Abstract
The directional information encoded in the waggle dances of absconding colonies of Apis florea shows how different sites are advertised during decision-making. Colonies of A. florea were observed from the inception of absconding until the swarm settled at a new nest site. The number of waggle dancers at the beginning of the absconding sequence was low, gradually increased and then declined shortly before liftoff. During the last 2 to 0.5 h before liftoff, the dances still indicated different directions. This significantly decreased in the last 0.5 h until only one or two dance directions were being advertised. All colonies reached a near consensus in the last 20 dances before liftoff. The swarm flight path is meandering so the actual distance flown is about twice that indicated by the dances. During the last 3 min the waggle dance in most colonies showed nest target angles that were closely clustered indicating that the final directions advertised were close to the chosen target site. In all absconding/migratory species of honeybees thus far studied, there is a special dance associated with absconding that appears not to select specific destinations but rather a particular direction in search of a new nesting area.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Migrations by animals are a widespread phenomenon among many disparate taxa in all biomes and are associated with changes in ambient environmental conditions (Baker, 1978; Milner-Gulland et al., 2011). For migrations to be successful over many generations, a non-random, direction-driven component must have evolved for this trait. The metabolic costs involved in migrations would prescribe the need for a distance component to be factored in insects’ decisions on where and how far to go in a particular case. These features are especially notable in the migrations, swarming and absconding by tropical species of honeybees (Hepburn, 2011). A requirement for honeybees and other social insects is that effective moves require group decisions which basically are based on consensual actions (Seeley, 2003, 2010; Schaerf et al., 2011).
All species of tropical honeybees studied to date that undergo seasonal movements have been found to perform modified waggle dances that are referred to as “absconding dances” or “migration dances”. Such dances have been reported for Apis florea (Koeniger and Koeniger, 1980; Beekman et al., 2008; Oldroyd et al., 2008; Schaerf et al., 2011), A. dorsata (Koeniger and Koeniger, 1980; Dyer and Seeley, 1994), A. cerana (Sasaki, 1991) and A. mellifera scutellata (Schneider and McNally, 1994; Lewis and Schneider, 2008). The dances differ from the more familiar waggle dances associated with foraging, in that the waggle runs are arrhythmic and slow paced, lack the return runs of foraging dances, and may not stimulate immediate recruitment (Sasaki, 1991; Dyer and Seeley, 1994; Schneider and McNally, 1994; Beekman et al., 2008). Also, the consecutive waggle runs performed by the same bee differ markedly in duration and communicate highly variable distances that far exceed the normal foraging range of the species. However, absconding dances maintain a consistent orientation and often communicate the same general direction, which tends to be the direction the entire colony travels when it absconds (Sasaki, 1991; Dyer and Seeley, 1994; Schneider and McNally, 1994). These observations suggest that absconding or migration dances lack a meaningful distance component, and function to prepare a colony to relocate in a general direction but not to a specific destination (Sasaki, 1991; Dyer and Seeley, 1994; Schneider and McNally, 1994). However, absconding dances remain one of the least understood aspects of tropical honeybee biology.
More recently, detailed analyses of honeybee dances in the context of absconding and migration by the red dwarf honeybee, A. florea, have reported very marked variations in the dance sequences and their communicative value that fundamentally differ from those associated with foraging (Sen Sarma et al., 2004; Beekman et al., 2008; Oldroyd et al., 2008; Duangphakdee et al., 2009, 2011; Diwold et al., 2011; Makinson et al., 2011). Most recently, Schaerf et al. (2011) reported a simple algorithm as a first approximation towards characterizing the intricacies associated with decision-making in the red dwarf honeybee, A. florea.
Understanding the complex processes of different stages of scouting for a new nest site during absconding/migrating by A. florea requires answers to a series of questions: (1) when do the scout bees begin searching for new nest sites? (2) When do the scout bees begin to recruit nestmates for new nest sites? (3) How many nest sites will have been reported to the colony by the scouts? (4) Do they reach agreement on a perspective site before liftoff? Does the swarm fly directly, in a straight line, to the chosen nest site? (5) Do the scouts reconnoitre the terrain and use dance communication to indicate a new nesting site before the swarm flies off? (An alternative would be that the bees use the dance to communicate the initial direction of flight, and then while airborne, search for a suitable nesting place). To answer these questions, individual bees within the swarms were examined in a behavioural nest-hunting context from the inception of swarming until it had reached and settled at a new nest site. Here, we report the results of observations and measurements that describe the overall pattern of absconding and new nest site selection in A. florea.
Methods
Study site and swarm preparation
The studies were performed from April to November 2007 and during July 2010 on Ratchaburi campus, King Mongkut’s University of Technology, Chom Bueng, Ratchaburi, Thailand (13°59′N, 99°51′E, altitude 86 m). About 30 colonies of A. florea were collected from a coconut estate at Maklong, Samut-Songkram (13°24′N, 100°00′E) and from the forest at Chom Bueng, Ratchaburi. However, as is usually the case, most colonies absconded before we could obtain a complete set of data, but we were able to obtain complete data sets for five colonies. The branches of trees with nests were carefully cut, placed in wooden boxes and transported to the campus. On arrival at the experimental area, each colony was suspended singly under an open-sided shelter constructed of wooden poles. Crossed wooden slats were attached horizontally to both sides of the shelter supports, about 1.2 m above the ground, for suspending the nests and to facilitate viewing. One day after the colony had settled, the brood comb was cut from the nest in the evening to stimulate absconding (Duangphakdee et al., 2009; Pirk et al., 2011). Removing the brood comb induces colonies to abscond because absconding colonies on the verge of flying off wait until the sealed brood has emerged and only then leave the natal nest (Woyke, 1976). As an aside it can be noted that A. florea show both disturbance-induced and seasonal absconding. Moreover, dance behaviour does not differ between the two forms of movement.
The locations of food sources within a 1-km radius of the nests were mapped to ensure that the bees’ dances for either food sources or for swarming could be distinguished. Three shelters were stationed at 10, 100, 150 m distance from the main food sources (both patches of flowers and artificial feeders) in different orientations (with respect to north) to minimize possible nest orientation effects. The shelters were positioned under the shady branches of trees to simulate their natural habitat. Conditions were provided as close to the natural habitat as possible, so we assume that the colonies would behave like natural absconding colonies.
Observations on the behaviour of colonies translocating to new nest sites
The colonies were monitored continuously from 08.00 to 17.00 hours every day on the morning following brood comb removal until the colonies absconded. We used a video camera (Sony Handycam DCR-DVD803E PAL) and DVD recorder (Sony DVD + RW, DPW60DSS2) to record observations. After swarm liftoff, we observed and followed the flight paths and behaviour of colony formations until the swarms settled at a new location. The behaviour of the airborne moving swarm towards the chosen nest site after liftoff was observed. The distances between the old and new nest sites were measured. A total of five colonies were successfully followed and systematically studied.
We tracked the total number of bees which performed absconding dances in each colony. The DVDs were analysed using the computer program power DVD with variable speed playback. We analysed the videos to obtain the following data: the compass directions (angles) of the nest sites indicated by the dances, the numbers of dances related to each direction indicated and the duration of the dances were observed for 3 min periods at 30 min intervals at 120, 90, 60, and 30 min and in the final 3 min before liftoff. Double counting was avoided by repeatedly reviewing each video at least five times to be sure that we counted each bee only once. Each colony was monitored from the time they moved to the new nest site until they had completely settled at new nest sites. For each colony, the last 20 waggle dances before liftoff were also analysed.
We were able to closely monitor each colony as it flew up from the old nest to abscond to the new one. They typically flew about 4–5 m above ground in a meandering fashion. We were able to walk along under the swarms, following their meandering, and whenever there was a deviation we marked that point with a coloured stone (Hansel and Gretel style). After the swarms settled we measured the total length of the meandering traverse and mapped it to scale. We then rolled thin threads from plasticine and fitted them to the meanders of the flight path drawn to scale and then carefully straightened the plasticine threads for total path length measurements.
Data analysis
Data from five colonies that absconded were analysed. Independent t tests and analysis of variance (ANOVA) were used to test for differences in the number of dances per min and the number of different dancers between time periods and colonies. Homogeneity of the variances between colonies and time periods was checked using Levene’s test. Statistical methods for circular distributed data were used to calculate the mean angles advertised by the dances during the time periods before liftoff for each colony. The angular accuracy of the dancers was determined by the mean angular deviation of the waggle angles, defined as s = (180°/π)√2(1 − r), where r is the measure of concentration of the angles (Zar, 2010). Approximate 95 % confidence intervals for population mean angles were also determined based on the von Mises distribution (Zar, 2010). The data were analysed using Statistica 9.0 (StatSoft, StatSoft Inc 2009).
Results
Sequence of absconding
Once their combs were removed, the colonies took about 16–26 h to reach a decision to move to a new nest site. In this set of observations four of five colonies absconded the day after brood removal (Table 1). That morning, the colonies showed low activity levels and only a few scouts still set out to forage for nectar and/or pollen. We observed the first waggle dances for nest sites 3–4 h after dawn at which time only the odd pollen or nectar forager was returning to the nest but no new foragers were recruited. After the first bees flew away from the old nest, the whole colony became airborne within 50.08 ± 4.90 s (n = 5), with a range of 41.6–54.0 s. The direct vector distances to new nest sites averaged 65.0 ± 11.05 m and ranged from 50 to 80 m (Table 1). However, because the swarming bees do not fly directly to the new nest site but meander, the average actual distance from old to new nests was 128 ± 23.61 m. So, the actual distance flown was approximately twice that of the direct distance.
Direction communication in the waggle dances
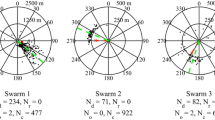
The apparent preferences for different sites that were advertised by the dances of the scout bees over a 2-h period for the five colonies are shown in Fig. 1. The plots show the compass directions indicated by the dances. Somewhere between 2 and 0.5 h prior to liftoff, the numbers of directions presented by waggle dancers still indicated different directions (4.0 ± 1.6). But on average, in the last 30 min before liftoff, the number of differing dancers decreased significantly until usually only one or at most two directions (1.8 ± 0.8) were advertised before colony liftoff (t test: t 8 = 2.75, p = 0.0251). In all cases, our colonies reached a near consensus in the last 20 dances before liftoff. At the beginning of the dance sequence (1.5 h) there were about 1.20 ± 0.18 dances per min with a range from 1 to 1.33 dances per min, but the dances then increased and reached a maximum of 3.87 ± 1.26 dances per min with a range of 2.67 to 5.66 dances per min at 30 min before liftoff. Between the last 30 min until just 3 min before liftoff, the number of dances decreased significantly to 2.47 ± 1.04 dances per min with a range of 1.33–4 dances (F 4,20 = 6.04, p = 0.0023; Fig. 2).
Achieving consensus
The mean angles advertised by the dances in the last 3 min before liftoff for each colony and the angular accuracy of the dancers determined by the mean angular deviation are given in Table 2. The accuracy of consensus represented by the angles advertised in the dances in the last 3 min for each colony was determined by the 95 % confidence intervals for the true mean angles: the 95 % confidence intervals for colonies 2, 4 and 5 enclose the nest site target angles (hypothesized angles) indicating that the advertised directions are close to that of the chosen target site. For colonies 1 and 3, the nest site target angles fell outside the 95 % confidence intervals indicating that the nest site target angles were significantly different from the advertised directions (Table 2; Fig. 3). The mean angular deviation as advertised by the waggle dances in the last 3 min before liftoff (6.4° ± 4.4°) was significantly less than the mean angular deviation of the waggle dances during the periods 120–30 min before liftoff (38.7° ± 20.1°) (t 23 = 3.53, p = 0.0018; Fig. 1). The variation in the angles advertised per time period decreased over the time periods as seen in Fig. 1.
The meandering flight paths of each colony. The closed arrows represent real flight paths from shelter (tail, point) to a new nesting site (closed circle). Sector of a circle shows an array of angles represented by the last 3 min before liftoff. (a construction less than 3 m high, b less than 7 m, D farm dam)
Swarm behaviour
The meandering flight path of each colony accurately reflected that sector of a circle containing those angles danced when consensus was reached in the dances of the last 3 min (Fig. 3). In three out of five colonies (colonies 2,4,5), the scouts advertised directions in very close alignment to a chosen site because the new nest sites were positioned in the array of angles presented by the last 3 min waggle dances. In colonies 1 and 3 the waggle direction was obviously different from the chosen nest site with angles of divergence of 27.8° and 58°, respectively. In all colonies we observed some dances with no visible direction component. These dances were similar to the so-called “round dance” described by Oldroyd et al. (2008) and the numbers of these round dances increased over time and were still present even just before liftoff.
Settling at a new nest site
Shortly after an absconding colony of A. florea approaches a tree which will be the new nest site, several bees settle on a specific twig and probably release a pheromone from the Nasonov gland. As shown in the sequence of Fig. 4, a few seconds later, the swirling mass of bees descend on that twig and after 4.45 ± 0.47 min (n = 4) the entire colony had clustered there.
Settling at a new nest site. When the whole colony has arrived at its chosen tree there is a general swirling of bees amongst the branches. a These bees presumably activate their Nasonov glands as several bees bend down their abdomens and fan their wings to possibly spread Nasonov pheromones, b the colony quickly joins them and a few seconds later, c the whole colony has aggregated into a colony cluster in 4.45 ± 0.47 min
Discussion
It seems apparent that in all absconding/migratory species of honeybees studied to date, there is a special dance associated with this type of colony movement that does not appear to select specific destinations but rather may help to prepare colonies to fly in a particular direction in search of a new nesting area. The proposed function of absconding dances in A. cerana, A. dorsata and A. m. scutellata is based partly on the observation that consecutive waggle runs by the same bee are of extreme duration, but highly variable, suggesting that no specific distance (and hence no specific site) is being communicated (Sasaki, 1991; Dyer and Seeley, 1994; Schneider and McNally, 1994). This is demonstrably the case for A. florea as recently shown in comparisons of the communicated distances with the total, meandering distances travelled by swarms before settling (Duangphakdee et al., in prep.). A. florea is the only honeybee species for which the distances actually travelled by absconding swarms has been determined.
Waggle dances for new nest sites usually begin during early morning after brood removal the previous evening and become completely focused on advertising these sites within 2–3 h after sunrise (observations consistent with those of Duangphakdee et al., 2009). The range of compass directions of the nest sites indicated by the dances usually decreased until only one or two directions were still being advertised before colony liftoff. The number of waggle dancers at the beginning of the absconding sequence was low and gradually increased over the next 2 h, but declined shortly before liftoff. The dwarf honeybees, A. florea, depart from the old nest in less than a minute, an observation also noted by others (Oldroyd et al., 2008; Duangphakdee et al., 2009). While the straight line distances to new nest sites averaged 65 ± 11.1 m, the swarming bees fly as a loose, meandering cluster, zigzaging about the straight line vector between old and new nests, thus approximately doubling the distance that need be flown.
The variation in the angles advertised per time period decreased over time, and the mean difference of the angles, based on potential new nest sites as advertised by the waggle dancers in the last 3 min before liftoff, was 21.1° ± 22.1° with a range of −14.3° to 75.0°. The accuracy of consensus measured by the degrees advertised in the dances in the last 20 dances varied. It is evident that only an approximate consensus is reached by absconding colonies of A. florea compared to those routinely reported for reproductive swarms of A. mellifera or A. cerana in which both direction and distance for a new nest sire are encoded in the waggle dances (Dyer, 2002; Seeley, 2003; Lewis and Schneider, 2008).
Cavity-nesting and open-nesting honeybee species really have very different objectives in both reproductive swarming and migratory translocations. A real consensus on a new site is reached in the former and the direction component is very accurate indeed (Seeley, 2003). This is as well as it must be if, for example, they are advertising a specific tree hollow and approximations that are off by 1°, this may prove disastrous. But, the relative angle of direction proposed by A. florea is very probably inconsequential and very much depends on a colony’s perception of what constitutes a “good” nest site.
In our examinations of hundreds of A. florea nest sites we came to the following conclusions: (1) species of tree is irrelevant; (2) crown structure is paramount; and (3) in free-standing trees with no other trees near (within perhaps 5 m), the bees utilize round, inverted vase, and oval canopied trees. Trees in a natural kiosk, copse or secondary forest are closely spaced together such that individual trees have a restricted flat crown. Four or five closely adjacent trees jointly present a continuous crown, so that a specific tree need not be sought but only a suitable crown continuum. In these circumstances, deviations from the straight line vector of the final dances, exacerbated by meandering will still provide a swarm with a high probability of reaching a “good” nest site. This interpretation is in total accord with that of Schaerf et al. (2011). Our results are also in agreement with those of Schaerf et al. (2011) and likewise support the interpretation of Makinson et al. (2011) that nest site selection has been essentially selected by the nesting requirements of each honeybee species. We conclude, as did Schaerf et al. (2011), that it is plausible, and maybe probable, that decision-making for a new site by A. florea is primarily directed towards synchronization of liftoff by the swarm, with a consensus on the general direction that swarm ultimately takes.
References
Baker R.R. 1978. The Evolutionary Ecology of Animal Migration. Holmes and Meier Publishers, Teaneck.
Beekman M., Gloag R.S., Even N., Wattanachaiyingcharoen W. and Oldroyd B.P. 2008. Dance precision of Apis florea: clues to the evolution of the honeybee dance language? Behav. Ecol. Sociobiol. 62: 1259-1265.
Diwold K., Schaerf T.M., Myerscough M.R., Middendorf M. and Beekman M. 2011. Deciding on the wing: in-flight decision making and search space sampling in the red dwarf honeybee A. florea. Swarm Intelligence 5: 121-141.
Duangphakdee O., Hepburn H.R. and Tautz J. 2011. The dance language. In: Honeybees of Asia (Hepburn H.R. and Radloff S.E., Eds), Springer-Verlag, Berlin, Heidelberg. pp 313-332.
Duangphakdee O., Radloff S.E., Pirk C.W.W. and Hepburn H.R. 2009. Sun angle time windows for absconding by the dwarf honeybee, Apis florea. J. Insect Physiol. 55: 1009-1012.
Dyer F.C. 2002. The biology of the dance language. Annu. Rev. Entomol. 4: 917-950.
Dyer F.C. and Seeley T.D. 1994. Colony migration in the tropical honeybee Apis dorsata F. (Hymenoptera: Apidae). Insect. Soc. 41: 129-140.
Hepburn H.R. 2011. Absconding, migration and swarming. In: Honeybees of Asia (Hepburn H.R. and Radloff S.E., Eds), Springer-Verlag Berlin, Heidelberg. pp 133-158.
Koeniger N. and Koeniger G. 1980. Observations and experiments on migration and dance communication of Apis dorsata in Sri Lanka. J. Apicult. Res. 19: 21-34.
Lewis L.A. and Schneider S.S. 2008. “Migration dances” in swarming colonies of the honey bee, Apis mellifera. Apidologie 39: 354-361.
Makinson J.C., Oldroyd B.P., Schaerf T.M., Wattanachaiyingcharoen W. and Beekman M. 2011. Moving home: nest-site selection in the red dwarf honeybee (Apis florea). Behav. Ecol. Sociobiol. 65: 945-958.
Milner-Gulland E.J., Fryxell J.M. and Sinclair A.R.E. 2011. Animal Migration: A Synthesis. Oxford University Press, Oxford.
Oldroyd B.P., Gloag R.S., Even N., Wattanachaiyingcharoen W. and Beekman M. 2008. Nest-site selection in the open-nesting honey bee Apis florea. Behav. Ecol. Sociobiol. 62: 1643-1653.
Pirk C., Crous K., Duangphakdee O., Radloff S. and Hepburn R. 2011. Economics of comb wax salvage by the red dwarf honeybee, Apis florea. J. Comp. Physiol. B 181: 353-359.
Sasaki M. 1991. Absconding dance in Apis cerana japonica: a slow and long tail-waggling motivates the whole colony to translocate. Proc. 11 th Int. Congr. IUSSI, Bangalore, pp 125-126.
Schaerf T.M., Myerscough M.R., Makinson J.C. and Beekman M. 2011. Inaccurate and unverified information in decision making: a model for the nest site selection process of Apis florea. Anim. Behav. 82: 995-1013.
Schneider S.S. and McNally L.C. 1994. Waggle dance behavior associated with seasonal absconding in colonies of the African honey bee, Apis mellifera scutellata. Insect. Soc. 41: 115-127.
Seeley T.D. 2003. Consensus building during nest-site selection in honey bee swarms: the expiration of dissent. Behav. Ecol. Sociobiol. 53: 417-424.
Seeley T.D. 2010. Honeybee Democracy. Princeton University Press, Princeton, New Jersey.
Sen Sarma M., Esch H. and Tautz J.2004. A comparison of the dance language in Apis mellifera carnica and Apis florea reveals striking similarities. J. Comp. Physiol. A 190: 49-53.
StatSoft Inc. 2009. STATISTICA (data analysis software system), version 9.0. www.statsoft.com.
Woyke J. 1976. Brood-rearing efficiency and absconding in Indian honeybees. J. Apicult. Res. 15: 133-143.
Zar J.H. 2010. Biostatistical Analysis. 5th ed. Prentice Hall, Inc., Upper Saddle River, New Jersey.
Acknowledgments
We would like to thank the Thailand Research Fund and Office of the Higher Education Commission for financial support (MRG5380128) and Colleen Hepburn for English editing.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Duangphakdee, O., Hepburn, H.R., Radloff, S.E. et al. Waggle dances in absconding colonies of the red dwarf honeybee, Apis florea . Insect. Soc. 59, 571–577 (2012). https://doi.org/10.1007/s00040-012-0254-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00040-012-0254-6