Abstract
Introduction
The occurrence of vascular injury during drilling is a rare but severe complication. Unfortunately, drilling protection systems are not available in all hospitals. Thanks to the development of 3D printing in recent years, sharing devices and materials to solve surgical problems has become easy and inexpensive. The objective of our work is to evaluate a universal, freely accessible, 3D-printable drilling protection device.
Materials and methods
A prospective, randomized, and triple-blind evaluation of excess drilling was evaluated in expert surgeons and resident surgeons without the use of the protection device. Subsequently, excess drilling was assessed using the device in both groups. The differences in drilling lengths between both groups with and without the device were analyzed to establish the reduction of excess drilling in both groups.
Results
The drilling in the expert surgeon without device group was 3.03 mm (SD 1.69 95% CI 2.40–3.66 p value = 9.89e−11), while the resident surgeon with device group performed excess drilling of 1.76 mm (SD 0.89 CI 95% 1.43–2.10). Results showed better results in the resident surgeon with device group than in the expert surgeons with device group (p value ˂ 0.01). The device improved drilling in both resident surgeons (5.77 mm) and expert surgeons (1.17 mm).
Conclusions
The device improves drilling in both expert surgeons and resident surgeons, showing greater benefits in the latter. The device is printable with any 3D printer, making it universally accessible, inexpensive, and effective, allowing expert surgeons to improve precision in high-risk situations and resident surgeons to improve their technique without increasing risk to the patient.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fractures of the proximal femur affect ~ 3.5% of men and 14.6% of women throughout their adult life [1]. Its incidence increases year by year due to the progressive aging of the world’s population, with a projected 100% increase in prevalence in the first half of the 21st century [2,3,4,5].
The primary treatment of hip fractures is surgery, with conservative treatment reserved for those patients at high surgical risk due to their comorbidities or for nondisplaced intracapsular fractures in selected patients [3, 6,7,8,9,10]. Of the surgical implant options available, sliding hip screws and intramedullary nails are the two most frequently used options [9, 11, 12], with the use of cannulated screws being much less frequent [13].
Vascular injury is one of the least frequent and most severe surgical complications, with a prevalence of 0.2–0.49% depending on the series [14]. When it occurs, it increases direct mortality by 6% and favors the appearance of new comorbidities in about 11% of patients [15]. In 42% of the cases, the deep femoral artery was the injured artery, and also the most frequently damaged vessel, followed by its perforating branches, which occurs in 22% of cases [15]. The intrapelvic arteries are traumatized less frequently (8.24% vs. 91.2% of the extrapelvic arteries), with the external iliac artery being the most frequently injured, only 2% of the time.
Pseudoaneurysm is the result of 67% of iatrogenic vascular lesions and is the most important in terms of frequency, followed by laceration with hemorrhage, which occurs 23% of the time [15, 16]. Thus, the symptoms are often not easily identifiable, thereby delaying diagnosis and, in some cases, may even go so far as a hospital discharge without it [16].
Between 30 and 60% of iatrogenic vascular lesions could easily be prevented [15, 16]. Although the traction of the minor trochanter fragment is a cause to consider [17], the drilling of the second cortex is responsible for about 20% of injuries, especially during the placement of the third and fourth nail plate screws and the short nail locking screw.
For many years, instrument kits have carried brand-specific devices designed to control the drilling of the second cortex. Because vascular injury is rare, surgeons often dispense with its use, which usually leads to its disappearance due to loss or its withdrawal due to disuse. This loss also occurs in university hospitals or hospitals with resident surgeons, where due to the infrequent use by experienced surgeons, younger surgeons do not have these aids, which implies an increased risk of vascular injury.
The objective of this article is to describe and evaluate an easily accessible device of universal use that is effective in controlling excessive drilling of the second cortex.
Materials and methods
Design
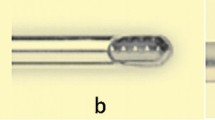
The design created by the bioengineering team of our hospital can be printed with any domestic 3D FFF (fused filament fabrication) printer and is explained in Fig. 1.
Description of the device. 1.1: screw-on-nut system with a lateral slit that allows drill insertion at any time without removing the motor. 1.2: during drill insertion, the screw rotates on the nut, which, on the one hand, firmly traps the drill and, on the other hand, modifies the length of the device to suit each patient. 1.3: an intraoperative image of the device during a short nail bolt placement. 1.4: detail of how once the first cortex has been drilled anddrill rests on the second cortex (image 1.6 is correlative to moment 1.4), the nut is turned to adjust its length ~1 cm from the end of the motor. In this way, the drill travels further until it passes the second cortex (image 1.5), thereby avoiding excess drilling when coming in contact with the device (image 1.6 and 1.7). The device was printed with medical poly lactic acid (Smartfil Medical, USP VI/ISO 10993-1 certified) and sterilized with a conventional 134° autoclave program. The parameters for the 3D impression are described in Table 1
After creating the design (3D Builder, Microsoft Corporation, Washington, USA), it was then converted into a conventional three-dimensional.stl format (Meshmixer® version 3.5.474) and configured for 3D printing in.gcode (Cura Software® version 4.3.0). Both documents were shared electronically for direct download via a web link.
For the evaluation of the device, a bi-cortical bone drill simulation system was designed, which is presented and explained in Fig. 2.
Description of the evaluation device. 2.1: in yellow, an overview of a simulated bi-cortical element with a thickness similar to the bone cortex. 2.2: complete lateral view in which the simulation of the two cortexes separated by a space and below the second cortex, represented in red a deformable plastic element in which excessive drilling is recorded. 2.3: in blue, the drill resting on the second cortex is represented. 2.4: the hole in the plastic device. Once drilling is carried out, drilling depth is measured using a screw gauge
Device evaluation
Cross-sectional study
Ten subjects were obtained by randomization using a table of random numbers, from a group of 20 trauma residents with < 10 surgeries per year as primary surgeon. Likewise, 10 subjects were obtained by randomization using a table of random numbers from a group of 20 expert surgeons with more than 50 surgeries per year as primary surgeon.
Each subject was asked to perform a bi-cortical drilling (Fig. 2) in triplicate, and in each attempt, the excessive drilling was evaluated to assess the learning curve. Subsequently, the same procedure was carried out, also in triplicate, using the evaluated protection device to study the learning curve of its use.
The excessive drilling in each attempt was evaluated, and differences in drilling lengths between expert surgeons without the device and resident surgeons with the device were defined as the main variable. Meanwhile, the differences between drilling lengths of expert surgeons with and without the device were defined as secondary data and were evaluated to analyze its usefulness in this group, and the same was carried with the resident. Finally, the learning curve of the use of the device was evaluated in both expert surgeons and resident surgeons (Fig. 3).
To evaluate the excessive drilling depth, a standard depth gauge for screws was used.
Statistics
The normal behavior of the data was analyzed (Shapiro–Wilk) to define the test used during the hypothesis testing for both the paired data (Student’s t test for paired data vs. Wilcoxon test) and independent data (Student’s t test vs. U of Mann–Whitney). In cases of comparison, multiple quantitative data between two groups were chosen through a variance analysis or Friedman’s test, based on the normal behavior of their data.
The statistics was carried out by an independent researcher with R statistics version 3.5.2.
Results
The expert surgeons drilling without device mean was 3.03 (SD 1.69 95% CI 2.40–3.66 p value = 9.89e−11) while that of residents with the device was 1.76 mm (SD 0.89 95% CI 1.43–2.10 p value = 1.169e−11), meaning that residents with the device performed drill lengths 1.26 mm smaller than those expert surgeons without the device, with this difference being statistically significant (95% CI 0.56–1.97, p value = 0.0007) (Fig. 4, 4.1).
The resident surgeon drilling without the device mean was 6.93 mm (SD 2.44 95% CI 6.01–7.84 p value = 1.415e−15), which means a higher drilling length of 5.17 mm compared to drilling with the device, with this increase being statistically significant (95% CI 4.2–6.1 p value = 5.322e−13) (Fig. 4, 4.2).
In the expert surgeon group, the drilling with the device mean was 1.86 mm (SD 0.89 CI 95% 1.53–2.20 p = 3.349e−12 value), this value being 1.17 mm lower than the mean value without the device, which implies a statistically significant improvement in drilling length (95% CI 0.46–1.87 p value = 0.001) (Fig. 4, 4.3).
Residents without the device showed higher drilling lengths than the experts without it, with a mean of 3.09 mm higher, this difference being statistically significant (95% CI 2.8–4.99) (Fig. 4, 4.4). With the use of the device, the differences between experts and residents were 0.1 mm; this difference was not significant [95% CI (−0.36) to (0.56) p value = 0.66] (Fig. 4, 4.5).
There was no difference in drilling length with the aid of the device between the first, second, and third drillings, in both expert surgeons (mean drilling p value = 0.62) and in resident surgeons (p value = 0.22) (Fig. 5, 5.1, 5.2). In drilling without the device, the expert surgeons did not show an improvement between the three attempts, with lengths remaining similar [3.8 (SD 2.29); 2.7 (SD 1.3); 2.6 (SD1.07) for the first, second and third attempts, respectively; p value = 0.21] (Fig. 5, 5.3). Meanwhile, the residents showed improvement in the drilling lengths from the first to the second attempt and did not improve between the second and the third attempt (Fig. 5, 5.4) [p value = 0.03; 8.5 (SD 3.02); 6.3 (SD 2.05); 6.0 (SD 1.33); for first and second attempts, respectively].
Diagram of drilling averages in the learning curve of the device for residents (5.1) and expert surgeons (5.2). 5.3 and 5.4 show the learning curve to avoid excessive drilling with the device in experts and residents, respectively. It should be noted that the first drillings were carried out without the device. This was done to facilitate overcoming the learning curve related to the evaluation device and subsequently analyze the learning curve of the protection device specifically
Discussion
The validation data we present obtained from the device show a reduction in the risk of vascular injury in trauma surgery. For less expert surgeons, it allows them to develop their learning curve without the risks of vascular injury exceeding those that an expert surgeon may have during their learning process. The device prevents excessive drilling without reducing the sensation perceived when the second cortex is passed, thereby reducing risks without interfering in the learning process.
In the case of expert surgeons, the device reduces the drilling distance compared to manual drilling. In situations where the risk of injury to vascular and nervous structures is high, this additional safety contribution may be useful, for example, in clavicle fractures to avoid injury to the axillary artery or in tibial plateau fractures to avoid injury to the popliteal artery [18,19,20].
The device has a low learning curve, and the study data show no difference in the results obtained since the first use, making it easy to use. At the same time, thanks to its design, it is easy to obtain thanks to 3D printing. 3D printing has brought about a revolution in orthopedic surgery and traumatology, especially in the field of preoperative planning [21,22,23]. This article is pioneer in describing a new 3D-printing application, which is sharing devices among orthopedic surgeons to find solutions to common problems.
Due to its design, the device can be obtained anywhere in the world with just a click, costs less than surgical gloves, or a scalpel, and can be shared and modified as if it were a conventional text document.
The main limitations of the study are having been carried out in a single center with only 20 participants and using the device designed by our bioengineers for the drilling. Furthermore, we must emphasize that the sample size was calculated before the start of the study and that the central hypothesis showed a statistically significant difference, which implies that a type II error was not made. On the other hand, the evaluation device (Fig. 2) showed similar behavior in terms of excessive drilling to that which has been demonstrated by other studies with elements validated for this purpose [18]. Besides, the possible advantages or disadvantages associated with this evaluation device were the same for both groups, for which the possible existing bias was distributed equally and should not have affected the results.
In conclusion, this device is one example of a new way of sharing surgical solutions among traumatologists through the use of 3D printing and is the first validation study of a device with these characteristics. Due to its simplicity, ease of use, accessibility, necessity, practicality, and efficacy, it is a device that can help resident surgeons develop their learning curve without putting patients at risk and can also help expert surgeons achieve more precision in those cases or situations with a high risk of iatrogenesis.
References
Sözen T, Özışık L, Başaran NÇ. An overview and management of osteoporosis. Eur J Rheumatol. 2017;4(1):46–56.
Kani KK, Porrino JA, Mulcahy H, Chew FS. Fragility fractures of the proximal femur: review and update for radiologists. Skeletal Radiol. 2019;48(1):29–45.
Bhandari M, Swiontkowski M. Management of acute hip fracture. N Engl J Med. 2017;377(21):2053–62.
Court-Brown CM, Duckworth AD, Clement ND, McQueen MM. Fractures in older adults. A view of the future? Injury. 2018;49(12):2161–6.
Rosengren BE, Karlsson MK. The annual number of hip fractures in Sweden will double from year 2002 to 2050: projections based on local and nationwide data. Acta Orthop. 2014;85(3):234–7.
LeBlanc KE, Muncie HLJ, LeBlanc LL. Hip fracture: diagnosis, treatment, and secondary prevention. Am Fam Physician. 2014;89(12):945–51.
Potera C. Seniors need hip fracture surgery within 24 hours. Am J Nurs. 2018;118(3):14.
Haviari S. Wait time for hip fracture surgery and mortality. JAMA. 2018;319(21):2234.
Roberts KC, Brox WT, Jevsevar DS, Sevarino K. Management of hip fractures in the elderly. J Am Acad Orthop Surg. 2015;23(2):131–7.
Chlebeck JD, Birch CE, Blankstein M, Kristiansen T, Bartlett CS, Schottel PC. Nonoperative geriatric hip fracture treatment is associated with increased mortality: a matched cohort Study. J Orthop Trauma. 2019;33(7):346–50.
Carulli C, Piacentini F, Paoli T, Civinini R, Innocenti M. A comparison of two fixation methods for femoral trochanteric fractures: a new generation intramedullary system vs sliding hip screw. Clin Cases Miner Bone Metab Off J Ital Soc Osteoporos Miner Metab Skelet Dis. 2017;14(1):40–7.
Stockton DJ, Dua K, O’Brien PJ, Pollak AN, Hoshino CM, Slobogean GP. Failure patterns of femoral neck fracture fixation in young patients. Orthopedics. 2019;42(4):e376–e380380.
Chughtai M, Khlopas A, Mont MA. Fracture fixation in the operative management of hip fractures (FAITH): an international, multicentre, randomised controlled trial. Lancet Lond Engl. 2017;389(10078):1519–27.
Zhang B-F, Cong Y-X, Wang P-F, Huang H, Wang H, Zhuang Y. Deep femoral artery branch pseudoaneurysm formation and injury after hip fracture surgery: a case series and a literature review. Medicine (Baltimore). 2018;97(6):e9872.
Barquet A, Gelink A, Giannoudis PV. Proximal femoral fractures and vascular injuries in adults: incidence, aetiology and outcomes. Injury. 2015;46(12):2297–313.
Neubauer T, Grechenig S, Leitner L, Auffarth A, Plecko M. Vascular complications in plating of the proximal femur: review. Arch Orthop Trauma Surg. 2016;136(4):539–51.
Tomčovčík L, Kováč P, Daňo J, Čuha R. Late laceration of the superficial femoral artery by an intertrochanteric fracture fragment. Hip Int J Clin Exp Res Hip Pathol Ther. 2011;21(2):273–5.
Clement H, Heidari N, Grechenig W, Weinberg AM, Pichler W. Drilling, not a benign procedure: laboratory simulation of true drilling depth. Injury. 2012;43(6):950–2.
McIntosh AL. Surgical treatment of adolescent clavicle fractures: results and complications. J Pediatr Orthop. 2016;36(Suppl 1):S41–43.
Bernhoff K, Björck M. Iatrogenic popliteal artery injury in non arthroplasty knee surgery. Bone Jt J. 2015;97-B(2):192–6.
Wang X-Z, Shi Z, Lu Q-L, Xie W, Chen L, Li X-G, et al. Treatment of old acetabular posterior wall fracture with 3D printing combined with composite plate internal fixation. Zhongguo Gu Shang China J Orthop Traumatol. 2019;32(5):395–400.
You W, Liu LJ, Chen HX, Xiong JY, Wang DM, Huang JH, et al. Application of 3D printing technology on the treatment of complex proximal humeral fractures (Neer3-part and 4-part) in old people. Orthop Traumatol Surg Res OTSR. 2016;102(7):897–903.
Tomaževič M, Kristan A, Kamath AF, Cimerman M. 3D printing of implants for patient-specific acetabular fracture fixation: an experimental study. Eur J Trauma Emerg Surg Off Publ Eur Trauma Soc. 2019
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Patient consent and ethical approval
The patient provided written informed consent for the publication of medical information and images.
Rights and permissions
About this article
Cite this article
Ferràs-Tarragó, J., Jordà-Gómez, P., Català-de-las-Marinas, J. et al. A new universal 3D-printable device to prevent excessive drilling in orthopedic surgery. Eur J Trauma Emerg Surg 48, 3887–3893 (2022). https://doi.org/10.1007/s00068-020-01465-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00068-020-01465-3