Abstract
Acid sphingomyelinase-induced ceramide release has been shown by many studies to induce apoptosis in response to various stimuli. However, the mechanisms of acid sphingomyelinase/ceramide-mediated death signaling following treatment with chemotherapeutic drugs have not been fully elucidated thus far. The present study demonstrates that treatment of glioma cells with clinically achievable doses of gemcitabine results in acid sphingomyelinase activation, lysosomal accumulation of ceramide, cathepsin D activation, Bax insertion into the mitochondria, and cell death. Pharmacological inhibition or genetic deficiency of acid sphingomyelinase prevented these events while overexpression of the enzyme sensitized cells to gemcitabine. Likewise, inhibitors of lysosomal functions also prevent gemcitabine-induced cell death. Our data indicate a critical role of the acid sphingomyelinase/ceramide system for gemcitabine-induced signaling and suggest that lysosomal ceramide accumulation mediates cell death induced by a chemotherapeutic drug.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Malignant gliomas are the most common primary brain tumors in adults. The most aggressive form of glioma, glioblastoma multiforme, is characterized by a poor prognosis for the patient, with a median survival of only 12 months. Due to a predominant tendency to invade surrounding tissue, gliomas cannot be resected completely and require, thus, chemo- and radiotherapy in addition to surgery. The current treatments proved to have only limited success; therefore, it is of great importance to define molecular mechanisms that mediate glioma cell death triggered by therapeutic drugs as an initial step towards improved efficiency of the respective stimuli.
Apoptotic stimuli can kill the cells via two main pathways: the extrinsic (receptor-initiated) or the intrinsic (mitochondria-initiated) pathways, which converge in type II cells [1–3]. The extrinsic pathway requires that upon binding of a death ligand to its receptor, the death-inducing signaling complex is formed, which leads to activation of initiator caspase 8 and effector caspases 3, 6, and 7, respectively [3]. The intrinsic pathway involves mitochondrial outer membrane permeabilization and release of mitochondrial molecules, such as cytochrome c. This leads to apoptosome formation, activation of initiator caspase 9, and execution of apoptosis mainly via caspase 3 [2]. At the level of mitochondria-initiated apoptosis pathways, proapoptotic Bcl-2 family members, such as Bax or Bak, and antiapoptotic molecules, such as Bcl-2 or Bcl-xL, are important regulators regarding the fate of the stimulated cells [4, 5].
Gemcitabine (29,29-difluoro 29-deoxycytidine) is an analog of cytosine arabinoside from which it differs structurally by its fluorine substituents on position 29 of the furanose ring. Originally investigated as an antiviral agent, it was then developed as an anticancer drug on the basis of its impressive in vitro and in vivo antitumoral activities. The evidence of the efficacy of gemcitabine in inhibiting the growth of human neoplasms was obtained in a broad range of solid and hematological cancer cell lines as well as in in vivo murine solid tumors and human tumor xenografts in nude mice. Currently, gemcitabine is used in treatment of pancreatic carcinoma, NSCLC, or breast cancer and has shown successful results in other solid tumors, such as ovarian cancer, mesothelioma, or head and neck carcinoma.
Over the last years, many groups demonstrated the involvement of sphingolipids, in particular, ceramide, in apoptosis induced by a wide variety of stimuli. Ceramide can be generated either by de novo synthesis via activation of ceramide synthase or by sphingomyelin hydrolysis following activation of acid, neutral, or alkaline sphingomyelinases [6, 7]. Although recent studies also involve neutral or alkaline sphingomyelinase in the induction of apoptosis [8, 9], most studies investigated the role of acid sphingomyelinase-released ceramide in cell death. Ceramide has been shown to play a significant role in signaling triggered by both intrinsic and extrinsic apoptosis-inducing stimuli, which suggests that the mechanisms of ceramide-mediated cell death are complex and diverse. It has been demonstrated that stimulation with members of the tumor necrosis factor (TNF) family of ligands, such as CD95 or TNF-related apoptosis-inducing ligand (TRAIL), results in rapid activation of acid sphingomyelinase and formation of ceramide-enriched platforms in the plasma membrane [10, 11]. Ceramide platforms serve to cluster CD95 or DR5 receptors, an event that was shown to be prerequisite for transmission of an efficient death signal into the cells [10–15]. The acid sphingomyelinase/ceramide pathway was proved to be involved also in cellular responses to γ-irradiation. Further studies established that γ-irradiation induces endothelial cell apoptosis in tumors or in the crypts of the gastrointestinal tract via the acid sphingomyelinase, and acid sphingomyelinase-deficient endothelial cells were resistant to irradiation doses of up to 50 Gy [16–19]. Similarly, ultraviolet (UV)-A and UV-C light activate acid sphingomyelinase and trigger ceramide release while cells deficient for acid sphingomyelinase fail to undergo apoptosis upon UV light radiation [20–23].
Several studies in the last years proposed acid sphingomyelinase and ceramide as mediators of chemotherapy-induced cell death. For instance, Morita and co-workers showed that oocytes derived from acid sphingomyelinase-deficient mice were resistant to doxorubicin stimulation [24] while other studies revealed activation of acid sphingomyelinase and subsequent ceramide level increase upon treatment of tumor cells with cisplatin [25]. Our group has recently demonstrated that overexpression of acid sphingomyelinase sensitizes transfected cells to doxorubicin and gemcitabine-induced cell death [26]. However, the mechanisms of chemotherapy-triggered cell death via acid sphingomyelinase/ceramide remain, thus far, poorly understood.
In the present study, we investigated the effect of clinically achievable concentrations of gemcitabine on acid sphingomyelinase activation and ceramide production in glioma cells. We examined the functional role of the acid sphingomyelinase/ceramide pathway regarding gemcitabine-induced apoptosis and focused on analyzing ceramide topology to identify mechanisms of gemcitabine-induced cell death. The results demonstrate that gemcitabine treatment leads to activation of the acid sphingomyelinase that correlates with a strong ceramide accumulation. Our studies employing amitriptyline and acid sphingomyelinase-deficient cells suggest a crucial role of acid sphingomyelinase/ceramide for gemcitabine-induced Bax activation and cell death. Furthermore, we identify accumulation of ceramide within lysosomes as a novel mechanism of cell death induced by a chemotherapeutic drug.
Materials and methods
Cell lines and primary tumor cells
The mouse astrocytoma cell line Spontaneous Murine Astrocytoma (SMA) 560 has been previously described [27]. To ensure tumorigenicity, 105 SMA 560 cells were injected intramuscularly into the leg of a VM/Dk mouse. The tumor was excised 14 days later and prepared into single-cell suspension by enzymatic digestion with 1 mg/ml Collagenase A (Roche, Mannheim, Germany) for 1 h at 37°C. The resulting cells, designated as SMA, were cultured and maintained in Dulbecco's Modified Eagle's Medium supplemented with 10% fetal calf serum, 10 mM N-2-Hydroxyethylpiperazine-N′-2-Ethanesulfonic Acid (HEPES) (pH 7.4), 2 mM l-glutamine, 1 mM sodium pyruvate, 100 mM nonessential amino acids, 100 units/ml penicillin, and 100 µg/ml streptomycin (all from Invitrogen, Karlsruhe, Germany). Primary human glioma cells were obtained from patients undergoing surgery. The ethics committee of the University Clinic Essen has approved these activities (reference no. 08-3709, August 2008), and informed written consent was obtained from all patients. The tumors were brought to single-cell suspensions and cultured as described previously. In addition, we stably transfected SMA cells with an expression vector of the acid sphingomyelinase (pJK-ASM), which controls expression of the ASM by an elongation factor promoter. Controls were transfected with the empty vector (pJK) alone. Cells were cultured as previously mentioned. Finally, we employed immortalized B-lymphocytes that were previously obtained from a patient with Niemann-Pick disease type A or a healthy control. All lymphocytes were cultured in RPMI-1640 supplemented as previously mentioned.
Reagents and antibodies
Monoclonal anticeramide antibodies were obtained from Glycobiotech (Kuekels, Germany). Anti-mouse Lamp1 and anti-Tim23 antibodies were from BD Pharmingen (San Diego, CA, USA). Anti-Bax and anti-cathepsin D antibodies were obtained from Upstate (Lake Placid, NY, USA). LysoSensor Green DND-189 was purchased from Invitrogen Molecular Probes (Karlsruhe, Germany). Amitriptyline, imipramine, bafilomycine, and chloroquine were from Sigma (Germany). All fluorescent secondary antibodies (Cy3-donkey anti-mouse IgM, Cy3-donkey anti-rabbit IgG, Cy5-donkey anti-mouse IgM, FITC-donkey anti-mouse IgG, and FITC-goat anti-rat IgG) were from Jackson Immunoresearch (West Grove, PA, USA).
Immunofluorescence assays
Cells were treated as indicated, washed once in HEPES/saline (H/S; 132 mM NaCl, 20 mM HEPES (pH 7.4), 5 mM KCl, 1 mM CaCl2, 0.7 mM MgCl2, 0.8 mM MgSO4), and fixed/permeabilized for 20 min at room temperature using BD Cytofix/Cytoperm kit (BD Biosciences, San Diego, CA, USA). When using LysoSensor Green DND-189, cells were stained prior to fixation according to the protocol provided by the manufacturer. Cells were then incubated with the indicated primary antibodies for 45 min at room temperature. Anti-ceramide and anti-Lamp1 antibodies were diluted 1:100 and 1:500, respectively. Anti-Bax and anti-Tim23 antibodies were diluted 1:250. The samples were washed again and incubated with the respective fluorescent secondary antibodies for 30 min at room temperature. All secondary antibodies were diluted 1:1,000. Cells were finally washed twice and either analyzed by flow cytometry or mounted in Fluoprep (BioMerieux, France) and analyzed by confocal microscopy. Laser scanning microscopy was performed on a LEICA TSP2 module linked to a LEICA DMIRE 2 microscope (Leica, Germany).
Acid sphingomyelinase activity
Acid sphingomyelinase activity was measured as previously described [28]. Tumor cells were incubated with 100 nM gemcitabine for the indicated times. The cells were then scraped and lysed in a buffer containing 0.1% Triton X-100 and 50 mM sodium acetate (pH 5.0) for 10 min on ice followed by three sonication cycles. The cell lysates were incubated with 0.05 mCi per sample of [14C]-labeled sphingomyelin (2 GBq/mmol; MP-Biomedicals, Irvine, CA, USA) for 30 min at 37°C. To this end, the substrate [14C]-labeled sphingomyelin was dried under vacuum, resuspended in 0.1% Triton X-100 and 50 mM sodium acetate (pH 5.0), and sonicated in a bath sonicator to promote the formation of liposomes. The reaction was terminated by extraction in CHCl3–CH3OH (2:1, v/v). The samples were centrifuged for 10 min at maximum speed, and an aliquot of the aqueous phase was measured by liquid scintillation counting (TriCarb Liquid Scintillation Analyzer, Perkin Elmer, USA) to quantify released phosphorylcholine. This method results in hydrolysis of less than 20% (range of 2% to maximal 20%) of [14C]-labeled sphingomyelin and is therefore suitable for a broad range of cells.
Apoptosis assays
Following stimulation, cells were stained with FITC-Annexin V and propidium iodide according to the manufacturer's instructions (Roche Molecular Biochemicals). Quantification was performed employing a FACS Calibur flow cytometer (Becton Dickinson, San Jose, CA, USA).
Ceramide measurement
To determine cellular ceramide levels by the DAG-kinase assay, cells were extracted in CHCl3:CH3OH:1 N HCl (100:100:1, v/v/v); the organic phase was collected, dried, and resuspended in 20 µl of a detergent solution consisting of 7.5% (w/v) n-octylglucopyranoside and 5 mM cardiolipin in 1 mM diethylenetriamine pentaacetic acid (DETAPAC). To promote the formation of micelles, samples were sonicated for 10 min in a bath sonicator. To start the kinase reaction, 50 µl of a buffer consisting of 100 mM imidazole/HCl (pH 6.6), 100 mM NaCl, 25 mM MgCl2, 2 mM EGTA, 2.8 mM DTT, 5 mM adenosine triphosphate (ATP), 10 µCi [32P] gamma-ATP, and 10 µl diacylglycerol (DAG) kinase (Calbiochem/Merck, Darmstadt, Germany) prediluted in 1 mM DETAPAC (pH 6.6) and 100 mM imidazole/HCl) were added, and the samples were incubated for 30 min at room temperature. The kinase reaction was stopped by extraction of the samples with 1 ml CHCl3:CH3OH:1 N HCl (100:100:1, v/v/v), 170 µl buffered saline solution (135 mM NaCl, 1.5 mM CaCl2, 0.5 mM MgCl2, 5.6 mM glucose, 10 mM HEPES, pH 7.2), and 30 µl of a 100 mM ethylenediaminetetraacetic acid (EDTA) solution. The lower phase was collected, dried, and dissolved in 20 µl CHCl3:CH3OH (1:1, v/v). Lipids were separated on a Silica G60 TLC plate (Merck, Darmstadt, Germany) with a buffer containing chloroform:acetone:methanol:acetic acid:H2O (50:20:15:10:5, v/v/v/v/v). The plates were dried and analyzed using a FLA-3000 Phospho-Imager (Fujifilm Life Sciences, Düsseldorf, Germany).
Mitochondrial and cytoplasmic fraction isolation
Stimulated or untreated SMA cells were collected and centrifuged, and the supernatant was removed. The pellet was incubated for 4 min on ice with 150 µl of a buffer containing 150 mM MgCl2, 10 mM KCl, and 10 mM Tris pH 6.7. Another 50 µl of a buffer containing 150 mM MgCl2, 250 mM sucrose, and 10 mM Tris pH 6.7 were added to the samples, which were then homogenized with 100 strokes in a Dounce homogenizer. Samples were centrifuged at 400×g, and the supernatant was transferred into fresh Eppendorf tubes followed by another centrifugation step at 5,500×g. The supernatant consisting of the cytoplasmic fraction was collected into fresh test tubes. An equal volume of a buffer containing 250 mM sucrose and 10 mM Tris pH 7.0 was added to the remaining pellet, which consisted of the mitochondrial fraction. Both mitochondrial and cytoplasmic fractions were then subjected to Western blot analysis as detailed below.
Cathepsin D analysis
Following stimulation, cells were washed once in HEPES/saline and lysed in 25 mM HEPES (pH 7.3), 0.1% sodium dodecylsulfate (SDS), 0.5% deoxycholate, 1% Triton X-100, 10 mM EDTA, 10 mM sodiumpyrophosphate, 10 mM NaF, 125 mM NaCl, and 10 μg/ml each of aprotinin/leupeptin. The cellular debris was removed by centrifugation at 14.000 rpm for 10 min. Samples were then subjected to Western blot analysis as detailed in the following section.
SDS-PAGE and Western blot analysis
Samples were incubated with SDS-sample buffer (final concentrations 50 mM Tris pH 6.8, 4% glycerin, 0.8% SDS, 1.6% ß-mercaptoethanol, and 0.04% bromophenol blue). Samples were boiled for 5 min at 95°C and analyzed by SDS-PAGE followed by transfer to nitrocellulose membranes (Amersham Biosciences, Piscataway, NJ, USA). The membranes were blocked for 45 min with Starting Block buffer (Thermo Scientific, Rockford, IL, USA) and incubated overnight at 4°C with the respective primary antibodies. The membranes were washed, incubated for 1 h at room temperature with alkaline phosphatase-conjugated goat–anti-rabbit IgG or goat–anti-mouse IgG (both 1:15.000, Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA) and developed with the CDP-Star chemoluminescence detection system (Perkin Elmer, Boston, MA, USA).
Statistical analysis
Data were expressed as mean values and standard deviations (SD). Statistical analysis was performed employing one-way analysis of variance (ANOVA) test with Dunnett's correction. P ≤ 0.05 was taken as the level of significance.
Results
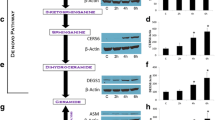
Several studies showed that expression and activity of acid sphingomyelinase is required for induction of apoptosis by some chemotherapeutic drugs, such as cisplatin or doxorubicin [24, 25]. Our group has recently demonstrated that cells transfected to overexpress acid sphingomyelinase become sensitive to gemcitabine-induced cell death [26]. In the present study, we stimulated SMA murine astrocytoma cells with clinically achievable doses of gemcitabine (100 nM) and determined the intrinsic activity of acid sphingomyelinase at different time points. We observed a biphasic enzyme activation pattern with an early but transient peak at 20–30 min posttreatment and a late, sustained increase starting 2 h after stimulation with gemcitabine (Fig. 1a).
Gemcitabine stimulation triggers acid sphingomyelinase activation and ceramide release (a). Treatment of SMA cells with 100 nM gemcitabine results in activation of acid sphingomyelinase (ASM) as determined by degradation of [14C]-labeled sphingomyelin to [14C]-labeled phosphorylcholine. The data are the mean±SD of three independent experiments. Significant differences (P ≤ 0.05, ANOVA) between stimulated and unstimulated samples are labeled by an asterisk. b SMA cells were stimulated with 100 nM gemcitabine for the indicated times and the levels of ceramide were determined by DAG-kinase assay, which phosphorylates ceramide to ceramide-1-phosphate in the presence of [32P]-gamma-ATP. Displayed is a typical result representative for five independent experiments
Acid sphingomyelinase activation leads to cleavage of sphingomyelin and an increase in cellular ceramide [7]. We therefore quantified ceramide levels biochemically in a time course after gemcitabine stimulation. The results show that treatment of SMA cells with gemcitabine induces a weak ceramide increase in response to the transient acid sphingomyelinase activation. However, a marked ceramide accumulation was observed starting 2 h posttreatment, which correlated with the later acid sphingomyelinase activation (Fig. 1b). These data indicate that clinically achievable doses of gemcitabine induce ceramide production via activation of the acid sphingomyelinase.
To clarify the biological significance of the acid sphingomyelinase/ceramide system for gemcitabine-induced apoptosis, we coincubated cells with amitriptyline and imipramine. Both drugs induce a rapid degradation of acid sphingomyelinase and are known to be functional inhibitors of the enzyme [29]. The drugs were applied 30 min prior to gemcitabine treatment, and cell death was assessed 48 h later by annexin V–propidium iodide staining. The results suggest that inhibition of the acid sphingomyelinase/ceramide pathway blocks cell death in SMA cells upon stimulation with gemcitabine (Fig. 2a). The drugs alone were without effect on cell viability (Fig. 2a). Control experiments were performed to assess the efficiency of amitriptyline and imipramine regarding inhibition of gemcitabine-induced ceramide release via the acid sphingomyelinase. To this end, cells were stimulated with 100 nM gemcitabine for 8 h in the presence or absence of 50 µM amitriptyline or 100 µM imipramine, respectively. Samples were stained employing Cy5-coupled anti-ceramide antibodies and analyzed by flow cytometry. The results show a significant decrease of gemcitabine-induced ceramide levels upon cotreatment with amitriptyline or imipramine (Fig. 2b), thus, indicate a crucial role of acid sphingomyelinase-mediated ceramide generation for gemcitabine signaling and cytotoxicity. Controls reveal that both drugs inhibited acid sphingomyelinase activity by approximately 80% (not shown).
The acid sphingomyelinase/ceramide pathway is important for gemcitabine-induced cell death. a Cotreatment of SMA cells with the acid sphingomyelinase inhibitors amitriptyline or imipramine prevents gemcitabine-induced apoptosis. Cell death was determined by flow cytometry analysis of annexin/propidium iodide-stained cells. Data are represented as percentage compared with the untreated sample, which was set at 100% survival. The results are the mean±SD of four independent experiments. Significant differences (P ≤ 0.05, ANOVA) between samples treated with gemcitabine alone and those treated with gemcitabine together with an acid sphingomyelinase inhibitor are labeled by an asterisk. b Cotreatment of SMA cells with the acid sphingomyelinase inhibitors amitriptyline or imipramine prevents gemcitabine-induced ceramide release. Ceramide was determined by flow cytometry analysis of cells stained with Cy5-coupled anti-ceramide antibodies. Displayed is a representative result out of three independent studies. c Overexpression of the acid sphingomyelinase in SMA cells sensitizes the cells to gemcitabine-induced cell death while deficiency of the acid sphingomyelinase in B lymphocytes lacking acid sphingomyelinase (derived from a Niemann-Pick disease type A patient) prevents gemcitabine-triggered cell death. Cell death was determined by FITC–annexin staining. Shown are the mean±SD of three independent studies (*P ≤ 0.05, ANOVA)
To further define the role of the acid sphingomyelinase for gemcitabine-induced cell death, we applied the drug to acid sphingomyelinase-overexpressing glioma cells (named SMA-ASM) or control-transfected cells (SMA-pJK) or human lymphocytes isolated from a Niemann-Pick disease type A patient that lacked expression of functional acid sphingomyelinase. The results show that acid sphingomyelinase-deficient cells were resistant to gemcitabine while over-expression of the acid sphingomyelinase sensitized the cells to the drug (Fig. 2c).
To characterize gemcitabine-induced apoptotic signaling via the acid sphingomyelinase/ceramide system, we investigated whether the proapoptotic molecule Bax is involved in this pathway. In viable cells, Bax is localized predominantly in the cytoplasm or is loosely associated with the mitochondrial compartment. Upon stimulation with different apoptotic stimuli, Bax is inserted into the mitochondrial membrane and signals initiation of the intrinsic apoptosis pathway [30]. We, therefore, stimulated SMA cells with gemcitabine for 24 h, and expression of Bax was assessed in the mitochondrial and cytoplasmic fractions, respectively. Western blot analysis demonstrates that gemcitabine treatment leads to a translocation of Bax from the cytoplasm into the mitochondria without a significant change of total cellular levels of Bax (Fig. 3a).
Gemcitabine treatment triggers the intrinsic apoptosis pathway via acid sphingomyelinase /ceramide. a SMA cells stimulated with gemcitabine respond with translocation of Bax from the cytoplasmic to the mitochondrial fraction. Cells were treated with 100 nM gemcitabine or left untreated for 24 h. Samples were collected; the mitochondrial and cytoplasmic fractions were separated, and each fraction was analyzed by SDS-PAGE followed by Western blotting using anti-Bax antibodies (upper panel). The blots were then stripped and reprobed using antibodies against the mitochondrial protein Tim23 (lower panel). Shown is a representative result out of three independent studies. b Cotreatment of SMA cells with the acid sphingomyelinase inhibitor amitriptyline blocks gemcitabine-induced Bax insertion into the mitochondria. Following stimulation, cells were fixed, permeabilized, stained with Cy3-anti-Bax and FITC–anti-Tim23 antibodies, and analyzed by confocal microscopy. The results are representative for four studies with analysis of each 150 cells per sample. c Primary human glioma cells treated with 1 µM gemcitabine for 72 h respond with translocation of Bax to the mitochondria as demonstrated by confocal microscopy analysis of cells stained with Cy3-anti-Bax and FITC–anti-Tim23 antibodies. The results are representative for three studies with analysis of each 150 cells per sample
Next, we employed the acid sphingomyelinase inhibitor amitriptyline to determine whether gemcitabine-induced Bax insertion into the mitochondria is downstream of acid sphingomyelinase. Confocal microscopy analysis of SMA cells stimulated with gemcitabine confirmed insertion of Bax into the mitochondria, an event that was blocked by pretreatment with amitriptyline in a dose-dependent manner (Fig. 3b).
To exclude that Bax insertion into the mitochondria following gemcitabine treatment is only a cell line-specific mechanism, we employed glioma cells isolated from human tumors shortly after surgery. Primary human glioma cells were incubated with 1 µM gemcitabine and stained 72 h later for Bax and mitochondrial marker Tim23. Although incubation times with gemcitabine and drug doses had to be adjusted in order to be lethal for these cells, primary cancer cells also presented a clear colocalization of Bax with the mitochondrial compartment upon stimulation with gemcitabine (Fig. 3c). The experiments were repeated employing two additional primary tumors with very similar results (data not shown).
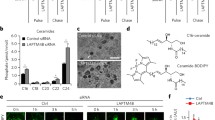
Our data indicate that acid sphingomyelinase and ceramide play a crucial role for cell death induced by gemcitabine. Next, we aimed to determine the exact subcellular localization of ceramide released upon gemcitabine treatment. To this end, SMA cells were stimulated with 100 nM gemcitabine, and immunofluorescence analysis was performed at different times posttreatment. The results confirm the biochemical quantification data and indicate a clear intracellular increase of ceramide (Fig. 4a). Eight hours after gemcitabine stimulation, ceramide localized to intracellular structures resembling vesicles (Fig. 4a, second panel). At later times, i.e., 12 and 18 h posttreatment, respectively, we observed an increased colocalization of ceramide with structures positive for the lysosomal marker Lamp1 and, thus, presumably a localization of ceramide within lysosomes (Fig. 4a, third and fourth panels).
Gemcitabine-induced ceramide accumulates in lysosomes (a). SMA cells were treated with 100 nM gemcitabine and fixed at the indicated times. Following permeabilization, samples were stained with Cy3-anti-ceramide and FITC–anti-Lamp1 antibodies. Confocal microscopy analysis indicates that gemcitabine induces a time-dependant accumulation of ceramide in lysosomes of treated cells. The results are representative for four studies with analysis of each 200 cells/sample. (b) Treatment of SMA cells with bafilomycine, which blocks lysosome functions, prevents gemcitabine-induced cell death. Displayed are the mean±SD of three independent studies (asterisks indicate significant differences compared with the respective control cells, P ≤ 0.05, ANOVA). (c) Gemcitabine induces an activation of cathepsin D, which is prevented by pretreatment of the cells with amitriptyline. Active cathepsin D is indicated by the detection of the shorter, active cathepsin D fragment. Shown is a representative Western blot of three experiments with similar results. (d) Primary human glioma cells treated with 1 µM gemcitabine for 48 h respond with release of ceramide in acidic organelles as indicated by confocal microscopy analysis of cells stained with Cy3-anti-ceramide and LysoSensor Green DND-189. The results are representative for three studies with analysis of each 150 cells per sample
Consistent with a role of lysosomal ceramide in cell death, inhibitors of lysosomal function, such as bafilomycine A or chloroquine, blocked gemcitabine-induced cell death (Fig. 4b and not shown). To further link the formation of lysosomal ceramide upon cellular treatment with gemcitabine to cell death, we determined whether gemcitabine triggers an activation of cathepsin D, which has been previously shown to mediate the effect of lysosomal ceramide on cell death [31, 32]. Our studies demonstrate that gemcitabine triggers an activation of cathepsin D, which is blocked by pretreatment with amitriptyline (Fig. 4c) providing a mechanistic link between lysosomal ceramide and cell death upon gemcitabine treatment.
We employed again human primary glioma cells to exclude potential cell line-specific mechanisms of the response to gemcitabine treatment. Primary glioma cells were stimulated with 1 µM gemcitabine for 48 h, incubated with LysoSensor Green DND-189 prior to ceramide staining, and analyzed by confocal microscopy. Since LysoSensor Green DND-189 has a low pK a value, a positive staining with this dye indicates acidic organelles. Our results show an almost complete colocalization of ceramide with the LysoSensor Green DND-189 fluorescent signal, which suggests that primary glioma cells also respond to gemcitabine treatment with an accumulation of ceramide in the lysosomal compartment (Fig. 4d). The experiments were repeated employing two additional primary tumors with very similar results (data not shown).
Discussion
The present study demonstrates that treatment of glioma cells with clinically achievable concentrations of gemcitabine triggers cell death via activation of acid sphingomyelinase and a subsequent increase of intracellular ceramide levels. Our study also characterizes a novel mechanism of gemcitabine-induced cell death and identifies accumulation of ceramide in lysosomes and insertion of proapoptotic molecule Bax into the mitochondria, respectively, as crucial events in this pathway.
Activation of acid sphingomyelinase and up-regulation of ceramide levels have been shown to play a role in signaling triggered by a variety of stimuli, such as members of the TNF family of ligands and receptors [10–12, 14], irradiation [16–19], several chemotherapeutic drugs [24–26], or pathogens [33–37]. The present study demonstrates an involvement of acid sphingomyelinase in gemcitabine-induced cell death since inhibition of the enzyme by amitriptyline or imipramine rescues cells from the lethal effect of the drug. Our study clarifies the kinetics of gemcitabine-induced acid sphingomyelinase activation as consisting of an early but transient activity peak followed by a sustained increase starting approximately 2 h posttreatment. The later activation correlates with the strong ceramide accumulation observed in our study. Interestingly, a very weak ceramide increase was observed in response to the early acid sphingomyelinase peak. This may be due to rapid consumption of ceramide at this level, suggesting that ceramide concentrations are kept at low levels to avoid cytotoxicity, at least in glioma cells.
Ceramide seems to be involved at several levels of the signaling pathways mediating cell death. Many studies agree on the role of ceramide in clustering of death receptors [10, 12, 14, 15]. It has been demonstrated that stimulation via CD95L/CD95R or TRAIL/DR5 leads to acid sphingomyelinase activation and ceramide production on the plasma membrane. Ceramide molecules tend to coalesce and form large ceramide-enriched membrane platforms, a prerequisite for signal amplification and induction of death via CD95 [10, 13, 14]. In addition, ceramide has been implicated in the activation of Rac-1 [38, 39] or PKC ζ [40, 41] leading to SAPK/JNK activation which, in turn, acts on targets such as c-jun, AP-1, or Bcl-2 [42–44]. In addition, the kinase suppressor of Ras (KSR) has been demonstrated to bind ceramide and to induce apoptosis via KSR, Ras, Raf-1, and BAD [45]. Another known ceramide target is phosphatase A2 (PPA2), which can negatively regulate prosurvival kinases such as Akt/PKB or antiapoptotic molecules such as Bcl-2 [46–49]. Recent studies from Zeidan and co-workers connect cisplatin-induced PPA2 activation via ceramide with ezrin dephosphorylation and changes in cell morphology and actin cytoskeleton [50].
Our study focused on determining the topology of ceramide upon stimulation with gemcitabine since this may lead to elucidating the mechanisms of gemcitabine-induced cell death. The results demonstrate that treatment of cells with gemcitabine for 8 h triggers an increased production of intracellular ceramide vesicles that colocalize with lysosomal structures at later times posttreatment. Furthermore, we demonstrate an activation of cathepsin D in glioma cells upon treatment with gemcitabine, which is prevented by amitripytline. The notion that cathepsin D mediates effects of lysosomal ceramide on cell death is consistent with findings of Heinrich and co-workers showing that an acid sphingomyelinase-derived ceramide specifically binds to and induces cathepsin D proteolytic activity in endolysosomal compartments and that acid sphingomyelinase-deficient cells derived from Niemann-Pick disease type A patients present decreased cathepsin D activity [31, 32]. Recently, the same group also demonstrated a TNF-induced activation of cathepsin D via acid sphingomyelinase and ceramide [31]. Cathepsin D, once activated, translocates to the cytosol and cleaves Bid, which leads to initiation of the intrinsic apoptosis pathway [31]. Since truncated Bid (tBid) is known to activate Bax, it is tempting to speculate that gemcitabine-induced acid sphingomyelinase activation and ceramide accumulation into the lysosomes, respectively, lead to cell death via a similar mechanism.
The notion that lysosomal ceramide kills cells via cathepsin D and Bax is supported by our data that demonstrate an inhibition of Bax translocation to mitochondria by amitriptyline.
Our studies show that 8 h incubation with gemcitabine results in the formation of ceramide in intracellular vesicles that do not costain with lysosomal markers, while 12–18 h incubation with gemcitabine induces a release of ceramide in lysosomes. Further, a detailed time course including very early time points (10 min to 4 h gemcitabine stimulation) showed that gemcitabine never induced ceramide in the outer leaflet of the cell membrane (data not shown) as previously observed for CD95. The ceramide observed early after gemcitabine might be released in endosomes that fuse with lysosomes. However, since the acid sphingomyelinase is primarily present in lysosomes and since gemcitabine induces a stable ceramide accumulation of ceramide in lysosomes for more than 6 hrs, it seems to be more likely that both vesicle populations are distinct and independent populations that respond to gemcitabine with the release of ceramide. While the later release of ceramide clearly occurs in classical lysosomes, the earlier ceramide accumulation might occur in other acidic vesicles that lack some of the typical lysosomal markers. However, a detailed analysis of the exact vesicle population in which ceramide accumulates requires very intensive cell fractionation and immunoelectron microscopy studies that are beyond the focus of the present study.
Previous publications have demonstrated that gemcitabine triggers activation of the acid sphingomyelinase and a release of ceramide via reactive oxygen species [26]. Here, we define for the first time the subcellular topology of ceramide release upon gemcitabine treatment and link gemcitabine to lysosomal acid sphingomyelinase and ceramide. Furthermore, we provide evidence for a novel mechanism of how gemcitabine triggers death via ceramide, the activation of lysosomal cathepsin D, and the stimulation of Bax proteins.
Understanding the exact molecular events triggered by stimuli currently used in cancer therapy may lead to a better modulation of the respective signaling cascades and to new and improved therapeutic strategies.
References
Fulda S, Debatin KM (2006) Extrinsic versus intrinsic apoptosis pathways in anticancer chemotherapy. Oncogene 25:4798–4811
Green DR (2005) Apoptotic pathways: ten minutes to dead. Cell 121:671–674
Lavrik I, Golks A, Krammer PH (2005) Death receptor signaling. J Cell Sci 118:265–267
Belka C, Budach W (2002) Anti-apoptotic Bcl-2 proteins: structure, function and relevance for radiation biology. Int J Radiat Biol 78:643–658
Daniel PT, Schulze-Osthoff K, Belka C, Güner D (2003) Guardians of cell death: the Bcl-2 family proteins. Essays Biochem 39:73–88
Brown DA, London E (1998) Functions of lipid rafts in biological membranes. Annu Rev Cell Dev Biol 14:111–136
Kolesnick RN, Goni FM, Alonso A (2000) Compartmentalization of ceramide signaling: physical foundations and biological effects. J Cell Physiol 184:285–300
Clarke CJ, Hannun YA (2006) Neutral sphingomyelinases and nSMase2: bridging the gaps. Biochim Biophys Acta 1758:1893–1901
Liu F, Cheng Y, Wu J, Tauschel HD, Duan RD (2006) Ursodeoxycholic acid differentially affects three types of sphingomyelinase in human colon cancer Caco 2 cells. Cancer Lett 235:141–146
Dumitru CA, Gulbins E (2006) TRAIL activates acid sphingomyelinase via a redox mechanism and releases ceramide to trigger apoptosis. Oncogene 25:5612–5625
Grassme H, Jekle A, Riehle A, Schwarz H, Berger J, Sandhoff K, Kolesnick R, Gulbins E (2001) CD95 signaling via ceramide-rich membrane rafts. J Biol Chem 276:20589–20596
Cremesti A, Paris F, Grassme H, Holler N, Tschopp J, Fuks Z, Gulbins E, Kolesnick R (2001) Ceramide enables fas to cap and kill. J Biol Chem 276:23954–23961
Grassme H, Cremesti A, Kolesnick R, Gulbins E (2003) Ceramide-mediated clustering is required for CD95-DISC formation. Oncogene 22:5457–5470
Grassme H, Schwarz H, Gulbins E (2001) Molecular mechanisms of ceramide-mediated CD95 clustering. Biochem Biophys Res Commun 284:1016–1030
Scheel-Toellner D, Wang K, Assi LK, Webb PR, Craddock RM, Salmon M, Lord JM (2004) Clustering of death receptors in lipid rafts initiates neutrophil spontaneous apoptosis. Biochem Soc Trans 32:679–681
Santana P, Pena LA, Haimovitz-Friedman A, Martin S, Green D, McLoughlin M, Cordon-Cardo C, Schuchman EH, Fuks Z, Kolesnick R (1996) Acid sphingomyelinase-deficient human lymphoblasts and mice are defective in radiation-induced apoptosis. Cell 86:189–199
Pena LA, Fuks Z, Kolesnick RN (2000) Radiation-induced apoptosis of endothelial cells in the murine central nervous system: protection by fibroblast growth factor and sphingomyelinase deficiency. Cancer Res 60:321–327
Paris F, Fuks Z, Kang A, Capodieci P, Juan G, Ehleiter D, Haimovitz-Friedman A, Cordon-Cardo C, Kolesnick R (2001) Endothelial apoptosis as the primary lesion initiating intestinal radiation damage in mice. Science 293:293–297
Garcia-Barros M, Paris F, Cordon-Cardo C, Lyden D, Rafii S, Haimovitz-Friedman A, Fuks Z, Kolesnick R (2003) Tumor response to radiotherapy regulated by endothelial cell apoptosis. Science 300:1155–1159
Charruyer A, Grazide S, Bezombes C, Müller S, Laurent G, Jaffrézou JP (2005) UV-C light induces raft-associated acid sphingomyelinase and JNK activation and translocation independently on a nuclear signal. J Biol Chem 280:19196–19204
Kashkar H, Wiegmann K, Yazdanpanah B, Haubert D, Krönke M (2005) Acid sphingomyelinase is indispensable for UV light-induced Bax conformational change at the mitochondrial membrane. J Biol Chem 280:20804–20813
Rotolo JA, Zhang J, Donepudi M, Lee H, Fuks Z, Kolesnick R (2005) Caspase-dependent and -independent activation of acid sphingomyelinase signaling. J Biol Chem 280:26425–26434
Zhang Y, Mattjus P, Schmid PC, Dong Z, Zhong S, Ma WY, Brown RE, Bode AM, Schmid HH, Dong Z (2001) Involvement of the acid sphingomyelinase pathway in UVA-induced apoptosis. J Biol Chem 276:11775–11782
Morita Y, Perez GI, Paris F, Miranda SR, Ehleiter D, Haimovitz-Friedman A, Fuks Z, Xie Z, Reed JC, Schuchman EH, Kolesnick RN, Tilly JL (2000) Oocyte apoptosis is suppressed by disruption of the acid sphingomyelinase gene or by sphingosine-1-phosphate therapy. Nat Med 6:1109–1114
Lacour S, Hammann A, Grazide S, Lagadic-Gossmann D, Athias A, Sergent O, Laurent G, Gambert P, Solary E, Dimanche-Boitrel MT (2004) Cisplatin-induced CD95 redistribution into membrane lipid rafts of HT29 human colon cancer cells. Cancer Res 64:3593–3598
Grammatikos G, Teichgraber V, Carpinteiro A, Trarbach T, Weller M, Hengge UR, Gulbins E (2007) Overexpression of acid sphingomyelinase sensitizes glioma cells to chemotherapy. Antioxid Redox Signal 9:1449–1456
Serano RD, Pegram CN, Bigner DD (1980) Tumorigenic cell culture lines from a spontaneous VM/Dk murine astrocytoma (SMA). Acta Neuropathol 51:53–64
Gulbins E, Kolesnick R (2000) Measurement of sphingomyelinase activity. Methods Enzymol 322:382–388
Hurwitz R, Ferlinz K, Sandhoff K (1994) The tricyclic antidepressant desipramine causes proteolytic degradation of lysosomal sphingomyelinase in human fibroblasts. Biol Chem Hoppe Seyler 375:447–450
Lalier L, Cartron PF, Juin P, Nedelkina S, Manon S, Bechinger B, Vallette FM (2007) Bax activation and mitochondrial insertion during apoptosis. Apoptosis 12:887–896
Heinrich M, Neumeyer J, Jakob M et al (2004) Cathepsin D links TNF-induced acid sphingomyelinase to Bid-mediated caspase-9 and -3 activation. Cell Death Differ 11:550–563
Heinrich M, Wickel M, Schneider-Brachert W, Hallas C, Tchikov V, Winoto-Morbach S, Wickel M, Schneider-Brachert W, Trauzold A, Hethke A, Schütze S (1999) Cathepsin D targeted by acid sphingomyelinase-derived ceramide. Embo J 18:5252–5263
Esen M, Schreiner B, Jendrossek V, Lang F, Fassbender K, Grassme H, Gulbins E (2001) Mechanisms of Staphylococcus aureus induced apoptosis of human endothelial cells. Apoptosis 6:431–439
Grassme H, Gulbins E, Brenner B, Ferlinz K, Sandhoff K, Harzer K, Lang F, Meyer TF (1997) Acidic sphingomyelinase mediates entry of N. gonorrhoeae into nonphagocytic cells. Cell 91:605–615
Grassme H, Jendrossek V, Riehle A, von Kürthy G, Berger J, Schwarz H, Weller M, Kolesnick R, Gulbins E (2003) Host defense against Pseudomonas aeruginosa requires ceramide-rich membrane rafts. Nat Med 9:322–330
Grassme H, Riehle A, Wilker B, Gulbins E (2005) Rhinoviruses infect human epithelial cells via ceramide-enriched membrane platforms. J Biol Chem 280:26256–26262
Jan JT, Chatterjee S, Griffin DE (2000) Sindbis virus entry into cells triggers apoptosis by activating sphingomyelinase, leading to the release of ceramide. J Virol 74:6425–6432
Brenner B, Koppenhoefer U, Weinstock C, Linderkamp O, Lang F, Gulbins E (1997) Fas- or ceramide-induced apoptosis is mediated by a Rac1-regulated activation of Jun N-terminal kinase/p38 kinases and GADD153. J Biol Chem 272:22173–22181
Yi F, Chen QZ, Jin S, Li PL (2007) Mechanism of homocysteine-induced Rac1/NADPH oxidase activation in mesangial cells: role of guanine nucleotide exchange factor Vav2. Cell Physiol Biochem 20:909–918
Lozano J, Berra E, Municio MM, Diaz-Meco MT, Dominguez I, Sanz L, Moscat J (1994) Protein kinase C zeta isoform is critical for kappa B-dependent promoter activation by sphingomyelinase. J Biol Chem 269:19200–19202
Bourbon NA, Yun J, Kester M (2000) Ceramide directly activates protein kinase C zeta to regulate a stress-activated protein kinase signaling complex. J Biol Chem 275:35617–35623
Basu S, Kolesnick R (1998) Stress signals for apoptosis: ceramide and c-Jun kinase. Oncogene 17:3277–3285
Jarvis WD, Grant S, Kolesnick RN (1996) Ceramide and the induction of apoptosis. Clin Cancer Res 2:1–6
Ruvolo PP (2001) Ceramide regulates cellular homeostasis via diverse stress signaling pathways. Leukemia 15:1153–1160
Basu S, Bayoumy S, Zhang Y, Lozano J, Kolesnick R (1998) BAD enables ceramide to signal apoptosis via Ras and Raf-1. J Biol Chem 273:30419–30426
Dobrowsky RT, Kamibayashi C, Mumby MC, Hannun YA (1993) Ceramide activates heterotrimeric protein phosphatase 2A. J Biol Chem 268:15523–15530
Ruvolo PP, Clark W, Mumby M, Gao F, May WS (2002) A functional role for the B56 alpha-subunit of protein phosphatase 2A in ceramide-mediated regulation of Bcl2 phosphorylation status and function. J Biol Chem 277:22847–22852
Ruvolo PP, Deng X, Ito T, Carr BK, May WS (1999) Ceramide induces Bcl2 dephosphorylation via a mechanism involving mitochondrial PP2A. J Biol Chem 274:20296–20300
Salinas M, Lopez-Valdaliso R, Martin D, Alvarez A, Cuadrado A (2000) Inhibition of PKB/Akt1 by C2-ceramide involves activation of ceramide-activated protein phosphatase in PC12 cells. Mol Cell Neurosci 15:156–169
Zeidan YH, Jenkins RW, Hannun YA (2008) Remodeling of cellular cytoskeleton by the acid sphingomyelinase/ceramide pathway. J Cell Biol 181:335–350
Conflict of interest statement
The authors declare that they have no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dumitru, C.A., Sandalcioglu, I.E., Wagner, M. et al. Lysosomal ceramide mediates gemcitabine-induced death of glioma cells. J Mol Med 87, 1123–1132 (2009). https://doi.org/10.1007/s00109-009-0514-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-009-0514-8