Abstract
The durability of five bamboo species from various origin against brown-, white- and soft-rot fungi was investigated in Kolle flasks in accordance with the European standards EN 350-1, EN 350-2 and EN 113. Considerable variability exists in the durability of the bamboo species. Guadua angustifolia was rather resistant to Trametes versicolor and Dendrocalamus asper against Chaetomium globosum. Among the brown-rot fungi, the four strains of Coniophora puteana and two strains of Gloeophyllum trabeum produced low mass loss (maximum 2.9 %). Of the white-rot fungi, T. versicolor yielded the highest decay (max. 15.3 %), whereas Schizophyllum commune was rather inactive (max. 3.2 %). Of the soft-rot fungi, Ch. globosum showed medium degradation (max. 9.6 %) and Paecilomyces variotii low decay (max. 3.1 %).
Zusammenfassung
Fünf Bambus-Arten verschiedener Herkunft wurden in Kolleschalen auf ihre Anfälligkeit für Braun-, Weiß- und Moderfäulepilze gemäß EN 350-1, EN 350-2 und EN 113 untersucht. Die Bambus-Arten unterschieden sich in ihrer Pilzanfälligkeit. Guadua angustifolia war besonders widerstandsfähig gegen Trametes versicolor und Dendrocalamus asper gegen Chaetomium globosum. Bei den Braunfäule-Erregern ergaben die vier Stämme von Coniophora puteana und beide Isolate von Gloeophyllum trabeum nur geringen Masseverlust (maximal 2,9 %). Von den beiden Weißfäulepilzen zeigte T. versicolor den stärksten Abbau (max. 15,3 % ), während Schizophyllum commune kaum aktiv war (max. 3,2 %). Bei den Moderfäulepilzen ergab Ch. globosum mittleren (max. 9,6 %) und Paecilomyces variotii geringen Abbau (max. 3,1 %).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Bamboo is a fast growing woody grass of increasing interest for the sustainable production of materials with many applications for buildings and industrial utilization. However, bamboo has generally a low natural durability and is easily attacked by fungi during storage, transport and final use. For applications it is important to know its susceptibility.
Like other lignocellulose materials, bamboos are subject to biodegradation by fungi under particular conditions which affects their quality (Hamid et al. 2003). The resistance of bamboo against decay fungi serves as an important parameter in bamboo uses. Several factors may influence bamboo natural durability, for example site, growth rate, age, portion of bamboo, extractives content and the microenvironment. The results obtained in this work, together with other data reported in literature on the decay of bamboo (Liese 1959, 1985; Banerjee and Mukhopadhyay 1962; Purushotham 1963; Abdurachim 1964; Wang and Hsieh 1968; Murphy et al. 1991; Razak et al. 2002, 2006; Hamid et al. 2003; Zhang et al. 2007; Kim et al. 2008, 2011; Suprapti 2010; Ma et al. 2010; Schmidt et al. 2011; Wei et al. 2012) may contribute to the characterization and appreciation of bamboo species. Such characterization is important not only for its correct utilization but also for the market promotion.
When comparing data, another difficulty is due to the different tests performed, such as field and laboratory tests. Also within the same test, the different standards (Asia, European and American) may differ by sample size, duration of tests and fungal strains. Therefore, it is important to test the bamboo species using the same standards. The aim of the study is to test bamboo durability using European standards as used for wood species.
2 Materials and methods
2.1 Investigated fungi
The ten strains of white-, brown- and soft-rot fungi used derive from the laboratory strain collection and are listed in Table 1.
2.2 Bamboo and wood samples
The five bamboo species tested are presented in Table 2. Culm sections were obtained from CONBAM, Geilenkirchen, Germany and the Bamboo Centre, Baden–Baden, Germany. Sections were proved with the curcuma-test (Peylo 2001) to ensure that no boron was present which is commonly used against moulding during storage and transport. Samples were prepared by cutting to dimensions of 5 (length) × 2.5 (width) × 0.5–3.5 cm (wall thickness). Wood samples from Fagus sylvatica and Pinus sylvestris were used as controls according to EN 113. All samples were put into a climate room at 20 ± 2 °C and 65 ± 5 % relative humidity (RH) for 4 weeks, weighed, wrapped in plastic (Sengewald Flexopeel, Germany) and send to BBF Sterilisationsservice GmbH, Kernen, Germany for gamma radiation.
2.3 Durability test
The durability experiment was performed with fungal pure cultures in Kolle flasks with white-, brown-, and soft-rot fungi according to the European standards EN 350-1 (1996), EN 350-2 (1997) and EN 113 (1996). The culture medium consisted of 2 % malt extract (Merck) and 1.5 % agar (Bacto) in distilled water. Flasks containing 48 ml medium were plugged with cotton, sterilized in the autoclave at 121 °C for 30 min, inoculated with a mycelial plug of the test fungus and incubated until mycelial growth covered the surface of the medium. Each two gamma ray sterilized samples were put aseptically in one flask and each three flasks were used for one fungal strain, with altogether 480 Kolle flasks. After 16 weeks of incubation at 20 ± 2 °C and 65 ± 5 % RH for the brown-rot and white-rot fungi and 28 ± 2 °C and 65 ± 5 % RH for the soft-rot fungi, the mycelium was removed from the sample surface, the samples were weighed for final wet weight determination and oven dried at 103 °C. Percentage of mass loss was calculated by weight comparison before and after incubation. Statistical differences were analyzed using variance analysis (ANOVA, SPSS software 2002).
2.4 Assessment of fungal growth and classification of bamboo durability
The development of fungal growth on the specimens was evaluated by the density and the percentage of the sample surface covered with mycelium (Table 3). Based on the average mass loss, bamboo durability against fungi was correlated to durability classes (Table 3) considering EN 350-1 (1996), Abdurachim (1975) and Djarwanto and Suprapti (2004).
3 Results and discussion
3.1 Growth of fungi and final moisture content of bamboo samples
The development of hyphal growth was measured by scoring the density of hyphal mats and the percentage coverage of the sample surface. After initial differences between the fungi within the first 2 weeks, most bamboo species were covered by mycelium after 16 weeks between two-thirds of the surface and total coverage (Table 3). An exception with low surface growth occurred on Guadua angustifolia. In general, coverage correlated with hyphal density. The density of mycelium is a specific feature for many fungi (Stalpers 1978). However, it must not relate to fungal activity (Schmidt 2006). Among the ten strains, Gloeophyllum trabeum 183 did not show any hyphae on the surface of samples from Bambusa maculata, Dendrocalamus asper and Guadua angustifolia. However, this strain produced some degradation (Fig. 1), obviously not by surface growth but by substrate mycelium. The final moisture content of the samples ranged from 39 to 101 %, which is generally a suitable range for fungal degradation. Former experiments showed fungal bamboo degradation in the moisture range from 24 to 187 % (Schmidt et al. 2011).
Mass loss caused by brown-rot fungi in 5 bamboo species after 16 weeks of incubation. Error bars represent mean ± standard deviation. A Bambusa maculata; B Dendrocalamus asper; C Gigantochloa atroviolacea; D Guadua angustifolia; E Phyllostachys pubescens China; F Phyllostachys pubescens Germany
Masseverluste durch Braunfäulepilze bei 5 Bambus-Arten nach 16 Wochen Inkubation. Fehlerbalken zeigen ± Standardabweichung. A Bambusa maculata; B Dendrocalamus asper; C Gigantochloa atroviolacea; D Guadua angustifolia; E Phyllostachys pubescens China; F Phyllostachys pubescens Deutschland
3.2 Durability of bamboo against brown-rot fungi
All five bamboo species were rather resistant to degradation by the various strains of the brown-rot fungi Coniophora puteana and Gloeophyllum trabeum (Fig. 1) without significant variation among the strains. Strain variation with regard to wood mass loss was shown for both species (Schmidt et al. 2002a, 2002b). Both fungi comprised the test strains to be used in EN 113 (1996) for wood samples. In this standard, C. puteana strain Ebw. 15 produces about 20 % mass loss in wood and wood products. Among the four C. puteana strains, the maximum mass loss of bamboo was 2.9 %. The two G. trabeum strains also revealed no significant difference in bamboo degradation, regardless of the species. Mass losses of the wood controls for the four C. puteana strains and two G. trabeum isolates ranged from 19.7 to 62.7 % of decay. This shows suitable incubation conditions for bamboo. The low activity against bamboo correlates with previous work by the authors (Schmidt et al. 2011; Wei et al. 2012) reporting maximum 5.7 % mass loss by both fungi for the same bamboo species of the same origin after 1 year of incubation in preserving jars. However, samples of Melocanna bambusoides after 6 month incubation in Kolle flasks had shown up to 13.7 % decay by C. puteana Ebw. 15 (Schmidt et al. 2011). To prove these earlier results, the degradation test according to EN 350 and EN 113 considered sample sterilization by gamma radiation. The highest mass loss of 3 % by G. trabeum is lower than the one reported by Schmidt et al. (2011) with maximum 5.7 % of decay after 1 year in preserving jars. However, both results contrast with Lee et al. (2006) who used potato dextrose agar in petri dishes and found 25 % degradation by G. trabeum in Phyllostachys pubescens. The reason for this discrepancy may be the use of different isolates and cultivation methods.
3.3 Durability of bamboo against white-rot fungi
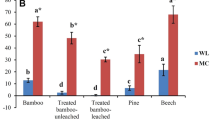
Among the two white-rot fungi, Trametes versicolor showed the greatest mass loss with 15.3 % for P. pubescens from China, followed by P. pubescens from Germany (12.3 %) and Gigantochloa atroviolacea (10.4 %) (Fig. 2). Guadua angustifolia was very resistant (2.3 %). Degradation of the Fagus sylvatica control was 26.7 %. Schizophyllum commune behaved rather inactive with maximum mass loss of only 3.2 %. Schmidt et al. (2011) obtained with S. commune a maximum of up to 9.1 % degradation in 6 months in Kolle flasks and a maximum of 5.6 % mass loss during 1 year in preserving jars. Abdurachim (1964) reported 15 % mass loss by S. commune. Low decay rates by S. commune were found by Suprapti (2010) and Kim et al. (2011). The beech wood control in this experiment showed also negligible decay (0.8 %), which corresponds to previous results (Schmidt and Liese 1978). However, this species is common on bamboo during storage and use (Liese 1985; Mohanan 1997; Liese and Kumar 2003). Kleist et al. (2002) showed that S. commune was the most successful coloniser among some fungi as it could penetrate bamboo via outer and inner culm walls, through cross section planes as well as through nodal ridges and longitudinally through nodes.
Mass loss caused by white-rot fungi in 5 bamboo species after 16 weeks of incubation. Error bars represent mean ± standard deviation. A Bambusa maculata; B Dendrocalamus asper; C Gigantochloa atroviolacea; D Guadua angustifolia; E Phyllostachys pubescens China; F Phyllostachys pubescens Germany
Masseverluste durch Weißfäulepilze bei 5 Bambus-Arten nach 16 Wochen Inkubation. Fehlerbalken zeigen ± Standardabweichung. A Bambusa maculata; B Dendrocalamus asper; C Gigantochloa atroviolacea; D Guadua angustifolia; E Phyllostachys pubescens China; F Phyllostachys pubescens Deutschland
3.4 Durability of bamboo against soft-rot fungi
The two soft-rot fungi, Chaetomium globosum and Paecilomyces variotii revealed significant differences with regard to bamboo decay, with higher mass loss by Ch. globosum than by P. variotii (Fig. 3). Highest mass loss was caused by Ch. globosum to P. pubescens from China (9.6 %), followed by Guadua angustifolia (8.1 %) and Bambusa maculata (7.5 %). Schmidt et al. (2011) reported up to 52.7 % decay by Ch. globosum after 6 months in Kolle flasks and maximum 38 % after 1 year in preserving jars. Suprapti (2010) measured 8.0 % mass loss. Kim et al. (2011) obtained up to 17.9 % decay for other soft-rot species. Maximum mass loss by P. variotii was only 3.1 %. The same strain of the species had shown maximum 3.9 % degradation in preserving jars, but another strain up to 19.7 % decay in Kolle flasks (Schmidt et al. 2011). The wood controls in this experiment revealed only rather low mass losses around 1 % which explains that soft-rot degradation of wood samples is tested by the specific standard EN 807 (2001).
Mass loss caused by soft-rot fungi in 5 bamboo species after 16 weeks of incubation. Error bars represent mean ± standard deviation. A Bambusa maculata; B Dendrocalamus asper; C Gigantochloa atroviolacea; D Guadua angustifolia; E Phyllostachys pubescens China; F Phyllostachys pubescens Germany
Masseverluste durch Moderfäulepilze bei 5 Bambus-Arten nach 16 Wochen Inkubation. Fehlerbalken zeigen ± Standardabweichung. A Bambusa maculata; B Dendrocalamus asper; C Gigantochloa atroviolacea; D Guadua angustifolia; E Phyllostachys pubescens China; F Phyllostachys pubescens Deutschland
3.5 Classification of bamboo durability
The bamboos were grouped into 5 durability classes as it is done for wood species. Table 3 shows the durability classification of all bamboo/fungus combinations derived from Figs. 1, 2, 3. The bamboo species investigated belong to 3 different classes. There were considerable differences. The resistances of the five bamboo species to the brown-rot fungi C. puteana and G. trabeum, the white-rot fungus S. commune and the soft-rot species P. variotii would group them into class II (durable). The soft-rot fungus Ch. globosum attacked the bamboos according to classes II and III (moderately durable). Considering white-rot decay by T. versicolor, the five bamboo species varied between classes II and IV (little durable).
4 Conclusion
Bamboo species differ in their susceptibility to fungal decay. With regard to the different groups of rot fungi, bamboo seems to be rather resistant against brown-rot fungi, whereas some soft-rot and white-rot fungi produce considerable deterioration. Results correspond to findings by other authors in laboratory studies and to observations in the practice, where bamboo is often colonized and deteriorated by fungi.
References
Abdurachim MRA (1964) Bamboo preservation in Indonesia. Rimba Indonesia 9:66–76
Abdurachim MRA (1975) Laboratory testing on the resistance of wood against fungi. Ministry of Agriculture, Jakarta
Banerjee S, Mukhopadhyay S (1962) A study on Merulius similis B. & BR. and the associated bamboo-rot. Öster Bot Zeitschr 109:197–212
Djarwanto, Suprapti S (2004) Laboratory testing on the resistance of wood against fungi. In: Herjanto E et al. (Eds) Prosiding Pertemuan dan Presentasi Ilmiah Standardisasi. Jakarta, pp 15–22
EN 113 (1996) Determination of the toxic values of wood preservatives against wood destroying Basidiomycetes cultured on agar medium. Beuth, Berlin
EN 350-1 (1996) Durability of wood and wood-based products. Natural durability of solid wood. Part 1: Guide to the principles of testing and classification of natural durability of wood. Beuth, Berlin
EN 350-2 (1997) Durability of wood and wood-based products. Natural durability of solid wood. Part 2: Guide to natural durability and treatability of selected wood species of importance in Europe. Beuth, Berlin
EN 807 (2001) Wood preservatives—Determination of the effectiveness against soft rotting micro-fungi and other soil inhabiting micro-organisms. Beuth, Berlin
Hamid NH, Abd Latif M, Sulaiman O (2003) Decay resistance of bamboo (Gigantochloa scortechinii) compared to 24 Malaysian hardwood. XII. World Forestry Congress 2003, Québec, Canada. www.fao.org/docrep/article/wfc/XII/0039-b4.htm
Kim JS, Lee KH, Cho CH, Koch G, Kim YS (2008) Micromorphological characteristics and lignin distribution in bamboo (Phyllostachys pubescens) degraded by the white rot fungus Lentinus edodes. Holzforschung 62:481–487
Kim JJ, Lee SS, Ra JB, Lee H, Huh N, Kim GH (2011) Fungi associated with bamboo and their decay capabilities. Holzforschung 65:271–275
Kleist G, Morris I, Murphy R (2002) Invasion and colonization of bamboo culm material by stain and decay fungi. The International Research Group on Wood Preservation, IRG Document No. IRG/WP 02-10453
Lee KH, Cho CH, Kim YS (2006) Micromorphology of bamboo fibers degraded by brown-rot fungus Gloeophyllum trabeum. The International Research Group on Wood Preservation, IRG Document No. IRG/WP 06-10576
Liese W (1959) Bamboo preservation and soft-rot. Food and Agriculture Organization Report to the Government of India, FAO Document No. 1106
Liese W (1985) Bamboos—biology, silvics, properties, utilization. Schriftenreihe Gesellschaft für Technische Zusammenarbeit, GTZ Document No. 180
Liese W, Kumar S (2003) Bamboo preservation compendium. International Network for Bamboo and Rattan, INBR Document No. 22
Ma X, Jiang M, Qin D (2010) The invasion channels of damage fungi in bamboo lumber. The International Research Group on Wood Preservation, IRG Document No. IRG/WG 10-10712
Mohanan C (1997) Diseases of bamboos in Asia. International Development Research Centre, New Delhi
Murphy RJ, Alvin KL, Tan YF (1991) Development of soft rot decay in the bamboo Sinobambusa tootsik. IAWA Bulletin n s 12:85–94
Peylo A (2001) Schnellanalyse von Holzschutzmitteln. Schützen & Erhalten:25–27
Purushotham A (1963) Utilization of bamboo. J Timber Dry Pres Assoc India 9:2–19
Razak W, Hashim WS, Murphy RJ (2002) SEM observation on the decay of bamboo Gigantochloa scortechinii exposed in tropical soil. J Trop For Prod 8:168–178
Razak W, Mahmud S, Tamizi M, Awang AMY (2006) Durability performance of Gigantochloa scortechinii through laboratory fungal decay tests. Res J Microbiol 1:198–202
Schmidt O (2006) Wood and tree fungi. Biology, damage, protection, and use. Springer, Berlin
Schmidt O, Liese W (1978) Biological variations within Schizophyllum commune. Material Organisms 11:215–230
Schmidt O, Grimm K, Moreth U (2002a) Molecular identity of species and isolates of the Coniophora cellar fungi. Holzforschung 56:563–571
Schmidt O, Grimm K, Moreth U (2002b) Molekulare und biologische Charakterisierung von Gloeophyllum-Arten in Gebäuden. Z Mykol 68:141–152
Schmidt O, Wei D, Liese W, Wollenberg E (2011) Fungal degradation of bamboo samples. Holzforschung 65:883–888
Stalpers J (1978) Identification of wood-inhabiting Aphyllophorales in pure culture. Studies Mycol 16:1–248
Suprapti S (2010) Decay resistance of five Indonesian bamboo species against fungi. J Trop For Sci 22:287–294
Wang SF, Hsieh RC (1968) Durability records of treated and untreated bamboo. Coop Bull Taiwan For Res Inst 15:26
Wei D, Schmidt O, Liese W (2012) Susceptibility of bamboo to fungi. IXth World Bamboo Congress, pp 235–245
Zhang X, Yu H, Huang H, Liu Y (2007) Evaluation of biological pretreatment with white rot fungi for the enzymatic hydrolysis of bamboo culm. Int Biodeterior Biodegrad 60:159–164
Acknowledgments
We thank Wolfgang Eberts (Bamboo Centre Germany) and Christoph Tönges (CONBAM, Germany) for providing bamboo sections and Marie-Therese Lenz for technical help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wei, D., Schmidt, O. & Liese, W. Durability test of bamboo against fungi according to EN standards. Eur. J. Wood Prod. 71, 551–556 (2013). https://doi.org/10.1007/s00107-013-0707-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00107-013-0707-2