Abstract
Six new genera, three tribes and one subfamily are added to the list of known hosts for the enigmatic endoparasitic fungus Myrmicinosporidium Hölldobler. Aphaenogaster senilis, Cataglyphis hispanica, Crematogaster auberti, Goniomma hispanicum, Messor barbarus, Tapinoma nigerrimum and Tapinoma simrothi were collected from olive groves and detected as infected with spores of the fungus. Pheidole pallidula and Tetramorium semilaeve were also found to be infected. The finding of seven hosts (seven genera, three subfamilies) from a single olive grove is an evidence that the fungus has a phylogenetically wide host spectrum and is, therefore, a generalist microparasite. Portugal is also a new country for Myrmicinosporidium.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
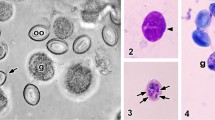
Schmid-Hempel (1998) noted a general lack of data of parasites and diseases in social insects. This is being currently addressed at theoretical level (Boomsma et al., 2005; Cremer et al., 2007) and supported with field data and experiments (Ugelvig and Cremer, 2007; Ugelvig et al., 2010; Reber and Chapuisat, 2011). A recent review of Holarctic entomoparasitic fungi on ants (Espadaler and Santamaria, 2012) also covered briefly an endoparasitic fungus, Myrmicinosporidium. However, subsequent work revealed an enormous extension of the host range of this fungus. A study of ants collected in pitfall traps in olive groves from Portugal showed new data that already rendered the previous study out of date. The subject is an enigmatic endoparasitic fungus, Myrmicinosporidium durum Hölldobler, whose lifecycle is only partly known (Sánchez-Peña et al., 1993) and that seems to be infrequently detected (Buschinger et al., 2004). The spores (diameter 47–61 μm) show a highly characteristic concave shape that is an artefact of alcohol fixation (Buschinger and Winter 1983); otherwise they are biconcave. Ant workers, queens and males may be infected, although behaviour seems to be only slightly impaired: infected workers are usually captured in pitfall traps, thus, foraging. Infected queens have also been captured in swarming (Buschinger et al., 2004). Those fungi seem to be close to Chytridiomycetes (Sánchez-Peña et al., 1993), although it remains to be studied where they belong within the fungi phylogeny.
We upgrade the previous knowledge with six new genera, three tribes, and one subfamily, which are added here to the list of known ant hosts. The fungus is also new for Portugal.
Materials and methods
For a wider project seeking to explore the usefulness of arthropods as tools for assessing the impact of agricultural practices, pitfall traps were used to characterize the ant assemblages of olive groves subjected to different management schemes. The study was carried out in Baixo Alentejo, Portugal in eight olive groves using intensive organic (O; n = 4) low density (LD; n = 2) and intensive high density (HD; n = 2) management systems (Table 1). Sixteen pitfall traps per olive grove, opened 1 week per month, for 3 months (June, September, November), were used to characterize the ant assemblages of olive groves subjected to the three different management schemes. The large olive groves are surrounded by a matrix of arable land and Quercus forest fragments. The climate is continental Mediterranean, with 605 mm of annual rainfall. Mean monthly temperatures reach a high of 32.3 °C in July and a low of 5.4 °C in January.
Ants were identified under a dissection microscope with a cold light source, using standard references (Collingwood and Prince, 1998; Seifert, 1992) and compared with our own collections. During the identification process, spores of the fungus Myrmicinosporidium were immediately detected in the first sample, which contained Tetramorium semilaeve (André) and Messor barbarus (Linnaeus). Spores were directly seen inside the legs and the distended gaster of the yellowish Tetramorium and also in the extremely dark gaster of the otherwise normally coloured Messor. After this discovery, the gaster of all specimens from species with dark cuticles was punched with forceps. If infected, spores appeared immediately out of the gaster. Light yellow coloured ants were directly inspected for spores. Samples are preserved at the Polytechnic Institute of Beja.
Results
Myrmicinosporidium infected workers were detected in 4 out of 8 olive groves, and in 16 samples out of 384. Those samples comprised 2,093 workers of which 79 had spores of the fungus (Table 2). Spores were detected only in June samples (8.VI.2011, leg. C. Gonçalves). No spores were detected in the ants captured in September (n = 1,423 workers) and November (n = 1,431 workers) using a similar trapping scheme. Prevalence in June: 3.7 % for total data, with much variability for each species. It is disputable whether specific prevalence should be defined as a percentage of infected ants within a trap or within a field. Further, we refrained from exploring this epidemiological characteristic.
Other species captured in the olive groves, but not detected as infected: Camponotus pilicornis Roger, Camponotus foreli Emery, Camponotus micans (Nylander), Camponotus sylvaticus (Olivier), Camponotus lateralis (Olivier), Camponotus cruentatus (Latreille), Cardiocondyla batesii Forel, Crematogaster scutellaris (Olivier), Crematogaster sordidula (Nylander), Formica cunicularia Latreille, Formica subrufa Roger, Hypoponera eduardi (Forel), Lasius lasioides (Emery), Lasius grandis Forel, Plagiolepis pygmaea (Latreille), Plagiolepis schmitzii Forel, Tapinoma madeirense Forel, Tetramorium forte Forel, Solenopsis sp.
Discussion
The seven new hosts noted here raise the number of known hosts to 35 species, belonging to 16 genera and 10 tribes (Table 3). A new subfamily, Dolichoderinae, is added to the Formicinae and Myrmicinae as subfamilies, where Myrmicinosporidium has been detected. Therefore, up to now only the formicoid clade (Moreau et al., 2006; Brady et al., 2006) contains examples of infected ants.
Feeding habits of the newly detected infected ants are highly variable: from granivores (Messor, Goniomma) to scavengers (Cataglyphis), mainly nectarivores (Crematogaster) and omnivores (Aphaenogaster, Tapinoma). Thus, the distinct methods of acquisition and composition of the different foods implied in these varied feeding regimes do not seem to be crucial in the cycle of the fungus, suggesting that neither food nor feeding habits are characteristic to the infection process. At present, no arboricolous ants have been found to be infected.
As all specimens were collected in pitfall traps, we assume that workers were foraging seemingly unaffected. Selection against virulence should be particularly strong in ants, where most transmission events will be within the colony (Boomsma et al., 2005). Little is known about the biology and specificity of Myrmicinosporidium. Its presence in the Palearctic, the Nearctic and the Southern hemisphere, and differences in spore size (Pereira, 2004) allowed speculation regarding the conspecificity of all Myrmicinosporidium populations. Espadaler and Santamaria (2012) expressed doubts over the conspecificity of all cases of Myrmicinosporidium, because of the wide phylogenetic range of host species. Now, with the new data revealed here, those doubts seem to be premature. Instead, we now believe that the detection within the small physical limits of one Portuguese olive grove (Póvoa de São Miguel) of seven hosts, belonging to seven genera, from five tribes, and three subfamilies argues against possible specific differentiation of Myrmicinosporidium on different hosts. A similar situation was already noted within the limits of an organic citrus grove in Spain (Espadaler and Santamaria, 2012), where four genera belonging to four tribes from two subfamilies were found to be parasitized with Myrmicinosporidium. This supports the hypothesis that Myrmicinosporidium is indeed a single gene pool, a generalist microparasite, and not a mixture of differentiated host lineages. Finally, it is worth mentioning that these two sites are under organic management. This is an agricultural method that promotes abundance and diversity of soil fungi (Hole et al., 2005; Birkhofer et al., 2008). Of course, we accept that these two sites (one in Portugal, one in Spain) do not offer a high number of replicates and our argument is merely a suggestion that the soil conditions created by organic management may promote the presence of Myrmicinosporidium infected ants. Experimental rearing of the fungus through cross-feeding controlled colonies, as performed by Buschinger and Kleespies (1999) with the neogregarine Mattesia geminata Jouvenaz and Anthony, and molecular studies, are required to clarify the phylogenetic position of Myrmicinosporidium. Only a genetic analysis will allow to determine whether the different host ant species are infected by same clones or different strains or maybe even cryptic species of the fungus.
References
Birkhofer K., Martijn Bezemer T., Bloem J., Bonkowski M., Christensen S., Dubois D., Ekelund F., Fließbach A., Gunst L., Hedlund K., Mäder P., Mikola J., Robin C., Setäla H., Tatin-Froux F., Van der Putten W.H. and Scheu S. 2008. Long-term organic farming fosters below and aboveground biota: Implications for soil quality, biological control and productivity. Soil Biol. Biochem. 40: 2297-2308
Bolton B. 2003. Synopsis and classification of Formicidae. Mem. Am. Entomol. Inst. 71: 1-370
Boomsma J.J., Schmid-Hempel P. and Hughes W.O.H. 2005. Life histories and parasite pressure across the major groups of social insects. In: Insect Evolutionary History (Fellowes M.D.E., Holloway G.J. and Rolff J., Eds), Royal Entomological Society, pp 139-175
Brady S.G., Schultz T.R. and Fisher B.L. 2006. Evaluating alternative hypotheses for the early evolution and diversification of ants. Proc. Natl Acad. Sci. U.S.A. 103: 18172-18177
Buschinger A., Beibl J., D’Ettorre P. and Ehrhardt W. 2004. Recent records of Myrmicinosporidium durum Hölldobler, 1933, a fungal parasite of ants, with first record North of the Alps after 70 years. Myrmecol. Nachr. 6: 9-12
Buschinger A. and Kleespies R.G. 1999. Host range and host specificity of an ant-pathogenic gregarine parasite, Mattesia geminata (Neogregarinida: Lipotrophidae). Entomol. Gen. 24: 93-104
Buschinger A. and Winter U. 1983. Myrmicinosporidium durum Hölldobler 1933, Parasit bei Ameisen (Hym., Formicidae) in Frankreich, der Schweiz und Jugoslawien wieder aufgefunden. Zool. Anz. 210: 393-398
Collingwood C.A. and Prince A. 1998. A guide to ants of continental Portugal (Hymenoptera: Formicidae). Bol. Soc. Port. Entomol., Supl. 5: 1-49
Cremer S., Armitage S.A.O. and Schmid-Hempel P. 2007. Social immunity. Curr. Biol. 17: R693-R702
Espadaler X. 1982. Myrmicinosporidium sp., parasite interne des fourmis. Etude au MEB de la structure externe. In: La Communication chez les Sociétés d’Insectes (De Haro A. and Espadaler X., Eds). Coll. Int. U.I.E.I.S., Barcelona, pp 239-241
Espadaler X. 1997. Pheidole williamsi (Hymenoptera, Formicidae) parasitized by Myrmiciniosporidium durum (Fungi) on San Salvador Island (Galápagos Islands). Sociobiology 30: 99-101
Espadaler X. and Santamaria S. 2012. Ecto- and endoparasitic fungi on ants from the Holarctic region. Psyche 2012 (168478): 1-10
García F. and Espadaler X. 2010. Nuevos casos y hospedadores de Myrmicinosporidium durum Hölldobler, 1933 (Fungi). Iberomyrmex 2: 3-9
Hole D.G., Perkins A.J.,Wilson J.D., Alexander I.H., Grice P.V. and Evans A.D. 2005. Does organic farming benefit biodiversity? Biol. Conserv. 122: 113-130
Hölldobler K. 1927. Über merkwürdige Parasiten von Solenopsis fugax. Zool. Anz. 70: 333-334
Hölldobler K. 1929. Über eine merkwürdige Parasitenerkrankung von Solenopsis fugax. Z. Parasit. 2: 67-72
Hölldobler K. 1933. Weitere Mitteilungen über Haplosporidien in Ameisen. Z. Parasit. 6: 91-100
Kanizsai O. 2010. Myrmicinosporidium durum, egy különös hangyapatogén. Proc. 3rd Carpathian Basin Myrmecol. Symposium: 5, Senete, Romania
Moreau C.S., Bell C.D., Vila R., Archibald S.B. and Pierce N.E. 2006. Phylogeny of the ants: Diversification in the age of angiosperms. Science 312: 101-104
Pereira R.M. 2004. Occurrence of Myrmicinopsoridium durum in red imported fire ant, Solenopsis invicta and other new ant hosts in eastern United States. J. Inv. Pathol. 84: 38-44
Reber A. and Chapuisat M. 2011. Diversity, prevalence and virulence of fungal entomopathogens in colonies of the ant Formica selysi. Insect. Soc. doi 10.1007/s00040-011-0209-3
Sánchez-Peña S.R., Buschinger A. and Humber R.A. 1993. Myrmicinosporidium durum, an enigmatic fungal parasite of ants. J. Inv. Pathol. 61: 90-96
Schmid-Hempel P. 1998. Parasites in Social Insects. Princeton University Press, Princeton, NJ
Seifert B. 1992. A taxonomic revision of the Palaearctic members of the ant subgenus Lasius s. str. (Hymenoptera: Formicidae). Abh. Ber. Naturkundemus. Görlitz 66: 1-66
Ugelvig L.V. and Cremer S. 2007. Social prophylaxis: group interaction promotes collective immunity in ant colonies. Current Biology 17: 1967-1971
Ugelvig L.V., Kronauer D.J.C., Schremp A., Heinze J. and Cremer S. 2010. Rapid anti-pathogen response in ant societies relies on high genetic diversity. Proc. R. Soc. B 277: 2821-2828
Acknowledgments
Comments by two reviewers added value to the manuscript. This study was financially supported by FEDER Funds through the Programa Operacional Factores de Competitividade—COMPETE and National Funds through the FCT—Fundação para a Ciência e Tecnologia, within project PTDC/AGR-PRO/111123/2009: The use of biological indicators as tools for assessing the impact of agricultural practices in the sustainability of olive groves. X.E. is currently supported by Ministerio de Ciencia y Tecnología MCYT-FEDER grant CGL2010-18182.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gonçalves, C., Patanita, I. & Espadaler, X. Substantial, and significant, expansion of ant hosts range for Myrmicinosporidium Hölldobler, 1933 (Fungi). Insect. Soc. 59, 395–399 (2012). https://doi.org/10.1007/s00040-012-0232-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00040-012-0232-z