Abstract
Objectives
The development of successful policies to reduce income-related inequalities in cervical cancer screening rates requires an understanding of the reasons why low-income women are less likely to be screened. We sought to identify important determinants contributing to inequality in cervical screening rates.
Methods
We analyzed data from 92,541 women aged 25–64 years, who participated in the World Health Survey in 2002–2003. Income-related inequality in Pap screening was measured using the concentration index (CI). Using a decomposition method for the CI, we quantified the contribution to inequality of age, education level, marital status, urbanicity and recent health-care need.
Results
There was substantial heterogeneity in the contributions of different determinants to inequality among countries. Education generally made the largest contribution (median = 15%, interquartile range [IQR] = 23%), although this varied widely even within regions (e.g., 5% in Austria, 28% in Hungary). The contribution of rural residence was greatest in African countries (median = 10%, IQR = 13%); however, there was again substantial within-region variation (e.g., 26% in Zambia, 2% in Kenya).
Conclusions
Considerable heterogeneity in the contributions of screening determinants among countries suggests interventions to reduce screening inequalities may require country-specific approaches.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cervical cancer disproportionately affects poor, socially marginalized women around the world. The major burden of cervical cancer is in developing countries, where over 80% of the 500,000 cases and 300,000 deaths each year occur (Sankaranarayanan and Boffetta 2010). Striking inequalities exist not only between better- and worse-off nations, but also between more and less socioeconomically advantaged groups within countries (Parikh et al. 2003). Between- and within-country inequalities in cervical cancer incidence and deaths primarily reflect inequalities in cervical screening coverage, as well as differences in risk factors for exposure to human papillomavirus (HPV) (IARC 2005).
Studies conducted in high-income countries, including those with universal health-care access, have found that women from socioeconomically disadvantaged backgrounds are less likely to receive cervical cancer screening (Baker and Middleton 2003; Maxwell et al. 2001; Moser et al. 2009; Puig-Tintoré et al. 2008; Siahpush and Singh 2002; Sutton and Rutherford 2005; Todorova et al. 2009; Eaker et al. 2001; Alves et al. 2009; Akers et al. 2007). Much more limited data exist on socioeconomic inequalities in cervical cancer screening within developing countries, where most research has relied on small sub-national samples (Chan et al. 2002; Martins et al. 2009; Arrossi et al. 2008; Lazcano-Ponce et al. 1997; Wellensiek et al. 2002). Cross-national comparisons of inequality in cervical screening have focused mainly on between-country inequalities in average screening rates. For example, a recent comparison of cervical screening coverage in 57 countries reported that 63 and 19% of women of age 25–64 years in high- and low-income countries, respectively, had been screened for cervical cancer in the past 3 years (Gakidou et al. 2008). Furthermore, in both high- and low-income countries, research on socioeconomic inequalities in screening has typically focused on comparisons of extreme socioeconomic groups rather than assessing screening across the entire socioeconomic spectrum. In cross-country comparisons, inequality measures that compare only extreme groups may lead to very different country rankings than methods that account for the entire distribution of socioeconomic position (Gwatkin et al. 2007).
We contribute to the literature on cross-national differences in cervical screening in two respects. First, we calculate and report cross-population comparable within-country summary measures of income-related inequality in cervical cancer screening that account for the full distribution of socioeconomic position. Second, we decompose income-related health inequalities within countries into constituent socio-demographic and behavioral determinants. This decomposition allows identification of the relative contributions of other factors in explaining income-related inequality on a country-by-country basis. Identifying determinants that contribute substantially to income-related inequality may reveal potential areas to intervene to reduce screening inequalities.
Methods
Data
The World Health Survey (WHS) is a large cross-sectional study conducted in 70 high-, middle-, and low-income countries in 2002–2003. It was designed to provide a comprehensive assessment of population health, including socio-demographics, adult and child morbidity and mortality, risk factors, health-care expenditures, and coverage of health interventions (Ustun et al. 2003). Using a multistate cluster design, random national samples were obtained for all participating countries except China, Comoros, the Republic of Congo, Côte d’Ivoire, India, and the Russian Federation.
Full details of the surveys are available on the WHS Web site (WHO). Briefly, adults aged 18 years and older who were living in private households were eligible to be interviewed. Face-to-face interviews were conducted in all countries except Australia, Israel, Luxembourg, and Norway. In these countries, computerized telephone interviews were employed. Interviewers were lay people with at least a high school education who had attended a training session. All questionnaires were translated into local languages and modified for cultural appropriateness according to standard World Health Organization (WHO) protocol.
Measures
The WHS included two questions related to cervical cancer screening that were asked of all women aged 18–70 years: (1) “When was the last time you had a pelvic examination, if ever?” and (2) “The last time you had the pelvic examination, did you have a PAP smear test?” Women were asked the latter only if they reported having a pelvic examination in the past 3 years. Interviewers provided prepared descriptions of pelvic examination and Pap smear test in cases where the interviewee seemed unsure of the procedures (WHO 2002) To maintain consistency with a previous analysis of cervical screening using the WHS data, our analyses included women between 25 and 64 years of age (Gakidou et al. 2008).
We used permanent income as our variable for socioeconomic status, which was estimated using the asset-based approach developed by Ferguson et al. (2003). This approach uses household ownership of assets (e.g., a car, a radio), access to services (e.g., electricity, drinking water), and known predictors of income (e.g., age, education) to estimate permanent household income. Assets for each country are chosen to estimate country-specific distributions of permanent income that can then be adjusted to an internationally comparable scale to enable cross-country comparisons. Asset-based measures of permanent income have been validated against reported household income and expenditures using household survey data from Greece, Peru, and Pakistan (Ferguson et al. 2003). This approach has also been applied in several previous studies (Gakidou et al. 2007; Gakidou and Vayena 2007; Pongou et al. 2006; Vapattanawong et al. 2007).
For the decomposition analysis, we examined variables that have been found to be associated with both cervical screening and permanent income: age, education level, marital status, and urbanicity (Palencia et al. 2010; Akers et al. 2007; Arrossi et al. 2008; Leyden et al. 2005; Albuquerque et al. 2009). We also included a variable indicating whether or not the woman reported needing health care for herself or her child within the past year (Winkler et al. 2008). Age and education were continuous variables, measured in years. Marital status and urbanicity were coded as indicator variables (never married vs. married/cohabitating, separated/divorced/widowed vs. married/cohabitating, and urban/peri-urban vs. rural residence).
Of the 110,658 women aged 25–64 years who participated in the WHS, data for both cervical screening and permanent income were available for 92,541 women. The surveys from Turkey and Norway had missing data on income, cervical screening, or both, and were excluded from the analysis. We also excluded Myanmar because income was missing for 81% of the eligible subjects. For the decomposition analysis, the sample size was reduced to 89,371 because we excluded women who had missing data on any of the five determinants. Netherlands, Australia, and Slovenia had missing data on urbanicity for all subjects, so we performed the decomposition analysis without urbanicity as a determinant for these three countries. Finally, unweighted analyses are reported for Austria, Denmark, Germany, Greece, Guatemala, Italy, The Netherlands, Slovenia, and the UK because probability-sampling weights were not available for these countries.
Statistical analysis
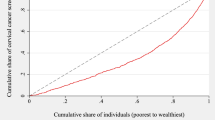
We measured within-country income-related inequalities in cervical screening rates using the concentration index (CI) (Wagstaff et al. 1991; Kakwani et al. 1997). The CI quantifies the magnitude of inequality portrayed by the concentration curve, a plot of the share of health accounted for by cumulative proportions of individuals ranked according to socioeconomic position. Figure 1 shows the relative concentration curves for cervical screening for Guatemala and Spain. The curves extend below the diagonal 45° line of equality for both countries, indicating the presence of inequality to the disadvantage of the poor. However, inequality was more prominent in Guatemala, where the poorest 50% of women obtained just over 20% of the country’s total cervical screenings, compared to just over 40% in Spain.
The CI is defined as twice the area between the concentration curve and the line of equality (Wagstaff et al. 1991). The index ranges from −1 to 1, with 0 representing complete equality. For a positive health variable such as cervical screening, positive values of the index indicate inequality to the disadvantage of the poor, and negative values indicate inequality to the disadvantage of the rich. The relative CI can be written as:
where μ is the overall screening rate, y i is the screening status of the ith individual, and R i is the ith individual’s rank in the income distribution (Wagstaff et al. 1991). We applied the normalization formula proposed by Wagstaff (2005) to correct for the fact that the maximum values of the CI are bounded by the overall prevalence when the health variable is binary. This was accomplished by dividing the CI by 1 − μ. This normalized CI was used to measure relative inequalities in other studies with binary health outcomes (Khadr 2009; Harper and Lynch 2007). We refer to the normalized CI as W. The absolute concentration index (ACI) is calculated by multiplying the CI by the mean of the health variable (μ). We report confidence intervals for W and ACI, with standard errors corrected for heteroskedasticity and autocorrelation of the rank variable (O’Donnell et al. 2007).
Wagstaff et al. (2003) showed that the CI can be decomposed by relating the health outcome y i to a set of k potential determinants, x ki :
where the β k are regression coefficients and ε i is an error term. Although the assumption of linearity of this equation is necessary for the decomposition, it can be extended to allow for binary health outcomes (Gravelle 2003).
The proportional contribution of each x k to overall socioeconomic inequality will be the same whether one considers decomposing relative or absolute inequality. Because we mainly focus on cross-country comparisons of absolute inequality, we decomposed the ACI using the following equation:
where β k are the regression coefficients from Eq. 2, ACI k is the absolute concentration index for x k , and ACIε represents the portion of inequality that cannot be explained by variation in the x k across income. The first term (β k ACI k ) thus measures the combined contribution to overall income-related inequality in screening of each determinant’s effect on screening and its association with income. Both the determinant’s effect on screening and its association with income can be either negative or positive, thus the contribution can also be negative or positive. For example, if higher education increases the likelihood of screening (positive β k ) and is more concentrated among the well off (positive ACI k ), education will make a positive contribution to the overall ACI. A negative contribution indicates that there is either no pro-rich inequality in the determinant (ACI k is negative) or that the determinant decreases the likelihood of screening (β k is negative).
Using this method, we examined the magnitude of income-related screening inequality due to differences in age, marital status, urbanicity, education, and a measure of recent health-care need. Because cervical screening was measured as a binary variable, we used a probit model with marginal effects in the decomposition analysis (O’Donnell et al. 2007). It has been noticed that the contributions obtained using the probit model may change depending on the reference category chosen for the determinants in the model (Yiengprugsawan et al. 2010). Marital status was our only variable with possible alternate reference categories; thus, we checked the decomposition results using the different reference categories, detecting no important differences. We proceeded with the probit model using currently married/cohabitating as the reference. The decomposition analysis was performed for all countries with statistically significant screening inequality (i.e., the 95% confidence interval for the CI excluded zero, the value signifying no inequality).
The WHS had missing data ranging from 0.4 to 35.2% by country for the questions on cervical screening. To address the missing data problem, we compared the demographic profiles of subjects with missing data on the cervical screening questions to those with data, finding no large discrepancies. We also performed sensitivity analyses for calculation of the concentration indices under four hypothetical scenarios. To do this, we generated four data sets by making assumptions about observations that were missing for Pap screening and income as follows: (1) unscreened, income = 10th percentile of income distribution; (2) unscreened, income = 90th percentile; (3) screened, income = 10th percentile; (4) screened, income = 90th percentile. All analyses were performed using Stata, version 11.
Results
There were wide variations in cervical screening rates among countries, ranging from <1% in Ethiopia and Bangladesh to 83% in Austria (Table 1). In 15 countries, all located in Asia or Africa, <5% of women had a Pap screen in the past 3 years. Country-specific means for each of the determinants included in the decompositions are also presented in Table 1.
Table 2 shows the normalized (W) and absolute concentration indices (ACI) along with their corresponding 95% confidence intervals for each of the 67 countries. There was statistically significant income-related screening inequality in 50 countries. Country rankings on inequality were highly dependent on whether we examined W or ACI. For example, although Laos had the largest W, it ranked 44th in terms of absolute inequality (ranks are found in the last two columns of Table 2).
The Americas had the largest inequality among regions (median ACI = 0.058, interquartile range [IQR] = 0.043), with Guatemala and Paraguay ranked number one and two out of all countries in terms of absolute inequality (ACI = 0.128 and 0.079, respectively). However, there was substantial variation even among countries in the Americas region, with Mexico and Ecuador having low levels of inequality (ACI = 0.023 and 0.035, respectively). In general, we observed substantial heterogeneity in the magnitude of inequality among countries, even within regions. For example, screening inequality in China (ACI = 0.067) was substantially greater than in Vietnam (ACI = 0.009), and in South Africa (ACI = 0.056) was much greater than in neighboring Zimbabwe (ACI = 0.013).
Table 3 presents the regression coefficients (β k ) and ACI for the determinants in the decomposition analysis. The β k for income are positive in all countries, indicating that there is still an association between income and cervical screening after controlling for the other determinants. In the majority of countries, higher education level, urban residence, younger age, and health-care need were associated with an increased probability of screening. Concentration indices for the determinants are mostly consistent across countries, demonstrating that rural residence, divorced/separated/widowed status, and, especially, lower education level, tend to be more concentrated among the poor.
The percent contributions of each of the determinants to overall income-related screening inequality are presented in Table 4. In general, there was heterogeneity in the contributions of different determinants to inequality, although some general trends did emerge. Besides income itself, education made the largest contribution to explaining inequality (median = 15%, IQR = 23%), although its contribution varied widely even within regions (e.g., 5% in Austria, 28% in Hungary). The contribution of rural residence was greatest in African countries (median = 10%, IQR = 13%), although there was again substantial within-region variation (e.g., 26% in Zambia, 2.3% in Kenya). With a few exceptions, income-related screening inequality in European countries was minimally explained by urban/rural residence (median = 0.3%, IQR = 5%). Many countries had small contributions for marital status; however, there were also a few countries that had large negative contributions (for example, Finland and Sweden with −38.8 and −34.4%, respectively). These negative contributions resulted because unmarried and divorced/widowed women were both more likely to be screened than married women (i.e., β k s were positive), but also more likely to be poor (negative concentration indices). Finally, with few exceptions, age generally made small contributions to screening inequality, largely because it had weak effects on the likelihood of screening.
The percentage of inequality unexplained by the determinants was <10% in 21 of the 50 countries examined in the decomposition analysis. For the most part, the unexplained portion of the ACI was large in eastern Mediterranean and southeast Asian countries, moderate in countries in Africa, the Western Pacific, and the Americas (median = 21, 9, and 11%, respectively), and smallest in European countries (median = 6.8%).
In the sensitivity analysis, we found that the ACIs for our complete case analysis were close to the ACIs for each of the four scenarios for the majority of countries (see Supplemental Table 1).
Discussion
This study confirms the existence of widespread pro-rich inequality in cervical screening around the world. In line with previous findings, our results point to a high degree of screening inequality in southern and eastern Europe and the Americas (Todorova et al. 2009; Palencia et al. 2010; Arrossi et al. 2008). We extend the results of a previous cross-country comparison of cervical screening that used the WHS data in two important ways (Gakidou et al. 2008). First, in contrast to the previous study, which presented only the prevalence of screening across countries and by income quintiles, we calculate summary measures of within-country inequalities that facilitate cross-country comparisons. Second, we go beyond quantifying the magnitude of screening inequality to explore sources of inequality using decomposition analyses.
We found Guatemala to have the greatest pro-rich inequality in absolute terms, followed by Paraguay, Portugal, and China. In general, our results suggest substantial heterogeneity both in the magnitude of inequality and the contributions of the determinants to inequality, even among countries in close geographic proximity and with similar economic and development status. In the decomposition analysis, income remained a strong determinant of inequality, with education also making substantial contributions in many countries. Other determinants such as urbanicity, age, and marital status were more variable, contributing to inequality in some countries but not in others. There were also several instances when a determinant made positive contributions in some countries (indicating it contributed to pro-rich inequality) and negative contributions in other countries (contributing to pro-poor inequality). These observations suggest that national environments (e.g., social, policy) may play an important role in conditioning the strength and direction by which specific individual socio-demographic determinants contribute to inequalities in screening.
This study is unique in how we measure socioeconomic inequalities in cervical screening. Unlike previous studies, which have relied primarily on descriptive statistics (Gakidou et al. 2008) and ratios comparing socioeconomic groups (Baker and Middleton 2003; Arrossi et al. 2008; Puig-Tintoré et al. 2008; Martins et al. 2009; Moser et al. 2009), we used the CI as a summary measure of inequality. The CI has several useful features: it accounts for the entire distribution of socioeconomic status, it is sensitive to the socioeconomic gradient in health so that pro-rich and pro-poor inequality corresponds to positive and negative values of the index for a positive health outcome, and it can be decomposed into a linear combination of the CIs of its determinants (Wagstaff et al. 1991, 2003). Furthermore, our decision to focus on absolute screening inequalities differs from the approach taken in most other studies (Alves et al. 2009; Arrossi et al. 2008; Baker and Middleton 2003; Martins et al. 2009; Moser et al. 2009; Puig-Tintoré et al. 2008; Todorova et al. 2009; Winkler et al. 2008). The ACI reflects two dimensions that are important in measuring the burden of socioeconomic inequalities at the population level: (1) the magnitude of socioeconomic inequality among women who receive screening and (2) the overall rates of screening in the population. As an example to illustrate the importance of these two dimensions, the relative indices (W) for France and Zambia were nearly identical (0.269 and 0.272, respectively), yet the prevalence of screening in the two countries was vastly different (76 and 3.4%, respectively). This difference is captured by the ACI, indicating that absolute inequalities in screening are five times larger in France (0.049) than Zambia (0.009). Another important advantage of the ACI is that, unlike W, it does not depend on whether one examines inequality in health (screening) or inequality in ill health (no screening) (Clarke et al. 2002).
This study has some limitations, in particular the reliance of the WHS on self-reported data. Self-reports of cervical screening may be inaccurate because women may have a limited ability to accurately report whether a Pap test was performed in the context of other gynecologic/obstetric care. For example, a meta-analysis of 37 studies from developed countries found that, compared to medical records, women tend to over-report their participation in Pap screening (Howard et al. 2009). In addition, the WHS only asked about Pap screening within the past 3 years. Although this shorter time interval probably reduces recall bias, it also means that we could not account for women who had been screened more than 3 years ago. This is unfortunate because several countries recommend screening at longer intervals than 3 years. For example, in Ecuador and South Africa, the recommendation is that women be screened once every 5 years and once every 10 years, respectively (IARC 2005). In fact, evidence suggests that even one Pap test every 10 years reduces the risk of cancer by 64% (IARC 1986). Finally, as previously stated, random national samples were not obtained in China, Comoros, the Republic of Congo, Côte d’Ivoire, India, and the Russian Federation. Thus, estimates of screening inequality cannot be taken as nationally representative in these countries.
With regard to the decomposition analysis, there were several countries where the proportion of the income-related CI that the determinants did not explain was large. These tended to be countries with low screening rates, mainly in Asia and Africa. It is possible that in these countries, our model did not capture some important determinants that predict screening inequality: for example, within-country geographic variability in screening availability, health insurance status, and knowledge and perceptions about screening (Forbes et al. 2002). To the extent that these unmeasured variables are correlated with income, there may be an overestimation of the residual effect of income in our study. While this is an important limitation to consider when interpreting some of the results, our decomposition model did explain a large proportion of inequality in many countries, particularly those in Europe and the Americas. Finally, because we were dealing with a binary health outcome, electing to preserve the absolute value judgment of the ACI, the bounds issue (the fact that the bounds of the CI depend on the mean of the health variable) was left unsolved, meaning that ACI may tend to be underestimated for high prevalence countries. Erreygers (2009) has also proposed a corrected CI, which may be useful if one wants to examine the relationship between average health and socioeconomic inequality (Van de Poel et al. 2008). However, for the purpose of cross-country comparisons of inequality, Erreygers’ index preserves the same ranking of distributions as the ACI (Erreygers 2009).
In this study, we present comparable measures of income-related inequality in cervical screening across a comprehensive list of high- and low-income countries from all regions of the world. The widespread inequalities we observed suggest that many countries should consider targeting interventions to improve screening among socioeconomically disadvantaged women. This requires an understanding of the reasons that low-income women are less likely to be screened and that is why the decomposition results are so valuable. Finally, we of course acknowledge that in several developing countries, average rates of screening are extremely low, to the point where even measuring inequality seems somewhat futile. However, our results do suggest that it may be beneficial for countries planning to implement or scale up cervical screening programs to specifically consider issues of equality when designing programs and policies.
References
Akers AY, Newmann SJ, Smith JS (2007) Factors underlying disparities in cervical cancer incidence, screening, and treatment in the United States. Curr Probl Cancer 31(3):157–181. doi:10.1016/j.currproblcancer.2007.01.001
Albuquerque KM, Frias PG, Andrade CL, Aquino EM, Menezes G, Szwarcwald CL (2009) Pap smear coverage and factors associated with non-participation in cervical cancer screening: an analysis of the cervical cancer prevention program in Pernambuco state, Brazil. Cad Saude Publica 25 Suppl 2:S301–S309. doi:10.1590/S0102-311X2009001400012
Alves C, Alves L, Lunet N (2009) Prevalence and determinants of cervical cytology use in an urban sample of Portuguese women. Eur J Cancer Prev. doi:10.1097/CEJ.0b013e328330eb47
Arrossi S, Ramos S, Paolino M, Sankaranarayanan R (2008) Social inequality in Pap smear coverage: identifying under-users of cervical cancer screening in Argentina. Reprod Health Matters 16(32):50–58. doi:10.1016/S0968-8080(08)32410-0
Baker D, Middleton E (2003) Cervical screening, health inequality in England in the 1990. J Epidemiol Community Health 57(6):417–423
Chan C, Ho SC, Chan SG, Yip YB, Wong FC, Cheng F (2002) Factors affecting uptake of cervical and breast cancer screening among perimenopausal women in Hong Kong. Hong Kong Med J 8(5):334–341
Clarke PM, Gerdtham U-G, Johannesson M, Bingefors K, Smith L (2002) On the measurement of relative and absolute income-related health inequality. Soc Sci Med 55(11):1923–1928
Eaker S, Adami HO, Sparen P (2001) Reasons women do not attend screening for cervical cancer: a population-based study in Sweden. Prev Med 32(6):482–491. doi:10.1006/pmed.2001.0844S0091-7435(01)90844-8
Erreygers G (2009) Correcting the concentration index. J Health Econ 28(2):504–515. doi:10.1016/j.jhealeco.2008.02.003
Ferguson BD, Tandon A, Gakidou E, Murray CJ (2003) Estimating permanent income using indicator variables. In: Murray CJL, Evans DB (eds) Health systems performance assessment: debates methods and empiricism. World Health Organization, Geneva, p 927
Forbes C, Jepson R, Martin-Hirsch P (2002) Interventions targeted at women to encourage the uptake of cervical screening. Cochrane Database Syst Rev (3):CD002834. doi:10.1002/14651858.CD002834
Gakidou E, Vayena E (2007) Use of modern contraception by the poor is falling behind. PLoS Med 4(2):e31. doi:10.1371/journal.pmed.0040031
Gakidou E, Oza S, Vidal Fuertes C, Li AY, Lee DK, Sousa A, Hogan MC, Vander Hoorn S, Ezzati M (2007) Improving child survival through environmental and nutritional interventions: the importance of targeting interventions toward the poor. JAMA 298(16):1876–1887. doi:10.1001/jama.298.16.1876
Gakidou E, Nordhagen S, Obermeyer Z (2008) Coverage of cervical cancer screening in 57 countries: low average levels and large inequalities. PLoS Med 5(6):e132
Gravelle H (2003) Measuring income related inequality in health: standardisation and the partial concentration index. Health Econ 12(10):803–819
Gwatkin DR, Rutstein S, Johnson K, Suliman E, Wagstaff A, Amouzou A (2007) Socio-economic differences in health, nutrition, and population within developing countries: an overview. Niger J Clin Pract 10(4):272–282
Harper S, Lynch J (2007) Trends in socioeconomic inequalities in adult health behaviors among US states, 1990–2004. Public Health Rep 122(2):177–189
Howard M, Agarwal G, Lytwyn A (2009) Accuracy of self-reports of Pap and mammography screening compared to medical record: a meta-analysis. Cancer Causes Control 20(1):1–13. doi:10.1007/s10552-008-9228-4
IARC (1986) Screening for squamous cervical cancer: Duration of low risk after negative results of cervical cytology and its implication for screening policies. IARC working group on evaluation of cervical cancer screening programmes. Br Med J (Clin Res Ed) 293(6548):659–664
IARC (2005) IARC handbooks of cancer prevention vol. 10: cervix cancer screening. Lyon, France
Kakwani N, Wagstaff A, van Doorslaer E (1997) Socioeconomic inequalities in health: measurement, computation, and statistical inference. J Econom 77(1):87–103
Khadr Z (2009) Monitoring socioeconomic inequity in maternal health indicators in Egypt: 1995–2005. Int J Equity Health 8:38. doi:10.1186/1475-9276-8-38
Lazcano-Ponce EC, Najera-Aguilar P, Buiatti E, Alonso-de-Ruiz P, Kuri P, Cantoral L, Hernandez-Avila M (1997) The cervical cancer screening program in Mexico: problems with access and coverage. Cancer Causes Control 8(5):698–704
Leyden WA, Manos MM, Geiger AM, Weinmann S, Mouchawar J, Bischoff K, Yood MU, Gilbert J, Taplin SH (2005) Cervical cancer in women with comprehensive health care access: attributable factors in the screening process. J Natl Cancer Inst 97(9):675–683. doi:10.1093/jnci/dji115
Martins LFL, Valente JG, Thuler LCS (2009) Factors related to inadequate cervical cancer screening in two Brazilian state capitals. Rev Saude Publica 43(2):318–325
Maxwell CJ, Bancej CM, Snider J, Vik SA (2001) Factors important in promoting cervical cancer screening among Canadian women: findings from the 1996–1997 national population health survey (nphs). Can J Public Health 92(2):127–133
Moser K, Patnick J, Beral V (2009) Inequalities in reported use of breast and cervical screening in Great Britain: analysis of cross sectional survey data. BMJ 338:b2025–b2025. doi:10.1136/bmj.b2025
O’Donnell O, van Doorslaer E, Wagstaff A, Lindelow M (2007) Analyzing health equity using household survey data: a guide to techniques and their implementation. World Bank, Washington
Palencia L, Espelt A, Rodriguez-Sanz M, Puigpinos R, Pons-Vigues M, Pasarin MI, Spadea T, Kunst AE, Borrell C (2010) Socio-economic inequalities in breast and cervical cancer screening practices in Europe: influence of the type of screening program. Int J Epidemiol. doi:10.1093/ije/dyq003
Parikh S, Brennan P, Boffetta P (2003) Meta-analysis of social inequality and the risk of cervical cancer. Int J Cancer 105(5):687–691. doi:10.1002/ijc.11141
Pongou R, Ezzati M, Salomon JA (2006) Household and community socioeconomic and environmental determinants of child nutritional status in Cameroon. BMC Public Health 6:98. doi:10.1186/1471-2458-6-98
Puig-Tintoré LM, Castellsagué X, Torné A, de Sanjosé S, Cortés J, Roura E, Méndez C, Bosch FX (2008) Coverage and factors associated with cervical cancer screening: Results from the AFRODITA study: a population-based survey in Spain. J Low Genit Tract Dis 12(2):82–89. doi:10.1097/LGT.0b013e3181599c16
Sankaranarayanan R, Boffetta P (2010) Research on cancer prevention, detection and management in low- and medium-income countries. Annals of Oncology: official journal of the European Society for Medical Oncology/ESMO. doi:10.1093/annonc/mdq049
Siahpush M, Singh GK (2002) Sociodemographic predictors of Pap test receipt, currency and knowledge among Australian women. Prev Med 35(4):362–368. doi:S0091743502910868
Sutton S, Rutherford C (2005) Sociodemographic and attitudinal correlates of cervical screening uptake in a national sample of women in Britain. Soc Sci Med 61(11):2460–2465. doi:10.1016/j.socscimed.2005.07.017
Todorova I, Baban A, Alexandrova-Karamanova A, Bradley J (2009) Inequalities in cervical cancer screening in Eastern Europe: perspectives from Bulgaria and Romania. Int J Public Health 54(4):222–232. doi:10.1007/s00038-009-8040-6
Ustun B, Chatterji S, Mechbal A, Murray CJ, WHS collaborating groups (2003) The world health surveys. In: Murray CJL, Evans DB (eds) Health systems performance assessment: debates methods and empiricism. World Health Organization, Geneva, pp 797–808
Van de Poel E, Hosseinpoor AR, Speybroeck N, Van Ourti T, Vega J (2008) Socioeconomic inequality in malnutrition in developing countries. Bull World Health Organ 86(4):282–291
Vapattanawong P, Hogan MC, Hanvoravongchai P, Gakidou E, Vos T, Lopez AD, Lim SS (2007) Reductions in child mortality levels and inequalities in Thailand: analysis of two censuses. Lancet 369(9564):850–855. doi:10.1016/S0140-6736(07)60413-9
Wagstaff A (2005) The bounds of the concentration index when the variable of interest is binary, with an application to immunization inequality. Health Econ 14(4):429–432
Wagstaff A, Paci P, Vandoorslaer E (1991) On the measurement of inequalities in health. Soc Sci Med 33(5):545–557
Wagstaff A, van Doorslaer E, Watanabe N (2003) On decomposing the causes of health sector inequalities with an application to malnutrition inequalities in Vietnam. J Econ 112(1):207–223
Wellensiek N, Moodley M, Moodley J, Nkwanyana N (2002) Knowledge of cervical cancer screening and use of cervical screening facilities among women from various socioeconomic backgrounds in Durban, Kwazulu Natal, South Africa. Int J Gynecol Cancer 12(4):376–382
WHO (2002) World health survey: guide to administration and question by question specifications. http://www.who.int/healthinfo/survey/instruments/en/index.html. Accessed 15 January 2010
Winkler J, Bingham A, Coffey P, Handwerker WP (2008) Women’s participation in a cervical cancer screening program in northern Peru. Health Educ Res 23(1):10–24. doi:10.1093/her/cyl156
Yiengprugsawan V, Lim LL, Carmichael GA, Dear KB, Sleigh AC (2010) Decomposing socioeconomic inequality for binary health outcomes: an improved estimation that does not vary by choice of reference group. BMC Res Notes 3:57. doi:10.1186/1756-0500-3-57
Acknowledgments
We thank the Institute for Health Metrics and Evaluation for providing us with the estimates of permanent income that were used in these analyses. This work was supported by the Canadian Institutes for Health Research (191612). At the time of this research, Sam Harper was supported by a Chercheur-boursier from the Fonds de la Recherche en Santé du Québec (FRSQ), and Spencer Moore was supported through a New Investigator Award from the CIHR-Institute of Aging.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
McKinnon, B., Harper, S. & Moore, S. Decomposing income-related inequality in cervical screening in 67 countries. Int J Public Health 56, 139–152 (2011). https://doi.org/10.1007/s00038-010-0224-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00038-010-0224-6