Abstract
The detection of quantitative trait loci (QTLs) associated with UV-B resistance in rice should allow their practical application in breeding for such a complex trait, and may lead to the identification of gene characteristics and functions. Considerable variation in UV-B resistance exists within cultivated rice (Oryza sativa L.), but its detailed genetic control mechanism has not been well elucidated. We detected putative QTLs associated with the resistance to enhanced UV-B radiation in rice, using 98 BC1F5 (backcross inbred lines; BILs) derived from a cross between Nipponbare (a resistant japonica rice variety) and Kasalath (a sensitive indica rice variety). We used 245 RFLP markers to construct a framework linkage map. BILs and both parents were grown under visible light with or without supplemental UV-B radiation in a growth chamber. In order to evaluate UV-B resistance, we used the relative fresh weight of aerial parts (RFW) and the relative chlorophyll content of leaf blades (RCC). The BIL population exhibited a wide range of variation in RFW and RCC. Using composite interval mapping with a LOD threshold of 2.9, three putative QTLs associated with both RFW and RCC were detected on chromosomes 1, 3 and 10. Nipponbare alleles at the QTLs on chromosome 1 and 10 increased the RFW and RCC, while the Kasalath allele at the QTL on chromosome 3 increased both traits. Furthermore, the existence of both QTLs on chromosomes 1 and 10 for UV-B resistance was confirmed using chromosome segment substitution lines. Plants with Kasalath alleles at the QTL on chromosome 10 were more sensitive to UV-B radiation than plants with them on chromosome 1. These results also provide the information not only for the improvement of UV-B resistance in rice though marker-associated selection, but also for the identification of UV-B resistance mechanisms by using near-isogenic lines.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The depletion of the stratospheric ozone layer due to the emission of chlorofluorocarbons and other trace gases has resulted in increased solar ultraviolet-B (UV-B; 280–320 nm) radiation on the earth's surface (Blumthaler and Ambach 1990). Increased levels of UV-B radiation cause reduction in plant biomass, leaf area, chlorophyll content and photosynthesis rate, although large variations in response exist between and within species. A sizable number of reviews have appeared during the last two decades highlighting the effects of UV-B on plants (Teramura 1983; Allen et al. 1998; Tevini and Teramura 1989; Mackerness 2000; Searles et al. 2001; Hollósy 2002). Although differences in UV-B resistance have been observed in rice (Oryza sativa L.) varieties (Teramura et al. 1991; Dai et al. 1992, 1997; Kumagai and Sato 1992; Barnes et al. 1993; Sato and Kumagai 1993), the genetic control mechanism for UV-B resistance has not been well elucidated yet.
Most Japanese lowland rice varieties, classified as japonica subspecies, are more resistant to enhanced UV-B radiation, while most of the indica varieties are more sensitive (Sato and Kumagai 1993). Among Japanese lowland rice varieties, Sasanishiki was more resistant to supplemental UV-B radiation than Norin 1, although these varieties are closely related (Kumagai and Sato 1992; Hidema et al. 1996). Further studies revealed that two or more genes controlled the difference of resistance to enhanced UV-B radiation between these rice varieties (Sato et al. 1994). Furthermore, the accumulation of UV-absorbing compounds in leaf tissues is one important factor contributing to the genetic differences in UV-B resistance between Sasanishiki and Norin 1 (Sato and Kumagai 1997). However, the number and chromosomal location of the genes associated with the difference of UV-B resistance has not been reported yet.
Recently, DNA markers and their linkage map on chromosomes are powerful tools and methods for mapping of quantitative trait loci (QTLs) (Tanksley 1993; Jansen 1996). A number of QTL analyses have been conducted to identify the loci controlling agronomic and physiological traits in various crop plants. In rice, molecular genetic maps and markers have facilitated the identification of QTLs for the trait of interest (McCouch and Doerge 1995; Yano and Sasaki 1997). Furthermore, DNA marker-assisted selection has made it possible to develop near-isogenic lines and chromosome segment substitution lines for QTL regions affecting agronomic and physiological traits including resistance to environmental stress (Lin et al. 1998; Lin et al. 2000; Miura 2001, 2002; Ma et al. 2002). In addition, molecular cloning of genes at QTLs has been achieved by a map-based strategy (reviewed by Yano 2001). Thus, natural variation has become a useful resource for understanding the genetic control mechanism of traits such as UV-B resistance.
The objectives of this study were: (1) to identify QTLs for resistance to enhanced UV-B radiation of rice, using backcross inbred lines (BILs) derived from a japonica × indica cross, and (2) to confirm whether the detected QTLs affected the tolerance to enhanced UV-B radiation using chromosome segment substitution lines (SLs).
Materials and methods
Plant materials and cultivation method
Ninety eight backcross inbred lines (BILs; BC1F5) developed from self-pollinating BC1F1 (Nipponbare/Kasalath//Nipponbare) plants for five generations by the single-seed descent method were used for QTL analysis in this study. The japonica cultivar Nipponbare (Japanese lowland rice) is grown in southeast Japan, whereas the indica rice cultivar Kasalath originated from Assam, India. Chromosome segment substitution lines (SL-15, SL-92 and SL-77) containing Kasalath chromosomal segments at two putative QTL regions were selected from advanced backcross progenies using Nipponbare as the recurrent parent and Kasalath as the donor parent by marker-assisted selection (M. Yano and S.Y. Lin, unpublished data).
The seeds of BILs and their parents were imbibed in distilled water at 30 °C for 2 days after surface-sterilization with 70% ethanol solution and 1% sodium hypochlorite solution. Germinated seeds were sown on synthetic culture soil (Gousei-Baido No. 3, Mtsui-Toatsu Co., Tokyo, Japan) in plastic pots (15 × 6 × 10 cm). This synthetic culture soil contained 0.7 g of N, 1.2 g of PO3 and 0.6 g of KO2/kg. These pots were placed on trays and then were transferred to a large growth cabinet (Koitotron type KG: Koito Ind. Co., Tokyo, Japan). Rice plants were grown for 21 days under visible light with or without supplemental UV-B radiation under a 12 h light and 12 h dark period kept at 25 °C and 17 °C, respectively. The rice plants grown in pots were watered every day to maintain a constant level of soil moisture in the trays. The pots were moved daily so that the rice plants received equal amounts of light.
Supplemental UV-B radiation
Visible light in the growth cabinet was supplied by a combination of high-intensity discharge lamps [Toshiba DR400/T(L), Toshiba Ltd. Co., Tokyo, Japan], fluorescent tubes (Toshiba FR80HWA) and tungsten lamps (Toshiba RF 220 V 200 WH). The source of UV-B radiation was supplied with UV-emitting fluorescent tubes (Toshiba FL20SE; Toshiba Electric, Tokyo, Japan). These lamps were wrapped with 0.1 mm of cellulose diacetate film (Cadillac Plastic Co., Baltimore, Md., U.S.A.), to eliminate UV radiation with wavelengths below 290 nm (Hidema et al. 1996). The film was replaced every 10 days, because the UV-B radiation declined due to UV-induced aging of the film. The UV-B fluorescent tubes were adjusted to a distance of 30 cm from the plant canopy every day, to maintain the radiation of supplemental UV-B at a constant level. The fluence rate of photosynthetically active radiation (PAR) was measured with a data logger (L1-1000, Li-Cor; U.S.A.) and an L1-190SA sensor (Li-Cor.). The fluence rate of PAR at the plant canopy was adjusted to about 130 μmol photons m–2 s–1. The irradiance of UV-B was measured with a spectroradiometer (SS-25; Japan Spectroscopic Co., Tokyo, Japan). The dose of biologically effective UV-B radiation (UV-BBE) was calculated from the generalized plant action-spectrum of Caldwell (1971). In the phytotron, daily UV-BBE (KJm–2 day–1) was 17.8 and 0.6 under visible light with and without supplemental UV-B radiation, respectively.
Evaluation of UV-B tolerance
At 21 days after sowing, the fresh weights of aerial parts were measured immediately after the plants were removed from the growth cabinet. Leaf tissues (5 cm from the third leaf tip) were taken from plants for the analysis of total chlorophyll content, and the fresh weight was immediately measured. Total chlorophyll content in these leaf blades was determined with a Shimazu UV-300 spectrophotometer (Shimazu Ltd. Co. Kyoto, Japan) according to the method of Wintermans and Mots (1965). The samples were then cut into small pieces and immersed in 10 ml of 95% ethanol in the dark at room temperature for 3 days. Total chlorophyll content (mg/g fresh weight of leaf blade) was calculated according to the following formula: 20.0 × A649 + 6.1 × A665, where A649 and A665 are the absorbance at 649 nm and 665 nm, respectively. Relative fresh weight (RFW) and relative chlorophyll content (RCC) of BILs and SLs including parents were calculated as follows: the ratio (%) of these traits (mean values) with supplemental UV-B radiation to those without supplemental UV-B radiation. QTL analysis was performed using RFW and RCC data from BILs and both parents. Statistical comparisons were made between mean values per plot for with UV-B radiation and without UV-B radiation using an unpaired t-test, with means separated by the least significant difference (LSD). The data were analyzed with the EXCEL statistical analysis 2000 package (SSRI Ltd. Co., Tokyo, Japan).
Genotype data and QTL detection
Genotype data of 245 RFLP markers (http://rgp.dna.affrc.go.jp/publicdata/genotypedataBILs/genotypedata.html ) were used for QTL analysis. Because a specific model for BILs was not available, we analyzed our data as an F2 backcross and treated heterozygous markers as missing data. Linkage analysis was performed by using Mapmaker/EXP 3.0 with the Kosambi function (Lander et al. 1987). QTL analysis was performed using composite interval mapping (CIM) with the QTL Cartographer version 1.16 computer program (Basten et al. 2002). Five markers with the highest P-value (selected using a forward-backward stepwise regression) were added as cofactors in the CIM procedure (model 6 with a window size of 10 cM). A significant threshold was determined using 1,000 permutations and the LOD-threshold of 2.9 was established.
Results and discussion
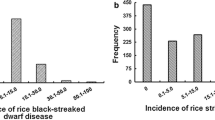
The effect of supplemental UVB radiation on the growth of rice was examined in Nipponbare, Kasalath and the BILs. Previously, supplemental UV-B radiation decreased biomass production and the total chlorophyll contents of leaf blades in UV-B-sensitive rice cultivars were more than the UV-B-resistant rice cultivars in a growth cabinet (Kumagai and Sato 1992, Sato and Kumagai 1993). The RFW of Nipponbare was 66%, while that of Kasalath was 19% (Fig. 1). The RCC of Nipponbare and Kasalath was 112% and 32%, respectively. The RFW and RCC of BILs observed continuous distribution with one peak ranging from 12% to 85% and from 8% to 122%, respectively (Fig. 1). The normal distribution of RFW, and the continuous distribution of RCC showed in BILs, suggested that multiple genes may regulate UV-B resistance in this population and that the alleles, which increased UV-B-resistance, were from Nipponbare.
Frequency distribution of relative fresh weight (RFW) and relative chlorophyll content (RCC) of backcross inbred lines (BILs). Both relative values (%) are the ratio of the value for the indicated parameter for plants grown with supplemental UV-B to the value for plants grown without supplemental UV-B radiation. The arrows indicate the column including the mean values of Nipponbare and Kasalath. RFW and RCC were calculated based on the mean values of seven plants
Based on composite interval mapping with a LOD-threshold of 2.9, a total of three putative QTLs were detected for both RFW and RCC (Table 1). Three putative QTLs for RFW were detected near the marker C1370 on chromosome 1, C746 on chromosome 3 and R2174 on chromosome 10. Furthermore, three putative QTLs for RCC, another indicator of UVB tolerance, were also detected on chromosomes 1, 3 and 10 (Table 1). These putative QTLs were located near the marker C122 on chromosome 1, C746 on chromosome 3 and R2174 on chromosome 10. Chromosomal locations of two QTLs for RCC on chromosomes 1 and 10 were coincident with those of the QTLs for RFW (Fig. 2). In these cases, the alleles from Nipponbare contributed to the increase in RFW and RCC (Table 1). On the contrary, Kasalath alleles at QTLs on chromosome 3 contributed to the increase in RCC (Table 1). This suggested that these putative QTLs located in three chromosomal regions were largely responsible for the difference in UVB tolerance between Nipponbare and Kasalath.
Chromosomal location of putative QTLs for UV-B resistance by composite interval mapping. Black and gray bars represent putative regions of QTLs for RFW and RCC, respectively. A reduction of 2.0 LOD value from the LOD peaks was used to define top and bottom borders of the confidence interval. Black and gray arrows indicate the nearest marker locus to the QTLs for RFW and RCC, respectively. Chromosomes with no QTLs are omitted from this figure
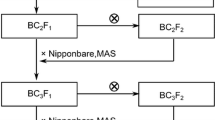
To confirm the existence of these QTLs on chromosomes 1, 3 and 10, we selected three substitution lines (SLs) from the advanced backcross progenies based on marker information for the putative QTLs (Fig. 3). In 01SL-92, 01SL-15 and 01SL-77, segments of chromosomes 1, 3 and 10 including three putative QTLs detected in the previous experiment were substituted with Kasalath segments. The effect of supplemental UV-B radiation on the fresh weight of aerial parts and chlorophyll content of leaves was examined in the parents and the SLs. Supplemental UV-B radiation reduced the RFW of Nipponbare by 26% and that of Kasalath by 80% (Table 2). The RFW of 01SL-92 and 01SL-77 was 15% and 28% lower than that of Nipponbare, respectively. Furthermore, supplemental UV-B radiation decreased the RCC of Nipponbare by 41% and that of Kasalath by 76%. 01SL-92 and 01SL-77 had a 3% and 20% lower RCC, respectively, than Nipponbare. On the contrary, the RCC of 01SL-15 was 10% higher than that of Nipponbare. Therefore, Kasalath alleles at QTLs located on chromosomes 1 and 10 reduced the RFW and RCC of Nipponbare, while the Kasalath allele at the QTL on chromosome 3 increased only the RCC of Nipponbare. Furthermore, the RFW and RCC of 01SL-77 were lower than those of 01SL-92, suggesting the major effect of the QTL on chromosome 10.
Graphical genotypes of chromosome segment substitution lines derived from a backcross of Kasalath to Nipponbare. Nipponbare was used as the recurrent parent. White and black bars on each chromosome indicate the Nipponbare and Kasalath segment, respectively. Nipponbare (open ellipses) and Kasalath (closed ellipses) QTL alleles contributed positively to RFW or RCC
Several mechanisms for resistance to enhanced UV-B radiation have been proposed, including physiological characters and morphological characters. Enhanced synthesis of UV-B absorbing compounds, leaf thickening, increased synthesis of cuticular wax and reduced leaf expansion reduce the UV-B quanta intercepted by leaf mesophyll tissues (Caldwell et al. 1983). We previously demonstrated that the accumulation of UV-B absorbing compounds in leaf blades was one of the characteristics determining the difference in UV-B resistance between the resistant rice variety Sasanishiki and the sensitive rice variety Norin 1 (Sato and Kumagai 1997). Subsequently, Hidema et al. (1997, 2001) and Hidema and Kumagai (1998) reported that the level of cyclobutyl pyrimidine dimer (CPD) photorepair might be a principal factor in determining the UV-B resistance in these rice cultivars. Furthermore, they also suggested that the difference in the DNA repair deficiency identified between the UV-B resistant variety Sasanishiki and the sensitive variety Norin 1 might have resulted from a defective photolyase (Hidema et al. 2000). On the other hand, enhanced free-radical oxygen scavenging-capacity to control oxidative damage incurred from UV-B radiation is also thought to contribute to UV-B tolerance (Elastner 1982). Dai et al. (1997) indicated that UV-B-induced injury might be associated with active oxygen species in rice. Near-isogenic lines for the QTLs detected in this study should be developed to characterize their association with UV-B absorbing compounds, with photolyase activity and with active oxygen species. The molecular mechanisms behind the reduction of chlorophyll content in leaf blades under UV-B radiation await future studies, including map-based cloning of the QTLs.
In field experiments conducted in four cropping seasons from 1994 to 1997, the decrease of the weight of fully ripened grains in rice plants grown under supplemental UV-B radiation was confirmed (Kumagai et al. 2001). The contribution of accumulated carbohydrate in the vegetative parts at heading to grain carbohydrate ranges from 0 to 40% under most conditions, while the contribution of photosynthesis after heading ranges from 60 to 100% (Yoshida 1981). Therefore, it is important to maintain the high photosynthetic activity in leaf blades after heading to increase the percentage of fully ripened grains. Leaf-discoloration of flag leaf after heading was more rapid in indica rice Kasalath than in japonica rice Nipponbare (Obara et al. 2001). One of the events during leaf discoloration is a decline in photosynthetic activity. Furthermore, leaf discoloration is faster in indica rice cultivars than in japonica ones and is also more rapid in warm regions than in cool regions (Yoshida 1981). The decrease in Rubisco, which is a key enzyme in photosynthesis, in the UV-sensitive rice cv Norin 1 grown under a high dose of UV-B radiation was exceptionally marked, and was not observed in UV-resistant rice cv Sasanishiki (Hidema et al. 1996). Therefore, the decrease of fully ripened grains in rice grown under enhanced UV-B radiation may primarily be due to decreased photosynthetic activity, indicating an inhibitory effect of a high dose of UV-B on photosynthesis. Our studies, which identified the QTLs for UV-B resistance, provide information to increase not only the percentage of fully ripened grains but also the yield of UV-B-sensitive rice. Furthermore, the reduction of fully ripened grain percentage is commercially very important, because the reduction might result in a change in grain protein content, which is a very important factor for the taste of rice grain. Therefore, information of QTLs for UV-B resistance in rice is important for future plant-breeding programs for producing high yielding and high quality rice.
References
Allen DJ, Nogues S, Baker NR (1998) Ozone depletion and increased UV-B radiation: is there a real threat to photosynthesis? J Exp Bot 49:1775–1788
Barnes PW, Maggard S, Holman SR, Vergara BS (1993) Intraspecific variation in sensitivity to UV-B radiation in rice. Crop Sci 33:1041–1046
Basten CJ, Weir BS, Zeng Z-B (2002) QTL Cartographer, version 1.16. A reference manual and tutorial for QTL mapping. Department of Statistics, North Carolina State University, Raleigh, North Carolina, USA
Blumthaler M, Ambach W (1990) Indication of increasing solar ultraviolet-B radiation flux in alpine regions. Science 248:206–208
Caldwell MM (1971) Solar UV irradiation and the growth and development of higher plants. In: Glese AC Photophysiology Vol. 6, Academic Press, New York, USA, pp 131–177
Caldwell MM, Robberecht R, Flint SD (1983) Internal filters: prospects for UV-acclimation in higher plants. Physiol Plant 58:445–450
Dai Q, Coronel VP, Vergara BS, Barnes PW, Quintos AT (1992) Ultraviolet-B radiation effects on growth and physiology of four rice cultivars. Crop Sci 32:1269–1274
Dai Q, Yan B, Huang S, Liu X, Peng S, Miranda MLL, Chavez AQ, Vergara BS, Olszyk DM (1997) Response of oxidative stress defense systems in rice (Oryza sativa) leaves with supplemental UV-B radiation. Physiol Plant 101:301–308
Elastner EF (1982) Oxygen activation and oxygen toxicity. Annu Rev Plant Physiol 35:15–44
Hidema J, Kumagai T (1998) UV-B-induced cyclobutyl pyrimidine dimer and photorepair with progress of growth and leaf age in rice. Photochem Photobiol 43:121–127
Hidema J, Kang H-S, Kumagai T (1996) Differences in the sensitivity to UVB radiation of two cultivars of rice (Oryza sativa L.). Plant Cell Physiol 37:742–747
Hidema J, Kumagai T, Sutherland JC, Sutherland BM (1997) Ultraviolet B-sensitive rice cultivar deficient in cyclobutyl pryimidine dimer repair. Plant Physiol 113:39–44
Hidema J, Kumagai T, Sutherland BM (2000) UV radiation-sensitive Norin 1 rice contains defective cyclobutane pyrimidine dimer photolyase. Plant Cell 12:1569–1578
Hidema J, Song I-K, Sato T, Kumagai T (2001) Relationship between ultraviolet-B sensitivity and cyclobutane pyrimidine dimer photorepair in rice. J Radiat Res 42:295–303
Hollósy F (2002) Effects of ultraviolet radiation on plant cells. Micron 33:179–197
Jansen RC (1996) Complex plant traits: time for polygenetic analysis. Trend Plant Sci 1:89–88
Kumagai T, Sato T (1992) Inhibitory effects of increase in near-UV radiation on the growth of Japanese rice cultivars (Oryza sativa L.) in a phytotron and recovery by exposure to visible radiation. Jpn J Breed 42:545–552
Kumagai T, Hidema J, Kang H-S, Sato T (2001) Effects of supplemental UV-B radiation on the growth and yield of two cultivars of Japanese lowland rice (Oryza sativa L.) under the field in a cool rice-growing region of Japan. Agr Ecosystems Environ 83:201–208
Lander ES, Green P, Abrahamson J, Barlow A, Daly MJ, Lincoln SE, Newburg L (1987) MAPMAKER: an interactive computer package for constructing primary genetic linkage maps of experimental and natural populations. Genomics 1:174–181
Lin HX, Yamamoto T, Sasaki T, Yano M (2000) Characterization and detection of epistatic interactions of three QTLs, Hd1, Hd2 and Hd3, controlling heading date in rice using nearly isogenic lines. Theor Appl Genet 101:1021–1028
Lin SY, Sasaki T, Yano M (1998) Mapping quantitative trait loci controlling seed dormancy and heading date in rice, Qryza sativa L. using backcross inbred lines. Theor Appl Genet 96:997–1003
Ma JF, Shen RF, Zhao ZQ, Wissuwa M, Takeuchi Y, Ebitani T, Yano M (2002) Response of rice to Al stress and identification of quantitative trait loci for Al tolerance. Plant Cell Physiol 43:652–659
Mackerness SAH (2000) Plant responses to ultraviolet-B (UV-B: 280–320 nm) stress: what are the key regulators? Plant Growth Reg 32:27–39
McCouch SR, Doerge RW (1995) QTL mapping in rice. Trends Genet 11:482–487
Miura K, Lin SY, Yano M, Nagamine T (2001) Mapping quantitative trait loci controlling low temperature germinability in rice (Oryza sativa L.). Breed Sci 51:293–299
Miura K, Lin SY, Yano M, Nagamine T (2002) Mapping quantitative trait loci controlling seed longevity in rice (Oryza sativa L.) Theor Appl Genet 104:981–986
Obara M, Kajiura M, Fukuta Y, Yano M, Hayashi M, Yamaya T, Sato T (2001) Mapping of QTLs associated with cytosolic glutamine synthetase and NADH-glutamate synthase in rice (Oryza sativa L.). J Exp Bot 52:1209–1217
Sato T, Kumagai T (1993) Cultivar differences in resistance to the inhibitory effects of near-UV radiation among Asian ecotype and Japanese lowland and upland cultivars of rice (Oryza sativa L.). Jap J Breed 43:61–68
Sato T, Kumagai T (1997) Role of UV-absorbing compounds in genetic differences in the resistance to UV-B radiation in rice plants. Breed Sci 47:21–26
Sato T, Kang HS, Kumagai T (1994) Genetic study of resistance to inhibitory effects of UV radiation in rice (Oryza sativa). Physiol Plant 91:234–238
Searles PS, Flint SD, Caldwell MM (2001) A meta analysis of plant field studies simulating stratospheric ozone depletion. Oecologia 127:1–10
Tanksley SD (1993) Mapping polygenes. Annu Rev Genet 27:205–233
Teramura AH (1983) Effects of ultraviolet-B radiation on the growth and yield of crop plants. Physiol Plant 58:415–427
Teramura AH, Ziska LH, Sztein AE (1991) Changes in growth and photosynthetic capacity of rice with increased UV-B radiation. Physiol Plant 83:373–380
Tevini M, Teramura AH (1989) UV-B effects on terrestrial plants. Photochem Photobiol 50:479–487
Wintermans JFGM, Mots A De (1965) Spectrophotometric characteristics of chlorophylls a and b and their pheophytins in ethanol. Biochem Biophys Acta 109:448–453
Yano M (2001) Genetic and molecular dissection of naturally occurring variations. Curr Opin Plant Biol 4:130–135
Yano M, Sasaki T (1997) Genetic and molecular dissection of quantitative traits in rice. Plant Mol Biol 35:145–153
Yoshida S (1981) Fundamentals of rice crop science. International Rice Research Institute, Manila
Acknowledgements
This work was supported by Grants-in-Aid (No. 10556075, 12480154) for Scientific Research from the Ministry of Education, Culture and Science, Japan (Y.F.T.K. and T.S.), and by a grant from the Ministry of Agriculture, Forestry and Fisheries of Japan (Rice Genome Project MP-1121) (T.U. and M.Y.). The authors thank Dr. Takeshi Hayashi (National Institute of Agrobiological Sciences), Dr. Jun Hidema (Graduate School of Life Science, Tohoku University) and Dr. Tadahiko Mae (Graduate School of Agricultural Science, Tohoku University) for their useful suggestions. We are grateful to Mr. Syoichi Musashi for his expert technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by D.J. Mackill
Rights and permissions
About this article
Cite this article
Sato, T., Ueda, T., Fukuta, Y. et al. Mapping of quantitative trait loci associated with ultraviolet-B resistance in rice (Oryza sativa L.). Theor Appl Genet 107, 1003–1008 (2003). https://doi.org/10.1007/s00122-003-1353-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-003-1353-6