Abstract
The domestication of tetraploid wheats started from their wild progenitor Triticum dicoccoides. In this paper, the geographical distribution of this progenitor is revised to include more sampling locations. The paper is based on a collection of wild and domesticated lines (226 accessions in total) analyzed by AFLP at 169 polymorphic loci. The collection includes the 69 wild lines considered by Mori et al. (2003) in their study on chloroplast DNA haplotypes of T. dicoccoides. The goal of the experiment was to reconsider which location thought to have generated the domesticated germplasm has the highest chance of being the actual site from which wild progenitors were sampled during domestication. Phylogenetic analysis of the nuclear AFLP databases indicates that two different genetic taxa of T. dicoccoides exist, the western one, colonizing Israel, Syria, Lebanon and Jordan, and the central-eastern one, which has been frequently sampled in Turkey and rarely in Iran and Iraq. It is the central-eastern race that played the role of the progenitor of the domesticated germplasm. This is supported by the cumulative results of the AFLP data from the collections of Ozkan et al. (2002) and of Mori et al. (2003), which indicate that the Turkish Karacadag population, intermixed with some Iraq-Iran lines, has a tree topology consistent with that of the progenitor of domesticated genotypes. The Turkish Kartal population belongs genetically to the central-eastern T. dicoccoides race but at the nuclear DNA level is less related to the domesticated gene pool. A general agreement between published work on tetraploid wheat domestication emerges from these results. A disagreement is nevertheless evident at the local geographical scale; the chloroplast DNA data indicate the Kartal mountains while AFLP fingerprinting points to the Karacadag Range as the putative site of tetraploid wheat domestication.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Two lines of evidence support the argument that the Fertile Crescent is the site of origin of Western agriculture. First, the geographical distributions of the wild progenitors of wheat (Triticum boeticum, T. urartu, T. dicoccoides, Aegilops tauschii), wild barley (Hordeum spontaneum) and wild rye (Secale vavilovii) intersect in this region (Nesbitt and Samuel 1996; Zohary and Hopf 2000; Salamini et al. 2002). Second, seeds of the wild species have been found in early archaeological sites of the region, followed in stratigraphic succession by the remains of domesticated forms (Moore et al. 2000; Gopher et al. 2002).
The domestication of the tetraploid wheats started from their wild progenitor T. dicoccoides. This species, with its A genome derived from T. urartu (Dvorak et al. 1993, 1998), has brittle ears that shatter at maturity. Domesticated emmer wheat, T. dicoccum, has hulled seeds and its AABB genome is common to other domesticated tetraploid wheats. Emmer was the most important crop in the Fertile Crescent until the early Bronze Age (Bar-Yosef 1998), and domesticated forms are present at early Neolithic sites (such as Tell Aswad, ~10,800 BP; Van Zeist and Bakker-Heeres 1982). Naked forms, such as the small grain T. parvicoccum (Kislev 1980), are also found in early Neolithic locations.
The geographical distribution of T. dicoccoides reported by Zohary and Hopf (2000) includes the western Fertile Crescent, the central part of southeastern Turkey and areas in eastern Iran and Iraq. Johnson (1975) reports that from southeastern Turkey to Iran and Iraq the species is progressively substituted by the wild tetraploid wheat T. araraticum. T. dicoccoides also grows on the basaltic rocky slopes of the Karacadag mountains in southeastern Turkey (Johnson 1975; Harlan and Zohary 1966).
AFLP data have indicated that domesticated tetraploid AABB wheats are most closely related to wild emmer populations from southeastern Turkey (Ozkan et al. 2002). In this study, 99 lines of wild emmer from Turkey, Israel, Jordan, Lebanon, Syria and Iran, as well as 19 wild emmer lines from a Karacadag population (southeastern Turkey), were studied. The genetic profile of 15 of the 19 Karacadag lines was consistent with their close genetic relationship to domesticated emmer. This was interpreted as more than a coincidence, considering that using the same technique the domestication of einkorn was also located to the Karacadag mountains in southeastern Turkey (Heun et al. 1997). Based on these and similar findings this area was later defined as the “core area” of the origin of agriculture (Lev-Yadun et al. 2000).
In the study by Ozkan et al. (2002) evidence was also provided that the hulled emmer and T. durum (free-threshing) tetraploid genotypes group separately on a phylogenetic tree, although the two groups merge into a common lineage soon before joining the T. dicoccoides line topologies. At least based on principal coordinate analysis, both a monophyletic and a diphyletic domestication of tetraploid wheats are possible (Salamini et al. 2004), with the former hypothesis having a higher probability. In addition, chloroplast DNA fingerprinting of wild and domesticated emmer wheat indicates that two distinct maternal lineages have been involved in the domestication process, suggesting that the event occurred at least twice (Mori et al. 2003). While referring again to southeastern Turkey as the domestication site for emmer wheat, these authors indicate that the Kartal mountains (280 km west of the Karacadag Range) host wild emmer lines having both chloroplast DNA haplotypes prevalent in domesticated emmer.
In this study, we use AFLP fingerprinting to analyze a new set of wild emmer lines, including 69 accessions from the collection of Mori et al. (2003), 7 lines from Dr. B. Gill, and 12 lines from Dr. A. Karagoz, and compared the results with those derived from 92 wild emmer lines previously characterized by Ozkan et al (2002). The reasoning behind this experiment was to reconsider which location of southern Turkey has the highest chance of being the actual site from which the wild progenitors of tetraploid wheats were sampled.
Materials and methods
Plant material
The nomenclature followed in this paper indicates with T. dicoccoides the wild lines of T. turgidum ssp. dicoccoides, with T. dicoccum the domesticated hulled emmer lines (T. turgidum ssp. dicoccum) and with T. durum the domesticated free-threshing hard wheat lines (T. turgidum ssp. durum) (see also Zohary and Hopf 2000).
A total of 224 wheat accessions were considered, including 179 accessions of T. dicoccoides, 22 of T. durum, 23 of T. dicoccum and 1 line of T. polonicum. The geographic origin and the source of the accessions are given in Tables 1 and 2. One hundred and thirty-one lines were those typed by Ozkan et al. (2002), of which 91 were wild T. dicoccoides accessions, 18 T. dicoccum emmer lines, and 22 T. durum hard wheat lines. Of the remaining lines analyzed in this paper, 69 were from the collection studied by Mori et al. (2003) and 7 and 12 lines were received from Dr. B. Gill (University of Kansas State, USA) and Dr. A. Karagoz (Department of Field Crops, Ankara), respectively. In addition, 5 T. dicoccum emmer lines were also obtained from Dr. A. Karagoz (Ankara University). The three collections had some lines in common based on their AFLP patterns.
Species identification of the 88 new lines characterized was carried out by field observations. Five plants were grown in pots and transplanted to the field after sampling material for DNA extraction.
Genomic DNA extraction and AFLP analyses
Young leaves were collected, lyophilized and kept at −70°C until use. DNA was extracted using the Qiagen DNA isolation kit (DNeasy Plant Mini, Qiagen, Hilden, Germany). AFLP procedure was performed according to Ozkan et al. (2002).
Data analysis
The AFLP gels were manually scored for the presence or absence of relevant bands. Each band was considered to represent a polymorphic locus. The genetic relationships among the taxa considered were established using the phylogenetic analysis package PHYLIP (Felsenstein 1989) and the multivariate analysis system NTSYS (Rohlf 1998). For the phylogenetic analysis concerning AFLP allele frequencies in populations or groups of lines, ten independent trees were constructed as described (Heun et al. 1997; Badr et al. 2000), using CONTML and distance matrix methods (Fitch and Margoliash 1967; Saitou and Nei 1987), and employing various measures of genetic distance (Cavalli-Sforza and Edwards 1967; Nei 1972; Wright 1978; Reynolds et al. 1983) calculated from AFLP allele frequency. To cluster single accessions (Fig. 1b), the neighbor-joining (NJ) method based on Jaccard distances was used as well as several other methods of tree building and genetic distance calculations (citations above).
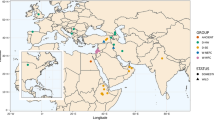
a Sampling locations of Triticum dicoccoides wild lines in the Fertile Crescent. b Genetic relationship among T. dicoccoides (wild emmer), T. dicoccum (hulled emmer) and T. durum (free-threshing hard wheat). AFLP phylogeny of 45 domesticated tetraploid wheats (23 T. dicoccum, 22 free-threshing T. durum varieties) and 88 wild emmer lines (69 from Mori et al. 2003 and 19 from A. Karagoz and B. Gill) with different sampling locations (Turkey I, Kartal populations; Turkey II, mainly Karakadag populations; Iraq-Iran III and III’, as in a; Israel IV as in a and IV’ as in Table 1; Syrian, Jordanian and Lebanese lines are included in group V). The neighbour-joining tree (Saitou and Nei, 1987) of Jaccard (1908) genetic distances is shown. Domesticated T. dicoccum lines were from Turkey, Rumania, Iran, India, German, and Italy. The hard wheat (T. durum) lines were from France, Palestine, Italy, Tunisia, Jordan, Cyprus, Spain, Syria, Greece, Ukraine, Tajikistan, and Mexico. c Genetic relationships of domesticated tetraploid wheats with wild T. dicoccoides groups sampled in different regions of the Fertile Crescent with origins already specified in b. Using programs of the PHYLIP package (1993), 10 independent trees were constructed as described (Heun et al. 1997; Badr et al. 2000) using CONTML (topology shown; Felsenstein 1981) and distance matrix methods (Fitch and Margoliash 1967; Saitou and Nei 1987) employing various measures of genetic distance (Cavalli-Sforza and Edwards 1967; Nei 1972; Wright 1978; Reynolds et al. 1983) calculated from AFLP allele frequencies between groups of lines from the geographic regions indicated. The number of tree-buiding methods generating the same topologies is indicated at branches. d, e Results of the principal coordinates analysis of AFLP data used for the phylogenetic reconstructions of domesticated to wild lines relationships in tetraploid wheats. The analyses were carried out using the computer package NTSYSpc (Rohlf 1998). The pairwise genetic distance were calculated according to the algorithm of Jaccard (1908).d Principal coordinates analysis of the data obtained by merging the AFLP fingerprints of the lines of Ozkan et al. (2002), of the Mori et al. (2003) and of the Gill and Karagoz collections. The first 3 axes explain 22.2% of the total variability. e The AFLP database concerns the 55 domesticated lines specified in b, the 69 wild lines studied by Mori et al. (2003) and the 7 and 12 lines obtained from Dr. B. Gill and Dr. A. Karagoz (some lines were duplicated in the 3 collections, therefore the actual results of the analysis reports the positions of 72 lines).The first three axes explain 28.5% of the total variability. f A total of 131 domesticated and wild accessions considered by Ozkan et al. (2002) were the basis for this figure. Group III’ and IV’ wild lines were not represented in this set of materials. The first three axes explain 24.8% of the total variability
Dates reported in the Introduction and the Discussion of this paper are calibrated years before present (cal BP). They refer to 14C dates that were transformed into calendar years of the absolute dendrochronological record using the data provided by Zohary and Hopf (2000) and by Moore at al. (2000) and were cross-checked for consistency with the data of Gopher et al. (2002) and Maier (1996).
Results
An attempt to assemble a large T. dicoccoides collection
Precisely where wild T. dicoccoides grows and how complete are the existing collections is still a matter of discussion. The distribution of T. dicoccoides (Zohary and Hopf 2000) includes the western Fertile Crescent, southeastern Turkey, and areas in eastern Iran and Iraq. From southeastern Turkey to Iran and Iraq the species is progressively substituted by the wild tetraploid wheat T. araraticum having the AAGG genome (Johnson 1975). T. dicoccoides accessions have been only rarely collected in Iran or Iraq, thus supporting Johnson’s conclusion. Stands of T. dicoccoides grow on the rocky slopes of the Karacadag Mountains (Harlan and Zohary 1966; Johnson 1975).
The study of Ozkan et al. (2002) has shown that in spite of their morphological similarity, T. dicoccoides and T. araraticum are easily distinguished at the molecular level. In the current study, the materials analyzed by Ozkan et al. (2002) were integrated with the accessions analyzed by Mori et al. (2003). Figure 1a summarizes, in a graphical representation, the T. dicoccoides geographical distribution based on the sampling locations of the wild lines analyzed for their species assignment using molecular techniques (Ozkan et al. 2002; Mori et al. 2003). Wild lines are included in groups I to V (including groups III’ and IV’, therefore defined also according to the phylogenies described in Fig. 1, but mainly based on geographical criteria). The figure highlights two aspects: (1) a number of wild lines have also been collected in the eastern part of the Fertile Crescent, and (2) the Kartal Mountains in southeast Turkey host rich natural stands of the species.
AFLP fingerprinting of the wild T. dicoccoides accessions studied by Mori et al. (2003)
The collection of wild T. dicoccoides lines (69 accessions) of Mori et al. (2003) was considered in this study, and 19 additional lines were obtained from Dr. B. Gill and Dr. A. Karagoz. Molecular fingerprinting was based on the same AFLP primer combinations as those used by Ozkan et al. (2002). In total, 169 polymorphic AFLP loci were scored for the presence or absence of amplified DNA fragments. The database was integrated with the AFLP data of the domesticated T. dicoccum and T. durum lines studied by Ozkan et al. (2002). The data were analyzed with several methods for genetic distance calculation and phylogenetic tree building. The topologies of the trees obtained with the different methods were consistently similar. A representative NJ tree is reported in Fig. 1b. As expected from previous experiments, the domesticated accessions of both T. dicoccum and T. durum have a lineage in common, but the two taxa diverge quite early after splitting from the wild lineage. Turkish wild accessions of group II (Karacadag Range) are, on average, highly related to the domesticated landraces. An interesting, and in part new, finding is that a small group of 8 lines from Iran and Iraq also appear to be genetically very similar to the domesticated gene pool. Turkish lines from the Kartal Mountain (group I), although more related to T. dicoccum and T. durum than the wild lines of the western Fertile Crescent, are not as highly related to the domesticated gene pool as the wild Karacadag and the Iraq-Iran lines. The Israeli, Jordanian, Lebanese and Syrian wild lines (groups IV, IV’ and V) are clearly separated from the other wild T. dicoccoides accessions and are genetically far less related to the domesticated lines (Fig. 1b). As in Ozkan et al. (2002), the Israeli T. dicoccoides lines are split in two groups (IV and IV’ in the figure).
The same conclusions can be drawn from Fig. 1c, which shows a consensus tree based on 10 different phylogenetic trees considering the relative frequency of the AFLP marker alleles in groups of wild lines (including Turkey I and II, Iran-Iraq III, Iran III’, the group IV Israeli lines, Israel IV’, the Jordanian, Syrian and Lebanese lines of group V) and in the two groups of domesticated genotypes.
Combining AFLP data from the two collections
Figures 1d, 1e and 1f reconsider the results of the same AFLP database used in Fig. 1b and 1c to illustrate the wild to domesticated genetic relationships. A different type of approach, the method known as principal coordinate (PC) analysis (Rohlf 1998), was used. The existence of different degrees of correlation among the allelic frequencies between pairs of genetic populations allows the extraction of successive components of total variability as axes along which the populations distribute. The similarity between the results of a phylogenetic tree and of a PC analysis should be close (Cavalli-Sforza and Piazza 1975). In PC analysis the first principal components frequently reflect the geographic distribution of the sample sites, therefore with this method genetic and geographical distances should be correlated and allow the alignment of the PC map with a geographic map. This is why PC analysis has been extensively used to follow the geographical distribution of human genes (Cavalli-Sforza et al. 1994). With the visual limitations imposed by the need to consider several two-dimensional maps to correlate all PC components extracted from a set of data, the analysis is useful to assign a geographical origin to crops, their mode of domestication included. PC maps supplement and uphold phylogenetic trees by presenting data in a different form; thus, when PC analysis produces the same information as phylogenetic trees, the overall result is highly reliable (Cavalli-Sforza et al. 1994). This is the case of the tetraploid wheat domestication data (Fig. 1d, 1e, 1f); the PC results support the findings reported in Fig. 1b and 1c. The field defined by the first three principal components suggests four major observations: (1) the relative topologies of domesticated lines and of their related groups of wild lines match those reported in the two trees of Fig. 1b and c, (2) the domesticated groups consisting of T. dicoccum and T. durum lines occupy distinct positions in the principal coordinate space, (3) the T. dicoccum landraces seem more related to the wild lines than those of T. durum, and (4) the wild lines of T. dicoccoides most related to both groups of domesticated tetraploid wheats were sampled in southeastern Turkey and in Iran-Iraq. All analyses carried out on the AFLP database of tetraploid wheats, while supportive of a common geographical origin of the hulled and free-threshing domesticated forms, do not exclude the possibility of a diphyletic domestication.
When the old set of AFLP data from Ozkan et al. (2002) was analyzed by the PC method (Fig. 1f) and compared to the results of Fig. 1e concerning the wild lines from Mori et al. (2003) and from B. Gill and A. Karagoz (see Materials and Methods), a very similar picture emerged, a finding strongly supporting the per se value of each of the two datasets. In Fig. 1f the Israel lines occupy two different topologies, a situation already evident from the work of Ozkan et al. (2002).
More information can, nevertheless, be extracted from the two AFLP databases when they are considered together and analyzed by the PC procedure (Fig. 1d). The first two principal components are extremely effective in dividing the T. dicoccoides lines of the western Fertile Crescent (groups IV, IV’ and V) from those of the central-eastern part (groups I, II, III and III’). Moreover, within the large central-eastern group (left part of the figure), the close relationships between the Turkish Karacadag lines (group II) and the domesticated accessions is evident. A looser but still significant genetic relatedness appears to characterize the genetic relationships among Turkish Kartal lines (group I) and the domesticated varieties.
Discussion
The publication of the paper by Heun et al. (1997) stimulated a discussion of southeastern Turkey as the place of origin of Western agriculture (Nesbitt and Samuel 1998). Later Lev-Yadun et al. (2000) summarized the evidence that the ranges of wild progenitors of cereals and legumes intersect in southeastern Turkey, circumscribing a core area that includes Karacadag, considered the cradle of agricultural innovation, a conclusion supported by the excavation of Neolithic sites in the core region at 10,500 cal BP (Lev-Yadun et al. 2000). The domestication of T. dicoccoides was traced back to the same core area by Ozkan et al. (2002) and was reported as monophyletic. In a recent comment by Salamini et al. (2004), the origin of domesticated tetraploid genotypes has been also referred to as possibly diphyletic, with two major phylogenetic tree topologies occupied by T. durum (free-threshing) and T. dicoccum (hulled) lines.
A diphyletic domestication was the conclusion made by Mori et al. (2003) who, based on chloroplast DNA (cpDNA) fingerprinting, have found two major organelle DNA lineages of domesticated emmer. When comparing the molecular phylogenies obtained from sets of polymorphisms recorded for different subcellular genomes, conclusions may differ due to variations in the rate of genome evolution. For plants, this is the case for comparisons involving cpDNA data and those obtained from nuclear markers. On average, the rate of variation observed in nuclear genomes is three times higher than for chloroplast genomes (Wolfe et al. 1987; Gaut et al. 1996; McDade et al. 2000).
Despite this, it was nevertheless interesting that the extensive investigation of Mori et al. (2003) did not reveal more that two cpDNA haplotypes which were selected during tetraploid wheat domestication. The first cpDNA lineage of Mori et al. (2003), consisting of a large group of haplotypes slightly different at some of several DNA sites, has counterparts in wild populations of the Kartal Dagi site, which is located at the border of the core area of Lev-Yadun et al. (2000). The most common group I haplotype variant (haplotype 10) is present in 39.6% of domesticated and in 4.0% of wild T. dicoccoides lines. The authors have, in addition, established that in the wild T. dicoccoides populations the second major chloroplast lineage of domesticated tetraploids is represented only by closely related haplotypes which are scattered at low frequency along the Fertile Crescent. This second major lineage is present only in 7.3% of the domesticated lines studied.
The research presented in this paper was mainly stimulated by the desire to compare the phylogenetic information from different sources. This is the reason why we used nuclear AFLP markers to analyze the same wild populations considered by Mori et al. (2003) and compared the results to those of Ozkan et al. (2002). The data reported in Fig. 1b justify this analysis; the two nuclear AFLP databases establish that two very different genetic taxa of T. dicoccoides exist, the western one colonizing primary habitats in Israel, Syria, Lebanon and Jordan, and the central-eastern race, frequently sampled in Turkey, and rarely in Iran and Iraq. Several phytogeographers have already reported the existence of two different T. dicoccoides races (Sachs 1953; Harlan and Zohary 1966; Rao and Smith 1968). However, only the central-eastern race played a role as progenitor of the domesticated germplasm (Fig 1c; Mori et al. 2003; Ozkan et al. 2002; Salamini et al. 2004). Moreover, the cumulative results from the two collections suggest that the Turkish Karacadag population, intermixed with some Iraq-Iran lines, has a topology consistent with that of the putative progenitor of domesticated genotypes. The Turkish Kartal population, belonging genetically to the central-eastern T. dicoccoides race, seems less related at the nuclear DNA level to the domesticated gene pool.
A general agreement on tetraploid wheat domestication emerges from the results presented in this paper, as well as from those reported by Mori et al. (2003) and Ozkan et al. (2002). A discrepancy is nevertheless evident on the local geographical scale, reflecting the difficulty of studying events which took place in the Neolithic; the chloroplast DNA data indicate the Kartal mountains while AFLP fingerprinting points to the Karacadag Range as the putative site of tetraploid wheat domestication. Similar discrepancies are usually noted when taxa are compared based on different single gene trees. In a well-presented review dealing with soybean interspecific differences, Doyle et al. (2003) introduce such a problem for cpDNA and nuclear DNA phylogenies, explaining that the incongruence of results may either depend on ancient introgressions or may be due to stochastic sorting of ancestral lineages. In our case, similar events have a certain probability of having played such a role since T. dicoccoides, although mainly self-fertilizing, also admits intra-specific crosses (Zohary and Hopf 2000). Thus, post-domestication introgression of wild germplasm into the domesticated gene pool, mediated by crosses in which the wild lines have accepted foreign pollen, may have resulted in the spreading of wild cpDNA haplotypes in domesticated lines. However, a diphyletic domestication is also supported by the data of Mori et al. (2003) and not only by the AFLP results; this should exclude a significant wild gene pool introgression into domesticated varieties.The detection of the opposite event, an introgression of domesticated germplasm into wild populations, was outside of the scope of this paper. However, the Mori et al. (2003) cpDNA haplotype 10 is a candidate for such a possibility, being very frequent in domesticated lines and rare in the wild gene pool.
An attempt to reconcile the T. dicoccoides nuclear and chloroplast DNA data may also be that the Karacadag region hosted representatives of the chloroplast group I haplotypes. As a matter of fact, one T. dicoccoides accession was indentified as carrying a type I chloroplast haplotype molecularly close to the haplotype 10 of Mori et al. (2003), which is largely present in domesticated accessions. However, the consideration of additional 48 wild lines from the Karacadag Mountains led the same authors to conclude in favour of the Kartal Mountains as source of the domesticated germplasm. When tested with nuclear DNA markers, the same T. dicoccoides accessions were less related to the cultivated gene pool than the Karacadag lines. This may result from incomplete information concerning the chloroplast DNA data; in the analysis only one free-threshing domesticated genotype was tested. If a larger collection of T. durum was to be analyzed, the results may provide evidence for the existence of more free-threshing specific chloroplast DNA haplotypes. Moreover, in the PC analysis presented in Fig. 1d, both domesticated sets of lines are more related to Karacadag than Kartal populations and thus we return to the belief that while the core area of Lev-Yadun et al. (2000) has played a clear role in the domestication of tetraploid wheat, the identification of a more precise location(s) within this area is still difficult. Factors contributing to keep this problem open are discussed in Salamini et al. (2002) and Mori et al. (2003).
References
Badr A, Muller K, Schafer-Pregl R, El Rabey H, Effgen S, Ibrahim HH, Pozzi C, Rohde W, Salamini F (2000) On the origin and domestication history of barley. Mol Biol Evol 17:499–510
Bar-Yosef O (1998) The natufian culture in the Levant, threshold of the origin of agriculture. Evol Anthropol 6:159–177
Cavalli-Sforza LL, Edwards AWF (1967) Phylogenetic analysis: models and estimation procedures. Evolution 32:550–570
Cavalli-Sforza LL, Piazza A (1975) Analysis of evolution: evolutionary rates, independence and treeness. Theor Popul Biol 8:127–165
Cavalli-Sforza LL, Menozzi P, Piazza A (1994) The history and geography of human genes. Princeton University Press, Princeton
Doyle JJ, Lucknow M (2003) The rest of the iceberg: legume diversity and evolution in a phylogenetic context. Plant Physiol 131:900–910
Dvorak J, Di Terlizzi P, Zhang HB, Resta P (1993) The evolution of polyploid wheats: identification of the A genome donor species. Genome 36:21–31
Dvorak J, Luo MC, Yang ZL, Zhang HB (1998) The structure of the Aegilops tauschii gene pool and the evolution of hexaploid wheat. Theor Appl Genet 67:657–670
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17:368–371
Felsenstein J (1989) PHYLIP - Phylogeny Inference Package (v3.2). Cladistics 5:164–166
Fitch WM, Margoliash E (1967) Construction of phylogenetic trees. Science 155:279–284
Gaut BS, Morton BR, McCaig BC, Clegg MT (1996) Substitution rate comparisons between grasses and palms: synonymous rate differences at the nuclear gene Adh parallel rate differences at the plastid gene rbcL. Proc Natl Acad Sci USA 93:10274–10279
Gopher A, Abbo S, Lev-Yadun S (2002) The “when”, the “where” and the “why” of the Neolithic revolution in the Levant. Doc Praehist 28:1–14
Harlan JR, Zohary D (1966) Distribution of wild wheats and barley. Science 153:1074–1080
Heun M, Schaefer-Pregl R, Klawan D, Castagna R, Accerbi M, Borghi B, Salamini F (1997) Site of Einkorn wheat domestication identified by DNA fingerprinting. Science 278:1312–1314
Jaccard P (1908) Nouvelle recherches sur la distribution florale. Bull Soc Vaud Sci Nat 44:223–270
Johnson BL (1975) Identification of the apparent B-genome donor of wheat. Can J Genet Cytol 17:21–39
Kislev ME (1980) Triticum parvicoccum sp. nov., the oldest naked wheat. Israel J Bot 28:95–107
Lev-Yadun S, Gopher A, Abbo S (2000) The cradle of agriculture. Science 288:1602–2603
Maier U (1996) Morphological studies of free-threshing wheat ears from a Neolithic site in southwest Germany, and the history of naked wheats. Veget Hist Archaeobot 5:39–55
McDade LA, Masta SE, Moody ML, Waters E (2000) Phylogenetic relationship among Acanthaceae: evidence from two genomes. Syst Bot 25:106–121
Moore AMT, Hillman GC, Legge AJ (2000) Village on the Euphrates. Oxford University Press, Oxford
Mori N, Ishii T, Ishido T, Hirosawa S, Watatani H, Kawahara T, Nesbitt M, Belay G, Takumi S, Ogihara Y, Nakamura C (2003) Origin of domesticated emmer and common wheat inferred from chloroplast DNA fingerprinting. 10th International Wheat Genetics Symposium, 1–6 September 2003, Paestum, Italy, pp 25–28
Nei M (1972) Genetic distance between populations. Am Naturalist 106:283–292
Nesbitt M, Samuel D (1996) From staple crop to extinction? The archaeology and history of the hulled wheats. In: Padulosi S, Hammer K, Heller J (eds) Hulled wheats. Proceedings of the 1st international workshop on hulled wheats, pp 41–100
Nesbitt M, Samuel D (1998) Wheat domestication: archaeological evidence. Science 279:1433
Ozkan H, Brandolini A, Schafer-Pregl R, Salamini F (2002) AFLP analysis of a collection of tetraploid wheats indicates the origin of emmer and hard wheat domestication in southeast Turkey. Mol Biol Evol 19:1797–1801
Rao PS, Smith LE (1968) Studies with Israel and Turkish accessions of Triticum L. emend. Var. dicoccoides (KORN) BOWDEN. Wheat Inform Serv 26:6–7
Reynolds JB, Weir BS, Cockerham CC (1983) Estimation of the coancestry coefficient: basis for a short-term genetic distance. Genetics 105:767–779
Rohlf FJ (1998) NTSYS-pc. Numerical taxonomy and multivariate analysis system. Applied Biostatistics, New York
Sachs L (1953) Chromosome behaviour in species hybrids with Triticum timopheevii. Heredity 7:49–58
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Salamini F, Ozkan H, Brandolini A, Schafer-Pregl R, Martin W (2002) Genetics and geography of wild cereal domestication in the near east. Nat Rev Genet 3:429–441
Salamini F, Heun M, Ozkan H, Wunder J (2004) On DNA markers, phylogenetic trees and mode of origin of crops. Genome 46:448–453
Van Zeist W, Bakker-Heeres JAH (1982) Archaeobotanical studies in the Levant. 1. Neolithic sites in the Damascus basin: Aswad, Ghoraifé, Ramad. Palaeohistoria 24:165–256
Wolfe KH, Li W-H, Sharp PM (1987) Rates of nucleotide substitution very greatly amomg plant mitochondrial, chloroplast, and nuclear DNAs. Proc Natl Acad Sci USA 84:9054–9058
Wright S (1978) Evolution and the genetics of populations. Variability within and among natural populations 4. University of Chicago Press, Chicago
Zohary D, Hopf M (2000) Domestication of plants in the old world. Oxford University Press, Oxford
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by H.C. Becker
Rights and permissions
About this article
Cite this article
Ozkan, H., Brandolini, A., Pozzi, C. et al. A reconsideration of the domestication geography of tetraploid wheats. Theor Appl Genet 110, 1052–1060 (2005). https://doi.org/10.1007/s00122-005-1925-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-005-1925-8