Abstract
The hydrolase Notum (also known as Wingful) has been implicated in shaping the distribution gradient of the morphogen Wingless in Drosophila by modifying the Wingless-binding heparan sulfate proteoglycan (HSPG) core glypicans Dally and Dally-like. Here we report on the expression of a Notum homolog during the embryonic development of the spider Cupiennius salei. Notum is expressed in two to three stripes in the posterior region of the germband where new segments are formed. At this location no Wingless expression is present, suggesting that Notum may be involved in the regulation of another HSPG-binding morphogen, possibly Hedgehog. In older segments, however, expression of Notum and Wingless roughly coincides. In the appendages, Notum is expressed in ventral mesodermal cells, directly adjacent to the Wingless expressing ectodermal cells. This could indicate a role for the mesoderm in regulating morphogen gradient formation in the ectoderm.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Morphogen gradients play an important role in pattern formation during animal development [reviewed in Gonzalez-Gaitan (2003)]. One example is the gradient formed by the morphogen Wingless (Wg) in the fruitfly Drosophila melanogaster. Wg plays a role in segmentation, wing and leg development, and several other processes (e.g., Kubota et al. 2003; Siegfried and Perrimon 1994). In most of these contexts, Wg is secreted by a small number of cells and is bound immediately to the surface of these cells by heparin sulfate proteoglycans (HSPGs) present on the cell membrane (Gonzalez et al. 1991; van den Heuvel et al. 1989; Reichsman et al. 1996; Binari et al. 1997; Tsuda et al. 1999; Lin and Perrimon 1999; Baeg et al. 2001). Drosophila has four different kinds of HSPG core glypicans, two of which, Dally and Dally-like (Dlp), have recently been subject to intensive studies (Desbordes and Sanson 2003; Franch-Marro et al. 2005; Han et al. 2004, 2005; Kirkpatrick et al. 2004; Kreuger et al. 2004; Lin and Perrimon 1999; Tsuda et al. 1999). Novel results indicate that Wg proteins bound to Dally are presented to Wg receptors present on the same cell, whereas Wg bound to Dlp is transported to neighboring cells (Franch-Marro et al. 2005; Han et al. 2005). Binding of Wg to HSPGs is therefore essential for both Wg signaling and Wg gradient formation. The hydrolase Notum (also known as Wingful) is an enzyme that appears to interfere with both of these processes by cleaving the HSPG core proteins, thus removing bound Wg protein from the cell surface and making it unavailable for signaling and gradient formation (Gerlitz and Basler 2002; Giraldez et al. 2002; Kirkpatrick et al. 2004; Kreuger et al. 2004). By controlling the level of available Wg, Notum has an important influence on Wg signal transduction and on the shape of the Wg gradient.
Homologs of Wingless from Drosophila have been isolated from a number of arthropod species (e.g., Nulsen and Nagy 1999; Damen 2002; Hughes and Kaufman 2002; Janssen et al. 2004; Prpic 2004). Although the available expression patterns have already suggested that the role of Wingless may be conserved among all arthropods, the existence of a Wg gradient has yet to be demonstrated in arthropods other than Drosophila. Among the prerequisites for a conserved mode of Wg gradient formation would be the presence of factors involved in distributing Wg (e.g., Dlp) or shaping its distribution gradient (e.g., Notum). Comparative studies of such factors in other arthropod species, however, are lacking. As a first step, we report here on the cloning and expression analysis of a Notum homolog in the spider Cupiennius salei.
Results and discussion
We have searched publicly accessible sequence databases (at http://www.ncbi.nlm.nih.gov/BLAST/ and at http://www.bioinformatics.ksu.edu/BeetleBase/index.html) for sequences similar to Drosophila melanogaster Notum using the tblastx algorithm (Altschul et al. 1997). A Notum homolog is present in the genome of Drosophila pseudobscura, a species closely related to D. melanogaster. Sequences similar to Notum are also present in the genomic sequence of the insects Anopheles gambiae (mosquito), Apis melifera (honeybee), and Tribolium castaneum (flour beetle). No Notum homolog has been found in the available genomic sequence from another insect, Bombyx mori (silk moth), possibly because the Bombyx genome sequence is still incomplete. Notum-like sequences were also found in the genomes of several vertebrates including human, chicken, mouse and rat. The fugu genome apparently contains four Notum-like genes, but no Notum-like sequence has been found in the sequence available from zebrafish. Notably, the two completely sequenced genomes of the nematode worms Caenorhabditis elegans and C. briggsae lack Notum-like genes, indicating gene loss in the Nematoda. Nonanimal proteins with similarity to Notum are plant pectin acetylesterases (Giraldez et al. 2002; Gerlitz and Basler 2002). We isolated a homolog of Notum from the spider Cupiennius salei using a set of degenerate primers (Notum fw: TNG CNG GNW SNW SNG CNG GNG G; Notum bw: GNG CYT CRT CRA ANA RCC AYT G). A phylogenetic analysis (Fig. 1) using all available arthropod Notum protein sequences (including the sequence from Cupiennius), the vertebrate Notum-like sequences, and a selection of plant pectin acetylesterases, shows that the arthropod Notum proteins and the vertebrate Notum-like sequences form two separate groupings with significant statistical support. The spider sequence is included in the group containing the insect Notum homologs, and the corresponding gene is therefore designated as Cs-Notum (GenBank accession number: AJ871012).
Phylogenetic analysis of arthropod Notum protein sequences. The analysis included partial protein sequences from Tribolium castaneum (Tc), Apis melifera (Am), Drosophila melanogaster and D. pseudobscura (Dm, Dp), Anopheles gambiae (Ag), Cupiennius salei (Cs), fugu, human, mouse, rat, chicken and two selected plant pectin acetylesterases (from Arabidopsis and rice). Shown is the unrooted puzzle tree computed from 1000 intermediate trees produced with Puzzle (implemented in PAUP 4.0b10) (Strimmer and von Haeseler 1996; Swofford 2002)
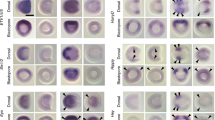
The spatio-temporal expression profile of Cs-Notum was then examined by whole-mount in situ hybridization. The expression level of Cs-Notum is extremely low and detection of specific signals was achieved only after staining for at least 20 h (the normal staining interval for strongly expressed genes like Distal-less or dachshund in Cupiennius is 1–2 h). Aside from expression in the stomodaeum (not shown), Cs-Notum mRNA is expressed during segment formation. Expression is seen in two or three discontinuous stripes at the posterior end in the region where new segments are formed (Fig. 2a–c, arrowheads). As it matures, each segment loses Cs-Notum expression, but expression is initiated again in older segments in the dorsal portion of the germband anterior to the parasegment borders (Fig. 2c, arrows). This expression pattern suggests a role of Notum in segment formation and segment border establishment. In the dorsal part of the older segments Notum may interact with Wg signaling, because Cs-Wingless is also expressed there (Damen 2002; Fig. 2d). However, Notum is unlikely to interact with Wg signaling in newly forming segments, because Cs-Wingless is not expressed there, but is expressed at the very posterior end of the germband (Damen 2002; Fig. 2d). Therefore, it is likely that in forming segments Notum interacts with another HSPG-binding morphogen. Among others, the morphogen Hedgehog has been shown to bind to HSPGs in Drosophila (The et al. 1999; Desbordes and Sanson 2003; Han et al. 2004). So far, we have not been able to isolate a hedgehog homolog from Cupiennius, but data from another chelicerate show that hedgehog is expressed in a segmental pattern in newly forming segments (Simonnet et al. 2004). We therefore speculate that the segmental Cs-Notum expression in forming segments may indicate an interaction of Notum with Hedgehog signaling in Cupiennius.
Expression of Cs-Notum during germband segmentation. a Posterior end of an embryo at approximately 90 h after egg laying (AEL). The very posterior end of the germband is indicated by the dotted line in all panels. Cs-Notum is expressed in two stripes (arrowheads) in the area where new segments are formed. b Posterior end of an embryo at approximately 150 h AEL. Three stripes of Cs-Notum expression can be seen (arrowheads). c Posterior end of an embryo at approx. 170 h AEL. Cs-Notum transcripts are present in three stripes in the area where the new segments form (arrowheads). Older segments have expression in their dorsal portion (arrows). d Simplified schematic drawing of the posterior end of the germband combining the expression patterns of Cs-Notum (black) and Cs-Wingless (gray); overlap of expression indicated by hatching. Please note that coexpression is inferred only from the fact that both genes are expressed anterior to the parasegment groove. Double-labeling was not possible due to technical problems with the extremely low level of Cs-Notum expression. All embryos are shown in ventral aspect, with anterior pointing to the left. Abbreviations: O3–O5, opisthosomal segment 2–5
Cs-Notum is expressed at low level in all prosomal appendages (Fig. 3). Expression of Cs-Notum is restricted to the ventral side of the appendages, suggesting a possible interaction of Notum with Wg gradient formation. However, Cs-Notum is expressed in the ventral mesoderm (Fig. 3c–f; arrow), while Cs-wg is expressed in the adjacent ventral ectoderm (Prpic et al. 2003). Thus, if the influence of Notum on Wg protein availability is conserved in the appendages of Cupiennius, then our data suggest that HSPGs and bound Wg are removed from the surface of Wg expressing ventral leg cells by Notum enzymes secreted from adjacent mesodermal cells. This could represent a fundamental difference between leg development in Cupiennius and leg imaginal disc development in Drosophila, where an influence of the mesoderm on pattern formation in the ectoderm has not yet been described.
Expression of Cs-Notum during appendage development. a, b Chelicera at approximately 150 and 200 h AEL, respectively. c, d Pedipalp at approximately 150 and 200 h AEL, respectively. e, f Walking leg at approximately 150 and 200 h AEL, respectively. Expression in the pedipalps and the legs is restricted to the ventral mesoderm (arrow in c–f), while the adjacent ectoderm does not express Cs-Notum (arrowhead in c–f). Expression in the chelicera is restricted to the proximal ventral mesoderm (a, b). All appendages are in anterior aspect and the proximal end is facing to the left. Abbreviations: che, chelicera; ped, pedipalp
The presence of Notum-like genes in the genomes of diverse arthropod species indicates an important, evolutionarily conserved role of this gene in the arthropods. The expression data in Cupiennius give support for an interaction of Notum with Wg signaling, since in older segments and in the developing appendages both genes are expressed in overlapping patterns or in directly adjacent tissues. However, the expression pattern of Notum in the newly forming segments is unanticipated, since no Wingless expression is found there. This may suggest the interaction of Notum with other HSPG-binding morphogens (e.g., Hedgehog).
References
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Baeg GH, Lin X, Khare N, Baumgartner S, Perrimon N (2001) Heparan sulfate proteoglycans are critical for the organization of the extracellular distribution of Wingless. Development 128:87–94
Binari RC, Staveley BE, Johnson WA, Godavarti R, Sasisekharan R, Manoukian AS (1997) Genetic evidence that heparin-like glycosaminoglycans are involved in Wingless signaling. Development 124:2623–2632
Damen WGM (2002) Parasegmental organization of the spider embryo implies that the parasegment is an evolutionary conserved entity in arthropod embryogenesis. Development 129:1239–1250
Desbordes SC, Sanson B (2003) The glypican Dally-like is required for Hedgehog signalling in the embryonic epidermis of Drosophila. Development 130:6245–6255
Franch-Marro X, Marchand O, Piddini E, Ricardo S, Alexandre C, Vincent JP (2005) Glypicans shunt the Wingless signal between local signalling and further transport. Development 132:659–666
Gerlitz O, Basler K (2002) Wingful, an extracellular feedback inhibitor of Wingless. Genes Dev 16:1055–1059
Giraldez AJ, Copley RR, Cohen SM (2002) HSPG modification by the secreted enzyme Notum shapes the Wingless morphogen gradient. Dev Cell 2:667–676
Gonzalez F, Swales L, Bejsovec A, Skaer H, Martinez Arias A (1991) Secretion and movement of Wingless protein in the epidermis of the Drosophila embryo. Mech Dev 35:43–54
Gonzalez-Gaitan M (2003) Endocytic trafficking during Drosophila development. Mech Dev 120:1265–1282
Han C, Belenkaya TY, Wang B, Lin X (2004) Drosophila glypicans control the cell-to-cell movement of Hedgehog by a dynamin-independent process. Development 131:601–611
Han C, Yan D, Belenkaya TY, Lin X (2005) Drosophila glypicans Dally and Dally-like shape the extracellular Wingless morphogen gradient in the wing disc. Development 132:667–679
Hughes CL, Kaufman TC (2002) Exploring myriapod segmentation: the expression patterns of even-skipped, engrailed, and Wingless in a centipede. Dev Biol 247:47–61
Janssen R, Prpic NM, Damen WGM (2004) Gene expression suggests decoupled dorsal and ventral segmentation in the millipede Glomeris marginata (Myriapoda: Diplopoda). Dev Biol 268:89–104
Kirkpatrick CA, Dimitroff BD, Rawson JM, Selleck SB (2004) Spatial regulation of Wingless morphogen distribution and signaling by Dally-like protein. Dev Cell 7:513–523
Kreuger J, Perez L, Giraldez AJ, Cohen SM (2004) Opposing activities of Dally-like glypican at high and low levels of Wingless morphogen activity. Dev Cell 7:503–512
Kubota K, Goto S, Hayashi S (2003) The role of Wg signaling in the patterning of embryonic leg primordium in Drosophila. Dev Biol 257:117–126
Lin X, Perrimon N (1999) Dally cooperates with Drosophila Frizzled 2 to transduce Wingless signalling. Nature 400:281–284
Nulsen C, Nagy LM (1999) The role of Wingless in the development of multibranched crustacean limbs. Dev Genes Evol 209:340–348
Prpic NM (2004) Homologs of Wingless and decapentaplegic display a complex and dynamic expression profile during appendage development in the millipede Glomeris marginata (Myriapoda: Diplopoda). Front Zool 1:6
Prpic NM, Janssen R, Wigand B, Klingler M, Damen WGM (2003) Gene expression in spider appendages reveals reversal of exd/hth spatial specificity, altered leg gap gene dynamics, and suggests divergent distal morphogen signaling. Dev Biol 264:119–140
Reichsman F, Smith L, Cumberledge S (1996) Glycosaminoglycans can modulate extracellular localization of the Wingless protein and promote signal transduction. J Cell Biol 135:819–827
Siegfried E, Perrimon N (1994) Drosophila Wingless: a paradigm for the function and mechanism of Wnt signaling. Bioessays 16:395–404
Simonnet F, Deutsch J, Queinnec E (2004) Hedgehog is a segment polarity gene in a crustacean and a chelicerate. Dev Genes Evol 214:537–545
Strimmer K, von Haeseler A (1996) Quartet puzzling: a quartet maximum likelihood method for reconstructing tree topologies. Mol Biol Evol 13:964–969
Swofford DL (2002) PAUP*. Phylogenetic analysis using parsimony (* and other methods), version 4. Sinauer Associates, Sunderland, MA
The I, Bellaiche Y, Perrimon N (1999) Hedgehog movement is regulated through tout-velou-dependent synthesis of a heparan sulfate proteoglycan. Mol Cell 4:633–639
Tsuda M, Kamimura K, Nakato H, Archer M, Staatz W, Fox B, Humphrey M, Olson S, Futch T, Kaluza V, Siegfried E, Stam L, Selleck SB (1999) The cell-surface proteoglycan Dally regulates Wingless signalling in Drosophila. Nature 400:276–280
van den Heuvel M, Nusse R, Johnston P, Lawrence P (1989) Distribution of the Wingless gene product in Drosophila embryos: a protein involved in cell–cell communication. Cell 59:739–749
Acknowledgements
We thank Diethard Tautz for his encouragement and support. We also thank Andrew Economou (UCL) for English language editing. The work of WD was supported in part by the Deutsche Forschungsgemeinschaft through SFB 572 of the Universität zu Köln. NMP was supported by a grant from the Deutsche Forschungsgemeinschaft (Grant TA99/19-2). The experiments described in this study comply with the current laws of the country in which they were performed (Germany)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prpic, NM., Damen, W.G.M. A homolog of the hydrolase Notum is expressed during segmentation and appendage formation in the Central American hunting spider Cupiennius salei. Naturwissenschaften 92, 246–249 (2005). https://doi.org/10.1007/s00114-005-0617-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00114-005-0617-1