead box P3 (FOXP3)+ T regulatory (Treg) activation in the intestine. However, the effects of intestinal microbiota in immune regulation and diabetes development in NOD mice are still poorly understood.
Methods
A colony of germ-free NOD mice was established to evaluate the effects of intestinal microbiota on regulatory immunity in the gut, and on the development of insulitis and diabetes in NOD mice.
Results
Diabetes developed in roughly equal numbers in germ-free and specific pathogen-free NOD mice. Insulitis was accentuated in germ-free NOD mice; yet insulin preservation was unaltered. Germ-free NOD mice showed increased levels of Il17 (also known as Il17a) mRNA in the colon, and of Th17 and Th1 cells in the mesenteric and pancreatic lymph nodes, while Foxp3 mRNA and FOXP3+ Tregs were reduced. In the islet infiltrates, FOXP3+CD4+ T cells were slightly increased in germ-free mice. B cells appeared less activated in the peritoneum and were less abundant in islet infiltrates.
Conclusions/interpretation
These results indicate that lack of intestinal microbiota promotes an imbalance between Th1, Th17 and Treg differentiation in the intestine. This imbalance is associated with accelerated insulitis, but intact recruitment of FOXP3+ Tregs into islets, suggesting: (1) a microbial dependence of local induction of Treg in the gut and draining lymph nodes; but (2) a potentially compensatory function of naturally occurring Tregs in the islets, which may help control diabetogenic T cells.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The pathogenesis of human type 1 diabetes is under genetic as well as environmental control. Genes and environment are in complex interaction and the diabetes risk has been mapped to multiple loci, many of which are associated with the function of the immune system. It is thus apparent that interactions between environment and the immune system are important in the development of diabetes [1]. Numerous studies indicate that in the NOD mouse model, microbial factors strongly affect the development of diabetes [2–4]. This is plausible, as microbes interact effectively with the immune system. However, the question of exactly how microbes regulate the immune system in diabetes remains unanswered. One important approach for resolving this issue is to model the development of diabetes in NOD mice lacking microbes.
Previous work with germ-free NOD mice has suggested that germ-free conditions significantly exacerbate the development of diabetes [5]. However, this aggravating effect has not been consistently observed in all germ-free NOD colonies [6]. It is clear that microbes are not a prerequisite for diabetes development in NOD mice. Rather, the prevalence of certain bacterial strains may be more relevant to the outcome of the disease. Innate immune mechanisms are important in determining the composition of microbiota. Thus absence of Cd1d (also known as Cd1d1), Nod2 or Myd88 qualitatively and quantitatively alters the microbiota compared with wild-type littermates [4, 7, 8]. Intriguingly, Myd88 knockout mice only develop diabetes under germ-free conditions, while the protective effect under conventional conditions has been demonstrated to result from beneficial microflora in these knockout animals [4].
Various opinions on the development and function of T regulatory (Treg) cells in germ-free mice have been presented [9–12]. Thus germ-free conditions significantly reduced relative and total numbers of Treg cells in the mesenteric lymph nodes (MLN) [9, 10]. Additionally, impairments of the suppressive function of germ-free Tregs have been demonstrated [9, 10]. Other evidence, in contrast, indicates that gut flora antigens play little role in the generation or suppressive activity of Treg cells [11].
The effects of microbiota on IL17 induction in the intestine seem to include multiple mechanisms and may differ depending on the location in the intestine [13–16]. Segmented filamentous bacteria, which colonise the small intestine, have been shown to efficiently induce ileal IL17 [13]. Furthermore, ATP, which can be derived from commensal bacteria, increases IL17 production in the colon of germ-free mice [12]. However, the mice colonised with segmented filamentous bacteria in the study by Ivanov et al. [13] exhibited low levels of ATP in the ileum and colon, demonstrating that IL17 induction can be triggered through various routes. It is also worth noting that commensal bacteria are not the only source of extracellular ATP. Indeed, although extracellular ATP levels under normal conditions are extremely low, it is released from cells under circumstances such as cell injury and T cell activation [14]. An increase in IL17 in the colon of mice under germ-free conditions has also been reported [15]. This increase depended on a lack of commensal-dependent IL25, which under normal circumstances would inhibit IL23 production and subsequently limit the expansion and/or survival of Th17 cells [15]. A recent communication suggests that IL17 is upregulated in the colon and MLN in young specific pathogen-free (SPF) NOD mice [16], but the effect of germ-free conditions on IL17 levels still remains undetermined in NOD mice.
Peritoneal B cells influence insulitis and diabetes development in NOD mice [17]. This pool of innate-type B cells is regulated by microbial factors and actively participates in the defence against invasion by intestinal microbiota. Recent reports indicate that peritoneal B cells in SPF NOD mice are abnormally activated, but the underlying reason for this remains unclear [18]. Investigation of peritoneal B cells in germ-free NOD mice could clarify whether activation of NOD peritoneal B cells is due to microbial factors or whether it depends on genetic factors that predispose the mice to diabetes.
The results presented herein indicate that NOD mice in germ-free and SPF conditions develop diabetes roughly at the same rate, regardless of the finding that germ-free NOD mice have an increased number of infiltrating cells in the pancreatic islets. B cells were less activated in the peritoneum and CD19 B cells were underrepresented in the islet infiltrates in germ-free mice, especially at an early age (6 weeks). Furthermore, IL17 upregulation was observed in the colon, MLN and pancreatic lymph nodes (PaLN), along with a reduction of Treg cells in the ileum, colon and MLN and PaLN of germ-free NOD mice. However, Treg cell numbers in the islet infiltrates were higher in germ-free than in SPF NOD mice, which may be an underlying reason for the relatively innocuous nature of germ-free NOD insulitis.
Methods
Mice
NOD mice were raised and bred in the central animal laboratory of Turku University. This NOD mouse colony originated from Bomholtgaard (presently Taconic, Bomholt, Denmark) and has been maintained at Turku University since 2001. The diabetes incidence in NOD mice at Turku University has for the last several years been 60–65% by 30 weeks of age, increasing to 70–75% by 1 year of age [16, 19]. Germ-free inbred Swiss mice were kindly provided by The Central Animal Laboratory of Radboud University, Nijmegen, the Netherlands. To establish a colony of germ-free NOD mice, sterile NOD pups, derived through Caesarean section, were cross-fostered with germ-free mice. Germ-free NOD mice used for experiments were of second or subsequent generations. The mice used for experiments were prediabetic females, aged 5 to 15 weeks as specified in individual experiments. All mice were maintained on a CRM (E) diet (Special Diets Services, Witham, UK). The food was additionally irradiated with 50 kGy and autoclaved.
Faecal samples from germ-free animals were collected weekly. These samples were analysed by PCR using 16S RNA primers for conserved bacterial sequences, as well as being cultured under aerobic and anaerobic conditions to monitor the sterility of the colony. The germ-free NOD mice displayed physiological traits typical for mice reared under germ-free conditions, such as lack of odour, extremely enlarged caecums [20] and a reduction of T cells in the lymph nodes [21] (electronic supplementary material [ESM] Figs 1 and 2). The diabetes incidence was monitored for germ-free NOD mice by analysis of glucosuria and for SPF NOD mice by blood glucose measurement. The two methods of diabetes monitoring were tested and found to be equally sensitive (C. Alam, unpublished observations). Two consecutive positive readings were regarded as diagnosing diabetes.
All animal experiments were approved by the National Laboratory Animal Care and Use Committee in Finland and conformed to the legal acts, regulations and requirements set by the European Union concerning protection of animals used for research.
Insulitis scoring
Pancreases from 6- and 13-week old SPF and germ-free NOD mice were dissected and fixed with formalin (10% [vol./vol.]). Paraffin-embedded sections, 4 to 5 μm thick, were prepared and stained with haematoxylin and eosin. Insulitis was assessed according to previous work [22] by giving a score from 0 to 3 for each islet, whereby 0 stood for no infiltration, 1 for peri insulitis, 2 for infiltration covering approximately half of the islet and 3 for full insulitis. We scored 50 islets per pancreas from 6- and 13-week-old prediabetic NOD and germ-free NOD mice. A mean insulitis score for a pancreas was calculated by dividing the sum of individual islet scores with the number of islets analysed in that pancreas.
Determination of lymphocyte subsets from lymph nodes
MLN, PaLN and peripheral lymph node cells from 6-week-old SPF and germ-free NOD mice were incubated with FITC-conjugated anti-CD8, anti-CD4 conjugated to phycoerythrin (Immunotools, Friesoythe, Germany) and biotinylated anti-CD45R/B220 (R&D Systems, Minneapolis, MN, USA) antibody followed by treatment with streptavidin and allophycocyanin (Invitrogen, Carlsbad, CA, USA). Flow cytometry was performed using a software package (FACS Calibur, Cell Quest; BD Biosciences, San Jose, CA, USA).
Assessment of intestinal histology
For histological studies of the colon, SPF and germ-free NOD mice and BALB/c mice were killed at the age of 4.5 to 5 weeks. Colons were excised, washed with PBS and fixed in 10% (vol./vol.) buffered formalin. After rehydration, 4 to 5 μm thick paraffin-embedded sections were stained with haematoxylin and eosin, and hyperplasia assessed by measuring the thickness of the epithelial crypts using light microscopy (Olympus, Berlin, Germany).
Real-time PCR for quantification of cytokine and Foxp3 gene expression
SPF and NOD mouse colon and ileum samples from 9- to 15-week-old mice were washed with ice-cold PBS (EuroClone, Wetherby, UK), cut into pieces (5 × 5 mm) and stored in RNA Later (Qiagen, Hilden, Germany). Total RNA was purified with a kit (RNEasy Mini Kit; Qiagen). RNA purity and quantification was determined using a spectrophotometer (Nanodrop Technologies, Wilmington, DE, USA). cDNA was synthesised using oligo-dT primers with a kit (DyNAmo cDNA Synthesis Kit; Finnzymes, Espoo, Finland). Levels of cytokines in colons and ileums of individual mice were evaluated with real-time quantitative PCR (Maxima SYBR Green qPCR Master Mix; Fermentas, St Leon-Rot, Germany) and a RotoGene cycler (Qiagen). The following primers were used: Gadph forward primer 5′-AACGACCCCTTCATTGAC-3′ and reverse primer 5′-TCCACGACATACTCAGCAC-3′; Il17 (also known as Il17a) forward primer 5′-TCCCTCTGTGATCTGGGAAG-3′ and reverse primer 5′-CTCGACCCTGAAAGTGAAGG-3′; and Foxp3 forward primer 5′-GGCCCTTCTCCAGGACAGA-3′ and reverse primer 5′-GCTGATCATGGCTGGGTTGT-3′. Ct-values were normalised to the endogenous housekeeping gene Gapdh and are expressed as copy numbers relative to the Gapdh copy numbers.
Intracellular cytokine and forkhead box P3 stainings
For cytokine staining, freshly isolated lymph node cells from MLN and PaLN of 9- to 15-week-old mice were pooled and incubated for 4 h at 37°C in RPMI 1640 (Gibco, Grand Island, NY, USA), supplemented with 10% (vol./vol.) FBS, 2 mmol/l l-glutamine and 100 U/ml penicillin and streptomycin (Sigma-Aldrich, St Louis, MO, USA), and 0.1 μmol/l phorbol 12-myristate 13-acetate (PMA), 1 μmol/l ionomycin and 10 μg/ml Brefeldin A (Sigma-Aldrich). After this stimulation, surface-staining was performed with FITC-conjugated anti-CD4 (Immunotools). For forkhead box P3 (FOXP3) staining, freshly isolated cells were stained directly with FITC-conjugated anti-CD4 and APC-conjugated anti-CD25 (Immunotools).
The cells were then fixed with 2% (vol./vol.) paraformaldehyde and permeabilised with 0.5% (vol./vol.) saponin. The intracellular staining was performed according to standard procedures with phycoerythrin-conjugated anti-IFN-γ, anti-IL17, anti-FOXP3 or appropriate isotype controls (eBioscience, San Diego, CA, USA). Fragment crystallisable block (eBioscience) was used to block non-specific binding. Samples were analysed immediately using FACS Calibur.
Peritoneal cell population and activation marker analysis
Peritoneal lavage cells from 9- to 15-week-old mice were counted and incubated with CD11b-FITC and biotinylated anti-CD45R/B220 (R&D Systems) antibody followed by treatment with streptavidin and allophycocyanin (Invitrogen), as well as phycoerythrin-conjugated CD69, CD40 or CD86 (Immunotools). Samples were analysed immediately using FACS Calibur.
Immunofluorescence staining
Pancreases from 6- and 13-week-old NOD and germ-free NOD mice were dissected, immersed in embedding medium (Sakura Finetek, Zoeterwoude, the Netherlands) and frozen in liquid nitrogen. Cryosections of 6 to 7 μm thickness were acetone-fixed and subsequently stained either with CD4-FITC and CD19-phycoerythrin (Immunotools), or with CD4-FITC, FOXP3-phycoerythrin (eBioscience) and anti-rabbit insulin H-86 (Santa Cruz Biotechnology, Santa Cruz, CA, USA) followed by donkey anti-rabbit fluor 647 (Invitrogen). Analysis was performed using a UV microscope (Olympus) or a confocal microscope (LSM510 META; Zeiss, Munich, Germany).
Results
Diabetes incidence and insulitis scores for SPF and germ-free NOD mice
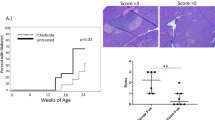
Persistent glucosuria in germ-free NOD mice developed at approximately the same time as persistent hyperglycaemia in SPF NOD mice, with 60% to 75% of all animals having diabetes by 30 weeks of age (Fig. 1a). Insulitis, however, developed earlier in germ-free NOD mice, with almost 20% of islets affected at 6 weeks of age, compared with only 5% in SPF NOD mice. At 13 weeks, almost 80% of islets were infiltrated in germ-free NOD mice, compared with <40% in SPF NOD mice. The average insulitis score was also significantly higher in germ-free NOD mice at 6 and at 13 weeks of age (Fig. 1b–e).
Diabetes incidence and insulitis scores in germ-free and SPF NOD colonies. a A Kaplan–Meier survival diagram for SPF NOD (continuous line) and germ-free (GF) NOD (broken line) mice colonies. The survival curves for both groups were not significantly different according to logrank test. Germ-free mice, n = 14, SPF mice, n = 10. b Average insulitis score for SPF and germ-free NOD mice at 6 weeks and (c) at 13 weeks of age. ***p < 0.001 by Student’s t test. d Per cent of islets with a given score in 6-week-old and (e) 13-week-old mice. White, insulitis score 0, no infiltration; light grey, insulitis score 1, peri-insulitis: dark grey, insulitis score 2, infiltration covering approximately half of the islet; black, insulitis score 3, full insulitis. b–e n = 4 mice per group, with 50 islets for each mouse scored
Il17 and Foxp3 mRNA levels in the ileum and colon, and colon histology
The mRNA levels of Il17 and Foxp3 were lower in ileum of germ-free than in that of SPF NOD mice. In the colon, Foxp3 expression was also lower in germ-free NOD mice, but Il17 expression was significantly higher in germ-free than in SPF NOD mice (Fig. 2a–d). Hyperplastic crypts have been consistently observed in colons of 4- to 5-week-old SPF NOD mice [16]. The crypt depth at this age is roughly double in SPF NOD mice, compared with BALB/c mice. In germ-free NOD mice, however, the crypts were very thin and uniform, showing no signs of hyperplasia (Fig. 2e, f).
Il17 and Foxp3 mRNA expression in ileum and colon of germ-free (GF) and SPF NOD mice measured with real-time PCR. a Foxp3 expression in ileum and (b) colon. c Il17 expression in ileum and (d) colon. Bars (a–d) represent means ± SEM; *p < 0.05 and **p < 0.01 by one-way ANOVA and Bonferroni’s post hoc test. e Average ± SEM colon crypt depth in 5-week-old mice as labelled; ***p < 0.001. f Representative images of crypt depth in mouse colons as indicated. Scale bars and diagonal bars, 300 μm. n = 4 mice per group for all experiments
Intracellular staining for IFNγ, IL17 and FOXP3 in pooled MLN and PaLN cells
In accordance with the RT-PCR results from ileum and colon samples, intracellular staining from pooled MLN/PaLN cells also showed a reduction of FOXP3 abundance; an average of 2% of CD4+ T cells were FOXP3+ in germ-free NOD MLN and PaLN, compared with 4% in SPF NOD mice (Fig. 3a, d). Moreover, intracellular staining for IFNγ and IL17 demonstrated a significantly higher percentage of IFNγ and IL17 CD4+ T cells in MLN and PaLN of germ-free than in that of SPF NOD mice (Fig. 3b, c, e).
Intracellular (IC) staining in MLN and PaLN of SPF and germ-free (GF) NOD mice. a Per cent CD4+CD25+FOXP3+ cells in MLN and PaLN of mice as indicated. b Per cent IL17+ CD4 T cells and (c) per cent IFNγ+CD4 T cells. Bars represent means ± SEM; **p < 0.01 by one way ANOVA and Bonferroni’s post hoc test; n = 4 mice per group. d Representative dotplot of the isotype, and of CD4+CD25+FOXP3+ staining in SPF and germ-free NOD mouse MLN and PaLN. e Representative dotplot of IFNγ and IL17 IC staining, gated on CD4+ cells in SPF and germ-free MLN and PaLN
Peritoneal B cells
The peritoneum harbours a substantial pool of B lymphocytes. In SPF NOD mice, a considerable reduction of B1 and B2 type B lymphocyte numbers has been detected [18]. The peritoneal B cells in SPF NOD mice also exhibit an increased activation status, as evidenced by multiple activation markers, including CD40, CD86 and CD69. In contrast to SPF NOD mice, germ-free NOD animals did not exhibit any reduction of peritoneal B1 or B2 cells at 5 weeks, but the number of these in the peritoneum was comparable with that in BALB/c mice. At a later time point (>17 weeks), peritoneal B cell numbers were also significantly higher in germ-free than in SPF NOD mice (Fig. 4a, b). The early activation marker CD69 and the costimulatory molecule CD40 were considerably lower on peritoneal B1 (CD11b+) and B2 (CD11b−) cells from germ-free NOD animals, as was levels of CD86 on germ-free NOD B1 cells (Fig. 4c–g). B2 cells showed little positivity for CD86 in SPF or germ-free NOD mice (not shown).
B cell numbers in the peritoneum of SPF and germ-free (GF) NOD, and expression of CD69 and co-stimulatory molecules CD40 and CD86. a Number of B1(B220+CD11b+) and (b) B2 cells (B220+CD11b−) at 5 and 17 weeks of age in SPF and germ-free peritoneum. Black bars, SPF NOD; white bars, germ-free NOD; striped bars, BALB/c. c Per cent CD69+ B1 cells and (d) B2 cells in the peritoneum of 5-week-old mice as labelled. e Mean fluorescence intensity (MFI) for CD40 on B1 and (f) B2 cells, and for CD86 (g) on B1 cells, respectively, in 5-week-old mice. Bars represent means ± SEM; *p < 0.05, **p < 0.01 and ***p < 0.01 by one-way ANOVA and Bonferroni’s post hoc test; n = 4 mice per group
Lower number of infiltrating CD19 cells in germ-free NOD mice
Insulitis was more severe at 6 and 13 weeks of age in germ-free NOD mice than in SPF NOD mice (Fig. 1). Immunohistochemical analysis revealed that the infiltrating cells were dominated by CD4+ T cells. Indeed, in SPF as well as germ-free NOD mouse pancreases, CD4+ T cells were more abundant than CD8+ T cells (result not shown). Approximately 15% of CD4+ islets in germ-free NOD mice were co-infiltrated with B cells at 6 weeks of age, compared with more than 40% in SPF NOD mice. At 13 weeks, the differences were less accentuated, but 40% of affected islets still lacked CD19 cells, compared with 30% in SPF NOD mice (Fig. 5a, b).
Relative numbers of infiltrating CD19/CD4 and CD4+FOXP3+ cells, and CD4 and insulin staining of pancreatic islets in 13-week-old SPF and germ-free (GF) NOD mice. a Per cent of CD19 B cells relative to the total number of CD4 T cells in islets. White bars, 0% CD19 B cells in CD4 T cell-infiltrated islets; hatched bars, < 25%; striped bars, 26–50%; black bars, > 50%. b Representative images of CD19 B cells (red) vs CD4 T cells (green) in islets as labelled. c Per cent CD4+ islets that were positive for FOXP3 in 13-week-old mice as labelled. Bars represent means ± SEM, *p < 0.05 by Student’s t test. A total of 59 islets from SPF NOD mice and 41 islets from germ-free NOD mice were analysed from four different SPF and three different germ-free NOD mice. d Representative images of CD4 (green) and FOXP3 (red) staining of islets from mice as indicated. White arrows indicate FOXP3-positive CD4 T cells. e Representative image of pancreatic islets stained for CD4+ (green) and insulin (blue) from 13-week-old mice as indicated
FOXP3 CD4+ cells and insulin staining of the pancreatic islets of germ-free NOD mice
Even though Foxp3 mRNA levels were lower in germ-free NOD ileum and colon, and FOXP3+ CD4 T cells from MLN and PaLN were scarcer, we observed a small but significant increase in FOXP3+ CD4 T cells in the islet infiltrates of germ-free NOD mice. FOXP3 positivity was observed in roughly 65% of islets with CD4+ infiltrating cells from germ-free animals and in 40% of CD4+ islets in SPF NOD mice (Fig. 5c, d). Moreover, insulin staining was preserved in the islets of 13-week-old germ-free NOD mice to the same extent as in age-matched SPF NOD mice (Fig. 5e).
Discussion
The results presented here suggest that even though insulitis developed at a faster rate in germ-free NOD mice, the diabetes incidence remained the same in germ-free and SPF NOD mice. Moreover, immune regulation in the colon, MLN and PaLN is altered in germ-free NOD mice, as can be seen from the diminished levels of FOXP3 and increased IL17 levels. Yet FOXP3 CD4 T cells were observed even more frequently in infiltrated pancreatic islets of germ-free than in those of SPF NOD mice. If lack of microbiota affects local induction of gut-induced Treg cells in the intestine and draining lymph nodes without inhibiting natural Treg cell development, it is conceivable that FOXP3 CD4 T cells in islets represent natural Treg cells. Indeed, studies on other germ-free mouse strains have shown that Treg cell numbers in the thymus or spleen are not affected by germ-free conditions, but, rather, the reduction is limited to MLN and Peyer’s patches [21]. The faster rate of insulitis development in young germ-free NOD mice may nevertheless be linked to the lack of antigen-induced FOXP3 Treg cells and/or the increased IL17 levels in intestine, and in MLN and PaLN during the initiation of islet-specific immune responses.
Despite the extensiveness of islet infiltration in germ-free NOD mice, insulin preservation in islets and transition to overt diabetes remained the same as in SPF NOD mice. An analysis of the infiltrating cells in the islets revealed a marked reduction in the ratio of CD19 B cells to CD4 T cells, as well as an increase in CD4+FOXP3+ cells in germ-free NOD mice. The importance of Treg cells for maintaining islet-infiltrating lymphocytes in an innocuous state and for preserving insulin staining in islets has been demonstrated in two different mouse models [23, 24]. Moreover, according to previous studies, depletion of B cells is accompanied by an expansion of Treg cells [25, 26]. It is hence tempting to speculate whether the reduction of infiltrating B cells in pancreatic islets of germ-free NOD mice contributes to the observed increase in Treg cells and subsequently to the (at least temporary) preservation of pancreatic islet lesions in an innocuous state in those mice. This could be an underlying reason for the similar diabetes incidence in SPF and germ-free NOD mice, despite the earlier and more extensive T cell infiltration into the islets.
Peritoneal B cells in NOD mice are overactivated and decreased in numbers in the peritoneal cavity [18]. However, in peritoneal B cells from germ-free NOD mice, no such signs of activation were observed. Levels of CD69, and of costimulatory molecules CD40 and CD86, which are abnormally increased in SPF NOD mice, were significantly lower on both B1 and B2 peritoneal cells in germ-free NOD mice. Peritoneal B cells characteristically migrate out of the peritoneum upon activation [27], so the reduced number of these cells in SPF NOD mice is indicative of cell activation. In germ-free NOD mice, however, B cell numbers were markedly higher than in SPF NOD mice. The increased number and the reduced surface expression of costimulatory molecules would thus indicate that peritoneal B cells in germ-free NOD mice may not exhibit the same heightened activation status as their SPF counterparts. This is intriguing, as it suggests that activation of peritoneal B cells in SPF NOD mice may be dependent on the presence of microbes. Peritoneal B cells and/or other innate B1 cells are thought to play an important role in the development of diabetes in the NOD and RIP-OVA transgenic mouse models, since B cells in the pancreatic infiltrates closely resemble peritoneal B1 cells [19, 26] and hypotonic lysis of peritoneal B cells has been reported to delay diabetes [17]. The results presented here raise the question of whether microbial stimulation may be needed for peritoneal B cells to become activated, efficiently infiltrate pancreatic islets and promote islet damage in NOD mice.
NOD mice reared under SPF conditions show several signs of colon inflammation, including increased IL17 levels and crypt hyperplasia at a young age [16]. Germ-free NOD mice, on the contrary, showed no signs of colon hyperplasia, indicating that this inflammatory change in SPF NOD mice is dependent on microbial factors. In fact, withdrawal of microbes greatly reduces inflammatory propensities in the colon in most experimental models of colitis [28]. In accordance with previous reports for other germ-free mouse strains [13], mRNA levels of Il17 in the germ-free NOD mouse ileum were lower than in SPF NOD mice. However, RT-PCR analysis of colon samples showed an increase in Il17 mRNA and intracellular staining of lymph nodes showed an increase in IL17+ and IFNγ+ CD4 T cells. This suggests that microbial factors are not necessary for IL17 induction in the colon of NOD mice. In fact, the differentiation of T cells in the colon as well as in gut- or pancreas-associated lymphoid tissues under germ-free conditions in NOD mice appears to be biased towards the autoimmunity-associated subsets Th17 and Th1. The effect of germ-free conditions on IL17 regulation in the intestine has been disputed. One view asserts that bacterial components or certain bacterial species are necessary for IL17 induction, particularly in the small intestine [13, 14], whereas an opposing view stipulates that germ-free conditions impede the regulatory mechanisms normally in place in the gut and actually induce IL17 [15]. Our results, based on RT-PCR, analysis indicate a different effect of germ-free conditions on Th17 levels in the ileum and colon, whereby IL17 is increased in the colon, but not in the ileum of germ-free NOD mice.
An early and much quoted study by Suzuki and colleagues indicated that germ-free conditions drastically aggravate diabetes in NOD mice [5]. However, not all reports support this. According to a study based on a sizeable germ-free NOD colony at Shionogi Aburahi Laboratories, Japan, 70.2% of female germ-free NOD mice became diabetic by 30 weeks of age, compared with 81.4% of female SPF NOD mice [6]. Moreover, regardless of the 100% diabetes incidence by 30 weeks in germ-free NOD female mice in their study, Wen and colleagues report that the incidence was not notably different from SPF NOD females, nor did the germ-free NOD mice develop diabetes earlier than their SPF counterparts [4]. The results presented herein also show that, despite the accelerated rate of islet infiltration in germ-free NOD mice, diabetes incidence was not significantly different in germ-free and SPF NOD mice by 30 weeks of age. The question of whether the unchanged diabetes incidence in germ-free NOD females witnessed in our colony or the considerably aggravated disease outcome observed by Suzuki et al. [5] is a more common effect of microbial deprivation in NOD mice will probably be answered as more germ-free NOD colonies of differing pedigree are established. It is also possible that the differences observed are due to factors such as genetic drift in colonies that have been reared for several years in a particular facility, like the NOD mice used in the present work. Finally, since the diabetes incidence in our colony of SPF NOD mice is as low as 70% by 30 weeks of age, it remains possible that a difference in incidence could have emerged if the mice had been monitored for hyperglycaemia beyond the typical 30 week follow-up period.
It is suggested that the absence of Treg cells and the increased levels of IL17 in the gut and in MLN and PaLN may be contributing factors to the increased islet infiltration in germ-free NOD mice. However, as islet infiltration in germ-free mice proceeded, Treg cells were detected more frequently in germ-free than in SPF mice, possibly explaining why the earlier and more extensive insulitis does not accelerate diabetes development in germ-free NOD mice. Whether the abundance of Treg cells in the pancreatic infiltrates resulted from a decreased number of infiltrating B cells deriving from reduced activation of peritoneal B cells in the absence of microbial stimulation is a question that will require further scrutiny. However, the results presented here certainly indicate that lack of microbiota alters gut immune regulation, B cell activation and the progression of insulitis. Ultimately, however, germ-free conditions in this study neither induced earlier diabetes development, nor did they protect our NOD mice from diabetes.
Abbreviations
- FOXP3:
-
Forkhead box P3
- MLN:
-
Mesenteric lymph node
- PaLN:
-
Pancreatic lymph node
- SPF:
-
Specific pathogen-free
- Treg:
-
T regulatory
References
Concannon P, Rich SS, Nepom GT (2009) Genetics of type 1A diabetes. N Engl J Med 360:1646–1654
Lee AS, Gibson DL, Zhang Y, Sham HP, Vallance BA, Dutz JP (2010) Gut barrier disruption by an enteric bacterial pathogen accelerates insulitis in NOD mice. Diabetologia 53:741–748
Vaarala O, Atkinson MA, Neu J (2008) The “perfect storm” for type 1 diabetes: the complex interplay between intestinal microbiota, gut permeability, and mucosal immunity. Diabetes 57:2555–2562
Wen L, Ley RE, Volchkov PY et al (2008) Innate immunity and intestinal microbiota in the development of type 1 diabetes. Nature 455:1109–1113
Suzuki T, Yamado T, Takao T, Fujimura T, Kawamura E, Shimizu M, Yamashita R, Nomoto K (1987) Diabetogenic effects of lymphocyte transfusion on the NOD or NOD nude mouse. Karger, Basel
Taniguchi H, Makino S, Ikegami H (2007) The NOD mouse and its related strains. CRC, Boca Raton
Nieuwenhuis EE, Matsumoto T, Lindenbergh D et al (2009) Cd1d-dependent regulation of bacterial colonization in the intestine of mice. J Clin Invest 119:1241–1250
Petnicki-Ocwieja T, Hrncir T, Liu YJ et al (2009) Nod2 is required for the regulation of commensal microbiota in the intestine. Proc Natl Acad Sci USA 106:15813–15818
Ishikawa H, Tanaka K, Maeda Y et al (2008) Effect of intestinal microbiota on the induction of regulatory CD25+ CD4+ T cells. Clin Exp Immunol 153:127–135
Ostman S, Rask C, Wold AE, Hultkrantz S, Telemo E (2006) Impaired regulatory T cell function in germ-free mice. Eur J Immunol 36:2336–2346
Min B, Thornton A, Caucheteux SM et al (2007) Gut flora antigens are not important in the maintenance of regulatory T cell heterogeneity and homeostasis. Eur J Immunol 37:1916–1923
Atarashi K, Nishimura J, Shima T et al (2008) ATP drives lamina propria T(H)17 cell differentiation. Nature 455:808–812
Ivanov II, Atarashi K, Manel N et al (2009) Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 139:485–498
Schenk U, Westendorf AM, Radaelli E et al (2008) Purinergic control of T cell activation by ATP released through pannexin-1 hemichannels. Sci Signal 1:ra6
Zaph C, Du Y, Saenz SA et al (2008) Commensal-dependent expression of IL-25 regulates the IL-23-IL-17 axis in the intestine. J Exp Med 205:2191–2198
Alam C, Valkonen S, Palagani V, Jalava J, Eerola E, Hanninen A (2010) Inflammatory tendencies and overproduction of IL-17 in the colon of young NOD mice are counteracted with diet change. Diabetes 59:2237–2246
Kendall PL, Woodward EJ, Hulbert C, Thomas JW (2004) Peritoneal B cells govern the outcome of diabetes in non-obese diabetic mice. Eur J Immunol 34:2387–2395
Alam C, Valkonen S, Ohls S, Tornqvist K, Hanninen A (2010) Enhanced trafficking to the pancreatic lymph nodes and auto-antigen presentation capacity distinguishes peritoneal B lymphocytes in non-obese diabetic mice. Diabetologia 53:346–355
Merinen M, Irjala H, Salmi M, Jaakkola I, Hanninen A, Jalkanen S (2005) Vascular adhesion protein-1 is involved in both acute and chronic inflammation in the mouse. Am J Pathol 166:793–800
Pesti L (1979) Intestinal microflora: elimination of germfree characteristics by components of the normal microbial flora. Comp Immunol Microbiol Infect Dis 1:141–152
Hrncir T, Stepankova R, Kozakova H, Hudcovic T, Tlaskalova-Hogenova H (2008) Gut microbiota and lipopolysaccharide content of the diet influence development of regulatory T cells: studies in germ-free mice. BMC Immunol 9:65
Maksimow M, Alam C, Hanninen A (2008) Incomplete killing and enhanced activation of islet-reactive CD8+ T cells by FasL-expressing dendritic cells limits protection from diabetes. Rev Diabet Stud 5:144–153
Chen Z, Herman AE, Matos M, Mathis D, Benoist C (2005) Where CD4+CD25+ T reg cells impinge on autoimmune diabetes. J Exp Med 202:1387–1397
Ryan GA, Wang CJ, Chamberlain JL et al (2010) B1 cells promote pancreas infiltration by autoreactive T cells. J Immunol 185:2800–2807
Fiorina P, Vergani A, Dada S et al (2008) Targeting CD22 reprograms B cells and reverses autoimmune diabetes. Diabetes 57:3013–3024
Hu CY, Rodriguez-Pinto D, Du W et al (2007) Treatment with CD20-specific antibody prevents and reverses autoimmune diabetes in mice. J Clin Invest 117:3857–3867
Ha SA, Tsuji M, Suzuki K et al (2006) Regulation of B1 cell migration by signals through Toll-like receptors. J Exp Med 203:2541–2550
Strober W, Fuss IJ, Blumberg RS (2002) The immunology of mucosal models of inflammation. Annu Rev Immunol 20:495–549
Acknowledgements
This work was supported by The Academy of Finland, The Päivikki and Sakari Sohlberg Foundation, Finland, The Finnish Diabetes Research Foundation, The Finnish Cultural Foundation, Emil Aaltonen Foundation, Finland and Oskar Öflund Foundation, Finland. We would also like to thank T. Hiltula-Maisala (Central Animal Laboratory, University of Turku) for maintenance of the germ-free mouse colony.
Duality of interest
The authors declare that there is no duality of interest associated with this manuscript.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM Fig. 1
Enlarged caecum in germ-free NOD mice. The caecums of germ-free (GF) NOD mice were grossly enlarged compared with SPF NOD mice. This enlargement is characteristic for mice raised under microbe-free conditions and is reduced upon bacterial colonisation [20]. The pictures are representative of the anatomy of the caecum (arrows) in SPF NOD mice (left) and germ-free NOD mice (right). The germ-free NOD mouse was dissected at the age of 8 weeks and was born when monocontamination of the isolator with an anaerobic Gram-positive rod was detected. The microbe was sequenced and identified as Clostridium lituseburense. Part of the results on cytokines and FOXP3 in the gut and lymph nodes, as well as islet immunohistochemistry are derived from mice potentially monocontaminated with this microbe. In contrast, results on diabetes incidence in the germ-free colony were derived from mice that became diabetic before contamination (PDF 56 kb)
ESM Fig. 2
T-lymphocyte deficiency in (a) PaLN, (b) MLN and (c) peripheral lymph nodes (Perif LN) of germ-free mice. a Per cent CD4/CD8/B220 cells in PaLN; (b) per cent CD4/CD8/B220 cells in MLN; and (c) per cent CD4/CD8/B220 cells in perif LN. The fraction of CD4 cells in MLN, PaLN and Perif LN of germ-free mice was only half that observed for conventional NOD mice. In contrast, the ratio of B lymphocytes (B220+) was higher in germ-free mice. Bar graphs present percentage of B and T cells in lymph nodes of SPF and germ-free (GF) NOD mice. Black bars represent B220 B cells, striped bars represent CD4 T cells and white bars represent CD8 T cells. The markedly smaller size of lymph nodes in germ-free mice, observed by us (C. Alam, unpublished observation) and others [21], must be acknowledged; hence, the total T cell difference between germ-free and conventional NOD mice was larger than twofold. Conversely, the total B cell numbers in the lymph nodes of NOD and germ-free NOD mice appear not to be significantly different (PDF 56 kb)
ESM Fig. 3
Peritoneal B cell staining for CD69, CD40 and CD86. Dotplots represent peritoneal B cell staining with anti-CD69, anti-CD40 and anti-CD86. Dotplots labelled ‘B1 cells’ are gated on CD11b+B220+ positive cells; plots for ‘B2 cells’ are gated on CD11b−B220+ peritoneal cells (PDF 85 kb)
Rights and permissions
About this article
Cite this article
Alam, C., Bittoun, E., Bhagwat, D. et al. Effects of a germ-free environment on gut immune regulation and diabetes progression in non-obese diabetic (NOD) mice. Diabetologia 54, 1398–1406 (2011). https://doi.org/10.1007/s00125-011-2097-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-011-2097-5