Abstract
Aims/hypothesis
The pathogenesis of diabetes and the success of islet transplantation depend on the control of pancreatic beta cell fate. The Notch signalling pathway is essential for normal prenatal pancreatic development, but the presence and function of this gene network in adult islets has received much less attention.
Methods
The presence of Notch signalling components was assessed in vitro using RT-PCR, western blotting and immunofluorescence. The functional consequences of altering Notch signalling on insulin secretion and programmed cell death were examined.
Results
Adult mouse islets, human islets and mouse insulinoma MIN6 cells possess key components of the Notch pathway. RT-PCR, western blotting and immunofluorescence indicated that the Notch target gene, neurogenin3 (Ngn3, also known as Neurog3), is also present in adult islet cells. Inhibiting Notch signalling with N-[N-(3,5-difluorophenacetyl-l-alanyl)]-S-phenylglycine t-butyl ester (DAPT) increased Ngn3 mRNA expression and protein levels in adult islets. The activated notch homologue 1 (NOTCH1) protein level was decreased upon serum withdrawal, as well as after treatment with a phosphatidylinositol 3-kinase inhibitor, or hydroxy-2-naphthalenylmethylphosphonic acid, an insulin receptor inhibitor. While islets cultured in DAPT did not exhibit defects in insulin secretion, indicating that differentiation is unaltered, inhibiting gamma-secretase-dependent Notch activation led to a dose-dependent increase in caspase-3-dependent apoptosis in both MIN6 cells and human islets. Conversely, gamma-secretase overactivity resulted in an accumulation of cleaved NOTCH1 and protection from apoptosis.
Conclusions/interpretation
Together these results show that the Notch/Ngn3 signalling network is intact and functional in adult islets. This pathway represents an attractive target for modulating beta cell fate in diabetes, islet transplantation and efforts to derive beta cell surrogates in vitro.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Notch signalling plays a critical role in many developmental processes, influencing differentiation, proliferation and apoptosis [1, 2]. The Notch pathway includes a conserved family of transmembrane receptors (NOTCH1–4) that interact with a number of specific ligands (the delta-like family; jagged 1 and 2) to regulate cell fate [1, 3]. Interaction between Notch receptors and these ligands leads to intracellular cleavage of Notch receptors by the gamma-secretase complex [4]. The cleaved Notch intracellular domain traffics to the nucleus where it interacts with transcription factors. This activates the expression of hairy and enhancer of split (e.g. Hes1) transcriptional repressors, which in turn repress expression of downstream target genes such as neurogenin 3 (Ngn3, also known as Neurog3) [3].
Several studies have illustrated the importance of the Notch pathway in the control of cell fate during development of both pancreatic endocrine and exocrine tissue [5–8]. It is well documented that the transient expression of the Notch target gene Ngn3 defines the endocrine precursor pool in prenatal development [5, 8, 9]. To the best of our knowledge detailed studies on the expression and regulation of Ngn3 in the adult pancreas have not been reported. While there is strong evidence that Notch regulates beta cell mass during prenatal development by controlling the number of Ngn3-positive endocrine precursor cells, little is known about the activity of this network in adult islets.
Although primarily involved in pancreatic embryogenesis, it is possible that the Notch pathway may retain a small but significant level of activity in the adult and may play a role in islet cell turnover, maintenance or gene expression. Indeed, Notch has been shown to regulate apoptosis in a number of mature cell types, including thymocytes, as well as some cancers [10–12]. Furthermore, Notch pathway components are re-expressed in exocrine cells after experimentally induced pancreas injury [13, 14]. Interestingly, a recent study demonstrated the ability of the cytokine IL-1β to rapidly increase the mRNA expression and protein levels of members of the Notch pathway in rat beta cells [15]. The aim of the present study was to determine whether the Notch pathway remains functional in adult islets. Our results show that Notch receptors, their ligands and their target genes are expressed in adult islets. We also find that inhibition of growth factor signalling reduced Notch activation in mouse and human islets. In addition, blocking the Notch pathway activity induced apoptosis in primary beta cells. We present evidence that this key ‘developmental’ pathway remains intact in adult islets. Our results suggest that the Notch pathway represents an attractive target in efforts to increase beta cell survival in diabetes or islet transplantation.
Methods
Reagents
The gamma-secretase inhibitor, N-[N-(3,5-difluorophenacetyl-l-alanyl)]-S-phenylglycine t-butyl ester (DAPT), was purchased from Sigma (St Louis, MO, USA); LY294002 and hydroxy-2-naphthalenylmethylphosphonic acid (HNMPA) were obtained from Calbiochem (La Jolla, CA, USA). All other reagents were from Sigma.
Cell culture
Primary islets were isolated from 3- to 6-month-old C57Bl6/J mice (Jax, Bar Harbor, MA, USA) using collagenase and filtration as described previously [16]. Human islets were provided with consent through the auspices of the Michael Smith Foundation for Health Research Centre (MSFHR) for Human Islet Transplant and Beta Cell Regeneration (Vancouver, BC, Canada), directed by G. Warnock. Our preparations of human islets were from donors aged 49–65 years (from both sexes), and cold ischaemia time was never more than 12 h. The human islet preparations typically stained >75% positive for beta cells using dithizone. After islet dispersion and culture >50% of the round ‘endocrine’ cells stained robustly for insulin (data not shown). Human and mouse islets were cultured in 35 × 10 mm Nunc suspension dishes (Nalge, Rochester, NY, USA) at 37°C and 5% CO2 in RPMI 1640 medium with glucose adjusted as indicated in each experiment. The medium was supplemented with 100 IU/ml penicillin–100 μg/ml streptomycin (10%, v/v) FCS and brought to pH 7.4 with NaOH [17, 18]. For experiments designed to test the effects of exogenous insulin, larger volumes of medium were used to reduce insulin build-up as in our previous studies [19]. The mouse-derived MIN6 cells were maintained and cultured as described [20] in DMEM medium containing 25 mmol/l glucose or 5 mmol/l glucose (as indicated), 10% FCS and 100 IU/ml penicillin–100 μg/ml streptomycin (Invitrogen, Burlington, ON, Canada).
Gene expression analysis
Total RNA was isolated from mouse primary islets or cells using the Qiagen RNeasy kit (Mississauga, ON, Canada). cDNA was reversed transcribed using Superscript III (Invitrogen). Primer sequences used for Notch1, -2, -3 and -4, Jag1 and -2, delta-like 1, 2, 3 and 4 (Dll1, -2, -3 and -4) and Hes1 have been described [21]. Primers for Ngn3 were: forward TGC AGC CAC ATC AAA CTC TC; reverse GGT CAC CCT GGA AAA AGT GA, and the expected product size was 139 bp. Primers for β-actin were: forward GGA AAT CGT GCG TGA CAT CAA AG; reverse ATC TGC TGG AAG GTG GAC AGT GAG, and the expected product size was 430 bp.
Analysis of programmed cell death
Islet apoptosis was measured using a previously described variation of a PCR-enhanced DNA-laddering protocol modified for use in small numbers of islets [17]. In order to compare data from separate gels, band intensity was normalised to the average of the control cultures. Cell death was also detected using propidium iodide (Sigma-Aldrich, Oakville, ON, Canada; 500 ng/ml) and Hoechst 33258 (Invitrogen; 500 ng/ml) after 30 min incubation with the dyes. Cells were visualised using an Axiovert 200M microscope (Zeiss, Thornwood, NY, USA) and imaged using a Coolsnap HQII camera (Photometric, Tucson, AZ, USA). Semi-automated counting of propidium iodide- and Hoechst-positive cells was performed using SlideBook software (Intelligent Imaging Innovations, Boulder, CO, USA).
Insulin secretion analysis by islet perifusion
After overnight culture with or without 10 μmol/l DAPT, groups of 100 size-matched islets were suspended with Cytodex microcarrier beads (Sigma-Aldrich) in the 300 μl plastic chambers of an Acusyst-S perifusion apparatus (Endotronics, Minneapolis, MN, USA). Under temperature- and CO2-controlled conditions, the islets were perifused at 0.5 ml/min with a Krebs–Ringer buffer containing (in millimole per litre) 129 NaCl, 5 NaHCO3, 4.8 KCl, 2.5 CaCl2, 1.2 MgSO4, 1.2 KH2PO4, 10 HEPES, 3 glucose. This buffer included 5 g/l RIA-grade BSA (Sigma). Prior to sample collection, islets were equilibrated under basal (3 mmol/l glucose) conditions for 1 h. Insulin secretion was measured by RIA (Rat Insulin RIA Kit; Linco Research, St Charles, MO, USA).
Immunoblot analysis
Western blots were performed according to standard methods. Briefly, human and mouse islets (cultured in groups of 140 in 15 ml of medium) were washed twice with 1× PBS before adding cell lysis buffer with protease inhibitor (Cell Signaling, Beverly, MA, USA). Whole cell lysates were sonicated and protein concentrations were determined by using the BCA protein assay (Pierce Biotechnology, Rockford, IL, USA). Protein lysates (15–30 μg) were subjected to PAGE electrophoresis, transferred to polyvinylidene fluoride membranes, which were then blocked with I-block solution (Tropix, Bedford, MA, USA), washed and probed with primary antibodies, including rabbit monoclonal anti-Notch1, rabbit monoclonal anti-cleaved caspase-3, rabbit polyclonal anti-B-cell leukaemia/lymphoma 2 (BCL2), mouse monoclonal anti-transformation related protein 53 (p53) and rabbit polyclonal anti-presenilin-1 (PSEN1; Cell Signaling). Mouse monoclonal antibody to β-actin was from Novus Biologicals (Littleton, CO, USA). Immunodetection was performed with ECL Western Blotting Detection Reagents from Amersham (Buckinghamshire, UK). Densitometric analysis employed either Image J (NIH, Bethesda, MD, USA) or Adobe Photoshop.
Immunofluorescence staining
Immunofluorescence analysis of dispersed islet cells was performed essentially as described [17, 22] using a Zeiss 200M microscope. Images were analysed and quantified using SlideBook software (Intelligent Imaging Innovations). Deconvolution was used to remove out-of-focus light in high-resolution images (×100 objective; 1.45 numerical aperture). Neurogenin 3 (NGN3) rabbit antiserum was kindly provided by M. German (University of California at San Francisco) or purchased from Cemines (Golden, CO, USA). Mouse monoclonal anti-glucacon antibody was from Sigma. Guinea pig anti-insulin antibody was from Linco Research. DAPI was used as a nuclear counterstain. Primary antibodies were omitted as negative controls in each case.
Transfection and infection of MIN6 cells
A full-length Psen1 clone in a CMV promoter vector (pCMV6-XL5-PSEN1) was purchased from Origene (Rockville, MD, USA). MIN6 cells were transfected with 1 μg of either pCDNA3 (as control) or pCMV6-XL5-PSEN1 using Lipofectamine 2000 (Invitrogen), according to the manufacturer’s instructions. Cells were incubated for 48 h at 37°C with 5% CO2 and subsequently harvested for western blot analysis as described above. Adenovirus expressing Ngn3 under CMV promoter was kindly provided by M. German (University of California at San Francisco). Adenovirus expressing beta-galactosidase (βGal) under CMV promoter was a gift from T. Kieffer (University of British Columbia). For western blot analysis, MIN6 cells were infected with either five multiplicities of infection (MOI) of adeno-βGal (control) or 1, 5 or 10 MOI of adeno-Ngn3 for 48 h.
Statistics
Results are presented as means ± SEM. Data were analysed by Student’s t test. Results were considered to be statistically significant when p values were <0.05.
Results
Notch pathway components are expressed in adult mouse islets
We examined whether key elements of the Notch signalling pathway are expressed in both MIN6 cells and primary mouse islets using RT-PCR. Our results demonstrated that many components of the Notch signalling pathway and Ngn3, an essential Notch target gene, were expressed in adult mouse islets (Fig. 1). All four Notch receptors (Notch1, -2, -3, -4) were expressed, with Notch1 the prominent receptor isoform in adult mouse islets and MIN6 cells. All four ligands tested were expressed at different levels. Dll1 was the most detectable Notch ligand in mouse islets and MIN6 cells. Hes1, a major target of Notch in prenatal development, was present at low abundance in MIN6 cells but was not detected in primary mouse islets.
Expression of Notch pathway components and related genes in adult mouse islets and MIN6 cells. a Total RNA was extracted from mouse islets and MIN6 cells and gene expression levels were analysed by semi-quantitative RT-PCR (1, MIN6 cells; 2, mouse islets). b Results are quantified using densitometry. Closed bars, MIN6; open bars, islets. Expression values for specific genes were normalised to β-actin. The values are means ± SEM from three experiments
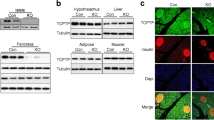
Next, we further examined the expression of Ngn3, a major downstream effector of Notch signalling that is essential for development of the endocrine pancreas [5–7]. Ngn3 mRNA was detected in adult mouse islets and MIN6 cells (Fig. 1). We also detected a modest amount of NGN3 protein in adult human islets, mouse islets, and in MIN6 cells using western blot analysis (Fig. 2a). We detected NGN3 protein using immunofluorescence labelling in dispersed adult mouse beta cells (Fig. 2b) and alpha cells (not shown), dispersed human islet cells (not shown) and in pancreatic sections from adult mice (Fig. 2c), but not in negative controls (not shown). NGN3 was produced in ∼60% of glucagon-expressing alpha cells. NGN3 was found in ∼20% of beta cells. NGN3 immunoreactivity was detected in both the cytoplasm and the nucleus of beta cells. High-resolution imaging and deconvolution (to remove out-of-focus light) revealed a punctate NGN3 staining pattern. The most intense NGN3 staining was observed in a few cytoplasmic structures, whereas less intense, but more abundant puncta were found in the nucleus (Fig. 2d). To verify the specificity of our NGN3 antibody staining, we overexpressed Ngn3 in MIN6 cells using an adenovirus. A dramatic increase in NGN3 staining was observed compared with cells infected with a βGal adenovirus (Fig. 2e). The intensity of NGN3 staining in the nuclei was 68 ± 5% relative to the staining in the cytoplasm (Fig. 2f). Together, the observation that Notch, its ligands and its target genes are expressed in adult islets suggests that the Notch pathway may retain some activity in normal tissues after prenatal pancreatic development is complete.
Ngn3 is expressed in mature islet cells. a NGN3 protein was detected in adult human islets, mouse islets and MIN6 cells using western blotting. b Immunofluorescent localisation of NGN3 (red) and insulin (green) in dispersed mouse beta cells (scale bar 30 μm). c Pancreatic sections from adult mouse were stained as above (scale bar 100 μm). d High-resolution deconvolution imaging of NGN3 (green) and nuclei (blue) in MIN6 cells was obtained with a ×100 objective (scale bar 5 μm). e, f Immunofluorescent localisation of NGN3 (red) in MIN6 cells infected with five MOI of an adenovirus vector overexpressing βGal or Ngn3 as indicated. DAPI was included to identify nuclei (blue). Scale bar = 20 μm. Images shown are representative of at least three preparations
Extracellular growth factor signalling affects the Notch pathway
As an initial step towards determining whether extracellular growth cues can modulate the Notch/neurogenin pathway in adult islets, we compared islets cultured with and without serum. In these experiments, removal of serum resulted in a significant (32%) decrease in cleaved NOTCH1 protein in mouse islets (Fig. 3a,b). Removal of serum also decreased cleaved NOTCH1 in human islets, and accordingly, increased NGN3 protein (Fig. 3c). This suggested that extracellular signals influence Notch activation in adult pancreatic islets.
Effects of extracellular signals on the Notch pathway. a Removal of serum reduced cleaved NOTCH1 protein in mouse islets. b Results are quantified using densitometry. Protein levels were normalised to β-actin. The values are means ± SEM of three experiments. *p < 0.05 vs control. c Removal of serum reduced cleaved NOTCH1 protein and increased NGN3 protein in human islets (n = 3). d Mouse islets were left untreated (control) or treated with inhibitors related to insulin/IGF-1 signalling for 24 h (50 μmol/l LY294002 [LY], 100 μmol/l HNMPA, 5 μmol/l DAPT). e Protein levels of cleaved NOTCH1 were detected by western blot analysis (n = 4). The values are means ± SEM of three experiments. *p < 0.05 vs control. f Human islets were left untreated (control) or treated with LY294002. g Protein levels of cleaved NOTCH1 were detected by western blot analysis (n = 3). The values are means ± SEM of three experiments. *p < 0.05 vs control
To investigate whether classic growth factor signalling pathways are involved in the regulation of Notch1 and Ngn3 in adult islets, we incubated mouse and human islets in medium containing inhibitors of signalling components common to insulin, IGF and other growth factors. We found that inhibition of phosphatidylinositol 3-kinase with LY-294002 reduced the cleaved NOTCH1 protein level in both primary mouse and human islets (Fig. 3d–g). In mouse islets this was associated with increased Ngn3 mRNA (data not shown). HNMPA, an inhibitor of the insulin receptor tyrosine kinase, also significantly reduced activated NOTCH1 protein in primary mouse islets (Fig. 3d,e). As a positive control, the gamma-secretase inhibitor DAPT also lowered NOTCH1 protein levels in mouse islets (Fig. 3d,e). Activation of Notch1 would be expected to negatively regulate Ngn3. Accordingly we observed an increase in Ngn3 mRNA expression and protein levels after blocking the gamma-secretase (see below). These results indicate that the Notch pathway can be modulated in adult primary islets by growth factor signalling, although they do not inform as to which specific growth factors are involved. We tested three candidate growth factors [insulin, IGF-I and fibroblast growth factor 10 (FGF10)] under the serum-free conditions with which we have previously observed robust effects of insulin [19], but were unable to discern significant differences in cleaved NOTCH1 at 48 h (data not shown). These results suggest that Notch may be regulated by a serum component other than insulin, IGF-I or FGF10, or that the effects of these hormones occur on a different time-scale from that tested.
Several studies have implicated Notch and NGN3 in the control of differentiation during fetal development that ultimately leads to mature beta cell function [4–7, 9]. We have shown that the active Notch fragment is reduced by the gamma-secretase inhibitor DAPT in beta cells. We next asked whether mature beta cell function requires intact Notch signalling. Glucose-stimulated insulin release is the primary function of differentiated beta cells. We found that blocking the Notch pathway with DAPT did not significantly inhibit glucose- or KCl-stimulated insulin release in primary mouse islets (Fig. 4). This suggests that the Notch pathway does not play a role in glucose signalling or insulin exocytosis in mature beta cells.
The Notch pathway regulates beta cell apoptosis
A number of reports have indicated the Notch pathway can suppress apoptotic cell death, depending on the cell context [1, 10, 23]. To determine whether Notch plays a role in the survival of mature beta cells, we examined the effect of Notch pathway inhibition on primary human and mouse islets and MIN6 cells. We took advantage of the gamma-secretase inhibitor DAPT, which reduces active Notch protein in many cell types [24–27], including primary islet cells (see above). Mouse islets and MIN6 cells treated with DAPT for 24 h showed a significant increase in apoptosis, measured with PCR-enhanced DNA ladders (Fig. 5a–c). MIN6 cells also exhibited a dose-dependent increase in cell death as evidenced by propidium iodide incorporation (Fig. 5d,e). This was accompanied by a dose-dependent increase in caspase 3 activation measured by western blot after 24 h (Fig. 6a), as well as 6 h and 12 h (data not shown). An increase in the number of cleaved caspase 3-positive MIN6 cells was confirmed in cultures treated with DAPT using immunofluorescence microscopy (Fig. 6c). DAPT also induced a dose-dependent increase in caspase 3 activation in human islets (Fig. 6b). Interestingly, DAPT did not induce apoptosis in MIN6 cells or human islets cultured in high glucose. In fact, DAPT was actually protective at some doses (Fig. 6a,b). We also examined other possible targets of the gamma-secretase, as well as proteins known to play a role in beta cell death. Levels of p53 were unchanged (Fig. 6d). No significant changes in the levels of BCL2 or BCL2-associated X protein (BAX) were identified (data not shown). Together, these results indicate that inhibiting Notch activation by ∼60% with DAPT (Fig. 3d,e) induces caspase 3-dependent beta cell apoptosis and therefore suggest that the Notch pathway is a regulator of programmed cell death in mature islet cells.
Blocking the Notch pathway induces apoptosis in mouse islets and MIN6 cells. MIN6 cells (a) and primary mouse islets (b) were treated with DAPT for 24 h as indicated and apoptosis was measured with PCR-enhanced DNA ladders. c Quantification of DNA ladders in mouse islets treated with DAPT for 24 h as indicated (n = 4 for 5 nmol/l and 5 μmol/l; n = 3 for 10 nmol/l). The values are means ± SEM of four experiments. *p < 0.05 vs control. d A representative image of MIN6 cells treated with DAPT in 5 and 25 mmol/l glucose for 24 h and subsequently stained with Hoechst 33258 and propidium iodide. Scale bar = 100 μm. e Propidium iodide-positive cells (red) were counted and reported as a percentage of Hoechst-positive cells (blue). Open bars, 5 mmol/l glucose; closed bars, 25 mmol/l glucose. The values are means ± SEM of three experiments. *p < 0.05 vs control
Blocking Notch induces caspase 3 activation. a MIN6 cells were treated with increasing concentrations of DAPT in 5 and 25 mmol/l glucose for 24 h and cleaved caspase 3 (CC3) protein levels were detected by western blot analysis. Results were quantified using densitometry and normalised to β-actin. The values are means ± SEM of three experiments. *p < 0.05 vs control. b Dose-dependent increase in CC3 was also observed in human islets cultured in 5 mmol/l glucose, but not 25 mmol/l glucose (n = 3). Open bars, 5 mmol/l glucose; closed bars 25 mmol/l glucose. The values are means ± SEM of three experiments. *p < 0.05 vs control. c Immunofluorescent staining for CC3 (red) in a field of MIN6 cells treated or not with 10 μmol/l DAPT for 24 h. DAPI was included to identify nuclei (blue). d p53 was not changed in mouse islets treated with 1 μmol/l DAPT for 24 h (n = 3). au, Arbitrary units
Next, we tested whether Ngn3, a well-characterised target of Notch in the embryonic pancreas, may also affect apoptosis in mature beta cells. First we confirmed that Ngn3 could be increased under conditions of inhibited NOTCH1. Indeed, treating mouse islets with DAPT resulted in a dose-dependent increase in Ngn3 mRNA (Fig. 7a). Similarly, NGN3 protein was increased in MIN6 cells treated with the gamma-secretase inhibitor (Fig. 7b,c). Based on our results up to this point, and the fact that NOTCH1 is a negative regulator of Ngn3, one would expect that NGN3 may be pro-apoptotic in adult beta cells. We tested this hypothesis using adenovirus-mediated overexpression of Ngn3. Overexpressing Ngn3, either approximately fourfold or 16-fold (Fig. 7d,e), resulted in a significant increase in apoptosis, as indicated by caspase 3 cleavage (Fig. 7d,f). These results are consistent with the idea that a signalling network involving the gamma-secretase, Notch and NGN3 is present and active in mature beta cells.
Regulation of Ngn3 by Notch signalling in adult islets. a Ngn3 mRNA level in mouse islets treated with DAPT for 24 h (n = 3). Values for Ngn3 mRNA were normalised to β-actin. *p < 0.05 vs control. b MIN6 cells were treated with increasing concentrations of DAPT in 5 mmol/l glucose for 24 h and NGN3 protein levels were detected by western blot analysis. c Results were quantified using densitometry and normalised to β-actin. The values are means ± SEM of three experiments. *p < 0.05 vs control. d MIN6 cells were infected for 48 h with increasing MOI of an adenovirus vector overexpressing beta galactosidase (βGal) or Ngn3 as indicated. NGN3 and cleaved caspase 3 (CC3) protein levels were detected by western blot analysis. e, f Results were quantified using densitometry and normalised to β-actin. The values are means ± SEM of three experiments. *p < 0.05 vs control
Finally, to complement our experiments with DAPT, we examined the effect of gamma-secretase overactivity. PSEN1, an essential component of the gamma-secretase, was overproduced approximately threefold in MIN6 cells (Fig. 8a,b,d,e). This resulted in a significant increase in cleaved/activated NOTCH1 (Fig. 8a,c) and a decrease in programmed cell death, as indicated by a decrease in caspase 3 cleavage (Fig. 8d,f) and propidium iodide incorporation (Fig. 8g). These results demonstrate that increasing NOTCH1 activation is associated with a protective effect in beta cells.
Overexpression of Psen1 increases NOTCH1 protein levels and reduces apoptosis. a MIN6 cells were transiently transfected with either an empty vector (control) or a Psen1 overexpressing clone (PS1 OE). Overexpression of Psen1 in MIN6 cells increased approximately threefold the levels of the PSEN1 (b; n = 3, *p < 0.05). Cleaved NOTCH1 protein (ICD) levels were also increased by Psen1 overexpression (c; n = 3, *p < 0.05). d Psen1 overexpression slightly reduced apoptosis as evidenced by reduced cleavage of caspase-3. e, f Results were quantified using densitometry and normalised to β-actin. The values are means ± SEM of three experiments. *p < 0.05. g MIN6 cells transiently transfected with either an empty vector or Psen1 clone were stained with Hoechst 33258 and propidium iodide (PI). Propidium iodide-positive cells and Hoechst-positive cells were counted and reported as a percentage of propidium iodide-positive cells (n = 3, *p < 0.05). CC3, cleaved caspase 3
Discussion
The present study was undertaken to determine whether the Notch pathway remains functional in adult islets. Our study produced four major findings. First, we have demonstrated that Notch pathway components and target genes are expressed in adult islets. Second, inhibition of growth factor signalling inhibits Notch activation in mouse and human islets. Third, blocking the Notch cleavage/activation at the level of the gamma-secretase induced apoptosis in adult islet cells, whereas increasing Notch activation was associated with an anti-apoptotic effect. Fourth, the Notch target gene Ngn3 is present in adult beta cells where it appears to play a pro-apoptotic role at very high levels. Together these findings illustrate the potential of manipulating Notch to control cell fate in adult islet cells.
Several studies have implicated Notch in the regulation of exocrine and endocrine cell fate during pancreatic development [5–7]. It was reported that Notch1 and Notch2 expression starts to decline in embryonic mouse pancreas after embryonic day 14.5 [28, 29]. Others have reported little or no expression of Notch pathway components in human adult pancreatic ductal epithelium [27], or in whole adult rat pancreas [14]. Notch was found in differentiated neurons of the adult retina and it was speculated that Notch might confer some degree of plasticity on post-mitotic neurons [30]. In the present study, we demonstrate the presence of Notch pathway components in adult pancreatic cells, using a combination of MIN6 cells, mouse islets and human islets. Each of these models has specific limitations and advantages. For example, human islets often contain substantial numbers of non-beta cells, whereas MIN6 cells may not be fully differentiated. Notwithstanding, the combination of all three models helps support a role for Notch in mature islet cells, including beta cells. Although the role of the Notch signalling pathway in the pancreas after birth remains poorly understood, these observations suggest that this ‘developmental’ network may be reactivated in adult beta cells, perhaps to control proliferation, differentiation or survival.
One potentially controversial finding of the present study was the detection of NGN3 and its modulation by Notch in adult islets. Ngn3 is required for the development of all endocrine cell lineages of the pancreas and has been designated as a marker of islet precursor cells [8]. NGN3 is first detected at embryonic day 9. Its levels peak at embryonic day 15.5, then decline to the point where little or no NGN3 is detected in the early postnatal pancreas [5, 9]. These findings do not preclude the re-emergence of NGN3 in more mature islets, and there are a few reports of Ngn3 expression in adult islets [31, 32]. We observed both Ngn3 mRNA and NGN3 protein in adult (3- to 6-month-old) mouse islets and human islets (49–65 years old), as well as in MIN6 cells. These findings support the possibility that NGN3 may play a role in the adult endocrine pancreas, perhaps in the regulation of cell fate. Whether NGN3-positive adult islet cells represent cells with ‘precursor potential’ remains to be addressed. While the RT-PCR and western blot data leave little doubt that NGN3 can be detected in adult islets, its identification in multiple cellular compartments using immunofluorescence microscopy is intriguing. Although substantial NGN3 immunoreactivity could be found within the nucleus, cytoplasmic staining was more intense. While NGN3 is typically found in the nucleus during development, cytoplasmic NGN3 staining has been observed by others in postnatal islet cells, and it was suggested that this may represent a transitional state [33]. Interestingly, although overproduction of NGN3 protein using a TAT protein transduction domain was effective in the control of downstream target genes, it primarily resided in the cytoplasm [34]. Together, with these other findings, our data suggest the possibility that NGN3 may reside in both the cytoplasm and nucleus, depending on whether cells are developing or mature. This idea is consistent with the observation that many transcription factors critical for adult pancreatic beta cell function (e.g. forkhead box O1 [Foxo1], cAMP response element binding [CREB] protein and pancreatic and duodenal homeobox 1 [Pdx1]) are known to traffic to and from the nucleus [35–37]. In our mature islet cells, therefore, it is possible that NGN3 could be further activated and that additional factors other than Notch may be required for its full action.
Our results support an important role for Notch signalling in cell-fate decisions in mature islet cells. Interestingly, the alterations in Notch have been associated with different human cancers including pancreatic cancer [11, 27, 38], suggesting malfunction of this pathway can have significant consequences in the pancreas. To uncover the function of Notch in adult islets we used DAPT, a gamma-secretase inhibitor, as well as Psen1 overexpression to increase gamma-secretase activity. DAPT is known to suppress Notch activation in many cell types [24-27] and we confirmed this in mature islets. DAPT induced caspase 3-dependent apoptosis in human and mouse islets, and MIN6 cells. Notably, DAPT evoked apoptosis in low-glucose, but not high-glucose conditions. This susceptibility to programmed cell death in the presence of low glucose is reminiscent of the situation in islets with reduced expression of the MODY gene Pdx1 [22], or increased levels of the type 2 diabetes susceptibility gene calpain 10 (Capn10) [17]. Further studies are required to characterise additional downstream mechanisms of DAPT-induced beta cell apoptosis.
It should be noted that while modulating Notch activation via the gamma-secretase is quite effective in our studies, it remains possible that additional targets of the gamma-secretase and/or gamma-secretase-independent targets of PSEN1 may have also been involved. Testing the specific role of Notch in beta cell function and survival will probably involve the simultaneous knockdown or knockout of all Notch isoforms in adult islet cells. Nevertheless, our results point to the importance of active Notch signalling in preventing apoptosis of beta cells in adult islets, specifically in low-glucose conditions. We also implicated growth factor signalling pathways in the control of Notch activity in adult islets, but we were unable to directly observe effects of specific growth factors under the conditions tested. Whether this reflects the requirement for multiple stimuli or other experimental parameters is not known. In the present study, we also examined islet function and found that gamma-secretase-dependent Notch cleavage is not involved in the maintenance of differentiated function of mature islets, assessed by glucose-stimulated insulin secretion (at least in the time-frame studied). This finding illustrates the possibility that programmed cell death can be in process in some islet cells, while total islet secretory function remains intact. Again, this is reminiscent of the situation in mice lacking one allele of Pdx1 [22].
It is well established that beta cell apoptosis is a critical event in the pathogenesis of diabetes and that beta cell death limits the potential of clinical islet transplantation [39–42]. In type 1 diabetes, a greater number of beta cells remain than previously appreciated and preventing ongoing beta cell death represents an intriguing therapeutic strategy for this disease. Our findings suggest Notch as a potential target for regulating beta cell death in islet transplantation. Further investigation of the role of Notch in mature beta cells may allow new strategies to improve cell culture conditions in clinical islet transplantation, perhaps by engineering modified apoptosis-resistant human islets.
Abbreviations
- BCL2:
-
B cell leukaemia/lymphoma 2
- DAPT:
-
N-[N-(3,5-difluorophenacetyl-l-alanyl)]-S-phenylglycine t-butyl ester
- HNMPA:
-
hydroxy-2-naphthalenylmethylphosphonic acid
- MOI:
-
multiplicity of infection
- NGN3:
-
neurogenin 3
- p53:
-
transformation related protein 53
- PSEN1:
-
presenilin 1
References
Artavanis-Tsakonas S, Rand MD, Lake RJ (1999) Notch signaling: cell fate control and signal integration in development. Science 284:770–776
Greenwald I (1998) LIN-12/Notch signaling: lessons from worms and flies. Genes Dev 12:1751–1762
Beatus P, Lendahl U (1998) Notch and neurogenesis. J Neurosci Res 54:125–136
Hardy J, Israel A (1999) Alzheimer's disease. In search of gamma-secretase. Nature 398:466–467
Apelqvist A, Li H, Sommer L et al (1999) Notch signalling controls pancreatic cell differentiation. Nature 400:877–881
Hald J, Hjorth JP, German MS, Madsen OD, Serup P, Jensen J (2003) Activated Notch1 prevents differentiation of pancreatic acinar cells and attenuate endocrine development. Dev Biol 260:426–437
Murtaugh LC, Stanger BZ, Kwan KM, Melton DA (2003) Notch signaling controls multiple steps of pancreatic differentiation. Proc Natl Acad Sci U S A 100:14920–14925
Habener JF, Kemp DM, Thomas MK (2005) Minireview: transcriptional regulation in pancreatic development. Endocrinology 146:1025–1034
Schwitzgebel VM, Scheel DW, Conners JR et al (2000) Expression of neurogenin3 reveals an islet cell precursor population in the pancreas. Development 127:3533–3542
Jang MS, Miao H, Carlesso N et al (2004) Notch-1 regulates cell death independently of differentiation in murine erythroleukemia cells through multiple apoptosis and cell cycle pathways. J Cell Physiol 199:418–433
Nair P, Somasundaram K, Krishna S (2003) Activated Notch1 inhibits p53-induced apoptosis and sustains transformation by human papillomavirus type 16 E6 and E7 oncogenes through a PI3K-PKB/Akt-dependent pathway. J Virol 77:7106–7112
Jang J, Choi YI, Choi J et al (2006) Notch1 confers thymocytes a resistance to GC-induced apoptosis through Deltex1 by blocking the recruitment of p300 to the SRG3 promoter. Cell Death Differ 13:1495–1505
Jensen JN, Cameron E, Garay MV, Starkey TW, Gianani R, Jensen J (2005) Recapitulation of elements of embryonic development in adult mouse pancreatic regeneration. Gastroenterology 128:728–741
Rooman I, De Medts N, Baeyens L et al (2006) Expression of the notch signaling pathway and effect on exocrine cell proliferation in adult rat pancreas. Am J Pathol 169:1206–1214
Darville MI, Eizirik DL (2006) Notch signaling: a mediator of beta-cell de-differentiation in diabetes? Biochem Biophys Res Commun 339:1063–1068
Salvalaggio PR, Deng S, Ariyan CE et al (2002) Islet filtration: a simple and rapid new purification procedure that avoids ficoll and improves islet mass and function. Transplantation 74:877–879
Johnson JD, Han Z, Otani K et al (2004) RyR2 and calpain-10 delineate a novel apoptosis pathway in pancreatic islets. J Biol Chem 279:24794–24802
Johnson JD, Misler S (2002) Nicotinic acid-adenine dinucleotide phosphate-sensitive calcium stores initiate insulin signaling in human beta cells. Proc Natl Acad Sci U S A 99:14566–14571
Johnson JD, Bernal-Mizrachi E, Alejandro EU et al (2006) Insulin protects islets from apoptosis via Pdx1 and specific changes in the human islet proteome. Proc Natl Acad Sci U S A 103:19575–19580
Ohsugi M, Cras-Meneur C, Zhou Y et al (2005) Reduced expression of the insulin receptor in mouse insulinoma (MIN6) cells reveals multiple roles of insulin signaling in gene expression, proliferation, insulin content, and secretion. J Biol Chem 280:4992–5003
Carson C, Murdoch B, Roskams AJ (2006) Notch 2 and Notch 1/3 segregate to neuronal and glial lineages of the developing olfactory epithelium. Dev Dyn 235:1678–1688
Johnson JD, Ahmed NT, Luciani DS et al (2003) Increased islet apoptosis in Pdx1+/− mice. J Clin Invest 111:1147–1160
Miele L, Osborne B (1999) Arbiter of differentiation and death: Notch signaling meets apoptosis. J Cell Physiol 181:393–409
Sastre M, Steiner H, Fuchs K et al (2001) Presenilin-dependent gamma-secretase processing of beta-amyloid precursor protein at a site corresponding to the S3 cleavage of Notch. EMBO Rep 2:835–841
Dovey HF, John V, Anderson JP et al (2001) Functional gamma-secretase inhibitors reduce beta-amyloid peptide levels in brain. J Neurochem 76:173–181
Micchelli CA, Esler WP, Kimberly WT et al (2003) Gamma-secretase/presenilin inhibitors for Alzheimer’s disease phenocopy Notch mutations in Drosophila. FASEB J 17:79–81
Miyamoto Y, Maitra A, Ghosh B et al (2003) Notch mediates TGF alpha-induced changes in epithelial differentiation during pancreatic tumorigenesis. Cancer Cell 3:565–576
Lammert E, Brown J, Melton DA (2000) Notch gene expression during pancreatic organogenesis. Mech Dev 94:199–203
Norgaard GA, Jensen JN, Jensen J (2003) FGF10 signaling maintains the pancreatic progenitor cell state revealing a novel role of Notch in organ development. Dev Biol 264:323–338
Ahmad I, Zaqouras P, Artavanis-Tsakonas S (1995) Involvement of Notch-1 in mammalian retinal neurogenesis: association of Notch-1 activity with both immature and terminally differentiated cells. Mech Dev 53:73–85
Gu G, Dubauskaite J, Melton DA (2002) Direct evidence for the pancreatic lineage: NGN3+ cells are islet progenitors and are distinct from duct progenitors. Development 129:2447–2457
Minami K, Okuno M, Miyawaki K et al (2005) Lineage tracing and characterization of insulin-secreting cells generated from adult pancreatic acinar cells. Proc Natl Acad Sci USA 102:15116–15121
Baeyens L, Bonne S, German MS, Ravassard P, Heimberg H, Bouwens L (2006) Ngn3 expression during postnatal in vitro beta cell neogenesis induced by the JAK/STAT pathway. Cell Death Differ 13:1892–1899
Dominguez-Bendala J, Klein D, Ribeiro M et al (2005) TAT-mediated neurogenin 3 protein transduction stimulates pancreatic endocrine differentiation in vitro. Diabetes 54:720–726
Dalle S, Longuet C, Costes S et al (2004) Glucagon promotes cAMP-response element-binding protein phosphorylation via activation of ERK1/2 in MIN6 cell line and isolated islets of Langerhans. J Biol Chem 279:20345–20355
Elrick LJ, Docherty K (2001) Phosphorylation-dependent nucleocytoplasmic shuttling of pancreatic duodenal homeobox-1. Diabetes 50:2244–2252
Kawamori D, Kaneto H, Nakatani Y et al (2006) The forkhead transcription factor Foxo1 bridges the JNK pathway and the transcription factor PDX-1 through its intracellular translocation. J Biol Chem 281:1091–1098
Sjolund J, Manetopoulos C, Stockhausen MT, Axelson H (2005) The Notch pathway in cancer: differentiation gone awry. Eur J Cancer 41:2620–2629
Biarnes M, Montolio M, Nacher V, Raurell M, Soler J, Montanya E (2002) Beta-cell death and mass in syngeneically transplanted islets exposed to short- and long-term hyperglycemia. Diabetes 51:66–72
Davalli AM, Scaglia L, Zangen DH, Hollister J, Bonner-Weir S, Weir GC (1996) Vulnerability of islets in the immediate posttransplantation period. Dynamic changes in structure and function. Diabetes 45:1161–1167
Moritz W, Meier F, Stroka DM et al (2002) Apoptosis in hypoxic human pancreatic islets correlates with HIF-1alpha expression. FASEB J 16:745–747
Stagner J, Mokshagundam S, Wyler K et al (2004) Beta-cell sparing in transplanted islets by vascular endothelial growth factor. Transplant Proc 36:1178–1180
Acknowledgements
We thank M. German and T. Kieffer for Ngn3-related reagents and advice. We thank J. Roskams for Notch reagents. We thank E. Bernal-Mizrachi for helpful discussions. This work was supported by operating grants to J. D. Johnson from the Canadian Institutes of Health Research (CIHR) and the Juvenile Diabetes Research Foundation (JDRF). J. D. Johnson was supported by salary awards from the Michael Smith Foundation for Health Research Centre (MSFHR), JDRF, CIHR and the Canadian Diabetes Association. V. Dror was a Stem Cell Network/JDRF Postdoctoral Fellow.
Duality of interest
The authors declare that there is no duality of interest associated with this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dror, V., Nguyen, V., Walia, P. et al. Notch signalling suppresses apoptosis in adult human and mouse pancreatic islet cells. Diabetologia 50, 2504–2515 (2007). https://doi.org/10.1007/s00125-007-0835-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-007-0835-5