Abstract
Aims/hypothesis
Insulin resistance in skeletal muscle is linked to mitochondrial dysfunction in obesity and type 2 diabetes. Emerging evidence indicates that reversible phosphorylation regulates oxidative phosphorylation (OxPhos) proteins. The aim of this study was to identify and quantify site-specific phosphorylation of the catalytic beta subunit of ATP synthase (ATPsyn-β) and determine protein abundance of ATPsyn-β and other OxPhos components in skeletal muscle from healthy and insulin-resistant individuals.
Methods
Skeletal muscle biopsies were obtained from lean, healthy, obese, non-diabetic and type 2 diabetic volunteers (each group n = 10) for immunoblotting of proteins, and hypothesis-driven identification and quantification of phosphorylation sites on ATPsyn-β using targeted nanospray tandem mass spectrometry. Volunteers were metabolically characterised by euglycaemic–hyperinsulinaemic clamps.
Results
Seven phosphorylation sites were identified on ATPsyn-β purified from human skeletal muscle. Obese individuals with and without type 2 diabetes were characterised by impaired insulin-stimulated glucose disposal rates, and showed a ∼30% higher phosphorylation of ATPsyn-β at Tyr361 and Thr213 (within the nucleotide-binding region of ATP synthase) as well as a coordinated downregulation of ATPsyn-β protein and other OxPhos components. Insulin increased Tyr361 phosphorylation of ATPsyn-β by ∼50% in lean and healthy, but not insulin-resistant, individuals.
Conclusions/interpretation
These data demonstrate that ATPsyn-β is phosphorylated at multiple sites in human skeletal muscle, and suggest that abnormal site-specific phosphorylation of ATPsyn-β together with reduced content of OxPhos proteins contributes to mitochondrial dysfunction in insulin resistance. Further characterisation of phosphorylation of ATPsyn-β may offer novel targets of treatment in human diseases with mitochondrial dysfunction, such as diabetes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Proteins in the mitochondrial oxidative phosphorylation (OxPhos) machinery play a crucial role in energy production, generation of reactive oxygen species and apoptosis. Through the concerted action of the electron transport chain and ATP synthase, OxPhos is responsible for the production of more than 90% of cellular ATP. Impaired mitochondrial OxPhos is involved in a wide range of human pathologies, including neurodegenerative diseases, cancer, diabetes and ageing [1]. Skeletal muscle, which is rich in mitochondria, accounts for ∼25% of whole body oxygen consumption in humans in the resting state [2]. Moreover, skeletal muscle is the major site of glucose uptake (∼80%) in response to insulin [3], and correspondingly is an important site of insulin resistance in obesity and type 2 diabetes [4–6]. Insulin resistance in skeletal muscle is characterised by impaired insulin stimulation of glucose transport and glycogen synthesis, and shows a strong correlation with intramyocellular lipid content [7, 8]. Recently, a number of studies have provided evidence for a reduced content and functional capacity of muscle mitochondria in obesity and type 2 diabetes [9–13], and it has been hypothesised that mitochondrial dysfunction could be responsible for the accumulation of lipid metabolites [14]. An accumulation of lipid metabolites could, in turn, partially explain the insulin signalling defects reported in skeletal muscle in obesity and type 2 diabetes [9, 14]. Microarray-based studies of muscle gene expression in patients with type 2 diabetes and high-risk individuals have consistently shown a coordinated downregulation of OxPhos genes and suggested that a reduced expression of the peroxisome proliferator-activated receptor, gamma, coactivator 1, alpha gene (PPARGC1A) could play a key role for impaired mitochondrial biogenesis [15–17]. Whether a similar coordinated downregulation is seen at the protein level and to what extent mitochondrial dysfunction involves altered post-translational modifications of OxPhos proteins remain to be clarified.
In higher organisms, reversible phosphorylation of enzymes plays a crucial role for hormonal regulation of cell signalling pathways, e.g. insulin signalling [18]. There is now evidence that a number of mitochondrial proteins including subunits in all respiratory complexes (I–V) of OxPhos are phosphorylated, and potentially regulated by several kinases and phosphatases localised to mitochondria [19, 20]. Using a two-dimensional gel-based proteomic approach, we previously reported reduced protein content and altered phosphorylation of the catalytic beta subunit in the ATP synthase complex (ATPsyn-β) in muscle of patients with type 2 diabetes [21]. In that study, a single phosphopeptide was identified with the potential phosphorylation site, Thr213, in the nucleotide-binding region of ATPsyn-β.
We recently developed a targeted approach for identifying and quantifying multiple phosphorylation sites on insulin receptor substrate 1 in human skeletal muscle using nanospray tandem mass spectrometry (MS/MS) [22–24]. Here, we applied a modification of this proteomic approach to discover novel phosphorylation sites on ATPsyn-β and to examine whether the phosphorylation of muscle ATPsyn-β at specific sites is altered in obesity and type 2 diabetes, or in response to physiological hyperinsulinaemia. Moreover, we studied whether changes in ATPsyn-β protein content are coordinated with changes in protein levels of other OxPhos proteins.
Methods
Study cohort
Ten healthy lean and ten obese volunteers were carefully matched to ten patients with type 2 diabetes participating in the study (Table 1). Patients with type 2 diabetes were treated either by diet alone or diet in combination with sulfonylurea, metformin or insulin, which were withdrawn 1 week prior to the study. Patients with type 2 diabetes were all negative for GAD65 antibody and without signs of diabetic retinopathy, nephropathy, neuropathy or macrovascular complications. The lean and obese controls had normal glucose tolerance and no family history of diabetes. All participants had normal results on screening blood tests of hepatic and renal function, and were instructed to refrain from strenuous physical activity for a period of 48 h before the experiment. Informed consent was obtained from all individuals before participation. The study was approved by the local ethics committee and was performed in accordance with the Declaration of Helsinki.
Metabolic studies and muscle biopsies
The euglycaemic–hyperinsulinaemic clamp studies were performed after an overnight fast as described in detail previously [5, 6]. In brief, a 2 h basal tracer equilibration period was followed by infusion of insulin at a rate of 40 mU m−2 min−1 for 4 h. Using this protocol, euglycaemia at a plasma glucose concentration of 5.0-5.5 mmol/l and physiological hyperinsulinaemia at a serum insulin concentration of ∼400 pmol/l were obtained in all groups during the insulin-stimulated steady-state period. The studies were combined with indirect calorimetry, and rates of total glucose disposal, glucose and lipid oxidation, and non-oxidative glucose metabolism were calculated as described [5, 6]. Serum insulin and plasma glucose, triacylglycerol, NEFA and adiponectin were assayed as described [5, 6]. A muscle biopsy from each individual was obtained from the vastus lateralis muscle in the basal state after the 2 h basal tracer equilibration period using a modified Bergström needle with suction under local anaesthesia. Muscle samples were immediately blotted free of blood, fat and connective tissue and frozen in liquid nitrogen within 30 s. The muscle biopsy specimens were homogenised while still frozen in an ice-cold buffer as previously described [25].
SDS-PAGE and immunoblotting
Fifty micrograms of muscle protein lysates were separated by 10% SDS-PAGE gels and transferred to nitrocellulose membranes. After blocking with TRIS-buffered saline plus 0.05% Tween 20 (TBST) with 5% non-fat dry milk for 1 h at 4°C, the membranes were washed with TBST, and then incubated for 1 h in TBST with 5% non-fat dry milk with primary antibodies against the proteins NADH-ubiquinone oxidoreductase chain 6 (ND6), succinate dehydrogenase complex, subunit A, flavoprotein (SDHA), ubiquinol-cytochrome c reductase core protein I (UQCRC1), ATP synthase beta (ATP5B) and ATP synthase alpha (ATP5A1) (Molecular Probes, Eugene, OR, USA) and β-actin (Cell Signaling Technology, Beverly, MA, USA) following the manufacturer’s recommendations. The protein bands on the membranes were quantified by densitometry (VersaDoc Imaging System, model 5000: BioRad Laboratories, Hercules, CA, USA). An internal control of previously extracted human muscle was used in each gel to normalise for variations in signals across different membranes. β-Actin was used to correct for potential differences in blood contamination of the muscle samples and assure equal loading.
Immunoprecipitation of ATPsyn-
β Protein G agarose beads (25 μg; SigmaAldrich, St Louis, MO) were prepared by washing twice with 1 ml PBS (GIBCO, cat. no. 10010-023; Invitrogen, Carlsbad, CA, USA). Mouse monoclonal ATP synthase β antibody (Abcam, Cambridge, UK) in 300 μl PBS was added to beads and mixed end over end for 1 h at room temperature. The antibody-conjugated beads were washed twice with 1 ml PBS while kept on ice. Two milligrams of muscle lysate protein diluted in 400 μl homogenisation buffer were mixed end over end with the antibody-conjugated beads overnight at 4°C, and washed with 1 ml PBS. The beads were then washed four times with 1 ml PBS and 15 µl SDS 2X sample buffer was added. Denaturing was performed by incubating the samples at 37°C for 30 min. Lower denaturing temperatures (i.e. 37°C) prevents the reduction of the disulphide bond between the immunoglobulin heavy chains, resulting in a shift in the IgG electrophoretic migration (from ∼50 KDa to ∼90–100 KDa), removing as much of the antibody as possible from the ∼50 kDa band of ATPsyn-β. The immunoprecipitates were resolved on 10% SDS polyacrylamide gels. For mass spectrometry analysis, gels were stained with Coomassie Blue for protein visualisation.
In-gel digestion
The band containing ATPsyn-β was excised and cut into 1 mm cubes for digestion with trypsin. Trypsin digestion of protein within gel slices and preparation of the samples for MS were performed as described previously [25, 26]. Three 10 fmol/µl peptides were added to serve as internal standards (Bradykinin Fragment 2-9, B1901, β-Sheet Breaker Peptide, S7563 and Anaphylatoxin C3a fragment, A8651; Sigma Chemical Co., St Louis, MO, USA).
Mass spectrometry
HPLC-electron spray ionisation (ESI)-MSn was performed on a hybrid linear ion trap (LTQ)-Fourier transform ion cyclotron resonance (FTICR) mass spectrometer (LTQ FT, Thermo Fisher; San Jose, CA, USA) fitted with a PicoView nanospray source (New Objective, Woburn, MA, USA) as described previously [25, 26].
A ‘top-ten’ data-dependent MS/MS approach was used to identify ATPsyn-β peptides and to obtain their HPLC retention times. In a top-ten scan protocol, a full scan spectrum (survey scan) is acquired followed by collision-induced dissociation (CID) mass spectra of the ten most abundant ions in the survey scan. Thus, a large number of tandem mass spectra are acquired during the course of each analysis. For the experiments used in the current report, the survey scan was acquired using the FTICR mass analyser in order to obtain high mass accuracy data. From this initial analysis, a list of potential phosphorylated peptides was generated based on detected serine/threonine-containing peptides of ATPsyn-β. For localisation of sites of phosphorylation, a scan protocol of one survey scan (FTICR), followed by six targeted MS/MS scans (CID spectra of specified m/z values that were acquired using the LTQ mass analyser). Ions in the target list are fragmented in the whole HPLC-ESI-MS/MS run (90 min) to ensure the detection of the phosphopeptides. Seven potential phosphopeptides were successfully monitored this way in a single run. Although di-phosphorylation and tri-phosphorylation were considered, only monophosphorylation was detected. For quantification, the following multi-segment strategy was employed: one survey scan, followed by six targeted CID scans. In order to assess the relative quantities of a large number of phosphopeptides in each experiment and yet still maintain acceptable mass analysis cycle times, the targeted m/z values were grouped into segments based on their expected HPLC retention times. Included in the target list were the 2+ charge states of six representative peptides of ATPsyn-β selected from the prominent ions reproducibly observed in the top-ten data-dependent tandem-MS analysis (see Electronic supplementary material [ESM] Table 1). These six representative peptides were used as internal standards for the amount of total ATPsyn-β protein content in the samples and used for relative quantification of the phosphopeptides. They were selected according to the following criteria: (1) detected by HPLC-ESI-MS with high intensity among ATPsyn-β peptides; (2) no missed cleavage observed; (3) no methionine in the sequence to avoid variability due to methionine oxidisation and no N-terminal Gln residues. The 2+ or 3+ ions of the phosphopeptides of interest were placed on the target list. The 2+ ions of the standard peptides were also placed on the target list. The CID spectra were acquired in the centroid mode. Experiments were performed to validate the MS peak area-based phosphorylation quantification approach (ESM Fig. 1).
Tandem mass spectra were extracted from Xcalibur ‘RAW’ files and charge states were assigned using the Extract_MSN script that is a component of Xcalibur 2.0 SR2 (Thermo Fisher; San Jose, CA, USA). The fragment mass spectra were then searched against the human SwissProt_v52.2 database (16,135 entries) using Mascot (Matrix Science, London, UK; version 2.1). The search variables that were used were: 10 ppm mass tolerance for precursor ion masses and 0.5 Da for product ion masses; digestion with trypsin; a maximum of two missed tryptic cleavages; variable modifications of oxidation of methionine and phosphorylation of serine, threonine and tyrosine. Cross-correlation of Mascot search results with X! Tandem was accomplished with Scaffold (version Scaffold-01_06_19; Proteome Software, Portland, OR, USA). Probability assessment of peptide assignments and protein identifications were made through use of Scaffold. Only peptides with ≥95% probability were considered.
Statistical analysis
Statistical analysis was performed using SSSP for Windows version 10.0. Results are given as means±SEM. Statistical evaluation of clinical and metabolic characteristics and OxPhos protein content was performed, where appropriate, by paired Student’s t test or one-way ANOVA using the Tukey’s post hoc testing. Differential phosphorylation of ATPsyn-β was assessed in eight experiments by calculating the relative changes between the lean healthy participants, the obese non-diabetic participants and the patients with type 2 diabetes before and after insulin stimulation within each experiment. Statistical comparison between groups was performed using the Kruskal–Wallis test (ANOVA) and the post-hoc Mann–Whitney U test. The effect of insulin within each group was assessed by the Wilcoxon signed-rank test. The relationships between continuous variables were examined by calculation of Pearson’s correlation coefficients. Differences between groups were considered statistically significant at p < 0.05.
Results
Clinical and metabolic characteristics
Patients with type 2 diabetes had significantly higher fasting levels of HbA1c, plasma glucose and serum insulin compared with both lean and obese individuals, whereas fasting plasma adiponectin and HDL-cholesterol concentrations were lower compared with lean individuals only (Table 1). Serum insulin levels were also significantly higher in obese than in lean individuals. There were no differences in fasting levels of plasma triacylglycerol or NEFA between the groups.
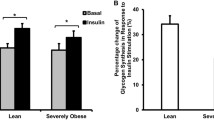
Basal rates of total glucose disposal, glucose oxidation, lipid oxidation and non-oxidative glucose metabolism were similar in the three groups. In the insulin-stimulated state during the euglycaemic–hyperinsulinaemic clamp studies, total glucose disposal, glucose oxidation and non-oxidative glucose metabolism were significantly reduced in type 2 diabetic patients compared with obese and lean controls, and in obese compared with lean individuals (Table 1). In the group of diabetic patients, the ability of insulin to suppress plasma levels of NEFA and rates of lipid oxidation was significantly impaired compared with both groups of non-diabetic individuals.
Identification of phosphorylation sites on human ATPsyn-β
Although we routinely obtained nearly 80% coverage of ATPsyn-β (Fig. 1a; ESM 1), only one phosphopeptide containing Thr213 was detected in a data-dependent analysis that did not include targeted MS/MS (see below). However, many ATPsyn-β peptides containing serine/threonine/tyrosine residues were detected in the data-dependent analysis. Since any peptide with serine/threonine/tyrosine residues has the potential to be phosphorylated, we predicted that low abundance phosphopeptides might be detected using a ‘targeted’ MS scan strategy based on addition of 80 Da (H3PO4–H2O) to the mass of each detected ATPsyn-β peptide that contained serine, threonine or tyrosine. Based on this, we constructed a hypothesis-driven strategy with various potential phosphopeptide m/z values in a target list, thereby greatly improving our ability to detect phosphopeptides [22–24]. This approach allowed us to detect six additional, distinct phosphorylation sites (Fig. 1b; Table 2). Among these, five sites have not been reported previously. From Scaffold analysis of the database search results, all phosphopeptides were identified at the 95% confidence level (ESM Tables 2, 3, 4, 5, 6, 7, 8 and 9). Three phosphorylated threonine residues, Thr213, Thr312 and Thr475 were detected. Thr213 is located in the nucleotide-binding region of the catalytic beta subunit of ATP synthase (Fig. 1b). Four phosphorylated tyrosine residues, Tyr230, Tyr269, Tyr361 and Tyr395 were also identified. Tandem mass spectra illustrating localisation of the sites of phosphorylation are given in Fig. 2 for the quantified phosphopeptides and in ESM Fig. 2 for the other phosphopeptides. NetworKIN analysis (www.networkin.info, accessed 25 June 2009) suggested protein kinase C, delta (PRKCD) and DNA-dependent protein kinase (PRKDC) as potential kinases for Thr312 phosphorylation. No kinases were predicted for the other sites.
Human ATPsyn-β is phosphorylated at multiple sites. Coverage map of peptides detected in tryptic digests of human muscle ATPsyn-β in three experiments using HPLC-nanospray-LTQ-FTICR MS/MS analysis (a). Detected peptides are shown in red, and phosphorylation sites are shown in green. The underlined sequence is the nucleotide-binding region, and the first 47 amino acids (italic) represent the mitochondrial precursor region. No phosphorylation sites were found in the precursor region (b). Among the three Pfam domains of ATPsyn-β (http://pfam.sanger.ac.uk, accessed 30 June 2009), no phosphorylation sites were found in the N-terminal domain (position 63–129). Six phosphorylation sites, Thr213, Tyr230, Tyr269, Thr312, Tyr361 and Tyr395, were found in the central domain (position 185–405), of which Thr213 is located in the nucleotide-binding region of ATPsyn-β. One phosphorylation site, Thr475, was found in the C-terminal domain (position 418–525)
Tandem mass spectra of quantified phosphopeptides. Tandem mass spectra of the phosphopeptides (a) pThr213 [MS/MS m/z 837.4 (3+)], (b) pTyr361 [MS/MS m/z 1265.6 (3+)] and (c) pThr475 [MS/MS m/z 707.0 (3+)] detected in tryptic digests of ATPsyn-β (encoded by ATP5B) immunoprecipitated from a human muscle biopsy. *Loss of H3PO4 (98 Da) from the indicated fragment. Mox, oxidised methionine
Effect of insulin resistance and insulin on phosphorylation of ATPsyn-β
As detailed in the ESM 1, we have validated the MS peak area-based approach for phosphorylation quantification. We applied this approach to investigate differential phosphorylation of ATPsyn-β among lean healthy, obese non-diabetic, and type 2 diabetic individuals under both basal conditions and after 4 h insulin infusion. ATPsyn-β was immunoprecipitated from 2 mg protein lysate from each muscle biopsy and subjected to HPLC-ESI-MS/MS. Basal phosphorylation of Thr213, which lies within the nucleotide-binding region of ATPsyn-β, was increased by 37% in obese non-diabetic participants (p = 0.003) and by 22% in patients with type 2 diabetes (p = 0.010) compared with lean individuals (ANOVA; p = 0.003; Fig. 3). Basal Tyr361 phosphorylation was increased by 32% in obese non-diabetic individuals (p = 0.028) and patients with type 2 diabetes (p = 0.021) compared with lean healthy controls (ANOVA; p = 0.031). No differences in basal Thr475 phosphorylation were observed between the groups. Interestingly, physiological hyperinsulinaemia for 4 h increased Tyr361 phosphorylation 46% in lean healthy individuals (p = 0.012), while a 24% decrease was seen in obese individuals (p = 0.036), and no change in patients with type 2 diabetes (Fig. 4). Insulin had no effect on Thr213 or Thr475 phosphorylation in any group.
Increased basal phosphorylation of ATPsyn-β at Thr213 and Tyr361 in insulin-resistant muscle. Site-specific phosphorylation of ATPsyn-β in skeletal muscle biopsies obtained from lean healthy, obese non-diabetic and type 2 diabetic individuals (n = 8 in each group) under basal conditions was quantified using targeted nanospray tandem mass spectrometry (see ESM 1). a Phosphorylation at Thr213 in the nucleotide-binding region; b phosphorylation at Tyr361 in the central domain; and c phosphorylation at Thr475 in the C-terminal domain of ATPsyn-β. Data are expressed as mean±SEM. **p ≤ 0.01 and † p < 0.03 vs lean controls
Insulin increases phosphorylation of ATPsyn-β at Tyr361 in healthy muscle. The effect of 4 h physiological hyperinsulinaemia (% change) on the site-specific phosphorylation of ATPsyn-β in skeletal muscle biopsies obtained from lean healthy and obese non-diabetic individuals, and patients with type 2 diabetes (n = 8 in each group) before and after an euglycaemic–hyperinsulinaemic clamp were assessed by targeted nanospray tandem mass spectrometry (see ESM 1). a Insulin-mediated change (%) in the phosphorylation at Thr213 in the nucleotide-binding region; b phosphorylation at Tyr361 in the central domain; and c phosphorylation at Thr475 in the C-terminal domain of ATPsyn-β. Data are expressed as mean±SEM. † p = 0.012 and ‡ p = 0.036
Coordinated reduction in abundance of ATPsyn-β and subunits of complex I, II, III and V
Immunoblotting was performed to examine whether changes in protein level of ATPsyn-β in obesity and type 2 diabetes might be coordinated with alterations in protein levels of other subunits in respiratory complexes I–V (Fig. 5). ATPsyn-β protein showed a significant trend to decrease from lean, to obese and type 2 diabetic individuals (ANOVA; p = 0.023). Similar significant decreasing trends were observed for protein levels of complex I (ND6; ANOVA; p = 0.004), complex II (SDHA; ANOVA; p = 0.003), complex III (UQCRC1; ANOVA; p = 0.021) and ATP synthase alpha (ATP5A1; ANOVA; p = 0.003). Post hoc analysis showed that there were no significant differences between obese non-diabetic individuals and patients with type 2 diabetes, whereas most of the subunits were significantly decreased in obese and diabetic individuals compared with lean controls. Correlation analysis showed that protein level of ATPsyn-β was significantly associated with the levels of proteins ND6, SDHA, UQCRC1 and ATP5A1 (r = 0.63–0.77, all p < 0.001), providing further evidence for a coordinated transcriptional and translational regulation of these OxPhos components.
Coordinated downregulation of ATPsyn-β and other OxPhos proteins in insulin-resistant muscle. Immunoblotting of OxPhos proteins in skeletal muscle biopsies obtained from lean healthy, obese non-diabetic and type 2 diabetic individuals under basal conditions (n = 10 in each group). Protein levels of: a complex I subunit, ND6 (a); a complex II subunit, SDHA (b); a complex III subunit, UQCRC1 (c); and two complex V subunits, ATP5B (e) and ATP5A1 (d). f Representative blots of the OxPhos proteins and actin in muscle from lean, obese and type 2 diabetic participants. The protein content is shown as arbitrary units and data are expressed as mean ± SEM. *p = 0.05, **p = 0.01 and † p = 0.06 vs lean controls. Cx, muscle from an internal control used to normalise for variation in signal intensities across gels; Le, lean; Ob, obese; T2D, type 2 diabetic
Discussion
Using our targeted MS/MS based approach for mapping of phosphorylation sites, we identified seven phosphorylation sites on human muscle ATPsyn-β. Quantification revealed a ∼30% increase in the basal phosphorylation of ATPsyn-β at Tyr361 and Thr213 in obesity and type 2 diabetes, and demonstrated a ∼50% increase in phosphorylation at Tyr361 in response to insulin in lean healthy individuals. Moreover, we found a coordinated downregulation of muscle ATPsyn-β and other OxPhos proteins in obesity and type 2 diabetes. Our results indicate that phosphorylation of human muscle ATPsyn-β may be an important regulatory mechanism of electron-coupled ATP synthesis, and that disturbances in this regulation in addition to reduced content of OxPhos proteins may contribute to the pathogenesis of insulin resistance and type 2 diabetes.
The F1 portion of the ATP synthase complex is located in the mitochondrial matrix and consists of five subunits with the stoichiometry α3β3γδε. The beta subunits contain the catalytic sites essential for the binding of ADP and Pi and synthesis of ATP. Here, we demonstrate that human ATPsyn-β is phosphorylated at multiple specific sites in vivo. These results extend our previous work [21] and that of several other studies that have used various phospholabelling techniques or phosphopeptide identification by MS, which have provided indirect evidence for serine/threonine/tyrosine phosphorylation of ATPsyn-β in yeast, plants and mammalian tissues and cell lines [27–35].
The present study provides the first mapping of phosphorylation sites on human ATPsyn-β. Recently, five specific phosphorylation sites were assigned to ATPsyn-β in rabbit heart muscle using phosphopeptide enrichment and MS/MS technology [27]. However, although all sites were targeted in our analyses, of these we could only confirm the phosphorylation at Thr312 in human skeletal muscle. In addition to Thr213 [21] and Thr312 [27], we identified five novel phosphorylation sites at Thr475, Tyr230, Tyr269, Tyr361 and Tyr395, respectively. These data suggest the possibility that reversible phosphorylation of human ATPsyn-β at specific serine/threonine/tyrosine residues regulates the catalytic activity of ATP synthase, and hence ATP synthesis. Moreover, phosphorylation of ATPsyn-β may play a role for its translocation and import into the mitochondrial matrix, assembly of the ATP synthase complex, dimerisation [32] and formation of OxPhos supercomplexes, stability and degradation [19]. Further studies are needed to address the precise functional consequences of these phosphorylations, and the mitochondrial kinases and phosphatases involved.
The present data represent, to our knowledge, the first evidence that a mitochondrial OxPhos protein may be differently phosphorylated in insulin resistant human muscle in vivo. The magnitude (∼30%) of the increase in basal phosphorylation of ATPsyn-β at Thr213 and Tyr361 corresponds to other defects in muscle in type 2 diabetes, e.g. impaired insulin-stimulated glycogen synthase activity [4, 5]. There are experimental data indicating that phosphorylation of ATPsyn-β, in particular at serine residues, lowers ATP synthase activity. Consumption of soy proteins, which have been linked to a decreased risk of type 2 diabetes and certain types of cancer, caused decreased phosphorylation of ATPsyn-β and increased ATP synthase activity in the liver of rats [31]. In rat renal cells, PRKCA-mediated serine phosphorylation of ATPsyn-β was associated with a decreased ATP synthase activity in vitro [30]. Moreover, in plant mitochondria, the binding of recombinant 14-3-3 protein to phosphorylated serine/threonine-residues on ATPsyn-β inhibited ATP synthase activity [36]. Of interest, Thr213 is located within the nucleotide-binding region, indicating that phosphorylation at this site may interfere with the binding of ADP and Pi to the catalytic sites on ATPsyn-β, and therefore could interfere with ATP synthesis. This suggests that increased phosphorylation at Thr213 could potentially contribute to impaired mitochondrial ATP synthesis in obesity and type 2 diabetes. However, further studies are warranted to investigate whether the observed changes in phosphorylation are paralleled by changes in ATP synthase activity and OxPhos in insulin resistant human skeletal muscle.
In this study we also demonstrated that at least one phosphorylation site on ATPsyn-β is altered by infusion of insulin. The ∼50% increase in phosphorylation of Tyr361 in lean individuals is a physiological relevant increase, and indicates that it may affect the rate of ATP production. A number of tyrosine-specific kinases and phosphatases have been localised to mitochondria, and a role for tyrosine phosphorylation in the regulation of mitochondrial function has gained increasing attention [19, 37]. Tyrosine phosphorylation of two subunits of cytochrome c oxidase (CcO) has been shown to either inactivate or activate complex IV activity [19]. Therefore, it is difficult to predict how insulin-induced Tyr361 phosphorylation of ATPsyn-β might affect ATP synthase activity in human skeletal muscle. There is evidence that TNF-α stimulation induces tyrosine phosphorylation of ATPsyn-β in murine fibroblasts [34], and that TNF-α inhibits OxPhos through tyrosine phosphorylation of subunit I of CcO in bovine and murine cells [38]. For a number of reasons, however, it is tempting to speculate that insulin-mediated Tyr361 phosphorylation of ATPsyn-β might stimulate OxPhos. First, 2 h of physiological hyperinsulinaemia increases muscle ATP production in healthy individuals, as evaluated by nuclear magnetic resonance spectroscopy [39, 40]. The same effect is seen in isolated muscle mitochondria obtained from healthy individuals after 4 h of insulin infusion, suggesting that this response it not solely due to increased substrate fluxes [41]. Analogous to the diminished effect of insulin on Tyr361 phosphorylation in insulin-resistant individuals in our study, there is an impaired effect of insulin on ATP production in patients with type 2 diabetes and high-risk individuals in these studies [39–41]. Second, the increased basal Tyr361 phosphorylation of ATPsyn-β could be a consequence of chronic hyperinsulinaemia and, as seen with increased basal tyrosine phosphorylation of the insulin receptor and IRS-1 in insulin-resistant muscle [4, 42], attenuate the ability to further increase Tyr361 phosphorylation in response to insulin. This could, however, also be a saturable effect, which does not necessarily imply a lack of insulin action. Taken together, our data suggest the possibility that insulin may regulate mitochondrial function through cell signalling in human muscle in vivo, and that these effects may be diminished in obesity and type 2 diabetes concomitant with reduced insulin-mediated glucose disposal. Additional studies are warranted to establish the precise role of these phosphorylation sites, as well as how they might affect mitochondrial function and insulin sensitivity in human muscle.
The finding that a ∼30–50% lower protein content of ATPsyn-β was coordinated with a similar decrease in other both nuclear- and mitochondrial-encoded OxPhos proteins clearly supports microarray-based studies of gene expression [15–17] and other reports that have provided evidence for a reduced mitochondrial content in skeletal muscle in type 2 diabetes and obesity [9, 10, 13, 43]. Together with the altered phosphorylation of ATPsyn-β, our data extend previous observations by showing that mitochondrial dysfunction coexists at the transcriptional, translational and post-translational level in insulin-resistant human muscle. Based on the data provided in the present study, we cannot exclude the possibility that these findings might be completely unrelated. Nevertheless, the finding of abnormal phosphorylation of ATPsyn-β at specific sites in obesity and type 2 diabetes could potentially explain the impaired respiration per mitochondrion, as recently reported [11, 12]. Based on previous studies of type 2 diabetic patients and matched controls, there is no clear evidence that an intrinsic defect in mitochondrial function is located exclusively to ATP synthase. In fact, it seems to be substrate specific and also present in uncoupled mitochondria, suggesting that a defect in complex I–IV contributes to mitochondrial dysfunction [11, 12]. This suggests the possibility that insulin and insulin resistance affects other subunits in complex I–IV and ATP synthase by phosphorylation or other post-translational modifications. Moreover, the changes in phosphorylation of ATPsyn-β and protein levels of ATPsyn-β and other OxPhos proteins were similar in type 2 diabetic and non-diabetic obese individuals. This strongly indicates that other factors contribute to the lower insulin-stimulated glucose metabolism in type 2 diabetes compared with obesity alone. There is evidence that a lower protein content of ATPsyn-β may affect beta cell and adipocyte function [44–47], and it is also a hallmark of most human carcinomas [48, 49]. Thus, aberrant phosphorylation of ATPsyn-β may have putative consequences in other tissues in insulin-resistant individuals as well as in other disorders.
In summary, our data demonstrate that a hypothesis-driven MS/MS-based approach for the identification and quantification of phosphorylated serine/threonine/tyrosine residues is well suited for the analysis of proteins from small human tissue samples. We identified seven phosphorylation sites on human ATPsyn-β in skeletal muscle. Of these, five phosphorylation sites are novel. Quantification allowed us to discover increased basal Tyr361 and Thr213 phosphorylation of ATPsyn-β in obesity and type 2 diabetes. The ability of physiological hyperinsulinaemia to increase Tyr361 phosphorylation in lean healthy individuals was absent in obesity and type 2 diabetes. These abnormalities in obesity and type 2 diabetes were seen together with impaired insulin-stimulated glucose disposal and a coordinated downregulation of OxPhos proteins. Our data indicate a role for perturbations in the phosphorylation of ATPsyn-β in mitochondrial dysfunction in skeletal muscle insulin resistance. Future characterisation of the phosphorylation of ATPsyn-β may offer novel targets of treatment and improved diagnosis and prognosis of human diseases characterised by mitochondrial dysfunction, such as diabetes.
Abbreviations
- ATPsyn-β:
-
ATP synthase beta subunit
- ATP5A1:
-
ATP synthase alpha
- ATP5B:
-
ATP synthase beta
- CID:
-
Collision-induced dissociation
- FTICR:
-
Fourier transform ion cyclotron resonance
- ND6:
-
NADH-ubiquinone oxidoreductase chain 6
- SDHA:
-
Succinate dehydrogenase complex, subunit A, flavoprotein
- TBST:
-
TRIS-buffered saline plus 0.05% Tween 20
- UQCRC1:
-
Ubiquinol-cytochrome c reductase core protein I
References
Wallace DC (2005) A mitochondrial paradigm of metabolic and degenerative diseases, aging, and cancer: a dawn for evolutionary medicine. Annu Rev Genet 39:359–407
Mottram RF (1955) The oxygen consumption of human skeletal muscle in vivo. J Physiol 128:268–276
Shulman GI, Rothman DL, Jue T, Stein P, DeFronzo RA, Shulman RG (1990) Quantitation of muscle glycogen synthesis in normal subjects and subjects with non-insulin-dependent diabetes by 13C nuclear magnetic resonance spectroscopy. N Engl J Med 322:223–228
Cusi K, Maezono K, Osman A et al (2000) Insulin resistance differentially affects the PI 3-kinase- and MAP kinase-mediated signaling in human muscle. J Clin Invest 105:311–320
Højlund K, Staehr P, Hansen BF et al (2003) Increased phosphorylation of skeletal muscle glycogen synthase at NH2-terminal sites during physiological hyperinsulinemia in type 2 diabetes. Diabetes 52:1393–1402
Højlund K, Frystyk J, Levin K, Flyvbjerg A, Wojtaszewski JF, Beck-Nielsen H (2006) Reduced plasma adiponectin concentrations may contribute to impaired insulin activation of glycogen synthase in skeletal muscle of patients with type 2 diabetes. Diabetologia 49:1283–1291
Højlund K, Beck-Nielsen H (2006) Impaired glycogen synthase activity and mitochondrial dysfunction in skeletal muscle. Markers or mediators of insulin resistance in type 2 diabetes. Curr Diabetes Rev 2:375–395
Levin K, Daa Schroeder H, Alford FP, Beck-Nielsen H (2001) Morphometric documentation of abnormal intramyocellular fat storage and reduced glycogen in obese patients with Type II diabetes. Diabetologia 44:824–833
Højlund K, Mogensen M, Sahlin K, Beck-Nielsen H (2008) Mitochondrial dysfunction in diabetes and obesity. Endocrinol Metab Clin North Am 37:713–731
Kelley DE, He J, Menshikova EV, Ritov VB (2002) Dysfunction of mitochondria in human skeletal muscle in type 2 diabetes. Diabetes 51:2944–2950
Mogensen M, Sahlin K, Fernström M et al (2007) Mitochondrial respiration is decreased in skeletal muscle of patients with type 2 diabetes. Diabetes 56:1592–1599
Phielix E, Schrauwen-Hinderling VB, Mensink M et al (2008) Lower intrinsic ADP-stimulated mitochondrial respiration underlies in vivo mitochondrial dysfunction in muscle of male type 2 diabetic patients. Diabetes 57:2943–2949
Ritov VB, Menshikova EV, He J, Ferrell RE, Goodpaster BH, Kelley DE (2005) Deficiency of subsarcolemmal mitochondria in obesity and type 2 diabetes. Diabetes 54:8–14
Lowell BB, Shulman GI (2005) Mitochondrial dysfunction and type 2 diabetes. Science 307:384–387
Mootha VK, Lindgren CM, Eriksson KF et al (2003) PGC-1alpha-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat Genet 34:267–273
Patti ME, Butte AJ, Crunkhorn S et al (2003) Coordinated reduction of genes of oxidative metabolism in humans with insulin resistance and diabetes: Potential role of PGC1 and NRF1. Proc Natl Acad Sci USA 100:8466–8471
Skov V, Glintborg D, Knudsen S et al (2007) Reduced expression of nuclear-encoded genes involved in mitochondrial oxidative metabolism in skeletal muscle of insulin-resistant women with polycystic ovary syndrome. Diabetes 56:2349–2355
Hunter T (2000) Signaling—2000 and beyond. Cell 100:113–127
Hüttemann M, Lee I, Samavati L, Yu H, Doan JW (2007) Regulation of mitochondrial oxidative phosphorylation through cell signaling. Biochim Biophys Acta 1773:1701–1720
Pagliarini DJ, Dixon JE (2006) Mitochondrial modulation: reversible phosphorylation takes center stage? Trends Biochem Sci 31:26–34
Højlund K, Wrzesinski K, Larsen PM et al (2003) Proteome analysis reveals phosphorylation of ATP synthase beta-subunit in human skeletal muscle and proteins with potential roles in type 2 diabetes. J Biol Chem 278:10436–10442
Yi Z, Luo M, Carroll CA, Weintraub ST, Mandarino LJ (2005) Identification of phosphorylation sites in insulin receptor substrate-1 by hypothesis-driven high-performance liquid chromatography-electrospray ionization tandem mass spectrometry. Anal Chem 77:5693–5699
Yi Z, Luo M, Mandarino LJ, Reyna SM, Carroll CA, Weintraub ST (2006) Quantification of phosphorylation of insulin receptor substrate-1 by HPLC-ESI-MS/MS. J. Am Soc Mass Spectrom 17:562–567
Yi Z, Langlais P, de Filippis EA et al (2007) Global assessment of regulation of phosphorylation of insulin receptor substrate-1 by insulin in vivo in human muscle. Diabetes 56:1508–1516
Højlund K, Yi Z, Hwang H, Bowen B et al (2008) Characterization of the human skeletal muscle proteome by one-dimensional gel electrophoresis and HPLC-ESI-MS/MS. Mol Cell Proteomics 7:257–267
Yi Z, Bowen BP, Hwang H et al (2008) Global relationship between the proteome and transcriptome of human skeletal muscle. J Proteome Res 7:3230–3241
Arrell DK, Elliott ST, Kane LA et al (2006) Proteomic analysis of pharmacological preconditioning: novel protein targets converge to mitochondrial metabolism pathways. Circ Res 99:706–714
del Riego G, Casano LM, Martín M, Sabater B (2006) Multiple phosphorylation sites in the beta subunit of thylakoid ATP synthase. Photosynth Res 89:11–18
Hopper RK, Carroll S, Aponte AM, Johnson DT et al (2006) Mitochondrial matrix phosphoproteome: effect of extra mitochondrial calcium. Biochemistry 45:2524–2536
Liu X, Godwin ML, Nowak G (2004) Protein kinase C-alpha inhibits the repair of oxidative phosphorylation after S-(1, 2-dichlorovinyl)-l-cysteine injury in renal cells. Am J Physiol Renal Physiol 287:F64–73
Mei J, Wood C, L’abbé MR et al (2007) Consumption of soy protein isolate modulates the phosphorylation status of hepatic ATPase/ATP synthase beta protein and increases ATPase activity in rats. J Nutr 137:2029–2035
Reinders J, Wagner K, Zahedi RP et al (2007) Profiling phosphoproteins of yeast mitochondria reveals a role of phosphorylation in assembly of the ATP synthase. Mol Cell Proteomics 6:1896–1906
Schulenberg B, Aggeler R, Beechem JM, Capaldi RA, Patton WF (2003) Analysis of steady-state protein phosphorylation in mitochondria using a novel fluorescent phosphosensor dye. J Biol Chem 278:27251–27255
Yanagida M, Miura Y, Yagasaki K, Taoka M, Isobe T, Takahashi N (2000) Matrix assisted laser desorption/ionization-time of flight-mass spectrometry analysis of proteins detected by anti-phosphotyrosine antibody on two-dimensional-gels of fibroblast cell lysates after tumor necrosis factor-alpha stimulation. Electrophoresis 21:1890–1898
Aponte AM, Phillips D, Hopper RK et al (2009) Use of (32)P to study dynamics of the mitochondrial phosphoproteome. J Proteome Res 8:2679–2695
Bunney TD, van Walraven HS, de Boer AH (2001) 14-3-3 protein is a regulator of the mitochondrial and chloroplast ATP synthase. Proc Natl Acad Sci USA 98:4249–4254
Salvi M, Brunati AM, Toninello A (2005) Tyrosine phosphorylation in mitochondria: a new frontier in mitochondrial signaling. Free Radic Biol Med 38:1267–1277
Samavati L, Lee I, Mathes I, Lottspeich F, Hüttemann M (2008) Tumor necrosis factor alpha inhibits oxidative phosphorylation through tyrosine phosphorylation at subunit I of cytochrome c oxidase. J Biol Chem 283:21134–21144
Petersen KF, Dufour S, Shulman GI (2005) Decreased insulin-stimulated ATP synthesis and phosphate transport in muscle of insulin-resistant offspring of type 2 diabetic parents. PLoS Med 2:e233
Szendroedi J, Schmid AI, Chmelik M et al (2007) Muscle mitochondrial ATP synthesis and glucose transport/phosphorylation in type 2 diabetes. PLoS Med 4:e154
Stump CS, Short KR, Bigelow ML, Schimke JM, Nair KS (2003) Effect of insulin on human skeletal muscle mitochondrial ATP production, protein synthesis, and mRNA transcripts. Proc Natl Acad Sci USA 100:7996–8001
Miyazaki Y, He H, Mandarino LJ, DeFronzo RA (2003) Rosiglitazone improves downstream insulin receptor signaling in type 2 diabetic patients. Diabetes 52:1943–1950
Rabøl R, Højbjerg PM, Almdal T et al (2009) Effect of hyperglycemia on mitochondrial respiration in type 2 diabetes. J Clin Endocrinol Metab 94:1372–1378
Maechler P, Wollheim CB (2001) Mitochondrial function in normal and diabetic beta-cells. Nature 414:807–812
Koh EH, Park JY, Park HS et al (2007) Essential role of mitochondrial function in adiponectin synthesis in adipocytes. Diabetes 56:2973–2981
Yang J, Wong RK, Wang X et al (2004) Leucine culture reveals that ATP synthase functions as a fuel sensor in pancreatic beta-cells. J Biol Chem 279:53915–53923
Choo HJ, Kim JH, Kwon OB et al (2006) Mitochondria are impaired in the adipocytes of type 2 diabetic mice. Diabetologia 49:784–791
López-Ríos F, Sánchez-Aragó M, García-García E et al (2007) Loss of the mitochondrial bioenergetic capacity underlies the glucose avidity of carcinomas. Cancer Res 67:9013–9017
Cuezva JM, Krajewska M, de Heredia ML et al (2002) The bioenergetic signature of cancer: a marker of tumor progression. Cancer Res 62:6674–6681
Acknowledgements
K. Højlund was funded by grants from the Danish Medical Research Council, the Novo Nordisk Research Foundation and the Danish Diabetes Association.
Duality of interest
The authors declare that there is no duality of interest associated with this manuscript.
Author information
Authors and Affiliations
Corresponding authors
Additional information
K. Højlund, Z. Yi and N. Lefort contributed equally to this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 24 kb)
ESM Table 1
(PDF 14 kb)
ESM Table 2
(PDF 15 kb)
ESM Table 3
(PDF 13 kb)
ESM Table 4
(PDF 9 kb)
ESM Table 5
(PDF 30 kb)
ESM Table 6
(PDF 29 kb)
ESM Table 7
(PDF 11 kb)
ESM Table 8
(PDF 28 kb)
ESM Table 9
(PDF 8 kb)
Fig. 1
Linearity of the MS peak area based quantification approach. In a serial dilution of immunopurified ATPsyn-β from human skeletal muscle the mean of the peak areas for the six reference peptides, MSrep6, showed an excellent linear response to the ATPsyn-β protein loading (a). The peak areas of three fragment ions, MSphos, for the phosphopeptide containing Thr475 also showed a good linear response to protein loading (b) (PDF 24 kb)
Fig. 2
Tandem mass spectra of 4 additional identified phosphopeptides. Tandem mass spectra of the phosphopeptides (a) pThr312, (b) pTyr230, (c) pTyr269 and (d) pTyr395 detected in tryptic digests of ATPsyn-β (ATP5B) immunoprecipitated from a human muscle biopsy. For pThr312 the b5, b6 and y9, y10 and y11 are unique. For pTyr230 only b4 and y9 can distinguish pTyr230 from pSer231. Despite detecting y9, we cannot completely rule out pSer231. For pTyr395 y5, y7, y8, y9, y10 and y11 are unique. *Loss of H3PO4, §loss of SOCH4, #loss of NH3, and &loss of H2O from the indicated fragments (PDF 630 kb)
Rights and permissions
About this article
Cite this article
Højlund, K., Yi, Z., Lefort, N. et al. Human ATP synthase beta is phosphorylated at multiple sites and shows abnormal phosphorylation at specific sites in insulin-resistant muscle. Diabetologia 53, 541–551 (2010). https://doi.org/10.1007/s00125-009-1624-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-009-1624-0