Abstract
How sterile, altruistic worker castes have evolved in social insects and how they are maintained have long been central topics in evolutionary biology. With the advance of kin selection theory, insect societies, in particular those of haplodiploid bees, ants, and wasps, have become highly suitable model systems for investigating the details of social evolution and recently also how within-group conflicts are resolved. Because insect societies typically do not consist of clones, conflicts among nestmates arise, for example about the partitioning of reproduction and the allocation of resources towards male and female sexuals. Variation in relatedness among group members therefore appears to have a profound influence on the social structure of groups. However, insect societies appear to be remarkably robust against such variation: division of labor and task allocation are often organized in more or less the same way in societies with high as in those with very low nestmate relatedness. To explain the discrepancy between predictions from kin structure and empirical data, it was suggested that constraints—such as the lack of power or information—prevent individuals from pursuing their own selfish interests. Applying a multilevel selection approach shows that these constraints are in fact group-level adaptation preventing or resolving intracolonial conflict. The mechanisms of conflict resolution in insect societies are similar to those at other levels in the biological hierarchy (e.g., in the genome or multicellular organisms): alignment of interests, fair lottery, and social control. Insect societies can thus be regarded as a level of selection with novelties that provide benefits beyond the scope of a solitary life. Therefore, relatedness is less important for the maintenance of insect societies, although it played a fundamental role in their evolution.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Our understanding of the mechanisms of evolution has long been governed by metaphors such as “Nature red in tooth and claw” (the poet Alfred Lord Tennyson 1850), the “struggle for life (Darwin 1859), the “selfish gene” (Dawkins 1976), or “the four billion year war” (Majerus et al. 1996), which stress the apparent predominance of competition and rivalry in natural selection. However, even Darwin recognized that cooperation is a second important driving force in evolution, and later authors, including the Russian anarchist Peter Kropotkin (1902), particularly emphasized the role of “mutual aid” in evolution. Cooperation among selfish biological entities is the foundation of the major evolutionary transitions to stages of higher complexity (independent replicators → chromosomes, prokaryotes → eukaryotes, individual cells → multicellular organisms, individuals → animal societies; Maynard Smith and Szathmáry 1995).
The transition from egoistic individuals to animal societies, in particular to the societies of eusocial bees, wasps, ants, and termites, has long puzzled evolutionary biologists. How, in a Darwinian world in which success comes only to those who produce larger numbers of more viable offspring than others, can genes spread that cause a honey bee worker to refrain from egg laying and to instead suicidally sting predators that threaten the society? Forty years ago, the British biologist William D. Hamilton had an idea that revolutionized our understanding of how evolution works and at the same time gave a very clever answer to this fundamental question. According to Hamilton’s theory of kin selection (Hamilton 1963, 1964), genes, which under certain conditions suppress an individual’s reproduction, can spread in a population when the respective individual helps its relatives to rear their offspring and in this way produces more replicas of the gene than by its own reproduction. This can be expressed as Hamilton’s rule, C<rB, where C is the cost to the helper (the average number of own offspring the helper could have produced instead of helping), B is the benefit to the recipient of the help (the number of the recipient’s offspring produced because of the helper’s assistance), and r is the relatedness between helper and recipient. Though alternative explanations of the origin of eusociality were proposed in the early days of kin selection theory (e.g., Lin and Michener 1972; Alexander 1974), it is undisputed today that kinship is a major force in the evolution of social behavior in the Hymenoptera (Hölldobler and Wilson 1990; Bourke and Franks 1995; Crozier and Pamilo 1996) and, more generally, also in most other group-living animals (e.g., Griffin and West 2003).
Haplodiploidy and eusociality
Due to haplodiploid sex determination, relatedness between sisters is particularly close in the Hymenoptera. As in other animals, mothers pass on copies of half of their genes to any given daughter, and sisters therefore differ to some extent in the maternally inherited genes. In contrast, sisters are identical in all their genes inherited from their father, because haploid males transmit copies of all their genes to their daughters. Sisters are therefore completely identical in the genes they inherited from their father and share 50% of the genes inherited from their mother, resulting in an overall relatedness of 1/2(1+0.5), i.e., 0.75. Because females transmit copies of only half of their genes to their own offspring (r=0.5), they are more closely related to their sisters than to their sons or daughters (see Table 1). Females can therefore gain extraordinarily high indirect fitness benefits from helping their mothers to rear female sexual sisters. Under certain initial conditions, e.g., when solitary virgins produce males and mated females in social groups produce females, this may lead to the evolution of a reproductive division of labor. Hamilton’s theory therefore sheds light on the circumstances that may have favored the multiple evolution of eusociality in ants, social bees, and wasps (e.g., West-Eberhard 1975; Wade 1979; Gadagkar 1985; Bourke and Franks 1995; Crozier and Pamilo 1996; Queller and Strassmann 1998).
Together with the advance of a gene-centered view of evolution (Dawkins 1976), the close genetic relationship among Hymenopteran sisters has sometimes led to an initial, unjustified fixation on genetic relatedness as the dominant factor underlying social evolution in insects. The findings of eusociality in clonal aphids (e.g., Aoki 1987) and of inbreeding and high relatedness in the colonies of naked mole-rats (Reeve et al. 1990; but see Braude 2000) and thrips (Chapman et al. 2000) may have added to this relatedness-biased interpretation of sociality. For example, in the 1970s, several authors attempted to explain the evolution of eusociality in diplodiploid termites by suggesting that relatedness asymmetries similar to those in the haplodiploid Hymenoptera may arise through tight linkage of genes to the sex chromosome or through repeated cycles of inbreeding and outbreeding (Bartz 1979; Lacy 1980). Neither is common in those clades of extant termites that are presumed to be closest to the ancestral species in which eusociality evolved (Crozier and Luykx 1985).
The development of highly variable genetic markers, which allow us to accurately determine nestmate relatedness (Queller et al. 1993), stimulated a large number of genetic studies, which substantiated that the 3/4 relatedness hypothesis is not true for all Hymenopteran societies and contributed to the view that relatedness cannot be everything. Nevertheless, the other parameters in Hamilton’s equation, the costs and benefits of helping, are still widely neglected in studies on social insects, probably because they are much more difficult to quantify (Bourke and Franks 1995).
Kinship not only plays a fundamental role in the origin of sociality, but the genetic composition of animal societies is also thought to affect numerous details of social life, such as the allocation of resources, the partitioning of reproduction, and the occurrence and expression of conflict in the society. Differences in kin structure, resulting from variation in the queen’s mating frequency or the number of reproductive queens per colony, might also influence the performance of the society under changing environmental conditions or its disease resistance (Wade 1982; Keller and Reeve 1994; Boomsma and Ratnieks 1996). Social organization might therefore be expected to strongly depend on the genetic relationships among colony members.
While a number of studies support a strong influence of kin structure in some contexts (e.g., Trivers and Hare 1976; Peters et al. 1999; Reeve et al. 2000; Tarpy 2003), others have failed to detect a clear association between social and genetic structure (e.g., Woyciechowski and Warakomska 1994; Heinze 1995; Brown and Keller 2000; Hammond et al. 2003). In the following, we describe how kin structure is expected to affect life history traits in social Hymenoptera and briefly examine the evidence for or against such an influence. We then summarize recent ideas about constraints that prevent individuals and societies from reacting to variation in kin structure. Applying multilevel selection theory, we show that such constraints may ultimately be beneficial, as is easily understood when societies are seen from a group-level perspective.
Kin structure and sex allocation
The combination of Fisher’s sex ratio theory and Hamilton’s kin selection theory, as pioneered by Trivers and Hare (1976), has proven extremely fruitful and initiated a large number of subsequent studies on sex allocation patterns in social Hymenoptera (for reviews, see e.g., Bourke and Franks 1995; Crozier and Pamilo 1996; Mehdiabadi et al. 2003). Whereas, in most animals, parents can maximize their fitness by allocating resources equally to male and female offspring and the evolutionary stable sex ratio is therefore almost generally 1:1 (Fisher 1930), the relatedness asymmetries (relatedness of workers to female vs male sexuals) resulting from haplodiploidy lead to changed sex-ratio optima for workers in social Hymenoptera. The close relatedness among sisters (0.75) is offset by the much lower relatedness to the haploid brothers (0.25; Table 1). If the mother produced a 1:1 sex ratio, a female would be similarly related to her mother’s as to her own offspring (0.5), and the inclusive fitness benefits for helpers arising from haplodiploidy would disappear (assuming that producing own offspring is as costly as helping). Female Hymenopteran helpers therefore favor a 3:1 sex ratio, i.e., they benefit from helping most if they can manipulate sex allocation so that three times more resources are invested in female than in male sexuals. Population-level sex ratios in Hymenoptera are indeed often female-biased, suggesting that workers control the allocation of resources to the brood (Trivers and Hare 1976; Bourke and Franks 1995; Crozier and Pamilo 1996). This finding also convincingly supports kin selection theory.
When a queen mates multiply (polyandry), the average relatedness of a worker to female sexuals decreases (because some of them are now the worker’s half-sisters with r=0.25, Table 1), while the relatedness to brothers remains unchanged. At high effective mating frequencies, the average relatedness of workers to female sexuals approaches 0.25, the relatedness asymmetry vanishes and the sex-ratio optima of queen and workers converge. The same applies when a colony contains several related queens (polygyny). Population-level sex ratios are indeed less female-biased in polygynous than in monogynous species (Trivers and Hare 1976; Bourke and Franks 1995; Crozier and Pamilo 1996).
Sex allocation ratios may differ among colonies when queen mating frequencies or the number of queens per nest vary between colonies within a population. Split sex ratio theory (Boomsma and Grafen 1990, 1991) predicts that societies with fewer queens than the population average and societies with queens with a lower than average mating frequency benefit most from focusing on the production of female sexuals, whereas the others produce males. In remarkable agreement with theory, single-queen nests of the primitively eusocial bee Augochlorella striata composed of a mother and her daughters reared a more female-biased sex ratio than colonies consisting only of sisters (Mueller 1991). Split sex ratios have also been documented in the facultatively polyandrous wood ant Formica truncorum, where colonies with a singly-mated queen consistently produced female sexuals over four consecutive years, while colonies with a multiply mated queen produced males (Sundström 1994). Similarly, in populations of facultatively polygynous Leptothorax acervorum, polygynous colonies produced a more male-biased sex ratio than monogynous colonies (Chan et al. 1999). Formica queens produce male and female sexual offspring in equal numbers, but male larvae disappear from colonies with a singly mated queen (Sundström et al. 1996). In Leptothorax, workers apparently do not destroy male brood in response to varying relatedness asymmetries but rear more sexuals and fewer workers from female larvae in monogynous colonies (Hammond et al. 2002).
This substantial support for the role of kin structure in sex allocation is at odds with a growing number of studies, which fail to detect the predicted variation in relatedness asymmetries associated with split sex ratios. For example, colony sex ratios vary with the number of queens, but not relatedness asymmetry, in the ant Pheidole pallidula (Fournier et al. 2003) and appear to be independent of kin structure in several other species (Helms 1999; Fjerdingstad et al. 2002). Finally, in dense populations of the monogynous, monandrous ant Leptothorax nylanderi genetically heterogeneous colonies may arise through colony take-over by a young founding queen. In such usurped colonies, workers care for sexuals to which they are not related and they thus do not benefit from manipulating resource allocation. Nevertheless, these heterogeneous colonies produce mostly males, whereas genetically homogeneous colonies produce female sexuals (Foitzik and Heinze 2000, Foitzik et al. 2003).
Such split sex ratios without the expected variation in relatedness asymmetries have been explained from variation in ecological parameters, such as resource availability (Nonacs 1986; Rosenheim et al. 1996), ongoing queen–worker conflict about sex allocation (Banschbach and Herbers 1996), and variable value of female sexuals (Brown et al. 2002). It must be acknowledged that sex allocation is a complex phenomenon that is likely affected by numerous parameters, but one might nevertheless wonder whether the present plethora of explanations for such a ubiquitous phenomenon is appropriate.
Kin structure and worker reproduction
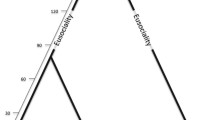
In most social Hymenoptera, workers cannot mate and lay fertilized eggs, but in many species they have retained the capability of producing males from unfertilized eggs. Workers are more closely related to their own sons (r=0.5) than to their brothers (r=0.25) and in monogynous, monandrous societies, a worker is even more closely related to the sons of other workers, her nephews (r=0.375), than to the sons of the queen (Hamilton 1972; Table 1). Instead of manipulating sex allocation, workers in a monogynous, monandrous society could therefore increase their inclusive fitness by producing their own sons (Fig. 1) or by helping to rear the sons of other workers. Note that worker reproduction was advantageous to the workers even if the production of one worker-derived male would need slightly more resources than the production of one queen-derived male. The queen, being more closely related to her own sons than to her grandsons, will typically be selected to prevent worker reproduction.
Egg-laying worker of the ant Platythyrea punctata. Workers are typically more closely related to their sons arising from unfertilized eggs than they are to their brothers. Note that in some populations of this species, unmated workers may produce diploid offspring from unfertilized eggs by thelytokous parthenogenesis (Heinze and Hölldobler 1995)
Kin theory does not make predictions as to how this queen–worker conflict about the origin of males will be resolved, but one might expect that the power of queens and workers varies with colony size. While in small societies, the queen can possibly prevent worker reproduction by direct aggression and egg eating (“queen policing”, Ratnieks 1988), we would expect workers to be free to reproduce in larger societies in which such direct queen control is not possible.
As shown above, multiple mating by the queen strongly changes nestmate relatedness and, as a result, also the workers’ interests about sex allocation. Polyandry affects the workers’ fitness optima concerning the origin of males in a similar way. Workers are always most closely related to their own sons, but at queen mating frequencies above 2, they are more closely related to their brothers than to the sons of an average worker nestmate. This should lead to worker policing, i.e., workers are expected to destroy the eggs laid by other workers (Ratnieks 1988; Monnin and Ratnieks 2001).
In agreement with kin structure, unsanctioned worker reproduction appears to be more common in monogynous, monandrous meliponine bees but extremely rare in monogynous, polyandrous honeybees (Tóth et al. 2002a, 2000b; but see Tóth et al. 2003). However, in ants, maternity analyses using microsatellites or other genetic markers give only scant evidence for workers being capable of producing males in colonies with a queen, regardless of colony size and queen number. With a few exceptions, the queen has “won” this conflict about the origin of males. In the presence of a functional queen, worker egg laying and male production appears to be restricted to a small minority of species (Bourke 1988; Choe 1988). For example, morphologically specialized “large workers” produce males in the subterranean ant Crematogaster smithi (Heinze et al. 1995). The queen and the brood presumably eat most of the eggs, but some survive and develop into males (Heinze et al. 2000).
In contrast, workers in queenright colonies almost generally appear to oppose egg laying by other workers, again regardless of queen mating frequency and colony size. In honeybees, the few workers that develop their ovaries in the presence of the queen are attacked by their nestmates and their eggs are eaten (Ratnieks and Visscher 1989; Visscher 1996; Ratnieks 1993), as predicted from the high mating frequency of honeybee queens. However, such worker policing has also been observed in a number of social insects, in which it is not always expected from relatedness alone, including hornets (Foster et al. 2002), ants with normal caste dimorphism (Iwanishi et al. 2003), ants without a morphologically differentiated queen caste (Monnin and Ratnieks 2001), clonal Cape honeybees (Pirk et al. 2003), and the clonal ant Platythyrea punctata (Hartmann et al. 2003) (Fig. 1). In particular in the latter species, where all individuals are genetically identical, worker policing is not associated with relatedness benefits (Greeff 1996).
Kin structure, division of labor, and disease resistance
Mating is risky and energetically expensive for the young queens, especially when it occurs during a nuptial flight. Multiple mating therefore poses unnecessary costs and in most species of social insects queens mate only once. However, queens of honeybees, vespine wasps, Pogonomyrmex harvester ants, and leaf-cutting ants mate regularly with numerous males (e.g., Villesen et al. 1999; Murakami et al. 2000; Palmer and Oldroyd 2000; Gadau et al. 2003; Wattanachaiyingcharoen et al. 2003), and in other taxa, some queens mate singly and others multiply (e.g., Sundström 1994; Sanetra and Crozier 2001; Fjerdingstad et al. 2002).
To explain this paradox, it was suggested that higher mating costs might be somehow compensated by benefits arising from the increased genetic variance among the offspring of polyandrous queens, such as an improved performance under a broader range of ecological conditions or a higher disease resistance (Sherman et al. 1988; Keller and Reeve 1994; Crozier and Fjerdingstad 2001). However, as with multiple mating in females of other animals (e.g., Keller and Reeve 1995; Yasui 1998), it has been surprisingly difficult to substantiate any benefits associated with polyandry in social insects. For example, multiply mated Lasius niger queens produced neither more nor qualitatively better sexual offspring than singly mated queens (Fjerdingstad et al. 2002, Fjerdingstad2004).
Some of the best support for the genetic variance hypothesis comes from studies with bees. Bumblebee colonies with singly inseminated queens fared worse when infected with the parasitic protist Crithidia bombi than colonies with queens that were artificially inseminated with a mixture of semen from many males. Unfortunately, most bumblebee queens mate only once in nature (Baer and Schmid-Hempel 1999). In obligatorily polyandrous honeybees, however, the evidence for advantages from multiple mating is at present ambiguous. It has long been recognized that patrilines may differ in their propensities to take over special tasks, such as pollen foraging, undertaking, or water collecting (Frumhoff and Baker 1988; Robinson and Page 1989; Robinson 1992). However, this apparent specialization does not always directly result in a benefit for polyandrous colonies. For example, in some studies, the genetic diversity of a colony had no effect on pollen diversity (Woyciechowski and Warakomska 1994), honey yield, or colony size (Neumann and Moritz 2000). In another study, multiple mating was found to be associated with larger pollen and honey stores and a higher production of sexuals (Fuchs and Schade 1994). Similarly, data concerning disease resistance are conflicting: in several studies infestation rates with various pathogens did not differ significantly between colonies with singly and multiply inseminated queens (Woyciechowski et al. 1994; Page et al. 1995), but Tarpy (2003) reported that the variance in prevalence of chalkbrood disease is lower in polyandrous colonies.
In general it appears that, at least in species in which multiple mating is facultative, mating frequency has little impact on colony performance other than concerning sex allocation. In contrast, the transition from single to obligatory multiple mating appears to be often correlated with a massive increase in mature colony size and presumably also other evolutionary innovations (Murakami et al. 2000; Villesen et al. 2002), giving genetic diversity a new role in colony homeostasis (see below). Nevertheless it is remarkable that the social structure of insect societies is quite stable against any effects from varying relatedness: division of labor, communication, or nest defense are organized in more or less the same way in societies of clonal or monogynous/monandrous social insects as in those of related, highly polygynous, or even unicolonial species with extremely low nestmate relatedness (Mirenda and Vinson 1981; Herbers 1982; Hölldobler and Wilson 1990; Hartmann and Heinze 2003).
Constraints, costs and levels of selection
To explain this discrepancy between predictions from kin structure and empirical data, researchers have repeatedly evoked constraints that prevent individuals from pursuing their own egoistical interests. These include physiological or morphological constraints: workers that over the course of evolution have lost their ovaries, perhaps because worker reproduction has been selected against in an ancestral polyandrous species, will no longer be able to produce males even when queens in the extant species mate only once. Workers may also lack the physical power to enforce their interests against the often much larger queen (e.g., Beekman et al. 2003), or they may not have the information necessary to manipulate sex allocation or to discriminately replace haploid, but not diploid queen-laid eggs by their own eggs (e.g., Keller and Nonacs 1993; Reeve 1998). Even the concept of inhibitive queen pheromones, abandoned by many researchers after Keller and Nonacs (1993) argued against the evolutionary stability of such manipulation, might be applicable under some conditions and could, for example, explain why hornet workers are incapable of enforcing their own reproductive interests against the queen (Foster et al. 2002).
Constraints may provide a proximate answer to the question of why kin structure is not sufficient to explain conflict resolution. Given the enormous plasticity of social insects, including the reversibility of sociality (Wcislo and Danforth 1997), we wonder whether some of these constraints would not disappear in evolution if they were indeed highly disadvantageous. For example, in the ant tribe Formicoxenini, worker egg laying in queenright colonies seems to be extremely rare in most species, but regular male production by workers appears to have evolved at least in the slave-maker Protomognathus americanus (Foitzik and Herbers 2001). The presence of rudimentarily or fully developed spermathecae in several other slave-making ants clearly shows that even severe anatomical constraints are sometimes not irreversible (Heinze 1996). Therefore, ultimate explanations are necessary to understand why the social structure of insect societies is robust against variations in relatedness. Such answers may be provided within a kin selection framework when the inclusive fitness interests of all nestmates are considered simultaneously, taking all costs and benefits of the various actions into account. That constraints may ultimately be beneficial is perhaps more intuitively understood when societies are seen from a group-level perspective.
The often-criticized idea of group selection has been the focus of one of the largest controversies in evolutionary biology (Reeve and Keller 1999). Its application to social insects is not new and has resulted in the fruitful, though much debated “superorganism concept” (e.g., Wilson 1971; Seeley 1989; Hölldobler and Wilson 1990; Moritz and Southwick 1992). Darwin (1871) had already applied group selection arguments to explain the evolution of altruism in social insects and humans. The scientists who initiated the modern study of ecology and behavior were influenced as much by pre-Darwinian notions of harmony as by the theory of natural selection. They often assumed that higher-level units function adaptively and that characters may evolve for the good of the group, species, or even the ecosystem (e.g., Wynne-Edwards 1962). In the 1960s, however, a consensus emerged that natural selection almost never operates above the level of the individual (Williams 1966). The process of group selection proposed by Darwin, though theoretically possible, was seen to be highly improbable in the real world and it was suggested that the concept of adaptation should be restricted to the level of the individual. This is where, for many scientists, the heretic group selection episode ends. It is often forgotten, however, that in the 1970s a theoretical framework of multilevel selection theory emerged that could withstand the earlier criticisms (Price 1970, 1972; Hamilton 1970; Wade 1985; Wilson 1997a, 1997b; Mayr 1997a). These models—often referred to as “new group selection”, “intrademic group selection”, or “trait-group selection”—partition fitness into within- and between-group components, generally by using a covariance approach (Price 1972; Frank 1997).
Insect societies and multilevel selection
The continued controversy about group selection is caused by the vague usage of the term “individual selection” (Wilson 1997b). In multilevel selection models it is defined as natural selection within groups (e.g., Haldane 1932; Maynard Smith 1964; Williams 1966; Price 1970, 1972). However, outside multilevel selection theory, “individual selection” has taken on a much broader range of definitions. Sometimes it is defined as an individual’s inclusive fitness, even though Hamilton (1975) realized that inclusive fitness theory includes a component of group selection. Using the terms “within-group selection” and “between-group selection” avoids such ambiguities (Wilson 1997b). Whenever a population is subdivided into groups, multilevel selection theory treats the single group as an evolutionary unit within which natural selection can occur. Within-group selection promotes traits that maximize the relative fitness of individuals within the group. The evolution of traits that are not favored by within-group selection may be explained by selection operating at a different level of the biological hierarchy. When this approach is used, between-group selection immediately becomes a significant and empirically well-documented evolutionary force [see Goodnight and Stevens (1997) for a review of laboratory experiments, Frank (1985, 1990) for sex ratio theory in Hymenoptera, and Frank (1996) for reduced virulence in parasites and diseases]. It also appears to be an ideal methodology to address the lack of a correlation between social structure and relatedness, which cannot be explained by within-group selection. Note that Hamilton’s kin selection theory and group selection are mathematically equivalent and in the end make the same predictions. However, it may be easier to understand the evolution of insect societies and why relatedness is not the only important parameter shaping social life by assuming a group perspective.
Social insects meet the requirements needed so that between-group selection can contend with the normally much stronger within-group selection; in particular, within-group variance is smaller than the between-group variances. This can be accomplished by several mechanisms (see, e.g., Price 1970; Leigh 1983, 1991):
-
1.
The exchange between groups needs to be low. Social insect colonies are indeed closed societies in which nestmate recognition reduces mixing between groups.
-
2.
All members of a group should have a common descent. Social insect colonies, with some rare exceptions, consist of families, mostly parent–offspring associations.
-
3.
The number of group members needs to be smaller than the number of competing groups to limit within-group variability relative to between-group variability. This latter requirement has, to our knowledge, not yet been considered for social insects. Insect societies may contain hundreds of thousands of individuals, while the number of colonies in a population is typically much smaller. However, what counts is only the number of reproductives within the society. Paralleling the germ–soma separation in multicellular organisms, the sterility of workers and soldiers in species with large colonies may help to reduce within-group variability. In contrast to previous explanations for the observed association between caste sterility and large colony size (e.g., Bourke 1999; Jeon and Choe 2003), sterility might thus be a cause rather than a consequence of large group sizes.
Hence, a multilevel selection approach might offer important insights about the robustness of insect societies against variations in relatedness. Furthermore, it solves the controversy about the level of selection (Keller and Reeve 1999; Reeve and Keller 1999) and links social insects with other major transitions in evolution that form new hierarchical levels. Such new levels of selection may arise when the cooperation among independent units provides benefits for all partners and represents very successful evolutionary breakthroughs that allow the occupation of new niches (Maynard Smith and Szathmáry 1995). According to Mayr (1997a, 1997b) the term “group selection” is justified under such conditions, because there are group phenotypes and the group as a whole shows adaptive traits that are more than the sum of the traits of individuals. Beneficial novelties linked to group living and emergent properties also exist in social insects.
Sociality in several animals, such as cooperatively breeding birds and mammals and colonial spiders, may be favored by various advantages of group living, such as an increased food availability or a reduced predation pressure (e.g., Solomon and French 1997; Emlen 1997; Cockburn 1998; Uetz 2001; Krause and Ruxton 2002). However, such groups do not generally meet the requirements for between-group selection as the variance within a group can be high compared with the variance between groups, and therefore they do not form units of selection. They often comprise open societies with individuals that are not derived from a common descent (Fig. 2). In contrast, social insects—where group living probably originated for the same reasons as in other animal groups—could evolve beyond simple groups because the common interests of group members were aligned through common ancestry, which also facilitated the evolution of altruism (Fig. 2). This stresses the importance of relatedness for the evolution of groups as new units in the biological hierarchy. Subsequently, factors such as intraspecific competition may have selected for increasing group size and new properties emerged that characterize the new level of selection and shifted the balance between relatedness and group advantages to group advantages as the driving forces in evolution. Examples for such emergent properties are given in the next section. Continued competition among groups might have favored the transition from single to obligatory multiple mating, as this appears to be often correlated with a massive increase in mature colony size (Murakami et al. 2000; Villesen et al. 2002).
Scenario for the evolution of group living. Predation pressure and competition for increased food acquisition favors the formation of animal groups. In most species these groups do not arise by common descent (different colors) and the groups are open (dashed circles) allowing migration between groups (bowed arrow). This contrasts with social insects (same color within circle; circles enclosed by solid line). Therefore, the former cannot become levels of selection as in the social insects where competition between groups arises (↔) and selects for increased group size. This leads to the evolution of novelties, such as division of labor and homeostasis, which present evolutionary breakthroughs that convert the groups to units of selection. Conflicts can arise within such groups through genetic variability (different colors), for example caused by polygyny, which may increase group size. Between-group selection now favors groups with mechanisms that prevent or suppress conflict (evolution of worker sterility, lack of kin discrimination)
Evolutionary breakthroughs and emergent properties in insect societies
Insect societies represent a level of organization above the individual organism which are characterized by their own specific morphology, behavior, internal organization, and life history pattern.
Division of labor, in which individuals specialize on subsets of the tasks performed by a colony, is one of the most prominent emergent features of social insect behavior (Fig. 3). This capacity to conduct all operations concurrently instead of sequentially clearly distinguishes a colony from individual organisms (Oster and Wilson 1978). As in engineering, performing operations in parallel makes systems more efficient and resilient against perturbations. Furthermore, behavioral flexibility within these colonies allows task-switching and provides the basis for an adaptive allocation of tasks according to needs (Gordon 1996; Beshers and Fewell 2001). Although it is not clear whether castes are a cause or effect of large colony size (Karsai and Wenzel 1998; Bourke 1999; Anderson and Ratnieks 1999; Anderson and McShea 2001), group living is certainly a prerequisite for division of labor to evolve. Intraspecific (Thomas and Elgar 2003) as well as interspecific analyses (Karsai and Wenzel 1998; Bourke 1999; Anderson and Ratnieks 1999; Anderson and McShea 2001) have shown that colony size and division of labor/task specialization are correlated.
Royal cell of the fungus-growing termite Macrotermes bellicosus with the queen, the king, and their sterile workers, showing extreme morphological differentiation, division of labor, and alignment of reproductive interests. All colony tasks, except for reproduction, are performed by the sterile workers and soldiers (not shown). The queen has evolved into an “egg-laying machine”
Social homeostasis makes insect colonies resilient against external disturbances. Division of labor with a large worker force guarantees redundancy and back-up systems (Oster and Wilson 1978). If some individuals fail or die, others can take over and continue the work. This assurance that investments are not wasted is even discussed as a driving factor in the evolution of insect sociality (Queller 1989, 1994; Gadagkar 1990).
The stabilization of climatic conditions in the nests of some species with large worker numbers enables them to inhabit habitats with extreme climates (Jeanne 1975; Kaspari and Vargo 1995). An active adjustment of nest temperature and humidity is known from honeybees and fungus-growing termites. Honeybees constantly regulate their core nest climate by fanning and the intake of water (Lindauer 1954; Heinrich 1993). In mound-building, fungus-growing termites, regulation is more indirect but also requires large worker numbers. The structure of Macrotermes bellicosus mounds is adapted to the local climate in such a way that nest temperature is kept at 30°C, with fluctuations of less than 2°C during the course of a year (Lüscher 1961; Korb 2003; Fig. 4). Termites thus provide optimal conditions for their symbiotic partner, a cultivated fungus. Similar mechanisms probably exist in the higher fungus-growing ants (Heinrich 1993) as well as in less spectacular cases such as wood ants (Coenen-Stass et al. 1980; Horstmann 1983, 1990) and several wasps (Jeanne 1975; Wenzel 1991).
Defendable fortress of a termite mound as an example of emergent properties on the colony level. The structure of Macrotermes bellicosus mounds is adapted to local conditions to achieve constant nest temperatures: a mound in the savanna, b mound in the forest (Korb and Linsenmair 2000)
The centralized nests of social insects largely improve their ability to defend the colony and the brood against predators. Living inside wood probably provides an even better protection, as indicated by a very low proportion of soldiers in all wood-living termites (Deligne et al. 1981; Henderson 1998). Some of the most protective nests are built by species with large colonies, which present a high premium for predators. The massive mounds of termites are like fortresses that impede the attack of generalist predators (Deligne et al. 1981; Noirot and Darlington 2000; Fig. 4).
Self-organization processes without centralized control add new qualities to colonies and make colonies qualitatively different from simple groups (Bonabeau et al. 1997; Queller and Strassmann 1998; Camazine et al. 2001). For example, pheromone trails become efficient recruiting mechanisms only above a certain threshold group size (Beekman et al. 2001) and the waggle and tremble dances of honeybees also present group-level adaptations that improve the foraging efficiency of the colony as a whole (Seeley 1995, 1997). Similar collective decisions are also made during a move to a new nest site, where colonies can quickly reach a decision about the single best nesting site among several alternatives (Seeley 1995; Franks et al. 2002). This sharing and collective processing of information by certain insect societies is one of the reasons warranting the epithet “superorganism” (Franks et al. 2002).
Conflict resolution in groups
As in other major transitions, group stability may provide the ultimate explanation for reproductive harmony in insect societies despite the existence of conflicts resulting from kin structure (Maynard Smith and Szathmáry 1995). Group-level adaptations can become so dominant that the exact values of relatedness only play a minor role. Between-group selection may thus result in the evolution of mechanisms that prevent the expression of within-group conflict. From a kin selection perspective, on average all individuals ultimately benefit from self-restraint or mutual policing (e.g., Ratnieks 1988; Ratnieks and Reeve 1992). However, information about such overall averages is like information about net forces (Sober and Wilson 2002). With a multilevel selection approach and a partitioning into a within- and a between-group components it might be easier to recognize and measure the relevant factors.
The resolution of conflicts among lower-level entities has been studied in the context of genomes and multicellular organisms (Buss 1987; Michod and Roze 1997; Michod 1999; Pomiankowski 1999). Conflicts always arise when the variability within groups increases and the reproductive interests of the group members are not aligned (Fig. 5). Multicellular organisms reduce the potential of variability within an organism by germ–soma separation (Buss 1987; Maynard Smith and Szathmáry 1995; Michod 1999). Likewise, the complete sterility of workers and soldiers in social insects may serve a similar function by increasing the heritability of fitness at the colony level while at the same time reducing the potential of conflict within insect societies. As there are only few individuals that can reproduce, the interests of all other nestmates are aligned to favor the well-being and reproduction of the reproducers (Fig. 3).
Conflict and conflict resolution in social insects. Social insect societies typically do not consist of clones due to sexual reproduction, which causes genetic variability. If individuals differ in relatedness patterns, conflict arises. Such conflicts can be resolved by reducing the potential for conflicts and aligning the interests of colony members through the evolution of sterile workers, a lack of mechanisms to discriminate among differently related nestmates, and/or a lack of information for distinguishing male and female offspring. Remaining conflicts that actually do break out in the colony are resolved by social control through colony members, which favors the evolution of pheromonal control as honest signaling or enforces the colony’s interest by policing
In multicellular organisms, cheating cells may occasionally arise, which multiply at the expense of the rest of the organism (Nunney 1999). In analogy to such cancer cells, the uncontrolled reproduction by certain thelytokous workers in the Cape honeybee Apis mellifera capensis, which finally leads to the death of the colony, has been referred to as “social cancer” (Oldroyd 2002). However, this comparison is misleading, as uncontrolled reproduction only occurs after Cape honeybees have invaded the nests of another subspecies, Apis mellifera scutellata (Martin et al. 2002). In contrast to cancer, uncontrolled reproduction in Cape honeybees therefore does not arise from within a colony and instead constitutes a case of parasitism (e.g., Neumann et al. 2001; Pirk et al. 2003).
Within an insect colony of sterile workers the reproductive interests of colony members are aligned and no conflict exists as long as there are no differences between individuals in relatedness patterns. Such differences can arise from polyandry/polygyny (e.g., Keller 1995, 1997) or haplodiploidy (e.g., Bourke and Franks 1995) (Fig. 5). Constraints, such as the lack of information on kinship and the inability to distinguish the sex of the offspring, may prevent the actual outbreak of conflict. Furthermore, such proximate constraints may be favored on the colony-level: selective, nepotistic brood rearing might be more time-consuming and less efficient than indiscriminate nursing, and colonies lacking within-colony favoritism may eventually outcompete colonies with kin discrimination (e.g., Page et al. 1989; Ratnieks and Reeve 1992).
When conflict cannot be prevented a priori, as in species with fertile workers, social control may ensure group stability (Fig. 5). As the interests of the majority of group members are aligned, they suppress selfish individuals that reduce the group’s performance and therefore the mean inclusive fitness of most individuals. In species with fertile workers, policing is an example of such social control (see above) and group-level considerations make it obvious why this even occurs in societies in which it is not expected from relatedness considerations alone (Ratnieks 1988). Similar mechanisms are well known from the gene–genome level (so-called ‘parliament of genes’; Leigh 1983), where selfish elements, such as segregation distorters or meiotic drive genes, are generally suppressed by other genes that enforce the common interest of the genome (Mercot et al. 1995; Atlan et al. 1997; Pomiankowski 1999).
When totipotent individuals compete for inheritance of the breeding position, for example in the ponerine ant genus Diacamma after the death of a gamergate (a mated worker, see, e.g., Peeters 1991; Cuvillier-Hot et al. 2002), or when the original king or queen are replaced by secondary reproductives in the lower termites Cryptotermes secundus or Kalotermes flavicollis (J. Korb, unpublished), costly overt conflict can be avoided through a “fair lottery” as in the above mentioned species: the first individual that reaches maturity or is in a sensitive stage between two molts after the death of the old reproductive takes over her role (Peeters and Higashi 1989; Lüscher 1956; J. Korb, unpublished). This mechanism is similar to a fair meiosis in sexually reproducing organisms: each gamete of an individual has an equal chance of receiving either allele at a heterozygous locus (Leigh 1991).
These considerations illustrate the above-mentioned constraints, such as the absence of ovaries or a lack of information to discriminate kin and to distinguish the sex of offspring, as mechanisms preventing the outbreak of conflict a priori. Their evolution can be explained by assuming that they increase the average fitness of all nestmates. However, such an averaging approach makes it difficult to elucidate the causal processes (Sober and Wilson 2002), which are more easily seen from a multilevel selection perspective (see above) (Fig. 5). However, once conflict breaks out in a colony, social control may, a posteriori, enforce the group interest.
Colony selection and kin selection
Because social insect groups are mostly kin groups, the evolution of mechanisms that prevent or resolve conflicts and thereby increase group efficiency can also be explained in terms of each individual maximizing its inclusive fitness. Indeed, many authors have noted the mathematical identity of kin selection and multilevel selection models (Wade 1978, 1979, 1980a, 1980b, 1985; Grafen 1984; Queller 1992; Dugatkin and Reeve 1994). The two approaches differ only in how they account for the fitness differences associated with a certain heritable trait: the kin selection approach concerns the population-wide average fitness difference between individuals with and without the trait, whereas the multilevel selection approach partitions this fitness difference into two components: the within and between group component. Both approaches should yield the same predictions about whether a trait will spread or disappear.
Partitioning selection into a within- and between-group component, however, offers the advantage of being able to highlight the group components that are less easily recognized from a kin selection perspective. Social insect societies are characterized by beneficial novelties that represent evolutionary breakthroughs and which appear to have gained more and more evolutionary importance, relative to relatedness, so that social insect colonies seem to be more sensitive to variations in those benefits than to variation in relatedness. For example, despite intracolonial conflict, much of the evolution of honeybee societies appears to be driven by selection operating at the colony level (Seeley 1995, 1997). Regarding colonies from a multilevel perspective reveals that “constraints” can in fact be adaptations that prevent or resolve conflict within colonies that are selected at the group level.
Although relatedness has played an important role in the evolution of insect societies, it is less important for their maintenance, because social insects have reached a new level of selection with novelties that provide benefits beyond the scope of a solitary life. Mechanisms have evolved to prevent or suppress selfish interests within the colonies, which arise through variation in relatedness. These conflict resolution mechanisms guarantee the stability of the group, as this is in the interest of most group members, and make insect colonies robust against variation in relatedness. Whether this will eventually lead to stable cooperative systems with zero relatedness is highly doubtful, given that open groups without relatedness are vulnerable to parasitism and cannot maintain or improve their performance (Queller 2000). Invasive ant species, such as the Argentine ant, Linepithema humile, which have been accidentally introduced by human activities into new habitats, have apparently lost their nestmate recognition systems to some degree and now form unicolonial populations (Giraud et al. 2002; Tsutsui et al. 2003). Instead of engaging in costly territorial fighting with conspecific non-nestmates, such supercolonies quickly dominate food sources and spread at the expense of native competitors. At present, invasive species thrive but, because nestmates are not more closely related than pairs of conspecific ants drawn at random from different nests (Krieger and Keller 2000), their success is presumably only a transient phenomenon that over the long run will decline (Queller 2000; Giraud et al. 2002).
References
Alexander RD (1974) The evolution of social behavior. Annu Rev Ecol Syst 5:325–383
Anderson C, McShea DW (2001) Individual versus social complexity, with particular reference to ant colonies. Biol Rev 76:211–237
Anderson C, Ratnieks FLW (1999) Task partitioning in insect societies. I. Effect of colony size on queuing delay and colony ergonomic efficiency. Am Nat 154:521–535
Aoki S (1987) Evolution of sterile soldiers in aphids. In: Itô Y, Brown JL, Kikkawa J (eds) Animal societies: theories and facts. Japan Scientific Societies, Tokyo, pp 53–65
Atlan A, Mercot H, Landre C, Montchamp-Moreau C (1997) The sex-ratio trait in Drosophila simulans: geographical distribution of distortion and resistance. Evolution 51:1886–1895
Baer B, Schmid-Hempel P (1999) Experimental variation in polyandry affects parasite load and fitness in a bumble-bee. Nature 397:151–153
Banschbach VA, Herbers JM (1996) Complex colony structure in social insects. II. Reproduction, queen-worker conflict, and levels of selection. Evolution 50:298–307
Bartz SH (1979) Evolution of eusociality in termites. Proc Natl Acad Sci USA 76:5764–5768
Beekman M, Sumpter DJT, Ratnieks FLW (2001) Phase transition between disordered and ordered foraging in Pharaoh’s ants. Proc Natl Acad Sci USA 98:9703–9706
Beekman M, Komdeur J, Ratnieks FLW (2003) Reproductive conflicts in social animals: who has power? Trends Ecol Evol 188:277–282
Beshers SN, Fewell JH (2001) Models of division of labor in social insects. Annu Rev Entomol 46:413–440
Bonabeau E, Theraulaz G, Deneubourg JL, Aron S, Camazine S (1997) Self-organization in social insects. Trends Ecol Evol 12:188–193
Boomsma JJ, Grafen A (1990) Intraspecific variation in ant sex ratios and the Trivers–Hare hypothesis. Evolution 44:1026–1034
Boomsma JJ, Grafen A (1991) Colony-level sex ratio selection in the eusocial Hymenoptera. J Evol Biol 3:383–407
Boomsma JJ, Ratnieks FLW (1996) Paternity in social Hymenoptera. Philos Trans R Soc Lond B 351:947–975
Bourke AFG (1988) Worker reproduction in the higher eusocial Hymenoptera. Q Rev Biol 63:291–311
Bourke AFG (1999) Colony size, social complexity and reproductive conflict in social insects. J Evol Biol 12:245–257
Bourke AFG, Franks NR (1995) Social evolution in ants. Princeton University Press, Princeton, N.J.
Braude S (2000) Dispersal and new colony formation in wild naked mole-rats: evidence against inbreeding as the system of mating. Behav Ecol 11:7–12
Brown WD, Keller L (2000) Colony sex ratios vary with queen number but not relatedness asymmetry in the ant Formica exsecta. Proc R Soc Lond B 267:1751–1757
Brown WD, Keller L, Sundström L (2002) Sex allocation in mound-building ants: the roles of resources and queen replenishment. Ecology 83:1945–1952
Buss L (1987) The evolution of individuality. Princeton University Press, Princeton, N.J.
Camazine S, Deneubourg JL, Franks N, Sneyd J, Theraulaz G, Bonabeau E (2001) Self-organization in biological systems. Princeton University Press, Princeton, N.J.
Chan GL, Hingle A, Bourke AFG (1999) Sex allocation in a facultatively polygynous ant: between-population and between-colony variation. Behav Ecol 10:409–421
Chapman TW, Crespi BJ, Kranz BD, Schwarz MP (2000) High relatedness and inbreeding at the origin of eusociality in gall-inducing thrips. Proc Natl Acad Sci USA 97:1648–1650
Choe JC (1988) Worker reproduction and social evolution in ants (Hymenoptera: Formicidae). In: Trager JC (ed) Advances in myrmecology. Brill, Leiden, pp 163–187
Cockburn A (1998) Evolution of helping behaviour in cooperatively breeding birds. Annu Rev Ecol Syst 29:141–177
Coenen-Stass D, Schaarschmidt B, Lamprecht I (1980) Temperature distribution and calorimetric determination of heat production in the nest of the wood ant, Formica polyctena (Hymenoptera, Formicidae). Ecology 61:238–244
Crozier RH, Fjerdingstad EJ (2001) Polyandry in social Hymenoptera: disunity in diversity? Ann Zool Fenn 38:267–285
Crozier RH, Luykx P (1985) The evolution of termite eusociality is unlikely to have been based on a male-haploid analogy. Am Nat 126:867–869
Crozier RH, Pamilo P (1996) Evolution of social insect colonies: sex allocation and kin selection. Oxford University Press, Oxford
Cuvillier-Hot V, Gadagkar R, Peeters C, Cobb M (2002) Regulation of reproduction in a queenless ant: aggression, pheromones and reduction in conflict. Proc R Soc Lond B 269:1295–1300
Darwin C (1859) On the origin of species by means of natural selection. John Murray, London
Darwin C (1871) The descent of man and selection in relation to sex. Appleton, New York
Dawkins R (1976) The selfish gene. Oxford University Press, Oxford
Deligne J, Quennedey A, Blum MS (1981) The enemies and defense mechanisms of termites. In: Hermann HR (Ed) Social insects, II. Academic Press, New York, pp 1–76
Dugatkin LA, Reeve HK (1994) Behavioral ecology and levels of selection: dissolving the group selection controversy. Adv Stud Behav 23:101–133
Emlen ST (1997) Predicting family dynamics in social vertebrates. In: Krebs JR, Davies NB (eds) Behavioural ecology, 4th edn. Blackwell Science, Oxford, pp 228–253
Fisher RA (1930) The genetical theory of natural selection. Clarendon, Oxford
Fjerdingstad EJ (2004) Multiple paternity and colony homeostasis in Lasius niger ants. Behav Ecol Sociobiol 55: 10.1007/s00265-004-0759-8
Fjerdingstad EJ, Gertsch PJ, Keller L (2002) Why do some social insect queens mate with several males? Testing the sex-ratio manipulation hypothesis in Lasius niger. Evolution 56:553–562
Foitzik S, Heinze J (2000) Intraspecific parasitism and split sex ratios in a monogynous, monandrous ant (Leptothorax nylanderi). Behav Ecol Sociobiol 47:424–431
Foitzik S, Herbers JM (2001) Colony structure of a slavemaking ant. I. Intracolony relatedness, worker reproduction, and polydomy. Evolution 55:307–315
Foitzik S, Strätz M, Heinze J (2003) Ecology, life history, and resource allocation in the ant, Leptothorax nylanderi. J Evol Biol 16:670–680
Foster KR, Gulliver J, Ratnieks FLW (2002) Why workers do not reproduce: worker policing in the European hornet Vespa crabro. Insectes Soc 49:41–44
Fournier D, Keller L, Passera L, Aron S (2003) Colony sex ratios vary with breeding system but not relatedness asymmetry in the facultatively polygynous ant Pheidole pallidula. Evolution 57:1336–1342
Frank SA (1985) Hierarchical selection theory and sex ratio. II. On applying the theory, and a test with fig wasps. Evolution 39:949–964
Frank SA (1990) Sex allocation theory for birds and mammals. Annu Rev Ecol Syst 21:13–55
Frank SA (1996) Models of parasite virulence. Q Rev Biol 71:37–78
Frank SA (1997) Foundations of social evolution. Princeton University Press, Princeton, N.J.
Franks NR, Pratt SC, Mallon EB, Britton NF, Sumpter DJT (2002) Information flow, opinion polling and collective intelligence in house-hunting social insects. Philos Trans R Soc Lond B 357:1567–1583
Frumhoff PC, Baker J (1988) A genetic component to the division of labour within honey bee colonies. Nature 333:358–361
Fuchs S, Schade V (1994) Lower performance in honeybee colonies of uniform paternity. Apidologie 25:155–168
Gadagkar R (1985) Evolution of insect sociality: a review of some attempts to test modern theories. Proc Indian Acad Sci (Anim Sci) 94:309–324
Gadagkar R (1990) Evolution of eusociality: the advantage of assured fitness returns. Philos Trans R Soc Lond B 329:17–25
Gadau J, Strehl C-P, Oettler J, Hölldobler B (2003) Determinants of intracolonial relatedness in Pogonomyrmex rugosus (Hymenoptera; Formicidae): mating frequency and brood raids. Mol Ecol 12:1931–1938
Giraud T, Pedersen JS, Keller L (2002) Evolution of supercolonies: the Argentine ants of southern Europe. Proc Natl Acad Sci USA 99:6075–6079
Goodnight CJ, Stevens L (1997) Experimental studies of group selection: what do they tell us about group selection in nature? Am Nat 150:S59–S79
Gordon DM (1996) The organization of work in social insect colonies. Nature 380:121–124
Grafen A (1984) Natural selection, kin selection and group selection. In: Krebs JR, Davies NB (eds) Behavioural ecology, 2nd edn. Blackwell Science, Oxford, pp 62–84
Greeff J (1996) Effects of thelytokous worker reproduction in kin selection and conflict in the Cape honeybee, Apis mellifera capensis. Philos Trans R Soc Lond B 351:617–625
Griffin AS, West SA (2003) Kin discrimination and the benefit of helping in cooperatively breeding vertebrates. Science 302:634–636
Haldane JBS (1932) The causes of evolution. Longmans Green, London
Hamilton WD (1963) The evolution of altruistic behavior. Am Nat 97:354–356
Hamilton WD (1964) The genetical evolution of social behaviour. I, II. J Theor Biol 7:1–52
Hamilton WD (1970) Selfish and spiteful behaviour in an evolutionary model. Nature 228:1218–1220
Hamilton WD (1972) Altruism and related phenomena, mainly in social insects. Annu Rev Ecol Syst 3:193–232
Hamilton WD (1975) Innate social aptitudes in man: an approach from evolutionary genetics. In: Fox R (ed) Biosocial anthropology. Malaby, London, pp 133–155
Hammond RL, Bruford MW, Bourke AFG (2002) Ant workers selfishly bias sex ratios by manipulating female development. Proc R Soc Lond B 269:173–178
Hammond RL, Bruford MW, Bourke AFG (2003) Male parentage does not vary with colony kin structure in a multiple-queen ant. J Evol Biol 16:446–455
Hartmann A, Heinze J (2003) Lay eggs, live longer: division of labor and life span in a clonal ant species. Evolution 57:2424–2429
Hartmann A, Wantia J, Torres JA, Heinze J (2003) Worker policing without genetic conflicts in a clonal ant. Proc Natl Acad Sci USA 100:12836–12840
Heinrich B (1993) The hot-blooded insect. Harvard University Press, Cambridge, Mass.
Heinze J (1995) Reproductive skew and relatedness in Leptothorax ants. Proc R Soc Lond B 261:375–379
Heinze J (1996) The reproductive potential of workers in slave-making ants. Insectes Soc 43:319–328
Heinze J, Hölldobler B (1995) Thelytokous parthenogenesis and dominance hierarchies in the ponerine ant, Platythyrea punctata (F. Smith). Naturwissenschaften 82:40–41
Heinze J, Cover SP, Hölldobler B (1995) Neither worker nor queen: an ant caste specialized on the production of male eggs. Psyche 102:173–185
Heinze J, Strätz M, Pedersen JS, Haberl M (2000) Microsatellite analysis suggests occasional worker reproduction in the monogynous ant Crematogaster smithi. Insectes Soc 47:299–301
Helms KR (1999) Colony sex ratios, conflict between queens and workers, and apparent queen control in the ant Pheidole desertorum. Evolution 53:1470–1478
Henderson G (1998) Primer pheromones and possible soldier caste influence on the evolution of sociality in lower termites. In: Vander Meer RK, Breed MD, Espelie KE, Winston ML (eds) Pheromone communication in social insects. Westview, Boulder, Colo., pp 314–330
Herbers JM (1982) Queen-number and colony ergonomics in Leptothorax longispinosus. In: Breed MD, Michener CD, Evans HE (eds) The biology of social insects. Westview, Boulder, Colo., pp 238–242
Hölldobler B, Wilson EO (1990) The ants. Harvard University Press, Cambridge, Mass.
Horstmann K (1983) Regulation der Temperatur in Waldameisennestern (Formica polyctena Förster). Z Naturforsch 38:508–510
Horstmann K (1990) Zur Entstehung des Wärmezentrums in Waldameisennestern (Formica polyctena Förster; Hymenoptera, Formicidae). Zool Beitr NF 33:105–124
Iwanishi S, Hasegawa E, Ohkawara K (2003) Worker oviposition and policing behaviour in the myrmicine ant Aphaenogaster smythiesi japonica Forel. Anim Behav 66:513–519
Jeanne R (1975) The adaptiveness of social wasp nest architecture. Q Rev Biol 50:267–287
Jeon J, Choe JC (2003) Reproductive skew and the origin of sterile castes. Am Nat 161:206–224
Karsai I, Wenzel JW (1998) Productivity, individual-level and colony-level flexibility, and organization of work as consequences of colony size. Proc Natl Acad Sci USA 96:8665–8669
Kaspari M, Vargo EL (1995) Colony size as a buffer against seasonality: Bergmann’s rule in social insects. Am Nat 146:610–632
Keller L (1995) Social life: the paradox of multiple-queen colonies. Trends Evol Ecol 10:355–360
Keller L (1997) Indiscriminate altruism: unduly nice parents and siblings. Trend Ecol Evol 12:99–103
Keller L, Nonacs P (1993) The role of queen pheromones in social insects; queen control or queen signal? Anim Behav 45:787–794
Keller L, Reeve HK (1994) Genetic variability, queen number, and polyandry in social Hymenoptera. Evolution 48:694–704
Keller L, Reeve HK (1995) Why do females mate with multiple males? The sexually selected sperm hypothesis. Adv Stud Behav 24:291–315
Keller L, Reeve HK (1999) Dynamics of conflicts within insect societies. In: Keller L (ed) Levels of selection in evolution. Princeton University Press, Princeton, N.J., pp 153–175
Korb J (2003) Thermoregulation and ventilation of termite mounds. Naturwissenschaften 90:212–219
Korb J, Linsenmair KE (2000) Ventilation of termite mounds: new results require a new model. Behav Ecol 11:486–494
Krause J, Ruxton GD (2002) Living in groups. Oxford University Press, Oxford
Krieger MJB, Keller L (2000) Mating frequency and genetic structure of the Argentine ant Linepithema humile. Mol Ecol 9:119–126
Kropotkin P (1902) Mutual aid: a factor of evolution. William Heinemann, London
Lacy RC (1980) The evolution of eusociality in termites: a haplodiploid analogy? Am Nat 116:449–451
Leigh EG (1983) When does the good of the group override the advantage of the individual? Proc Natl Acad Sci USA 80:2985–2989
Leigh EG (1991) Genes, bees and ecosystems: the evolution of a common interest among individuals. Trends Ecol Evol 6:257–262
Lin N, Michener CD (1972) Evolution of sociality in insects. Q Rev Biol 14:131–159
Lindauer M (1954) Temperaturregulierung und Wasserhaushalt im Bienenstaat. Z Vergl Physiol 36:391–432
Lüscher M (1956) Die Entstehung von Ersatzgeschlechtstieren bei der Termite Kalotermes flavicollis (Frabr.). Insectes Soc 3:119–128
Lüscher M (1961) Air-conditioned termite nests. Sci Am 204(1):138–145
Majerus M, Amos W, Hurst G (1996) Evolution: the four billion year war. Longman, Harlow, UK
Martin SJ, Beekman M, Wossler TC, Ratnieks FLW (2002) Parasitic Cape honeybee workers, Apis mellifera capensis, evade policing. Nature 415:163–165
Maynard Smith J (1964) Group selection and kin selection. Nature 201:1145–1146
Maynard Smith J, Szathmáry E (1995) The major transitions in evolution. WH Freeman, Oxford
Mayr E (1997a) The objects of selection. Proc Natl Acad Sci USA 94:2091–2094
Mayr E (1997b) This is biology. Belknap, Harvard University Press, Cambridge, Mass.
Mehdiabadi N, Reeve HK, Mueller UG (2003) Queens versus workers: sex-ratio conflict in eusocial Hymenoptera. Trends Ecol Evol 18:88–93
Mercot H, Atlan A, Jacques M, Montchamp-Moreau C (1995) Sex-ratio distortion in Drosophila simulans: co-occurrence of a meiotic drive and a suppressor of drive. J Evol Biol 8:283–300
Michod RE (1999) Individuality, immortality, and sex. In: Keller L (ed) Levels of selection in evolution. Princeton University Press, Princeton, N.J., pp 53–74
Michod RE, Roze D (1997) Transitions in individuality. Proc R Soc Lond B 264:853–857
Mirenda JT, Vinson SB (1981) Division of labor and specification of castes in the red imported fire ant Solenopsis invicta. Anim Behav 29:410–420
Monnin T, Ratnieks FLW (2001) Policing in queenless ponerine ants. Behav Ecol Sociobiol 50:97–108
Moritz RFA, Southwick EE (1992) Bees as superorganisms: an evolutionary reality. Springer, Berlin Heidelberg New York
Mueller UG (1991) Haplodiploidy and the evolution of facultative sex ratios in a primitively eusocial bee. Science 254:442–444
Murakami T, Higashi S, Windsor D (2000) Mating frequency, colony size, polyethism and sex ratio in fungus-growing ants (Attini). Behav Ecol Sociobiol 48:276–284
Neumann P, Moritz RFA (2000) Testing genetic variance hypotheses for the evolution of polyandry in the honeybee (Apis mellifera L.). Insectes Soc 41:271–279
Neumann P, Radloff SE, Moritz RFA, Hepburn HR, Reece SL (2001) Social parasitism by honeybee workers (Apis mellifera capensis Escholtz): host finding and resistance of hybrid host colonies. Behav Ecol 12:419–428
Noirot C, Darlington JPEC (2000) Termite nests: architecture, regulation and defence. In: Abe T, Bignell DE, Higashi M (eds) Termites: evolution, sociality, symbioses, ecology. Kluwer Academic, Dordrecht, The Netherlands, pp 121–140
Nonacs P (1986) Sex-ratio determination within colonies of ants. Evolution 40:199–204
Nunney L (1999) Lineage selection: natural selection for long-term benefit. In: Keller L (ed) Levels of selection in evolution. Princeton University Press, Princeton, N.J., pp 238–252
Oldroyd BP (2002) The Cape honeybee: an example of a social cancer. Trends Ecol Evol 17:249–251
Oster GF, Wilson EO (1978) Caste and ecology of social insects. Princeton University Press, Princeton, N.J.
Page RE, Robinson GE, Fondrk MK (1989) Genetic specialists, kin recognition and nepotism in honey-bee colonies. Nature 338:576–579
Page RE, Robinson GE, Fondrk MK, Nasr ME (1995) Effects of worker genotypic diversity on honey bee colony development and behavior (Apis mellifera L.). Behav Ecol Sociobiol 36:387–396
Palmer KA, Oldroyd BP (2000) Evolution of multiple mating in the genus Apis. Apidologie 31:235–248
Peeters C (1991) The occurrence of sexual production among ant workers. Biol J Linn Soc 44:141–152
Peeters C, Higashi S (1989) Reproductive dominance controlled by mutilation in the queenless ant Diacamma australe. Naturwissenschaften 76:177–180
Peters JM, Queller DC, Imperatriz-Fonseca VL, Roubik DW, Strassmann JE (1999) Mate number, kin selection and social conflicts in stingless bees and honeybees. Proc R Soc Lond B 266:379–384
Pirk CWW, Neumann P, Ratnieks FLW (2003) Cape honeybees, Apis mellifera capensis, police worker-laid eggs despite the absence of relatedness benefits. Behav Ecol 14:347–352
Pomiankowski A (1999) Intragenomic conflict. In: Keller L (ed) Levels of selection in evolution. Princeton University Press, Princeton, N.J., pp 121–152
Price GR (1970) Selection and covariance. Nature 277:520–421
Price GR (1972) Extension of covariance selection mathematics. Ann Hum Genet 35:485–490
Queller DC (1989) The evolution of eusociality: reproductive head starts of workers. Proc Natl Acad Sci USA 86:3224–3226
Queller DC (1992) Quantitative genetics, inclusive fitness, and group selection. Am Nat 139:540–558
Queller DC (1994) Extended parental care and the origin of eusociality. Proc R Soc London B 256:105–111
Queller DC (2000) Pax Argentinica. Nature 405:519–520
Queller DC, Strassmann JE (1998) Kin selection and social insects. BioScience 48:165–178
Queller DC, Strassmann JE, Hughes C (1993) Microsatellites and kinship. Trends Ecol Evol 8:285–288
Ratnieks FLW (1988) Reproductive harmony via mutual policing by workers in eusocial Hymenoptera. Am Nat 132:217–236
Ratnieks FLW (1993) Egg laying, egg removal, and ovary development by workers in queenright honey bee colonies. Behav Ecol Sociobiol 32:191–198
Ratnieks FLW, Reeve HK (1992) Conflict in single-queen Hymenopteran societies: the structure of conflict and processes that reduce conflict in advanced eusocial species. J Theor Biol 158:33–65
Ratnieks FLW, Visscher PK (1989) Worker policing in the honeybee. Nature 342:796–797
Reeve HK (1998) Game theory, reproductive skew, and nepotism. In: Dugatkin LA, Reeve HK (eds) Theory and animal behaviour. Oxford University Press, Oxford, pp 118–145
Reeve HK, Keller L (1999) Levels of selection: burying the units-of-selection debate and unearthing the crucial new issues. In: Keller L (ed) Levels of selection in evolution. Princeton University Press, Princeton, N.J., pp 3–14
Reeve HK, Westneat DF, Noon WA, Sherman PW, Aquadro CF (1990) DNA “fingerprinting” reveals high levels of inbreeding in colonies of the eusocial naked mole-rat. Proc Natl Acad Sci USA 87:2496–2500
Reeve HK, Starks PT, Peters JM, Nonacs P (2000) Genetic support for the evolutionary theory of reproductive transactions in social wasps. Proc R Soc Lond B 267:75–79
Robinson GE (1992) Regulation of division of labor in insect colonies. Annu Rev Entomol 37:637–655
Robinson GE, Page RE Jr (1989) Genetic determination of nectar foraging, pollen foraging, and nest-site scouting by honey bee colonies. Behav Ecol Sociobiol 24:317–323
Rosenheim JA, Nonacs P, Mangel M (1996) Sex ratios and multifaceted parental investment. Am Nat 148:501–535
Sanetra M, Crozier RH (2001) Polyandry and colony genetic structure in the primitive ant Nothomyrmecia macrops. J Evol Biol 14:368–378
Seeley TD (1989) The honey bee colony as a superorganism. Am Sci 77:546–553
Seeley TD (1995) The wisdom of the hive. Harvard University Press, Cambridge, Mass.
Seeley TD (1997) Honey bee colonies are group-level adaptive units. Am Nat 150: S22–S41
Sherman PW, Seeley TD, Reeve HK (1988) Parasites, pathogens, and polyandry in social Hymenoptera. Am Nat 131:602–610
Sober E, Wilson DS (2002) Perspectives and parameterizations: Commentary on Benjamin Kerr and Peter Godfrey-Smith’s “Individualist and multi-level perspectives on selection in structured populations”. Biol Philos 17:529–537
Solomon NG, French JA (1997) Cooperative breeding in mammals. Cambridge University Press, Cambridge
Sundström L (1994) Sex ratio bias, relatedness asymmetry and queen mating frequency in ants. Nature 367:266–268
Sundström L, Chapuisat M, Keller L (1996) Conditional manipulation of sex ratios by ant workers: a test of kin selection theory. Science 274:993–995
Tarpy DR (2003) Genetic diversity within honeybee colonies prevents severe infections and promotes colony growth. Proc R Soc Lond B 270:99–103
Thomas ML, Elgar MA (2003) Colony size affects division of labour in the ponerine ant Rhytidoponera metallica. Naturwissenschaften 90:88–92
Tóth E, Queller DC, Imperatriz-Fonseca VL, Strassmann JE (2002a) Genetic and behavioral conflict over male production between workers and queens in the stingless bee Paratrigona subnuda. Behav Ecol Sociobiol 53:1–8
Tóth E, Strassmann JE, Nogueira-Neto P, Imperatriz-Fonseca VL, Queller DC (2002b) Male production in stingless bees: variable outcomes of queen–worker conflict. Mol Ecol 11:2261–2267
Tóth E, Strassmann JE, Imperatriz-Fonseca VL, Queller DC (2003) Queens, not workers, produce the males in the stingless bee Schwarziana quadripunctata quadripunctata. Anim Behav 66:359–368
Trivers RL, Hare H (1976) Haplodiploidy and the evolution of the social insects. Science 191:249–263
Tsutsui ND, Suarez AV, Grosberg RK (2003) Genetic diversity, asymmetrical aggression, and recognition in a widespread invasive species. Proc Natl Acad Sci USA 100:1078–1083
Uetz GW (2001) Understanding the evolution of social behavior in colonial web-building spiders. In: Dugatkin LA (Ed) Model systems in behavioral ecology. Princeton University Press, Princeton, N.J., pp 110–132
Villesen P, Gertsch PJ, Frydenberg J, Mueller UG, Boomsma JJ (1999) Evolutionary transition from single to multiple mating in fungus-growing ants. Mol Ecol 8:1819–1825
Villesen P, Murakami T, Schultz TR, Boomsma JJ (2002) Identifying the transition between single and multiple mating of queens in fungus-growing ants. Proc R Soc Lond B 269:1541–1548
Visscher PK (1996) Reproductive conflict in honey bees: a stalemate of worker egg-laying and policing. Behav Ecol Sociobiol 39:237–244
Wade MJ (1978) Kin selection: a classical approach and a general solution. Proc Natl Acad Sci USA 75:6154–6158
Wade MJ (1979) The evolution of social interactions by family selection. Am Nat 113:399–417
Wade MJ (1980a) Kin selection: its components. Science 210:665–667
Wade MJ (1980b) An experimental study of kin selection. Evolution 34:844–855
Wade MJ (1982) The effect of multiple inseminations on the evolution of social behavior in diploid and haplodiploid organisms. J Theor Biol 95:351–368
Wade MJ (1985) Soft selection, hard selection, kin selection, and group selection. Am Nat 125:61–73
Wattanachaiyingcharoen W, Oldroyd BP, Wongsiri S, Palmer K, Paar J (2003) A scientific note on the mating frequency of Apis dorsata Fabricius. Apidologie 34:85–86
Wcislo WT, Danforth BN (1997) Secondarily solitary: the evolutionary loss of social behavior. Trends Ecol Evol 12:468–474
Wenzel JW (1991) Evolution of nest architecture. In: Ross KG (ed) The social biology of wasps. Cornell University Press, Ithaca, N.Y., pp 480–519
West-Eberhard MJ (1975) The evolution of social behavior by kin selection. Q Rev Biol 50:1–33
Williams GC (1966) Adaptation and natural selection: a critique of some current evolutionary thoughts. Princeton University Press, Princeton, N.J.
Wilson DS (1997a) Introduction: multilevel selection theory comes of age. Am Nat 150:S1–S4
Wilson DS (1997b) Altruism and organism: disentangling the themes of multilevel selection theory. Am Nat 150:S122–S134
Wilson EO (1971) The insect societies. Belknap, Harvard University Press, Cambridge, Mass.
Woyciechowski M, Warakomska Z (1994) Workers’ genetic diversity has no relation to pollen diversity in a honey bee colony (Apis mellifera L.). J Ethol 12:163–167
Woyciechowski M, Król R, Figurny E, Stachowicz M, Tracz M (1994) Genetic diversity of workers and infection by the parasite Nosema apis in honey bee colonies (Apis mellifera L). In: Lenoir A, Arnold G, Lepage M (eds) Les insectes sociaux. Université Paris Nord, Paris, p 347
Wynne-Edwards VC (1962) Animal dispersion in relation to social behavior. Oliver and Boyd, Edinburgh
Yasui Y (1998) The ‘genetic benefits” of female multiple mating revisited. Trends Ecol Evol 13:246–250
Acknowledgements
Our studies were supported by DFG (HE-1623/15 and KO-1895/2). We thank three referees for their valuable comments on a first draft of our manuscript. This paper is dedicated to Ernst Mayr, with our best wishes for his 100th birthday.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Prof. Ernst Mayr
Rights and permissions
About this article
Cite this article
Korb, J., Heinze, J. Multilevel selection and social evolution of insect societies. Naturwissenschaften 91, 291–304 (2004). https://doi.org/10.1007/s00114-004-0529-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00114-004-0529-5