Abstract
A disomic chromosome addition line of tobacco, Nicotiana tabacum L., was established previously that possesses a single chromosome pair from N. africana [Merxm. and Buttler]. This addition chromosome carries a gene that confers increased resistance to severe strains of potato virus Y (PVY). Methods to increase the probability of gene transfer from alien chromosomes to tobacco (2n=48) are desired. In the research described here, the PVY resistance gene was transferred to a tobacco chromosome from the N. africana addition chromosome in seven independent cases. One introgression event was obtained using conventional backcrossing of the disomic addition line to N. tabacum cv. Petite Havana, while the remaining six events were obtained using a scheme that involved exposure of explants of the addition line to tissue culture. Twenty-six derived 2n=48 individuals heterozygous for PVY resistance were found to exhibit 24 bivalents or 23 bivalents + 2 univalents at metaphase I. Ovular transmission rates for the PVY resistance factor ranged from 25% to 52%, while pollen transmission rates were much lower, ranging from 0 to 39%. Fifty-one random amplified polymorphic DNA (RAPD) markers specific for the intact addition chromosome were identified and used to characterize derived 2n=48/PVY-resistant genotypes. Variability was observed among these plants with respect to the total number of N. africana RAPD markers that were present, which is an indication that crossing over was occurring within each of the seven introgressed chromosome segments. A limited molecular marker-assisted backcrossing experiment allowed for selection of a 2n=48/PVY-resistant individual that possessed only 6 of the 51 original N. africana RAPD markers. In vitro culture is potentially a valuable system for increasing the rate of alien gene transfer in tobacco, and the successful transfer of PVY resistance from N. africana may allow for an increased level and range of resistance to this virus in tobacco.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cultivated tobacco, Nicotiana tabacum L. (2n=48), is susceptible to crop loss caused by potato virus Y (PVY), a pathogen of great economic importance to the tobacco industry worldwide (Lucas 1975; Delon et al. 1993). Resistance to many isolates is conferred by the irradiation-induced recessive gene va from cv. Virgin A Mutante (Koelle 1961) and by its allelic forms found in several other cultivars (Wernsman 1992a). While partial resistance has also been found in gametoclonal variant NC602 (Witherspoon et al. 1991), no one source provides complete resistance to all strains of PVY, and additional sources of resistance would be valuable for increasing the range and level of resistance in N. tabacum .
Lucas et al. (1980) found an accession of N. africana [Merxm. and Buttler] (2n=46) to be immune to three strains of PVY, and Wernsman (1992b) subsequently developed a chromosome addition line, NC152 (2n=50), which possesses a pair of homologous chromosomes from N. africana . The addition line does not exhibit the extreme levels of PVY resistance of N. africana per se, but it does carry a gene(s) that provides resistance to the necrotic effects of severe strains of PVY (Campbell et al. 1994). NC152 may be resistant to some va -breaking isolates and possesses tolerance to several isolates of tobacco etch virus (unpublished observations). Resistance acts in an additive nature as the disomic addition line exhibits less stunting than the monosomic addition line.
The usual objective in introgression is to transfer a gene of interest from an alien species to a chromosome of the cultivated species’ genome with minimal amounts of accompanying foreign chromatin. This is often complicated by strong preferential bivalent pairing between the chromosomes of the recipient species’ genome and low levels of homology between the donor and recipient chromosomes. Gerstel et al. (1979) reported chromosome pairing in N. tabacum × N. africana hybrids to be of the “low-variable” type. Efforts by Witherspoon (1987) were not successful in isolating genotypes in which the N. africana addition chromosome of NC152 had recombined with a chromosome of the N. tabacum genome.
There is a strong need to develop methods that will facilitate alien gene transfer when the affinity for chromosome pairing is low. Strategies for effecting recombination with addition or substitution chromosomes in wheat have included the use of pollen or seed irradiation, mutations affecting homoeologous chromosome pairing, and whole-arm substitution brought about by misdivision of univalent chromosomes during meiosis (Riley et al. 1968; Sears 1981, 1993; Knott 1987; Friebe et al. 1996). In vitro culture frequently induces chromosome breakage and fusion in plant species (Lee and Phillips 1988; Kaeppler et al. 1998, 2000). Translocations were found to be the most frequent chromosomal abnormality in maize and oat plants regenerated from tissue culture (Benzion et al. 1986; Kaeppler et al. 1998). Such observations led Larkin and Snowcroft (1981) and Larkin et al. (1989) to propose in vitro culture as a possible means for introgressing desirable alien genes into the genomes of a cultivated crop species.
The first objective of this investigation was to determine the value of tissue culture relative to simple backcrossing as a means of transferring genetic material from the N. africana chromosome of NC152 to the N. tabacum genome. The goal was to identify multiple independent events in which the PVY resistance gene was transferred to a N. tabacum chromosome. Campbell et al. (1994) previously tagged the addition chromosome with a mutant dhfr transgene conferring resistance to the antibiotic methotrexate. Consequently, the determination of ovular dhfr transmission rates in BC1F1 families was used in an efficient preliminary screen to identify potential interchromosomal recombination events.
Tobacco breeding lines possessing disease resistance genes transferred from wild relatives have often had reduced commercial value, a consequence likely due to deleterious alien genes flanking the gene of interest (Chaplin et al. 1966; Legg et al. 1981; Johnson 1999; Linger et al. 2000). Young and Tanksley (1989) found the traditional backcross method to be very ineffective in reducing the size of an alien DNA segment flanking a disease resistance gene in tomato. This problem can be exacerbated when the donor chromosome is from a different species. The authors recommended the use of marker-assisted backcrossing to identify those individuals in which desirable recombination events had occurred. A second objective of this investigation was to identify a set of random amplified polymorphic DNA (RAPD) markers associated with the intact N. africana chromosome in NC152. This information was then used to determine the relative amounts of N. africana chromatin present in 48-chromosome PVY-resistant plants derived from multiple independent introgression events. Markers were used to determine the potential for crossing over within transferred chromosome segments and to select for reduced fragment sizes during backcrossing of the PVY resistance gene to an elite cultivar.
Materials and methods
Starting plant material and the generation of BC1F1 families
The PVY-resistant (PVYR) disomic chromosome addition line NC152 (2n=50) was developed by adding a single pair of N. africana chromosomes to PVY-susceptible (PVYS) tobacco cultivar McNair 944 (Wernsman 1992b). The addition chromosome was subsequently tagged with a mutant dhfr transgene that confers resistance to the antibiotic methotrexate (Mtx) to create line NC152-dhfr -996 (Campbell et al. 1994). BC1F1 families were derived from this line using two different schemes in order to determine the potential value of tissue culture for promoting recombination between the alien chromosome and the N. tabacum genome. Non-tissue culture-derived families were produced by first crossing 144 individual NC152-dhfr -996 plants with the PVYS/2n=48 cv. Petite Havana. Individual F1 plants were then backcrossed as females to Petite Havana to produce 144 independent BC1F1 families which, in most cases, were expected to segregate for the presence of the addition chromosome.
For the tissue culture regime, leaf discs from 12 different NC152-dhfr -996 plants were placed in tissue culture. Two hundred and sixty 1-cm2 leaf discs were initially placed on solid MS culture medium (Murashige and Skoog 1962) for 2 days and then transferred to shoot regeneration medium comprised of MS inorganic salts supplemented with 4.0 mg l−1 indole acetic acid (IAA), 2.5 mg l−1 kinetin, 30 g l−1 sucrose, 250 mg l−1 cefotaxime, 0.5 mg l−1 Mtx, 100 mg l−1 kanamycin, and 7 g l−1 agar. The discs were transferred to fresh medium every 14–21 days. Regenerated shoots were removed periodically and transferred to rooting medium consisting of MS inorganic salts plus 30 g l−1 sucrose and 7 g l−1 agar. One hundred and forty-one rooted R0 plants derived from 121 different leaf discs were selected and transferred to soil-filled pots in a growth room. Only one or two R0 plants per explant were retained, thus ensuring a high number of independent regenerates. These regenerates were then crossed to Petite Havana. Individual F1 plants were then backcrossed as females to Petite Havana to produce 141 BC1F1 families derived from tissue culture.
Identification of putative interchromosomal recombination events
The relative positions of dhfr and the PVY resistance factor on the addition chromosome were not known a priori. Due to the subtelomeric structure of the chromosome, however, it was predicted that they resided on the same chromosome arm. In the absence of interchromosomal recombination, it was expected that dhfr would be transmitted to BC1F1 individuals at a frequency of approximately 7–10% (Campbell et al. 1994; Lewis and Wernsman 2001). A much greater percentage of MtxR plants (close to 50%) was expected if a N. africana chromosome segment carrying dhfr had been transferred to a N. tabacum chromosome. The percentage of MtxR plants in each BC1F1 family was determined by germinating 121–847 surface-sterilized seeds per family on sterile medium consisting of MS inorganic salts (Murashige and Skoog 1962) supplemented with 1 mg l−1 Mtx and 7 g l−1 agar in 100×15-mm petri plates. A Chi-square test was used to determine if the frequency of putative introgression events obtained using the tissue culture scheme was significantly different from that observed for the non-tissue culture scheme.
It was hypothesized that dhfr had been transferred to a N. tabacum chromosome in families with 40–60% MtxR plants. Data were collected to determine if the PVY resistance gene was also transferred to a N. tabacum chromosome in these families. One hundred plants from each of these families were tested for resistance to both PVY and Mtx approximately 30 days after seeding (two true-leaf stage). Chi-square tests were used to determine if segregation differed significantly from 1:1 ratios.
The whole-plant response to Mtx was determined by placing three 0.70-cm-diameter surface-sterilized leaf discs per plant on solid MS culture medium supplemented with 4.0 mg l−1 IAA, 2.5 mg l−1 kinetin, 30 g l−1 sucrose, 7 g l−1 agar, and 1 mg l−1 Mtx. Approximately 14 days after plating, leaf discs were classified as MtxR (proliferation of shoots on medium) or MtxS (death of leaf disc).
PVY inoculation procedure
Plants were inoculated at the two true-leaf stage (approximately 30 days old) as described by Witherspoon et al. (1991) using a strain of PVY NN (NC isolate 78) maintained by our program. The nomenclature used to describe this strain reflects the necrotizing effects that it has on genotypes with or without the root-knot nematode resistance gene Rk (Gooding and Tolin 1973; Gooding and Lapp 1980). Plants were maintained in a growth room at 26±3°C and were evaluated 11–15 days post-inoculation. PVY NN causes severe veinal necrosis 8 days after inoculation on genotypes possessing no genetic resistance. While variation for amounts of stunting, leaf crinkling, and mosaic was usually present among the inoculated plants, plants were classified as PVYR or PVYS according to whether or not veinal necrosis was present.
Identification of RAPD markers associated with the intact N. africana addition chromosome
A total of 1,216 random decamer primers (Operon Technologies, Alameda, Calif. and University of British Columbia, Vancouver, B.C., Canada) were screened for their ability to reveal RAPD polymorphisms between NC152-dhfr -996 and a bulk of three 2n=48/PVYS tobacco lines (McNair 944, K326, and Petite Havana). DNA was isolated according to Johnson et al. (1995), except that a BIO 101 FastPrep machine (BIO 101, Holbrook, N.Y.) was used for tissue grinding. RAPD reactions were performed using a PTC-100 MJ Research Programmable Thermal Controller (MJ Research, Watertown, Mass.). Reactions were conducted in 22-μl volumes containing 15 ng genomic DNA, 10 mM Tris-HCl, pH 8.3, and 50 mM KCl (1× Stoffel buffer), 200 μM of each dNTP, 1.65 U Amplitaq DNA polymerase Stoffel fragment (Perkin Elmer, Foster City, Calif.), 4 mM MgCl2, 22 μg bovine serum albumin, and 20 ng of primer. Amplifications were performed for a total of 37 cycles. Template DNA was initially denatured at 94°C for 1 min and then subjected to 16 cycles of 92°C for 30 s, 50–0.5°C per cycle for 30 s, and 72°C for 2 min, followed by 21 cycles of 92°C for 1 min, 40°C for 1 min, and 72°C for 2 min; the final step was 5 m at 72°C. Reaction products were electrophoresed on 1.5% agarose gels containing 3 μl of 10 mg ml−1 ethidium bromide. The gels were run for 5–6 h at 60–70 V and visualized using a UV transilluminator. Allele sizes were determined using rflpscan ver. 2.1 gel analysis software (Scanalytics, Billerica, Mass.). The association of polymorphic RAPD markers with the N. africana chromosome was verified by genotying 24 BC1F1 individuals derived from the cross NC152-dhfr -996/McNair 944//McNair 944. These plants were also phenotyped for resistance to Mtx. Cosegregation analysis was performed on these genotypic and phenotypic data.
Isolation and characterization of 2n=48/PVYR plants
For BC1F1 families possessing putative introgression events, an additional 24 plants were tested for resistance to Mtx and PVY NN. The chromosome number and meiotic pairing were determined for each PVYR plant by examining pollen mother cells at metaphase I according to Burns (1982). Determinations were made on at least eight countable cells per genotype. To estimate the relative amounts of N. africana chromatin present in 2n=48/PVYR types, three to five 2n=48/PVYR individuals per family were genotyped at those RAPD loci where polymorphism existed between NC152-dhfr -996 and the bulk of three 2n=48/PVYS lines. Preference was given to the selection of MtxS/2n= 48/PVYR plants, although one MtxR/2n=48/PVYR plant was included per family. Each of the 2n= 48/PVYR plants that was genotyped was also crossed as a male and as a female to PVYS var. K326 to determine the pollen and ovular PVYR transmission rates. One hundred plants from each cross were inoculated with PVYNN at the two-leaf stage and evaluated using previously described methods.
Marker-assisted backcrossing
Progenies from crosses between a selected MtxS/2n=48/PVYR BC1F1 plant and K326 were used to initiate backcrossing of the PVY resistance factor to K326 and to determine the potential for using molecular markers for the selection of genotypes possessing reduced amounts of N. africana chromatin. The BC1F1 plant was crossed both as a male and a female to K326 and 40 progeny (20 from each cross) were initially genotyped. Only those RAPD markers that were present in the parental BC1F1plant were screened on progenies.
Results
Segregation of Mtx and PVY resistance in BC1F1 families
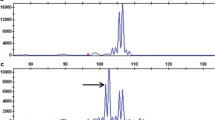
One hundred and forty-four BC1F1 families were generated using the non-tissue culture scheme. The percentage MtxR plants in each family was used as a preliminary screen to identify putative interchromosomal recombinatory events. The mean percentage survival was 7.6% with a range of 0.0–49.6%. While most all of the families exhibited dhfr transmission rates in the range of what was expected based on previous experience (Fig. 1), one family had a percentage survival that was conspicuously higher. The 49.6% survival rate in this family suggested that dhfr might have been transferred to a N. tabacum chromosome. This family is hereafter referred to as BC1F1 Family#1.
One hundred and forty-one BC1F1families were generated from tissue culture of 121 different NC152-dhfr-996 leaf discs. Consequently, the group consisted of at least 121 independently derived BC1F1 families. Average percentage survival was 9.2%, with a range 0.0–49.6%. Six families exhibited notably higher percentages (40.5–49.6%) of MtxR individuals (Fig. 2). These six percentages were also interpreted as possibly being caused by transfers of the N. africana chromosome segment bearing dhfr to a chromosome of the N. tabacum genome. Each of these six families was derived from a different leaf disc and are hereafter referred to as BC1F1 Families#2–7. A Chi-square test indicated that the frequency of putative introgression events obtained using the tissue culture scheme was significantly higher than that observed for the non-tissue culture scheme (χ2=25.9, P< 0.001).
To gain insight into whether the PVY resistance gene was also transferred to a N. tabacum chromosome in BC1F1 Families#1–7, 100 plants from each family were tested for both resistance to PVY NN and Mtx. The percentage transmission of PVY-resistance to the BC1F1individuals in these families ranged from 38% to 53% (Table 1), which is consistent with the resistance gene residing on a N. tabacum chromosome. The frequency of recombination between dhfr and the PVY resistance gene ranged from r=0.12 to r=0.24 (Table 1). These results were highly supportive of the hypothesis that a chromosome segment bearing both dhfr and the PVY resistance gene had been transferred to a N. tabacum chromosome in each of the seven families and that it was possible to obtain crossing over within the introgressed segments.
Identification of RAPD markers associated with the intact N. africana addition chromosome
Of 1,216 random decamer primers, 85 were initially identified that amplified a band that was present for NC152-dhfr -996 but absent for the 2n=48 bulk (McNair 944, Petite Havana, K326). Based upon repeatability and ease of interpretation, 48 primers that amplified 51 markers were selected for further use (Table 2). Twenty-four BC1F1 individuals from the cross NC152-dhfr -996/McNair 944//McNair 944 were then tested for the presence of these 51 RAPD markers and for dhfr . Complete cosegregation was observed between all RAPD markers and dhfr . These data, in combination with the strong segregation distortion that was observed (2 MtxR : 22 MtxS), were consistent with all markers residing on the addition chromosome.
Isolation and characterization of 2n=48/PVYR plants
PVYR/2n=48 plants were isolated from BC1F1 Families#1–7. Almost all PVYR plants possessed 48 chromosomes. While most 2n=48 plants had cells exhibiting 24 bivalents (Fig. 3a), cells with 23 bivalents + 2 univalents were also observed (Fig. 3b). Three to five PVYR/2n =48 individuals from each family were scored for the presence or absence of each of the 51 N. africana RAPD markers. Variability was observed among these plants with respect to the total number of N. africana RAPD markers that were present (range: 10–51, Fig. 4). MtxR plants possessed much greater numbers of N. africana markers (range: 45–51) than did MtxS plants (range: 10–25). Simple selection against dhfr apparently allowed for the elimination of a large fraction of alien germplasm. Based on these marker data, it was not possible to determine whether there were substantial differences among the seven introgression events with respect to the amount of N. africana chromatin that was initially transferred to the N. tabacum genome. Almost all of the genotyped plants had unique marker profiles, suggesting that a fair amount of crossing over was occurring within the introgressed segments. This statement might be biased, however, as preference was given to genotyping MtxS plants. The frequency of PVYR/MtxS plants within the BC1F1 families was actually low (Table 1), and it is not known whether this is due to a close linkage between dhfr and the PVYR gene or due to infrequent crossing over within the introgressed alien segments.
Each BC1F1 plant that was genotyped was also crossed both as a male and a female to K326 to determine the ovular and pollen transmission rates for PVY resistance. The average ovular transmission for the 26 BC1F1 plants was 39.9% (range: 25–52%) (Table 3). Pollen transmission was extremely low in most cases and averaged 7.0%. One plant, (BC1F1 Family#3, plant 65-8), transmitted PVY resistance through the pollen 48% of the time. This data point was out of line with the other pollen transmission rates given the relatively high number of N. africana alleles (17) that were present for this plant. It is noteworthy that the two plants with the next two highest pollen transmission rates were those possessing the fewest N. africana RAPD markers. Plant 12-3 (BC1F1 Family#1) possessed only ten N. africana markers and had a pollen transmission rate of 39%.
Marker-assisted backcrossing
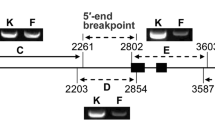
An experiment was undertaken to determine if the amount of N. africana chromatin present in BC1F1 plant 12-3 could be reduced further through molecular marker-assisted backcrossing. This plant was chosen because it possessed the least number of N. africana RAPD markers (ten) and exhibited a high transmission of PVY resistance through the pollen, indicating that it most likely carried the smallest introgressed N. africana segment. Twenty PVYR plants from the cross plant 12-3/K326 and 20 PVYR plants from the reciprocal cross were genotyped at those marker loci where a N. africana allele was present in plant 12-3 (in total ten loci). Of 40 plants, a single additional recombinant was identified that possessed only six of the 51 markers that were initially identified for the intact N. africana chromosome.
Discussion
The data presented in this paper demonstrate that a chromosome segment providing an increased level of resistance to a severe strain of PVY was transferred to tobacco from a N. africana addition chromosome in seven independent cases. The introgression lines do not exhibit the extreme type of resistance that is observed in N. africana per se or in F1 hybrids between N. africana and N. tabacum . Instead, the introgression lines develop mosaic symptoms with no necrosis when inoculated with PVY NN. Multiple genes on several chromosomes may condition the full resistance that is observed in N. africana . Alternatively, the resistance factor may function differently within the genomic background of N. tabacum .
Six introgression events were obtained through a system involving tissue culture, and one event was obtained using conventional backcrossing without the use of in vitro culture. Banks et al. (1995) also observed an increased rate of transfer of a barley yellow dwarf virus resistance gene to wheat from a Thinopyrum intermedium (Host) Barkw. & D.R. Dewey addition chromosome using methods involving tissue culture as compared to a procedure using genetically induced homoeologous pairing. The data provided by these investigators do not reveal whether these gene introgressions occurred via translocation or simple homoeologous crossing over. The increased number of introgression events obtained from schemes that include the exposure of explants to tissue culture points to a possible role of tissue culture-induced chromosome breakage. The precise mechanism of such breakage is not known. Analyses of chromosome structural changes in tissue culture-derived plants have revealed that breakpoints are not random. In grass species, most tissue culture-induced breakages occur within centromeric chromatin or between heterochromatic knobs and the centromeres (McCoy et al. 1982; Lapitan et al. 1984; Johnson et al. 1987; Lee and Phillips 1987; Jorgensen and Anderson 1989; Hang and Bregitzer 1993). Such observations led to the hypothesis that late replication of heterochromatic regions in culture causes chromosome bridge formation and subsequent breakage (Lee and Phillips 1988; Peschke and Phillips 1992; Kaeppler et al. 2000). Late replication may be due to the disruption of normal cell-cycle controls that prevent cell division prior to the completion of DNA replication (Phillips et al. 1994). Changes in genome methylation status also occur during culture and may play a role in chromosome breakage (Phillips et al. 1994; Kaeppler et al. 1998).
The positions of the RAPD markers identified in this research relative to each other on the N. africana chromosome are not known. Few studies have examined the distribution of RAPD markers in Nicotiana , most likely because of a general lack of polymorphism in N. tabacum and a lack of interest in the non-cultivated species. A linkage map based on an interspecific N. longflora /N. plumbaginifolia population exhibited a fair distribution of RAPD markers across the genome (Lin et al. 2001). A N. africana mapping population would provide further insight on the distribution of our RAPD markers, but only a single N. africana accession is available. Although markers are most useful when their map positions are known, it was assumed that the RAPD markers in our case provided, at the least, a fair estimate of the relative amounts of alien chromatin present in the derived materials.
Marker data indicated that crossing over was occurring within each of the introgressed N. africana chromosome segments. This suggests that each segment was integrated into a position that permitted synapsis with a N. tabacum counterpart possessing at least some degree of structural and sequence similarity. Additional data (not shown) based on intercrosses among derived homozygous lines have since demonstrated that at least three of the seven introgression events involved the same N. tabacum recipient chromosome. Similar results were observed in work in wheat by Banks et al. (1995). Here, six of seven “tissue culture-induced” translocations involved the same recipient chromosome (Banks et al. 1995; Hohmann et al. 1996). These results lend support to the possibility that some form of homoeologous recombination was involved with these gene transfers. Tissue culture-induced translocations might be considered to be similar to irradiation-induced translocations in that they likely result from breaks and reunions between random or semi-random chromosomes. In vitro culture might also, however, influence introgression by providing circumstances that increase the opportunity for homoeologous crossing over (i.e. chromosome loss, alteration of factors controlling normal bivalent pairing, etc.).
The amount of apparent crossing over that was occurring within the introgressed segments was unexpected given the presumed taxonomic distance between N. tabacum and N. africana . Previous work found very little affinity between the chromosomes of N. tabacum and N. africana (Gerstel et al. 1979; Witherspoon 1987). N. tabacum is a 2n=48 amphidiploid from section Genuinae that originated in South America (Goodspeed 1954), while N. africana (2n=46) is the only Nicotiana species to have been found on the continent of Africa. It has not been assigned to a section (Reed 1991), but cytological and biochemical observations suggest a relationship with the species of section Suaveolentes found in Australia and a few islands of the South Pacific (Merxmuller and Buttler 1975; Burk and Durbin 1978; Gerstel et al. 1979; Chen and Wildman 1981; Burns 1982). Evidence suggests a particularly close taxonomy with N. fragrans Hook. (Gerstel et al. 1979; Reed 1991).
The recombination that was occurring within the alien segments was used to our advantage. Molecular markers allowed the selection of PVYR genotypes possessing relatively smaller amounts of alien chromatin. Without genotypic selection, it is unlikely that a PVYR plant possessing this small number of N. africana markers would have been selected within the number of backcross generations that are usually used to transfer a disease resistance gene to an elite breeding line. Other investigations have demonstrated that recombination is often suppressed, but not necessarily prohibited, in chromosome segments introgressed from wild relatives (Paterson et al. 1990; Messeguer et al. 1991; Causse et al. 1994; Ganal and Tanksley 1996; Liharska et al. 1996). Additional effort may allow for further marker-based selection of PVYR tobacco genotypes possessing minimal amounts of N. africana genetic material. Closely linked markers may also be valuable in work to pyramid this resistance mechanism with other resistance genes. With further development, derived materials may allow for an increased level and range of genetic resistance to PVY in tobacco.
References
Banks PM, Larkin PJ, Bariana HS, Lagudah ES, Appels R, Waterhouse PM, Brettell RIS, Chen X, Xu HJ, Xin ZY, Qian YT, Zhou XM, Cheng ZM, Zhou GH (1995) The use of cell culture for subchromosomal introgressions of barley yellow dwarf virus resistance from Thinopyrum intermedium to wheat. Genome 38:395–505
Benzion G, Phillips RL, Rines HW (1986) Case histories of genetic variability in vitro: oats and maize. In: Vasil IK (ed) Cell culture and somatic cell genetics of plants, vol 3. Academic Press, New York, pp 435–448
Burk LG, Durbin RD (1978) The reaction of Nicotiana species to tentoxin. J Hered 69:117–120
Burns JA (1982) The chromosomes of Nicotiana africana Merxm.: a recently discovered species. J Hered 73:115–118
Campbell KG, Wernsman EA, Fitzmaurice WP, Burns JA (1994) Construction of a designer chromosome in tobacco. Theor Appl Genet 87:837–842
Causse MA, Fulton TM, Cho YG, Ahn SN, Chunwongse J, Wu K, Xiao J, Yu Z, Ronald PC, Harrington SE, Second G, McCouch SR, Tanksley SD (1994) Saturated molecular map of the rice genome based on an interspecific backcross population. Genetics 138:1251–1274
Chaplin JF, Matzinger DF, Mann TJ (1966) Influence of the homozygous and heterozygous mosaic-resistance factor on quantitative character of flue-cured tobacco. Tob Sci 10:81–84
Chen K, Wildman SG (1981) Differentiation of fraction 1 protein in relation to age and distribution of angiosperm groups. Plant Syst Evol 138:89–113
Delon R, Fisher CR, Blancard D (1993) Survey on tobacco viruses. CORESTA Inf Bull No 4:72–79
Friebe B, Jiang J, Raupp WJ, McIntosh RA, Gill BS (1996) Characterization of wheat-alien translocations conferring resistance to diseases and pests: current status. Euphytica 91:59–87
Ganal MW, Tanksley SD (1996) Recombination around the Tm2a and Mi resistance genes in different crosses of Lycopersicon peruvianum . Theor Appl Genet 92:101–108
Gerstel DU, Burns JA, Burk LG (1979) Interspecific hybridizations with an African tobacco, Nicotiana africana Merxm. J Hered 70:342–344
Gooding GV, Lapp NA (1980) Distribution, incidence and straits of potato virus Y in North Carolina. Tob Sci 24:91–94
Gooding GV, Tolin SA (1973) Strains of potato virus Y affecting flue-cured tobacco in the southeastern United States. Plant Dis Rep 57:200–204
Goodspeed TH (1954) The Genus Nicotiana. Chronica Botanica, Waltham, Mass.
Hang A, Bregitzer P (1993) Chromosomal variations in immature embryo-derived calli from six barley cultivars. J Hered 84:105–108
Hohmann U, Badaeva K, Busch W, Friebe B, Gill BS (1996) Molecular cytogenetic analysis of Agropyron chromatin specifying resistance to barley yellow dwarf virus in wheat. Genome 39:336–347
Johnson ES (1999) Identification and marker-assisted selection of a major gene for Phytophthora resistance, its origin, and effect on agronomic characters in tobacco. PhD thesis, North Carolina State University, Raleigh, N.C.
Johnson SS, Phillips RL, Rines HW (1987) Possible role of heterochromatin in chromosome breakage induced by tissue culture in oats (Avena sativa L). Genome 29:439–446
Johnson ES, Miklas PN, Stavely JR, Martinez-Cruzado JC (1995) Coupling-phase and repulsion-phase RAPDs for marker-assisted selection of PI 181996 rust resistance in common bean. Theor Appl Genet 90:659–664
Jorgensen RB, Anderson B (1989) Karyotype analysis of regenerated plants from callus cultures of interspecific hybrids of cultivated barley (Hordeum vulgare L). Theor Appl Genet 77:343–352
Kaeppler SM, Phillips RL, Olhoft P (1998) Molecular basis of heritable tissue culture-induced variation in plants. In: Jain SM, Brar DS, Ahloowalia BS (eds) Somaclonal variation and induced mutations in crop improvement. Current plant science and biotechnology in agriculture, vol 32. Kluwer, Dordrecht, pp 465–484
Kaeppler SM, Kaeppler HF, Rhee Y (2000) Epigenetic aspects of somaclonal variation in plants. Plant Mol Biol 43:179–2000
Knott DR (1987) Transferring alien genes to wheat. In: Heyne EG (ed) Wheat and wheat improvement, 2nd edn. American Society of Agronomy, Madison, Wis. pp 462–471
Koelle G (1961) Genetische analyse einer Y-virus (Rippenbraun) resistenten Mutante der Tabaksorte Virgin A. Zuchter 31:71–72
Lapitan NLV, Sears RG, Gill BS (1984) Translocations and other karyotypic structural changes in wheat x rye hybrids regenerated from tissue culture. Theor Appl Genet 68:547–554
Larkin PJ, Snowcroft WR (1981) Somaclonal variation—a source of variability from cell cultures for plant improvement. Theor Appl Genet 60: 197–214
Larkin PJ, Banks PM, Bhati R, Brettell RIS, Davies PA, Ryan SA, Snowcroft WR, Spindler LH, Tanner GJ (1989) From somatic variation to variant plants: mechanisms and applications. Genome 31:705–711
Lee M, Phillips RL (1987) Genomic rearrangements in maize induced by tissue culture. Genome 29:122–128
Lee M, Phillips RL (1988) The chromosomal basis of somaclonal variation. Annu Rev Physiol Plant Mol Biol 39:413–437
Legg PD, Litton CC, Collins GB (1981) Effects of the Nicotiana debneyi black root rot resistance factor on agronomic and chemical traits in burley tobacco. Theor Appl Genet 60:365–368
Lewis RS, Wernsman EA (2001) Efforts to initiate construction of a disease resistance package on a designer chromosome in tobacco. Crop Sci 41:1420–1427
Liharska T, Koornneef M, van Wordragen M, van Kammen A, Zabel P (1996) Tomato chromosome 6: effect of alien chromosomal segments on recombination frequencies. Genome 39:485–491
Lin TY, Kao YY, Lin S, Lin RF, Chen CM, Huang CH, Wang CK, Lin YZ, Chen CC (2001) A genetic linkage map of Nicotiana plumbaginifolia / Nicotiana longiflora based on RFLP and RAPD markers. Theor Appl Genet 103:905–911
Linger LR, Wolff ML, Wernsman EA (2000) Comparison of transgenic versus interspecific sources of TMV resistance conferred by the N gene. In: 39th Tobacco Workers’ Conf. Williamsburg, Virginia
Lucas GB (1975) Diseases of tobacco, 3rd edn. Biological Consulting Assoc, Raleigh, N.C.
Lucas GB, Gooding GV Jr, Sasser JN, Gerstel DU (1980) Reaction of Nicotiana africana to black shank, granville wilt, root knot, tobacco mosaic virus, and potato virus Y. Tob Sci 24:141–142
McCoy TJ, Phillips RL, Rines HW (1982) Cytogenetic analysis of plants regenerated from oat (Avena sativa) tissue cultures; high frequency of partial chromosome loss. Can J Genet Cytol 24:37–50
Merxmuller H, Buttler KP (1975) Nicotiana in der afrikanischen Namib-ein pflanzengeographisches und phylogenetisches. Ratsel Mitt Bot Munchen 12:91–104
Messeguer R, Ganal MW, de Vicente MC, Young ND, Bolkan H, Tanksley SD (1991) High resolution RFLP map around the root-knot nematode resistance gene (Mi ) in tomato. Theor Appl Genet 82:529–536
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio-assays with tobacco tissue cultures. Physiol Plant 15:473–497
Paterson AH, de Verna JW, Lanini B, Tanksley SD (1990) Fine mapping of quantitative trait loci using selected overlapping recombinant chromosomes in an interspecific cross of tomato. Genetics 124:735–742
Peschke VM, Phillips RL (1992) Genetic implications of somaclonal variation in plants. Adv Genet 30:41–75
Phillips RL, Kaeppler SM, Olhoft P (1994) Genetic instability of plant tissue cultures: breakdown of normal controls. Proc Natl Acad Sci USA 91:5222–5226
Reed SM (1991) Cytogenetic evolution and aneuploidy in Nicotiana. In: Tsuchiya T, Gupta PK (eds) Chromosome engineering in plants: genetics, breeding, evolution, part B. Elsevier, Amsterdam, pp 483–505
Riley R, Chapman V, Johnson R (1968) The incorporation of alien disease resistance in wheat by genetic interference with the regulation of meiotic chromosome synapsis. Genet Res 12:198–219
Sears ER (1981) Transfer of alien genetic material to wheat. In: Evans LT, Peacock WP (eds) Wheat science—today and tomorrow. Cambridge University Press, Cambridge, pp 75–89
Sears ER (1993) Use of radiation to transfer alien segments to wheat. Crop Sci 33:897–901
Wernsman EA (1992a) Sources of resistance to virus diseases. CORESTA Inf Bull 1992-3/4, pp 113–119
Wernsman EA (1992b) Varied roles for the haploid sporophyte in plant improvement. In: Stalker HT, Murphy JP (eds) Plant breeding in the 1990s. Proc Symposium Plant Breed 1990s. CAB Int, Wallingford, pp 461–484
Witherspoon WD (1987) Utilization of the haploid sporophyte as the selection unit in tobacco breeding. PhD thesis, North Carolina State University, Raleigh, N.C.
Witherspoon WD, Wernsman EA, Gooding GV Jr, Rufty RC (1991) Characterization of a gametoclonal variant controlling virus resistance in tobacco. Theor Appl Genet 81:1–5
Young ND, Tanksley SD (1989) RFLP analysis of the size of chromosomal segments retained around the Tm-2 locus during backcross breeding. Theor Appl Genet 77:353–359
Acknowledgements
This work was supported in part through a CORESTA study grant provided to the author.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P.S. Heslop-Harrison
Rights and permissions
About this article
Cite this article
Lewis, R.S. Transfer of resistance to potato virus Y (PVY) from Nicotiana africana to Nicotiana tabacum: possible influence of tissue culture on the rate of introgression. Theor Appl Genet 110, 678–687 (2005). https://doi.org/10.1007/s00122-004-1893-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-004-1893-4