Abstract
Social insect colonies are models for complex systems with sophisticated, efficient, and robust allocation of workers to necessary tasks. Despite this, it is commonly reported that many workers appear inactive. Could this be an artifact resulting from the simplified laboratory conditions in most studies? Here, we test whether the time allocated to different behavioral states differs between field and laboratory colonies of Temnothorax rugatulus ants. Our results show no difference in colony time budgets between laboratory and field observations for any of the observed behaviors, including ‘inactivity’. This suggests that, on the timescale of a few months, laboratory conditions do not impact task allocation at the colony level. We thus provide support for a previously untested assumption of laboratory studies on division of labor in ants. High levels of inactivity, common in social insects, thus appear to not be a laboratory artifact, but rather a naturally occurring trait.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Task allocation and division of labor in complex systems are common research themes in behavioral ecology (Krebs and Davies, 2009), but also in diverse fields such as computer sciences (Johnson, 2012), robotics (Gerkey and Matarić, 2004), logistics (Zhang and Chen, 2011), sociology (Durkheim, 1997), and economics (Becker and Murphy, 1992). Social insect colonies are highly successful, evolved, self-organized collectives which are often used as model systems for research on task allocation. In part, this is due to their tractability. Colonies can easily be maintained, observed, and manipulated in the laboratory and, as a result, studies are frequently performed in the laboratory (Schmid-Hempel, 1990). Some task allocation studies occur in the field, but these usually focus on a small subset of workers, typically foragers (Wilson, 1983; Gordon, 2002), and do not allow a full view of the colony’s task profile in natural conditions. Perhaps the most puzzling in-nest observation, made in laboratory studies, is that behavioral inactivity appears to be highly common in social insect colonies, typically occupying more than 50 % of colony task time (bumble bees: Jandt et al., 2012; honey bees: Lindauer, 1952; ants: Cole, 1986; Herbers and Cunningham, 1983; Schmid-Hempel, 1990; wasps: Gadagkar and Joshi, 1984; termites: Rosengaus and Traniello, 1993). Producing so many extraneous workers seems non-adaptive, or at least counterintuitive. It has been suspected that high levels of inactivity could result from decreased workload and task availability due to simplified living conditions in the laboratory (Schmid-Hempel, 1990). Indeed, laboratory conditions have been shown to affect behavior and potentially introduce artifacts in other contexts (reviewed for vertebrates: Calisi and Bentley, 2009; but rarely tested in invertebrates: Markow, 1988).
Here, we aim to test whether colony time budgets in the laboratory, and in particular the level of inactivity in workers, are representative of colony time budgets in the field for colonies of Temnothorax rugatulus ants, a commonly used laboratory species (Pinter-Wollman et al., 2012). Our results show that there are no significant differences in how field and laboratory colonies allocate time to tasks. Most surprisingly, laboratory and field colonies also had comparably high levels of inactivity.
Methods
Colonies of Temnothorax rugatulus ants were collected in the Santa Catalina Mountains near Tucson, Arizona, USA, in 2012. Of the 13 total colonies collected, 7 (1 collected in May, 1 in August, and 5 in September) were used to quantify time on tasks in the field, and 4 (2 collected in May and 2 in August) in the laboratory. Mean and median colony sizes were 40.75 and 42 workers in the lab, and 42 and 34 workers in the field.
Field colonies were collected, brought to the laboratory to be moved to semi-artificial nests (glass slide taped to a piece of flagstone separated by a piece of balsa wood; Fig. 1), and brought back to the field for observation. In the field, Temnothorax rugatulus ants typically nest in small rock crevices, and artificial nests are meant to emulate these (as for Temnothorax albipennis, Franks et al., 2003). Laboratory colonies were collected, moved to artificial nests (cardboard used as a spacer between two glass slides) and then filmed. Nest construction details can be found in supplementary materials (Fig. S-1).
All ants kept in the laboratory were given ad libitum food (2-mL Eppendorf tube of honey water every week and 10 frozen adult Drosophila flies) and water, kept on a 12-h light regimen (lights on at 8 a.m. and off at 8 p.m.) at constant temperatures (approx. 21 °C) and approx. 15–25 % relative humidity. Once colonies were in the field they were left without added food or water, but they were able to forage freely.
Field data were collected during 2 days each in late September and early October. Each colony was brought back to its collection site, set out in its semi-artificial nest, and left undisturbed for 30 min. In-nest activity was filmed with an HD camera using a red LED light source (Fig. 1). Typically, each colony was film 3 × 5 min (though some field colonies could only be filmed once or twice). Colonies were left overnight in the field and filmed again the next day. To limit potential effects of circadian rhythm, all field videos were taken between 2 p.m. and 7 p.m. and compared to 4 p.m. laboratory videos. Laboratory videos were taken with the HD camera in October 2012.
The behavioral state of each ant was recorded every second by an observer analyzing the videos. Tasks are broadly classified as either “active” (e.g. brood care), “undifferentiated” (walking inside the nest with no clear task), or “inactive” (completely immobile), comparable to Cole (1986). A detailed list of behavior classifications is shown in Table 1. Foraging could not be directly observed in the field, so foraging rate was estimated by counting the instances of workers leaving the nest relative to colony size. Videos were analyzed by multiple observers and spot checked by a single person to ensure uniformity of behavioral classification.
Statistical analyses were performed in R (Version 3.0.3, package ‘nlme’ v3.1-115). Colony-level time spent on a task was defined as the mean proportion of observed time spent on that task across all ants.
Results
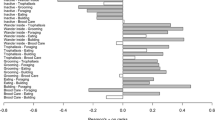
There were no significant differences in the proportion of time field and laboratory colonies spent active, wandering inside, or inactive (Fig. 2, left side; p = 0.56, p = 0.40, and p = 0.63, respectively. Breaking down overall activity into specific tasks also shows no significant differences in how colonies allocate time between the laboratory and the field (Fig. 2, right side; building p = 0.10, brood care p = 0.98, grooming p = 0.93, trophallaxis p = 0.46). The task ‘Eating’ (feeding on dead insects inside nest) could not be tested because of its absence from most observations (67 %). These results were the same when first and second (after overnight outside) recordings of field colonies were analyzed separately. However, colonies spent more time on grooming on the second day of observations (p = 0.02) and, as a result, were also more active on the second day (p < 0.01).
Lab and field colonies do not differ significantly in the proportion of time dedicated to activity, wandering inside (undifferentiated), or inactivity. Specific tasks within total active time also do not differ significantly. Boxplots show quartiles (box), median (horizontal line), and extremes (whiskers) for colony time on tasks in the lab (light gray) and in the field (dark gray). Linear mixed-effects models–fixed-effect: treatment (lab or field); random effect: colony. *Insufficient data to test ‘Eating’
Although foraging time could not be directly observed in the field, foraging rates for laboratory and field colonies were estimated by counting the instances of workers leaving the nest relative to colony size (mean foraging rate = [workers observed leaving nest/colony size/min] = 0.0162 and 0.0158 for field and laboratory, respectively, ANOVA p = 0.93).
Variance of field and laboratory data were not significantly different for most tasks (Bartlett’s test p > 0.05), except for wandering inside and trophallaxis which were greater in the field (Wandering inside: s lab = 0.60, s field = 0.146, p < 0.0001; Trophallaxis: s lab = 0.005, s field = 0.012, p < 0.01, respectively), and building which was greater in the laboratory (s lab = 0.025, s field = 0.001, p < 0.0001).
Discussion
Our results show that simplified artificial conditions experienced by ant colonies in a laboratory do not significantly impact how the colony allocates time to different activities in the ant Temnothorax rugatulus. Although time spent foraging in the field could not be directly measured, foraging rates, estimated by the number of ants seen leaving the nest divided by colony size, between field and laboratory colonies were comparable. Most surprisingly, the high levels of inactivity observed in laboratory colonies were also observed in field colonies (average proportion of time inactive 76.9 and 76.3 %, respectively) and are consistent with values found in the literature for other Temnothorax ants (Herbers and Cunningham, 1983; Cole, 1986).
Contrary to the popular belief that social insects are hardworking (as in Aesop’s Fable ‘The Grasshopper and the Ant’), many species have been shown to display high levels of inactivity in the laboratory (Hölldobler and Wilson, 1990; Schmid-Hempel, 1990). Simplified living conditions and lack of challenge in the laboratory (e.g. ad libitum food, controlled climate, and lack of predators or competitors) have been proposed as possible explanations (Schmid-Hempel, 1990). If so, high levels of inactivity would be a laboratory artifact and not an evolved trait. Our results show that time budgets are not significantly different between field and laboratory colonies; thus, we can reject this hypothesis.
Foraging in particular might be expected to require more time in the field. No studies directly compare foraging activity in the field and in the laboratory. Some studies measure foraging intensity (i.e. number of foragers/h) for different species of ants in the laboratory (e.g. Gordon, 1983) and in the field (e.g. Gordon, 2002), but colony size, and thus per capita foraging rate, is typically unknown in field studies. Gordon et al. (2005) report anecdotal evidence of lower foraging activity in the lab in the ant Pogonomyrmex barbatus. Here, food for laboratory colonies was located at 3–4 cm (~20 body lengths) from the nest perimeter, while the median foraging distance in the field for Temnothorax rugatulus ants is 1.5 m (~800 body lengths) (Bengston and Dornhaus, 2013). However, despite this, the foraging rates measured here control for colony size and did not differ between field and laboratory colonies.
The similarity in colony task allocation between the field and the laboratory was unexpected. Many studies show changed behavior for animals in captive environments (Carlstead, 2010). For example, changes in the amount and types of food available can change foraging behaviors (McFarland, 1989), or a lack of exposure to predators can affect anti-predator behavior (Moodie and Chamove, 2005). Similar changes in behavior could be expected in social insects as well, and so it is surprising that to see no change at all in how the colony allocates its time.
However, much of the research on the effects of captivity on behavior focuses on vertebrates, particularly mammals, and so may not necessarily apply to insects. Endotherms have been shown to have more control over their metabolic rate than ectotherms (Garland Jr et al., 1987). Thus, insects may not be able to internally adjust their activity levels to fit new living conditions because of physiological constraints. Scale may also be a factor as the laboratory may be less different from natural conditions for small animals than for large ones. For social insects, the social environment may be more important than external living conditions, and the interactions and tasks inside the nest may be quite similar in the laboratory and in the wild.
Our results provide evidence that colonies of Temnothorax rugatulus ants kept in the laboratory maintain similar patterns of colony-level task allocation as colonies in the field. This increases our confidence that inactivity is indeed high in social insect colonies and not an artifact of laboratory studies (Lindauer, 1952; Herbers and Cunningham, 1983; Gadagkar and Joshi, 1984; Cole, 1986; Schmid-Hempel, 1990; Jandt et al., 2012; Pinter-Wollman et al., 2012). Ant and other social insect colonies thus contain many workers that are not working (Schmid-Hempel, 1990; Jandt et al., 2012; Lindauer, 1952; Cole, 1986; Herbers and Cunningham, 1983; Gadagkar and Joshi, 1984; Rosengaus and Traniello, 1993), apparently at odds with the idea that colonies are evolutionarily tightly optimized systems. The adaptive explanations for this remain elusive.
References
Becker G.S. and Murphy K.M. 1992. The division of labor, coordination costs, and knowledge. Quart. J. Econ. 107: 1137-1160
Bengston S.E. and Dornhaus A. 2013. Colony size does not predict foraging distance in the ant Temnothorax rugatulus: a puzzle for standard scaling models. Insect. Soc. 60: 93-96
Calisi R.M. and Bentley G.E. 2009. Lab and field experiments: Are they the same animal? Hormones and Behavior 56: 1-10
Carlstead K. 2010. Effects of captivity on the behavior of wild mammals. In: Wild Mammals in Captivity, University of Chicago Press, Chicago, IL
Cole B.J. 1986. The social behavior of Leptothorax allardycei (Hymenoptera, Formicidae): time budgets and the evolution of worker reproduction. Behav. Ecol. Sociobiol. 18: 165-173
Durkheim E. 1997. The Division of Labor in Society. Simon and Schuster, New York, NY
Franks N.R., Mallon E.B., Bray H.E., Hamilton M.J. and Mischler T.C. 2003. Strategies for choosing between alternatives with different attributes: exemplified by house-hunting ants. Anim. Behav. 65: 215-223
Gadagkar R. and Joshi N.V. 1984. Social organisation in the Indian wasp Ropalidia cyathiformis (Fab.) (Hymenoptera: Vespidae). Z. Tierpsychol. 64: 15-32
Garland Jr T., Else P.L., Hulbert A.J. and Tap P. 1987. Effects of endurance training and captivity on activity metabolism of lizards. Am. J. Physiol. - Regul. Integr. Comp. Physiol. 252: R450-R456
Gerkey B.P. and Matarić M.J. 2004. A formal analysis and taxonomy of task allocation in multi-robot systems. Int. J. Robotics Res. 23: 939-954
Gordon D.M. 1983. Daily rhythms in social activities of the harvester ant, Pogonomyrmex badius. Psyche: A Journal of Entomology 90: 413-423
Gordon D.M. 2002. The regulation of foraging activity in red harvester ant colonies. Am. Nat. 159: 509-518
Gordon D.M., Chu J., Lillie A., Tissot M. and Pinter N. 2005. Variation in the transition from inside to outside work in the red harvester ant Pogonomyrmex barbatus. Insect. Soc. 52: 212-217
Herbers J.M. and Cunningham M. 1983. Social organization in Leptothorax longispinosus Mayr. Anim. Behav. 31: 759-771
Hölldobler B. and Wilson E.O. 1990. The Ants. Belknap Press of Harvard University Press, Cambridge, MA
Jandt J., Robins N., Moore R. and Dornhaus A. 2012. Individual bumblebees vary in response to disturbance: a test of the defensive reserve hypothesis. Insect. Soc. 59: 313-321
Johnson S. 2012. Emergence: The Connected Lives of Ants, Brains, Cities, and Software. Simon and Schuster, New York, NY
Krebs J.R. and Davies N.B. 2009. Behavioural Ecology: An Evolutionary Approach. John Wiley & Sons, Hoboken, NY
Lindauer M. 1952. Ein Beitrag zur Frage der Arbeitsteilung im Bienenstaat. J. Comp. Physiol. A: Neuroethology, Sensory, Neural, and Behavioral Physiology 34: 299-345
Markow T.A. 1988. Reproductive behavior of Drosophila melanogaster and D. nigrospiracula in the field and in the laboratory. J. Comp. Psychol. 102: 169
McFarland D. 1989. Problems of Animal Behaviour. Longman Scientific & Technical, Harlow, Essex; New York
Moodie E.M. and Chamove A.S. 2005. Brief threatening events beneficial for captive tamarins? Zoo Biology 9: 275-286
Pinter-Wollman N., Hubler J., Holley J.-A., Franks N. and Dornhaus A. 2012. How is activity distributed among and within tasks in Temnothorax ants? Behav. Ecol. Sociobiol. 66: 1407-1420
Rosengaus R.B. and Traniello J.F.A. 1993. Temporal polyethism in incipient colonies of the primitive termite Zootermopsis angusticollis: A single multiage caste. J. Insect Behav. 6: 237-252
Schmid-Hempel P. 1990. Reproductive competition and the evolution of work load in social insects. Am. Nat. 501-526
Wilson E.O. 1983. Caste and division of labor in leaf-cutter ants (Hymenoptera: Formicidae: Atta). Behav. Ecol. Sociobiol. 14: 47-54
Zhang J. and Chen G. 2011. The influence of logistics development on manufacturing division. In: Artificial Intelligence, Management Science and Electronic Commerce (AIMSEC), 2011 2nd International Conference, pp 791-794
Acknowledgments
We thank Alex Downs, Andrew Scott, Mary Levandowski, Matthew Velazquez, Neil Hillis, and Nicole Fischer for their help with data collection and ant maintenance. We also thank the entire Dornhaus lab for their ongoing feedback. Research supported through the GIDP-EIS and EEB Department at University of Arizona, as well as NSF Grants no. IOS-1045239, IOS-0841756, and DBI-1262292 (to A.D.).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Charbonneau, D., Hillis, N. & Dornhaus, A. ‘Lazy’ in nature: ant colony time budgets show high ‘inactivity’ in the field as well as in the lab. Insect. Soc. 62, 31–35 (2015). https://doi.org/10.1007/s00040-014-0370-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00040-014-0370-6