Abstract
Anthropogenic alteration of terrestrial shorelines can have pronounced effects on marine environments at the upland-marsh boundary. Possible terrestrial development effects on several physical and biological variables of high-marsh habitats were examined along developed and undeveloped shorelines in an ocean-dominated, southeastern US estuary. Analyses of sediment characteristics suggested development of the upland boundary affected physical conditions within the high-marsh. For example, pore water salinities were greater along undeveloped shorelines during a non-drought period even after rain events. Significant floral and faunal differences also existed between shoreline treatments. Black needle rush stems were significantly taller and marsh periwinkle densities significantly greater, but eastern coffee bean snail densities were significantly reduced along developed shorelines. Benthic infaunal community abundance and composition also were significantly different between shoreline treatments with sand fly larvae, human pest precursors, either only present or present in greater densities along developed shorelines. Sediment respirometry experiments indicated significant differences in heterotrophic and autotrophic processes occurring between shoreline treatments. Greater sediment surface temperatures along developed shorelines provided one possible mechanism driving high-marsh responses to boundary alteration. The history and extent of shoreline development along with a tendency in ocean-dominated southeastern marshes to resist change likely influenced current ecological conditions within our high-marsh study areas. A greater understanding of the driving mechanisms producing localized effects on salt marshes and recognizing regional differences in marsh resistance to change will facilitate predictions of shoreline development consequences and help in proposing effective management strategies for coastal boundaries.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The coastal transition zone (CTZ) is a critical environmental boundary or interface between terrestrial/freshwater and marine systems (e.g., Levin et al. 2001; Talley et al. 2003). Disproportionate to the limited aerial extent globally, the coastal region including the CTZ provides an extensive range of necessary human and ecosystem services (Costanza et al. 1997). Similar to most boundaries (Gosz 1992; Wiens et al. 1985; Yarrow and Marín 2007), the CTZ is defined primarily by abrupt physical gradients (e.g., salinity) and changes in material flux (e.g., nutrients) that occur over local, 10s of meters, to landscape scales, 100s of kilometers. For example, in the southeastern USA, a typical local-scale species gradient from the glycophytic Morella cerifera (wax myrtle) to the halophytic Spartina alterniflora (cord grass) is contained within the landscape scale gradient between the terrestrial watershed and contiguous estuarine environment. Within estuarine river systems, faunal compositions are dependent on changes in two opposing gradients, one from freshwater to marine and the other from marine to freshwater (Attrill and Rundle 2002), which produce a dynamic boundary within estuaries that moves up- and down-stream with changes in climate. Increasingly, both natural and anthropogenic boundaries are recognized as profoundly influencing ecosystem dynamics (e.g., Cadenasso et al. 2003; Ries et al. 2004). Both the alteration of natural boundaries (e.g., removal of native vegetation) and creation of anthropogenic boundaries (e.g., highways, seawalls) are dependent on human activities. Worldwide, coastal environments are being transformed into human-dominated habitats creating and altering boundaries at an increasing rate (Beach 2002). In the USA >50% of the population lives or works within coastal counties (Vernberg et al. 1992), and future population growth along the South Atlantic Bight (e.g., North Carolina, South Carolina and Georgia) is expected to exceed that of all other regions (Bricker et al. 1999). The anticipated population-driven development and resultant changes to terrestrial-marine boundaries could significantly affect one of the region’s most ecologically and economically important habitats, the estuarine salt marsh.
Previous research along the eastern USA coast typically has examined the effects of landscape-scale terrestrial development within associated tidal creeks and bays. Nutrient (Mallin et al. 2000a; Valiela and Bowen 2002; Lewitus et al. 2004; White et al. 2008), fecal coliform (Mallin et al. 2000b; DiDonato et al. 2009), and phytoplankton changes in water quality (White et al. 2004) all have been attributed to watershed development. Differences in tidal creek sediment characteristics (Sanger et al. 1999a, b; Van Dolah et al. 2008) and benthic faunal communities (Lerberg et al. 2000; Bilkovic et al. 2006) also have been correlated to landscape-scale change resulting from suburban, urban and industrial alterations of coastal watersheds. Although less studied, local development of the terrestrial-marine boundary also can affect adjacent marine habitats (Wigand et al. 2001; Bertness et al. 2002; Pennings et al. 2002; Silliman and Bertness 2004; Bertness and Silliman 2008; Gedan et al. 2009; Wigand et al. 2010). In the rapidly developing Southeastern USA coastal region, both unknown local- and well-studied landscape-scale changes (e.g., Lerberg et al. 2000; Holland et al. 2004; Lewitus et al. 2004) are likely to have major affects on the terrestrial-marine boundary and adjacent marsh habitats.
Direct, local-scale development of the terrestrial-marine boundary may affect a variety of high marsh physical and biological characteristics. Removal of natural vegetation as a result of shoreline stabilization, suburban landscaping, or other development could alter thermal regimes, surface and below-ground water movements, nutrient and organic inputs, and other factors that contribute to the flux between terrestrial and marine environments. Small scale horizontal differences in high-marsh temperatures resulting from alterations to the thermal environment, such as changes in shading, can influence heat stress levels experienced by intertidal organisms (Jost and Helmuth 2007). Altered anthropogenic nutrient inputs can affect salt marsh plant productivity and benthic microbial activity (Morris 1991; Kiehl et al. 1997; Levine et al. 1998; Morris and Bradley 1999; Van Wijnen and Bakker 1999; Sundareshwar et al. 2003; Hines et al. 2006; Deegan et al. 2007). Nutrient additions also can alter typical patterns of plant zonation and modify normal competitive interactions and community structure (Levine et al. 1998; Emery et al. 2001; Bertness et al. 2002; Pennings et al. 2005; Crain 2007; McFarlin et al. 2008; but also see Brewer 2003). Even the community structure of marsh fauna may be influenced by elevated nutrient inputs (Vince et al. 1981; Stiven and Kuenzler 1982; Silliman and Zieman 2001; Denno et al. 2003; but also see Meany et al. 1976). Anticipated differences in freshwater flux during precipitation events (e.g., Corbett et al. 1997; Wahl et al. 1997) may result in changes in sediment pore-water salinities that affect plant production (Mendelssohn and Morris 2000) and community structure (Pennings and Callaway 1992). Separate from the landscape-scale effects of watershed development, alteration of the terrestrial-marine boundary likely will have major consequences for the dynamics of high-marsh environments.
This paper focuses on identifying the physical (e.g., sediment organic content, pore water salinity, surface temperature) and biological parameters (e.g., plant biomass and richness, macrofauna density and composition, benthic respiration) of a typical southeastern USA high-marsh community that may be affected by development of the associated terrestrial shoreline. Landscape-scale effects are factored out by stratifying selection of developed and undeveloped shorelines within a single marine-dominated estuary surrounded by a watershed of limited aerial extent. Development effects on the high-marsh associated with both barrier land spits and mainland areas were measured. Shoreline development was expected to increase the flux of freshwater and nutrients as in previous studies (Wahl et al. 1997; Bertness et al. 2002; Silliman and Bertness 2004), which would lead to increased autotrophic and heterotrophic production and potentially alter floral and faunal community structure. Results provide the first extensive study of local-scale terrestrial development effects on the high-marsh environment in a region experiencing rapid population growth and increasing terrestrial-marine boundary alteration.
Methods
Study area
The study was conducted within the ocean-dominated estuary of Murrells Inlet, South Carolina (33:32.7′ N, 79:2.7 W) where salinities average > 30 ppt (Blood and Vernberg 1991). Within the inlet, approximately equal semidiurnal tides, mean 1.37 m, vary in timing, ca. 30 min delay, but not height of high tide from north to south. Freshwater input is restricted to a relatively small, local watershed (ca. 35.5 km2) without major riverine influence. Mainland drainage into the estuary can be divided into eight sub-basins, some of which are emptied by small creeklets (Tufford et al. 2003). The estuary covers ca. 26.7 km2 between the barrier land spits and mainland, almost half of which is salt marsh. A primary inlet stabilized by the construction of two weir jetties in 1977 splits the estuary roughly in half. The cities of Murrells Inlet and Garden City are located on the estuary and have a combined population of 14,876 individuals (2000 USA census). Murrells Inlet housing density in 2000 was 165.8 units km−2. Surrounding Georgetown and Horry County populations have increased by up to 36.5% between 1990 and 2000 (1990 and 2000 USA census). Huntington Beach State Park, an 1,100 ha undeveloped area, forms the southern boundary of the inlet.
Regional rainfall averages 138.4 cm year−1 with 115 days year−1 of precipitation, but surrounding counties experienced intermittent drought conditions throughout the study period. The South Carolina Department of Natural Resources, Land, Water, and Conservation Division, State Climate Office declared statewide severe to extreme drought conditions ended in April 2003, 2 months prior to the study’s start. Incipient drought conditions were declared for Georgetown and Horry counties in June 2004 and downgraded to normal in July 2004. Reduced rainfall amounts from 12 to 25 cm below normal between September and November 2004, encompassing the study’s last sampling dates, prompted a November 2004 advisory but no declaration of drought conditions. Average daily temperatures range from a low of 2.3°C in January to a high of 32.4°C in July.
The position of terrestrial-wetland boundary locations (n = 8) studied within Murrells Inlet are shown in Fig. 1. Location names, inlet position, development status including type and approximate time since development, coordinates, and sampling dates are found in Table 1. All boundary locations were selected primarily based on a visual assessment of CTZ development status. Undeveloped locations (n = 4) were identified by the presence of an extensive overhang of trees and dense stand of shrubs, typically consisting of native oaks (e.g., Quercus virginiana), wax myrtle (Morella cerifera), yaupon (Ilex vomitoria), and southern red cedar (Juniperus silicicola) (Fig. 2, top). Developed locations (n = 4) had >90% of the boundary vegetation removed (Fig. 2, bottom), including native trees and shrubs that often were replanted with grass or cultivated shrubbery when the type of development was residential housing. A contiguous length (ca. 50 m) of shoreline and high marsh unbroken by tidal creeks, without an upland berm, and with a minimum of 0.2 km of salt marsh between open water and the shoreline boundary was selected at each location. Selection of developed locations with consistently similar characteristics was hampered by a lack of accessible, multiple shorelines of sufficient length within Murrells Inlet having the same extent (>90% of natural vegetation removed), type (e.g., residential, commercial), and age of boundary alteration. The extent of shoreline development did not vary among developed locations, but differences in the type and age of development (Table 1) could affect our results and conclusions (see “Discussion”).
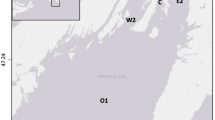
Map of South Carolina indicating location of Murrells Inlet (inset), and Google® satellite image of inlet showing sampling locations. Refer to Table 1 for location abbreviations
Wetland areas adjacent to the terrestrial boundary at each location consisted of salt marsh species typical of southeastern USA high intertidal elevations; black needle rush (Juncus roemerianus), salt hay (Spartina patens), marsh elder (Iva frutescens), and sea oxeye (Borrichia frutescens). Marsh samples from each location were collected from an area <50 m long, to avoid possible edge effects at the ends of developed or undeveloped boundaries, and <15 m wide, to remain in close proximity to the CTZ. All salt marsh areas sampled were shoreward of the short S. alterniflora zone in the high marsh just adjacent (<15 m) to the terrestrial boundary. Additionally, in October 2003, samples were collected from within J. roemerianus stands located directly adjacent to the boundary to reduce potential variation associated with sampling within a mixed plant community.
Variables sampled
Various physical and biological variables were sampled between June 2003 and September 2004, but data for all variables were not collected at all locations on all dates (Table 1). Sample variation and boundary status effects initially were evaluated in June 2003. Based on June results, a more extensive study was conducted in October 2003 to document boundary treatment differences. Follow-up samples in March and September 2004 were collected to examine significant or unexplained results from the October 2003 study or additional variables. Differences among dates likely affected the variables sampled but not the proportional effects of boundary development on the variables. Only data from individual sampling dates were analyzed statistically to minimize any potential to confound date with boundary development effects (see “Analyses”).
Up to five plots (0.25 m2) were positioned haphazardly within the ca. 50 by 15 m sampling area at each developed and undeveloped boundary location. All snails, exclusively Littoraria irrorata and Melampus bidentatus, were collected and a haphazard subsample of stem heights (n = 20) was measured from each plot. Live and standing-dead plant stem densities were enumerated from one 0.0625 m2 sub-plot within each plot. Plant biomass was determined by removing all above-ground material from each sub-plot, determining dry mass (60°C for 4 days), and calculating ash-free dry mass (AFDM) after loss from ignition (450°C for >4 h). Within the same high-marsh elevation, sediments were sampled (n = 5) using a 2.1 cm diameter corer to a depth of 5 cm. Individual cores, either whole or divided into 0–2 and 2–5 cm sections (June 2003 only), immediately were frozen at −20°C for later determination of pore water salinity and sediment grain size and organic content. Pore water salinity (ppt) was measured both in the lab and field. Salinities in the lab were calculated by adding 20–30 ml of distilled water to a thawed sediment sample, repeatedly agitating over 48 h, allowing the sediment to settle, measuring the supernatant salinity with a refractometer, and correcting for dilution. Field salinities were measured using an YSI 85 salinometer within 2–3 surface wells dug to a depth of 30 cm at each site. Sediment grain sizes were determined by measuring the dry mass of wet-sieved standard soil size fractions. The sediment organic content or AFDM (mg g−1) was calculated as the difference between sediments dried at 60°C and then ashed at 450°C for >4 h. An additional five sediment cores were collected using a 2.1 cm diameter corer to a depth of 5 cm to identify benthic infauna. Core samples were preserved with 10% formaldehyde, processed on a 500-μm sieve in the lab, and all organisms identified to major taxon and enumerated.
Sediment samples for benthic respiration incubations were collected in a 10.2 cm diameter acrylic corer to a depth of 3 cm. Only at the barrier island location, three sites 0.0625 m2 in area were selected haphazardly within developed and undeveloped locations. At each site, three sediment cores were collected and mixed mechanically, any root or rhizome material removed, and dispensed into 250-ml acid-washed Pyrex glass bottles. Sediments were slurried with the addition of 30% by volume of either 0.45 μm filtered seawater (control), filtered seawater with 20 μM nitrogen as ammonium sulphate and 1.25 μM phosphorus as sodium diphosphate, or filtered seawater with 200 μM glucose. Samples were placed within a Percival climate controlled chamber at 28°C and incubated under either light or dark conditions. Benthic respiration measurements were collected multiple times (n = 4) during the 3 h incubation using a Columbus Instruments 30 channel respirometer.
Sediment surface temperatures were determined during a 2-week period in September 2004 using HOBO® and Stowaway Tidbit® temperature loggers. Loggers were placed above the highest high tide level on the terrestrial side of the boundary at Murrells Inlet mainland locations (Table 1). Sites where loggers were located differed mainly in the presence or absence of vegetation >1.5 m in height.
Analyses
Data from our initial June 2003 samples were used to conduct a prospective or a priori power analysis using G*Power v3.0 (Faul et al. 2007). Mean differences and variation between developed and undeveloped locations for each variable measured were used to calculate initial effect sizes and estimate appropriate sample sizes to detect significant shoreline treatment effects. Sample sizes were estimated for 2-tailed tests with an error probability or α = 0.05, power or 1 − β = 0.80, and based on the calculated effect sizes from the June samples. Future sample sizes and approaches were adjusted based on the results of power analyses and a qualitative assessment of collection and processing costs.
Sediment grain size distributions were analyzed separately for each inlet side using the Kolmogorov–Smirnov Z-test for two independent samples. Sediment pore water salinity and organic content, plant density, height and biomass, snail and benthic infaunal abundance, and benthic respiration rate data all were analyzed using parametric ANOVA models including blocked one-way, nested and two-way analyses. Data were transformed when necessary to satisfy critical assumptions of the ANOVA models. Inlet side (barrier, mainland) in October 2003 and sample dates without rain in March 2004 (see Table 1) were treated as block effects, except for the analysis of benthic respiration which was conducted only at the barrier location. Significant differences between undeveloped barrier and mainland locations, given the range of potential differences between inlet sides (e.g., groundwater discharge, soil properties), and between dates without major rainfall events were anticipated, but inlet side or date differences were not the focus of the current study. Our primary factor of interest was shoreline development.
Differences in the taxonomic composition of benthic infauna were analyzed using PERMANOVA, permutational multivariate analysis of variance (Anderson 2005). The PERMANOVA program provides an ANOVA-like test based on P values generated from permutations of a distance matrix derived from the original species by sample data (Anderson 2001, 2005; McArdle and Anderson 2001). Barrier and mainland location compositional differences were analyzed separately, abundance data were 4th root transformed, and distances were based on Bray-Curtis dissimilarity measures (Anderson 2005).
Results
Prospective power analyses for June 2003 initial data indicated that detectable effect sizes ranged between 0.5 or medium and 0.8 or large (see Cohen 1988), but the total samples required to detect differences at a 0.05 significance level with 80% power often were large (Table 2). In a number of instances (e.g., L. irrorata, 3–5 mm AFDM and pore water) the large effect sizes were detectable with relatively few samples (Table 2). Based on initial results, we decided to increase sample sizes where possible to guarantee a minimum of five samples per location for each measurement variable, to increase the number of locations (n = 2–3), and to combine 0–2 and 2–5 cm sediment depth data into a single sample in an effort to reduce among sample variability and increase the ability to detect reasonable effect size differences between shoreline treatments.
Analyses of sediment characteristics in October 2003 provided suggestive evidence for the effects of boundary development. No significant difference in sediment particle size distributions (Fig. 3) existed between developed and undeveloped locations for either barrier (Z = 0.577, P > 0.05) or mainland locations (Z = 0.289, P > 0.05). Except for the occasional size-class disparity (e.g., >0.125 mm), differences in developed and undeveloped sediment grain size fractions were similar between barrier and mainland locations (Fig. 3). Sediment AFDM exhibited significant block (location) effects (F 1, 17 = 5.03, P < 0.04) but not a significant shoreline development effect (F 1, 17 = 1.67, P > 0.05). Mainland compared to barrier sediments contained greater amounts of organic matter, but among sample variation limited detection of a significant development effect on sediment AFDM (Fig. 4). Sediment pore water salinities in October 2003 also exhibited a significant block or location effect (F 1, 16 = 14.83, P < 0.005) but not a significant shoreline development effect (F 1, 16 = 0.87, P > 0.05). Barrier sediment salinities were two times mainland values, but no consistent difference between developed and undeveloped locations was identified (Fig. 4). In situ comparison of mainland pore water salinities in March 2004 on two dates 5 days apart after 7+ days without rain resulted in a marginally significant date block effect (F 1, 20 = 4.10, P < 0.057), no significant shoreline development effect (F 1, 4 = 4.84, P > 0.05) but a significant site effect (F 4, 20 = 4.61, P < 0.009). Although unable to detect significant differences, undeveloped location salinities were ca. two times that of developed locations (Fig. 5). Comparison of salinity before and after a 0.64–1.3 cm rain event in March 2004 resulted in nonsignificant development (F 1, 2 = 3.89, P > 0.05), rain (F 1, 2 = 5.17, P > 0.05) or interaction effects (F 1, 2 = 0.05, P > 0.05). However, among site effects again were significant (F 2, 35 = 4.83, P < 0.03). The rainfall event did not affect pore water salinities significantly, but undeveloped locations consistently had greater pore water salinities before and after the rain event (Fig. 5).
Characteristics of the predominant high-marsh plant at all locations, J. roemerianus, generally did not exhibit the effects of shoreline development. Live and dead stem densities were not significantly different between locations (F 1, 17 = 7.34, P > 0.05 and F 1, 17 = 8.96, P > 0.05, respectively) or between shoreline treatments (F 1, 17 = 0.35, P > 0.05 and F 1, 17 = 0.48, P > 0.05, respectively). Live stems tended to be more abundant at all developed locations, but dead stems were more numerous at mainland undeveloped locations (Fig. 6). Stem AFDM also was not significantly different between locations (F 1, 17 = 0.34, P > 0.05) or shoreline treatments (F 1, 17 = 0.03, P > 0.05). However, stem heights were significantly different between locations (F 2, 388 = 1,765.15, P < 0.001) and shoreline treatments (F 1, 8 = 6.95, P < 0.04). Stem heights were greater at both mainland and barrier developed locations (Fig. 6).
Boundary development also had varying effects on resident marsh fauna. Snail densities exhibited a significant location (F 1, 17 = 10.52, P < 0.006) and shoreline development effect (F 1, 17 = 13.73, P < 0.003) for L. irrorata but no significant location (F 1, 17 = 2.87, P > 0.05) or shoreline development effect (F 1, 17 = 2.95, P > 0.05) for M. bidentatus. Periwinkles virtually were absent from mainland locations and densities were greater at developed barrier locations (Fig. 7). However, M. bidentatus densities were greater at undeveloped barrier locations (Fig. 7). Benthic infaunal abundances were significantly different by location (F 1, 17 = 13.18, P < 0.003) and shoreline treatment (F 1, 17 = 4.73, P < 0.045), but only the barrier island undeveloped site exhibited greater infaunal abundances, ca. fourfold, compared to the developed site (Fig. 7). The similarity of taxonomic compositions analyzed by PERMANOVA also was significantly different between developed and undeveloped shorelines for barrier (F 1, 8 = 4.38, P < 0.02) and mainland locations (F 1, 8 = 7.83, P < 0.02). Compositions were more similar among barrier developed, 42.5%, compared to undeveloped shorelines, 28.4%, but were less similar among mainland developed, 45.1%, compared to undeveloped shorelines, 53.2%. Both barrier and mainland undeveloped shorelines had greater abundances of oligochaetes while developed shorelines had greater abundances of ceratopogonid larvae (Table 3). Mainland developed shorelines also, in general, had a greater number of Diptera larvae (e.g., Ceratopogonidae, Hexatoma, Chironomidae).
The responses of benthic autotrophic and heterotrophic communities at the barrier location varied between undeveloped and developed shorelines (Fig. 8). A significant interaction between sediment and shoreline treatments affected respiration rates under natural light conditions (F 2, 12 = 5.41, P < 0.03). Heterotrophic consumption was greater in developed compared to undeveloped control sediments but was decreased in N & P addition sediments (Fig. 8). Dark incubations resulted in no significant interaction (F 2, 12 = 1.67, P > 0.05) or sediment treatment effects (F 2, 12 = 1.90, P > 0.05) but a significant shoreline treatment effect (F 1, 12 = 28.82, P < 0.001). Undeveloped shoreline heterotrophic oxygen consumption was greater in all sediment treatments (Fig. 8).
The maximum and range of sediment surface temperatures varied over the 2-week period sampled at mainland developed and undeveloped locations (Fig. 9). A significant interaction between date and boundary development existed for maximum temperatures (F 13, 56 = 1.94, P < 0.05), but while temperatures varied between 25 and 40°C, on no date did undeveloped exceed developed (Fig. 9). No significant interaction (F 13, 56 = 1.79, P > 0.05) but significant date (F 13, 13 = 17.08, P < 0.001) and boundary treatment effect (F 1, 13 = 26.79, P < 0.001) existed for daily temperature ranges. Day–night temperatures varied between 5 and 18°C, but the range was always greater at the developed locations (Fig. 9).
A summary of the ecological attributes measured and shoreline alteration effects determined in the present study are compared with observed or predicted effects of shoreline development from previous studies in Table 4. Shoreline development has or is expected to increase freshwater inflow and both inorganic and organic loading in the high salt marsh environment (see Wahl et al. 1997; Bertness et al. 2002; Silliman and Bertness 2004). Expected or predicted changes occurred in only 50% of the ecological attributes measured in Murrells Inlet (Table 4). Typically significant differences between developed and undeveloped shorelines were not detected (e.g., plant density, benthic species richness), but in a few instances (e.g., sediment organic content) the observed trend contradicted expectations (Table 4).
Discussion
The alteration of terrestrial shoreline characteristics affected a number of ecological attributes in the high-marsh environment within an ocean-dominated, southeastern USA estuary. Faunal densities and composition, sediment surface temperatures, benthic microbial respiration rates and plant height all exhibited predictable, significant differences between developed and undeveloped shorelines. Densities of L. irrorata, J. roemerianus stem height, and both temperature maxima and range were significantly greater while night-time microbial oxygen consumption was significantly reduced at developed shoreline locations. Increased densities of L. irrorata can have cascading effects within salt marshes (Silliman and Zieman 2001; Silliman and Bertness 2002), and anthropogenic development can lead to consumer control of marsh productivity (Bertness et al. 2008; Bertness and Silliman 2008). Although few effects of consumer control were evident at Murrells Inlet locations (Walters et al. pers. obs.), L. irrorata densities even along developed shorelines were <50% of the numbers reported in studies where consumers do affect marsh productivity (Silliman and Zieman 2001; Bertness and Silliman 2008). Autumn marsh periwinkle densities from undeveloped locations also were low compared to spring snail densities of just the year before (Hutchens and Walters 2006). Typically undeveloped, high marsh shorelines in northern South Carolina tend to have low L. irrorata densities but increased biomass in spring relative to mid- and low-marsh elevations (Hutchens and Walters 2006). Summer recruitment and growth in blue crabs, Callinectes sapidus, a major predator on L. irrorata (Schindler et al. 1994; Lewis and Eby 2002), could account for observed snail density differences between spring and autumn. Increased crab numbers or size could increase predation pressure on snail populations in the fall of the year, but would not account for density differences between adjacent developed and undeveloped shorelines. However, crab predation might reduce overall autumn snail densities within Murrells Inlet marshes so that snail populations along developed shorelines do not increase to densities sufficient to produce catastrophic cascade effects on the marsh.
The benthic infauna was dominated by oligochaetes and nematodes at all marsh locations, but barrier island side densities were greater along the undeveloped shoreline. Average total metazoan densities at all shorelines (66–401 ind/10 cm2) were ca. 50% less than densities in the preserved North Inlet marshes (Bell 1980). Taxonomic similarity among samples varied most at undeveloped locations possibly indicating a more patchily distributed fauna. The most conspicuous difference between developed and undeveloped locations was the order of magnitude greater proportion of larval ceratopogonids collected along developed shorelines. Ceratopogonid larvae were absent from barrier undeveloped shorelines but comprised 13% of total metazoan abundance (14 ind/10 cm2) at the developed shoreline. Increased densities of larvae suggest that altered shoreline conditions may lead to unanticipated environmental management issues for Murrells Inlet residents and tourists. Adult ceratopogonids (sand flies, no-see-ums) are serious mammalian pests and can vector livestock diseases (e.g., blue tongue) as a result of the female’s biting behavior. Developed locations also contained two other groups of Diptera flies absent from undeveloped locations: Chironomidae or midge flies and Hexatoma (Tipulidae) or crane flies. Coastal environments that vary widely in salinity (e.g., oligohaline marshes to saline pools) typically contain increased densities of nematocerous dipterans, both biting and non-biting species (LaSalle and Bishop 1987; Giberson et al. 2001). Although not statistically significant between shoreline treatments, salinities varied most along developed shorelines (from <10 to >35 ppt) and the variation may be what enables greater densities of Diptera larvae to persist. Whatever the reason, shoreline development appears to contribute to an increase in insect pests and the potential need for greater management efforts (e.g., pesticide spraying).
Shoreline development resulted in consistently greater daily temperature maxima and greater day–night ranges at the sediment surface. Intertidal temperature patterns can be variable and unpredictable both spatially and temporally (Helmuth and Hofmann 2001; Helmuth et al. 2002; Helmuth et al. 2006), but differences in temperature experienced by organisms lead to dramatic differences in distributions, selective pressure and behavior (Helmuth et al. 2006; Jost and Helmuth 2007; Schneider and Helmuth 2007; Pincebourde et al. 2008). Although only documented over a relatively short, 2-week period, the persistent temperature difference between developed and undeveloped marsh shorelines is likely to affect organisms typically inhabiting the high marsh. Ribbed mussels, Geukensia demissa, experienced thermal stress and mortality under natural conditions along an undeveloped high marsh shoreline in North Inlet, ca. 25 km south of Murrells Inlet (Jost and Helmuth 2007). High marsh mussels were uncommon in our samples, but temperature differences between developed and undeveloped locations may have affected the coffee bean snail, Melampus bidentatus, another high marsh resident. The coffee bean snail has no operculum and is believed to bury in the sediments or invade crab burrows to minimize thermal stress (McMahon and Russell-Hunter 1981). Coffee bean snails experience increased mortality (McMahon and Russell-Hunter 1981) at air temperatures similar to or below (ca. 40°C) the body temperatures that cause increased mortality in ribbed marsh mussels (Jost and Helmuth 2007). Melampus bidentatus were significantly more abundant along undeveloped shorelines, especially on the barrier island side of Murrells Inlet. The greater thermal stress of developed shorelines may result in either increased mortality or emigration possibly explaining the observed significant difference in snail densities.
Oxygen consumption within sediments, primarily the result of heterotrophic bacteria, was affected by shoreline development. Although consumption rates suggested heterotrophy predominated production pathways along both barrier island shorelines, both heterotrophic (dark) and autotrophic production rates (light–dark) were greater at the undeveloped location. Heterotrophic bacterial production in salt marsh soils generally is understood to be phosphorus limited, but continued phosphorus loading may shift the bacterial population to a carbon-limited status (Sundareshwar et al. 2003). Typical of many environments, the structure of salt marsh microbial communities is sensitive to increases in anthropogenic pollutants (Cao et al. 2006). Darby and Turner (2008) also suggest that phosphorus loading and the resulting competition with microbes can decrease belowground Spartina tissue production and be detrimental to long-term salt marsh maintenance. Although difficult to reconcile definitively based on the comparison of barrier island shorelines, the greater oxygen demand, greater metazoan abundance, and the trend towards greater organic matter in undeveloped sediments suggests the presence of a more productive benthic food web.
The significantly greater heights of J. roemerianus stems suggest shoreline development is associated with increased production in the adjacent salt marsh. Juncus roemerianus is a generalist species tolerating a wide range of conditions (Woerner and Hackney 1997; Brinson and Christian 1999), but previous research indicates that biomass production is greater when grown at relatively low soil salinities (Pennings et al. 2005). Compared to J. roemerianus populations further from the shoreline, Touchette (2006) also demonstrated that high marsh plants primarily fed by upland freshwater were less stressed and had greater biomass. Although runoff from areas adjacent to our study locations was not measured directly, salinity results from before and after a rain event suggest that development and the concomitant increased freshwater runoff may be responsible for decreased pore water salinity and improved growing conditions for J. roemerianus. In New England, shoreline development is associated with dramatic changes in plant community composition, including the aggressive invasion by Phragmites australis, a non-native reed (Bertness et al. 2002, 2008, 2009; Silliman and Bertness 2004). Although present in the area (Luken and Walters 2009), shoreline development in Murrells Inlet has not resulted in an invasion by P. australis (Walters et al. pers. obs.). A number of factors including limits on chance dispersal and lack of habitat disturbance (e.g., wrack) could account for the absence of P. australis in Murrells Inlet. Alternatively, shoreline development in southeastern marshes may lead to more subtle changes that modify natural environmental gradients and trophic structure but do not necessarily alter the composition of the salt marsh plant community.
The increased freshwater runoff associated with shoreline development also is accompanied typically with increased nitrogen loading (Bowen and Valiela 2001; Bertness et al. 2002, 2009; Wigand et al. 2003) and alteration in the timing of release of mineral nitrogen to associated near shore habitats (Wahl et al. 1997; Gorman et al. 2009). In communities comprised of S. alterniflora and S. patens, nitrogen loading will alter plant growth, change vegetation structure and modify competitive interactions (Bertness et al. 2002; Wigand et al. 2003). The common presence of a J. roemerianus zone adjacent to the upland/marsh boundary in many southeastern marshes suggests a relatively high degree of plant diversity as a result of natural gradients in flooding and salinity (Pennings et al. 2005). As the closest marsh plant to the terrestrial boundary, the J. roemerianus zone may be viewed as a secondary buffer that receives and processes the various effects of shoreline development and possibly mitigates anticipated consequences on other marsh species. The regional differences in resistance to shoreline development, specifically the types and prevalence of plant species that may serve as buffers to changing environmental conditions, may explain many of the unanticipated results in our current study.
Even given the possible regional differences, almost 50% of the predicted effects of shoreline development were not observed in Murrells Inlet. Aside from variables not measured or not measured with sufficient power, a number of factors previously not considered in studies of shoreline development may have contributed to our inability to predict consistently the effects of shoreline development in southeastern marshes: the extent and history of shoreline modification. The extent of shoreline modification can vary from no alteration or no removal of the native salt-shrub thicket bordering a marsh to severe alteration or removal of all native vegetation and bulk-heading, channeling, ditching or other modification of the normal tidal flow taking place. Differences also can occur within a modification category. In suburbanized Murrells Inlet, housing developments along the marshes can range from no alteration of the native vegetation, at least directly along the marsh/upland boarder, to complete removal of all native vegetation and alternative landscaping including extensive planting of turf grasses. Depending on the mindset or economy, individual households where complete removal of native vegetation occurs, additionally may experience weekly, monthly, seasonally, or no nitrogen supplements in the form of fertilizers along with the possible additional application of pesticides and herbicides. Although each Murrells Inlet developed shoreline represented a residential alteration of the existing habitat, the extent of the boundary’s modification ranged from minimal alteration to complete removal of native vegetation. Differences in the extent of shoreline modification likely made detection of predicted effects difficult.
Any ecological effects of shoreline development also are likely dependent on the history of boundary alteration. Significant high-marsh changes may not have had time to take place along newly altered shorelines depending on the variable being measured. Based on salt marsh restoration studies, plant biomass (Broome et al. 1986; Roman et al. 2002), genetic diversity (Travis et al. 2002), fish utilization (Roman et al. 2002; Able et al. 2004), and sediment salinities (Walters 2009) can change rapidly within newly planted marshes, while changes in sediment organic content (Craft et al. 1999), nutrient cycling (Thompson et al. 1995; Craft et al. 1999), infauna (Sacco et al. 1994; Levin et al. 1996), and plant canopy (Zedler 1993) tend to require significant time if not decades. Ecological changes in Narragansett Bay salt marshes only were detected after two decades of urbanization (Wigand et al. 2010). The developed shoreline we studied, OMD1, consisted of recently built (<5 years) and ongoing building and therefore was a newly developed shoreline along which high-marsh changes may not have been expected to have occurred. In contrast, the barrier island shoreline was developed for at least 20 years and any differences between shoreline locations (e.g., benthic infaunal densities) may be explained in part by the age of boundary development. The same may be true for any differences between observed and expected boundary development effects. Basing predicted effects on studies from Northeastern marshes, historically experiencing greater impacts from increasing populations, may not be justified. Southeastern marshes may be at the very beginning of the shoreline development trajectory leading ultimately to negative effects on high-marsh environments.
Historically, humans have either directly or indirectly altered salt marsh environments (e.g., Gedan et al. 2009), making an accurate assessment of cumulative historical effects difficult. From the earliest days of European colonization, South Carolina marshes have been ditched, dredged, filled and altered to support numerous commercial or residential pursuits (Gedan et al. 2009). Although overt direct impacts to South Carolina salt marshes are now less common, residential development adjacent to salt marshes is on the increase; there are few signs that development of land adjacent to salt marshes will be curtailed in the future as such lots offer numerous amenities (e.g., scenic vistas, recreation opportunities, investment return). Our research suggests that development of the marsh shoreline may produce subtle changes to the adjacent salt marsh. These subtle changes can be contrasted with the more dramatic changes in northeastern USA marshes that presumably are linked to residential development (Bertness et al. 2002, 2008, 2009; Silliman and Bertness 2004).
Future research might focus on two different but related salt marsh issues. First, our data suggest regional differences in the resistance (stability) of salt marshes. Although salt marshes are relatively simple systems and share many of the same characteristics, understanding why regional differences occur is important to making effective management decisions. Experimental results suggest salinity is an important factor affecting the speed of marsh recovery following disturbance (Crain et al. 2008), and regional differences in factors affecting salinity (e.g., drought periods) may influence the impacts of shoreline development. Second, the effects of small changes to marshes may be cumulative and eventually bring the system to a point where the marsh will experience a major change. The invasion of northeast salt marshes by Phragmites australis is one example where small, cumulative changes in the nutrient status of marshes eventually set the stage for a larger ecosystem change (Silliman and Bertness 2004). Both lines of research are important for understanding how to manage residential development of shorelines in order to minimize potential negative effects on coastal marshes.
References
Able KW, Nemerson DM, Grothues TM (2004) Evaluating salt marsh restoration in Delaware Bay: analysis of fish response at former salt hay farms. Estuaries 27:58–69
Anderson MJ (2001) Permutation tests for univariate or multivariate analysis of variance and regression. Can J Fish Aquat Sci 58:626–639
Anderson MJ (2005) PERMANOVA: a FORTRAN computer program for permutational multivariate analysis of variance. Department of Statistics, University of Auckland, New Zealand
Attrill MJ, Rundle SD (2002) Ecotone or ecocline: ecological boundaries in estuaries. Estuar Coast Shelf Sci 55:929–936
Beach D (2002) Coastal sprawl: the effects of urban design on aquatic ecosystems in the United States. Pew Oceans Commission, Arlington, VA. http://www.pewoceans.org/reports/water_pollution_sprawl.pdf
Bell SS (1980) Meiofauna–macrofauna interactions in a high salt marsh habitat. Ecol Monogr 50:487–505
Bertness MD, Silliman BR (2008) Consumer control of salt marshes driven by human disturbance. Conserv Biol 22:618–623
Bertness MD, Ewanchuk PJ, Silliman BR (2002) Anthropogenic modification of New England salt marsh landscapes. Proc Natl Acad Sci USA 99:1395–1398
Bertness MD, Cain CM, Holdredge C, Sala N (2008) Eutrophication and consumer control of New England salt marsh primary productivity. Conserv Biol 22:131–139
Bertness MD, Silliman BR, Holdredge C (2009) Shoreline development and the future of New England salt marsh landscapes. In: Silliman BR, Grosholz ED, Bertness MD (eds) Human impacts on salt marshes: a global perspective. University of California Press, Berkeley, pp 137–148
Bilkovic DM, Roggero M, Hershner CH, Havens KH (2006) Influence of land use on macrobenthic communities in nearshore estuarine habitats. Estuaries Coasts 29:1185–1195
Blood ER, Vernberg FJ (1991) Characterization of the physical, chemical and biological conditions and trends in Winyah Bay and North Inlet estuaries: 1970–1985. Characterization of the physical, chemical and biological conditions and trends in three South Carolina estuaries, vol. 2. South Carolina Sea Grant Consortium, Charleston, p 117
Bowen JL, Valiela I (2001) The ecological effects of urbanization of coastal watersheds: historical increases in nitrogen loads and eutrophication of Waquoit Bay estuaries. Can J Fish Aquat Sci 58:1489–1500
Brewer JS (2003) Nitrogen addition does not reduce belowground competition in a salt marsh clonal plant community in Mississippi (USA). Plant Ecol 168:93–106
Bricker SB, Clement CG, Pirhalla DE, Orlando SP, Farrow DRG (1999) National estuarine eutrophication assessment: effects of nutrient enrichment in the nation’s estuaries. National Oceanic and Atmospheric Administration, National Ocean Service, Special Projects Office and the National Centers for Coastal Ocean Science, Silver Spring, Maryland, 71 pp
Brinson MM, Christian RR (1999) Stability of Juncus roemerianus patches in a salt marsh. Wetlands 19:65–70
Broome SW, Seneca ED, Woodhouse WW (1986) Long-term growth and development of transplants of the salt-marsh grass Spartina alterniflora. Estuaries 9:63–74
Cadenasso ML, Pickett STA, Weathers KC, Jones CG (2003) A framework for a theory of ecological boundaries. BioScience 53:750–758
Cao Y, Cherr GN, Córdova-Kreylos AL, Fan TW-M, Green PG, Higashi RM, LaMontagne MG, Scow KM, Vines CA, Yuan J, Holden PA (2006) Relationships between sediment microbial communities and pollutants in two California salt marshes. Microb Ecol 52:619–633
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2nd edn. Lawrence Erlbaum Assoc, Hillsdale, p 567
Corbett CW, Wahl M, Porter DE, Edwards D, Moise C (1997) Nonpoint source runoff modeling: a comparison of a forested watershed and an urban watershed on the South Carolina coast. J Exp Mar Biol Ecol 213:133–149
Costanza R, d’Arge R, de Groot R, Farber S, Grasso M, Hannon B, Limburg K, Naeem S, O’Neill RV, Paruelo J, Raskin RG, Sutton P, van den Belt M (1997) The value of the world’s ecosystem services and natural capital. Nature 387:253–260
Craft C, Reader J, Sacco JN, Broome SW (1999) Twenty-five years of ecosystem development of constructed Spartina alterniflora (Loisel) marshes. Ecol Appl 9:1405–1419
Crain CM (2007) Shifting nutrient limitation and eutrophication effects in marsh vegetation across estuarine salinity gradients. Estuaries Coasts 30:26–34
Crain CM, Albertson LK, Bertness MD (2008) Secondary succession dynamics in estuarine marshes across landscape-scale salinity gradients. Ecology 89:2889–2899
Darby FA, Turner RE (2008) Below- and aboveground biomass of Spartina alterniflora: response to nutrient addition in a Louisiana salt marsh. Estuaries Coasts 31:326–334
Deegan LA, Bowen JL, Drake D, Fleeger JW, Friedrichs CT, Galván KA, Hobbie JE, Hopkinson C, Johnson DS, Johnson JM, LeMay LE, Miller E, Peterson BJ, Picard C, Sheldon S, Sutherland M, Vallino J, Warren RS (2007) Susceptibility of salt marshes to nutrient enrichment and predator removal. Ecol Appl 17(Supplement):S42–S63
Denno RF, Gratton C, Döbel H, Finke DL (2003) Predation risk affects relative strength of top-down and bottom-up impacts on insect herbivores. Ecology 84:1032–1044
DiDonato GT, Stewart JR, Sanger DM, Robinson BJ, Thompson BC, Holland AF, Van Dolah RF (2009) Effects of changing land use on the microbial water quality of tidal creeks. Mar Pollut Bull 58:97–106
Emery NC, Ewanchuk PJ, Bertness MD (2001) Competition and salt-marsh plant zonation: stress tolerators may be dominant competitors. Ecology 82:2471–2485
Faul F, Erdfelder E, Lang A-G, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39:175–191
Gedan KB, Silliman BR, Bertness MD (2009) Centuries of human-driven change in salt marsh ecosystems. Annu Rev Mar Sci 1:117–141
Giberson DJ, Bilyj B, Burgess N (2001) Species diversity and emergence patterns of Nematocerous flies (Insecta: Diptera) from three coastal salt marshes in Prince Edward Island, Canada. Estuaries 24:862–874
Gorman D, Russell BD, Connell SD (2009) Land-to-sea connectivity: linking human-derived terrestrial subsidies to subtidal habitat change on open rocky coasts. Ecol Appl 19:1114–1126
Gosz JR (1992) Ecological functions in a biome transition zone: translating local responses to broad-scale dynamics. In: Hansen AJ, diCastri F (eds) Landscape boundaries: consequences for biotic diversity and ecological flows. Springer, New York, pp 55–75
Helmuth B, Hofmann GE (2001) Microhabitats, thermal heterogeneity and physiological gradients of stress in the rocky intertidal zone. Biol Bull 201:374–384
Helmuth B, Harley CDG, Halpin P, O’Donnell M, Hofmann GE, Blanchette C (2002) Climate change and latitudinal patterns of intertidal thermal stress. Science 298:1015–1017
Helmuth B, Broitman BR, Blanchette CA, Gilman S, Halpin P, Harley CDG, O’Donnell MJ, Hofmann GE, Menge B, Strickland D (2006) Mosaic patterns of thermal stress in the rocky intertidal zone: implications for climate change. Ecol Monogr 76:461–479
Hines J, Megonigal JP, Denno RF (2006) Nutrient subsidies to belowground microbes impact aboveground food web interactions. Ecology 87:1542–1555
Holland AF, Sanger DM, Gawle CP, Lerberg SB, Santiago MSS, Riekerk GHM, Zimmerman LE, Scott GI (2004) Linkages between tidal creek ecosystems and the landscape and demographic attributes of their watersheds. J Exp Mar Biol Ecol 298:151–178
Hutchens JJ, Walters K (2006) Gastropod abundance and biomass relationships with salt marsh vegetation within ocean-dominated South Carolina, USA estuaries. J Shellfish Res 25:947–953
Jost J, Helmuth B (2007) Morphological and ecological determinants of body temperature of the Atlantic ribbed mussel, Geukensia demissa, and their effects on mussel mortality. Biol Bull 213:141–151
Kiehl K, Esselink P, Bakker JP (1997) Nutrient limitation and plant species composition in temperate salt marshes. Oecologia 111:325–330
LaSalle MW, Bishop TD (1987) Seasonal abundance of aquatic Diptera in two oligohaline tidal marshes in Mississippi. Estuaries 10:303–315
Lerberg SB, Holland AF, Sanger DM (2000) Responses of tidal creek macrobenthic communities to the effects of watershed development. Estuaries 23:838–853
Levin LA, Talley D, Thayer G (1996) Succession of macrobenthos in a created salt marsh. Mar Ecol Prog Ser 141:67–82
Levin LA, Boesch DF, Covich A, Dahm C, Erseus C, Ewel KC, Kneib RT, Moldenke A, Palmer MA, Snelgrove P, Strayer D, Weslawski JM (2001) The function of marine critical transition zones and the importance of sediment biodiversity. Ecosystems 4:430–451
Levine JM, Brewer JS, Bertness MD (1998) Nutrients, competition and plant zonation in a New England salt marsh. J Ecol 86:285–292
Lewis DB, Eby LA (2002) Spatially heterogeneous refugia and predation risk in intertidal salt marshes. Oikos 96:119–129
Lewitus AJ, Kawaguchi T, DiTullio GR, Keesee DM (2004) Iron limitation of phytoplankton in an urbanized vs. forested southeastern U.S. salt marsh estuary. J Exp Mar Biol Ecol 298:233–254
Luken JO, Walters K (2009) Management of plant invaders within a marsh: an organizing principle for ecological restoration. In: Indergit (ed) Management of invasive weeds. Springer, New York, pp 61–76
Mallin MA, Burkholder JM, Cahoon LB, Posey MH (2000a) North and South Carolina coasts. Mar Pollut Bull 41:56–75
Mallin MA, Williams KE, Esham EC, Lowe RP (2000b) Effect of human development on bacteriological water quality in coastal watersheds. Ecol Appl 10:1047–1056
McArdle BH, Anderson MJ (2001) Fitting multivariate models to community data: a comment on distance-based redundancy analysis. Ecology 82:290–297
McFarlin CR, Brewer JS, Buck TL, Pennings SC (2008) Impacts of fertilization on a salt marsh food web in Georgia. Estuaries Coasts 31:313–325
McMahon RF, Russell-Hunter WD (1981) The effects of physical variables and acclimation on survival and oxygen consumption in the high littoral salt-marsh snail, Melampus bidentatus Say. Biol Bull 161:246–269
Meany RA, Valiela I, Teal JM (1976) Growth, abundance and distribution of larval tabanids in experimentally fertilized plots on a Massachusetts salt marsh. J Appl Ecol 13:323–332
Mendelssohn IA, Morris JT (2000) Eco-physiological controls on the productivity of Spartina alterniflora Loisel. In: Weinstein MP, Kreeger DA (eds) Concepts and controversies in tidal marsh ecology. Kluwer, Dordrecht, pp 59–80
Morris JT (1991) Effects of nitrogen loading on wetland ecosystems with particular reference to atmospheric deposition. Annu Rev Ecol Syst 22:257–279
Morris JT, Bradley PM (1999) Effects of nutrient loading on the preservation of organic carbon in wetland sediments. Limnol Oceanogr 44:699–702
Pennings SC, Callaway RM (1992) Salt marsh plant zonation: the relative importance of competition and physical factors. Ecology 73:681–690
Pennings SC, Wall VD, Moore J, Pattanayek M, Buck TL, Alberts JJ (2002) Assessing salt marsh health: a test of the utility of five potential indicators. Wetlands 22:405–414
Pennings SC, Grant MB, Bertness MD (2005) Plant zonation in low-altitude salt marshes: disentangling the roles of flooding, salinity and competition. J Ecol 93:159–167
Pincebourde S, Sanford E, Helmuth B (2008) Body temperature during low tide alters the feeding performance of a top intertidal predator. Limnol Oceanogr 53:1562–1573
Ries L, Fletcher RJ, Battin J, Sisk TD (2004) Ecological responses to habitat edges: mechanisms, models, and variability explained. Annu Rev Ecol Syst 35:491–522
Roman CT, Raposa KB, Adamowicz SC, James-Pirri M-J, Catena JG (2002) Quantifying vegetation and nekton response to tidal restoration of a New England salt marsh. Restor Ecol 10:450–460
Sacco JN, Seneca ED, Wentworth TR (1994) Infaunal community development of artificially established saltmarshes in North Carolina. Estuaries 17:489–500
Sanger DM, Holland AF, Scott GI (1999a) Tidal creek and salt marsh sediments in South Carolina Coastal Estuaries: I. Distribution of trace metals. Arch Environ Contam Toxicol 37:445–457
Sanger DM, Holland AF, Scott GI (1999b) Tidal creek and salt marsh sediments in South Carolina Coastal Estuaries: II. Distribution of organic contaminants. Arch Environ Contam Toxicol 37:458–471
Schindler DE, Johnson BM, MacKay NA, Bouwes N, Kitchell JF (1994) Crab:snail size-structured interactions and salt marsh predation gradients. Oecologia 97:49–61
Schneider KR, Helmuth B (2007) Spatial variability in habitat temperature drives patterns of selection between an invasive and native mussel species. Mar Ecol Prog Ser 339:157–167
Silliman BR, Bertness MD (2002) A trophic cascade regulates salt marsh primary productivity. Proc Natl Acad Sci USA 99:10500–10505
Silliman BR, Bertness MD (2004) Shoreline development drives the invasion of Phragmites australis and the loss of New England salt marsh plant diversity. Conserv Biol 18:1424–1434
Silliman BR, Zieman JC (2001) Top-down control of Spartina alterniflora production by periwinkle grazing in a Virginia salt marsh. Ecology 82:2830–2845
Stiven AE, Kuenzler EJ (1982) The response of two salt marsh mollusks, Littorina irrorata and Geukensia demissa, to field manipulations of density and Spartina litter. Ecol Monogr 49:151–171
Sundareshwar PV, Morris JT, Koepfler EK, Fornwalt B (2003) Phosphorus limitation of coastal ecosystem processes. Science 299:563–565
Talley DM, North EW, Juhl AR, Timothy DA, Conde D, de Brouwer JFC, Brown CA, Campbell LM, Garstecki T, Hall CJ, Meysman FJR, Nemerson DM, Souza Filho PW, Wood RJ (2003) Research challenges at the land-sea interface. Estuar Coast Shelf Sci 58:699–702
Thompson SP, Paerl HW, Go MC (1995) Seasonal patterns of nitrification and denitrification in a natural and a restored salt marsh. Estuaries 18:399–408
Touchette BW (2006) Salt tolerance in a Juncus roemerianus brackish marsh: spatial variation in plant water relations. J Exp Mar Biol Ecol 337:1–12
Travis SE, Proffitt CE, Lowenfeld RC, Mitchell TW (2002) A comparative assessment of genetic diversity among differently-aged populations of Spartina alterniflora on restored versus natural wetlands. Restor Ecol 10:37–42
Tufford DL, Samarghitan CL, McKellar HN, Porter DE, Hussey JR (2003) Impacts of urbanization on nutrient concentrations in small southeastern coastal streams. J Am Water Resour Assoc 39:301–312
Valiela I, Bowen JL (2002) Nitrogen sources to watersheds and estuaries: role of land cover mosaics and losses within watersheds. Environ Pollut 118:239–248
Van Dolah RF, Riekerk GHM, Bergquist DC, Felber J, Chestnut DE, Holland AF (2008) Estuarine habitat quality reflects urbanization at large spatial scales in South Carolina’s coastal zone. Sci Total Environ 390:142–154
Van Wijnen HJ, Bakker JP (1999) Nitrogen and phosphorus limitation in a coastal barrier salt marsh: the implications for vegetation succession. J Ecol 87:265–272
Vernberg FJ, Vernberg WB, Blood E, Fortner A, Fulton M, McKellar H, Michener W, Scott G, Siewicki T, El Figi K (1992) Impact of urbanization on high-salinity estuaries in the southeastern United States. Neth J Sea Res 30:239–248
Vince SW, Valiela I, Teal JM (1981) An experimental study of the structure of herbivorous insect communities in a salt marsh. Ecology 62:1662–1678
Wahl MH, McKellar HN, Williams TM (1997) Patterns of nutrient loading in forested and urbanized coastal streams. J Exp Mar Biol Ecol 213:111–131
Walters K (2009) Integrating structural and functional metrics to assess the early success of salt marsh restoration. In: Pardue GH, Olvera TK (eds) Ecological restoration. Nova Science Publishers, Hauppauge, pp 111–139
White DL, Porter DE, Lewitus AJ (2004) Spatial and temporal analyses of water quality and phytoplankton biomass in an urbanized versus a relatively pristine salt marsh estuary. J Exp Mar Biol Ecol 298:255–273
White DL, Porter DE, Lewitus AJ, Keesee J (2008) Spatial gradient analyses of nutrients and chlorophyll a biomass in an urbanized lagoonal estuary: a comparison between wet and dry years. J Coastal Res 24:649–659
Wiens JA, Crawford CS, Gosz JR (1985) Boundary dynamics: a conceptual framework for studying landscape ecosystems. Oikos 45:421–427
Wigand C, Comeleo R, McKinney R, Thursby G, Chintala M, Charpentier M (2001) Outline of a new approach to evaluate ecological integrity of salt marshes. Human Ecol Risk Assess 7:1541–1554
Wigand C, McKinney RA, Charpentier MA, Chintala MM, Thursby GM (2003) Relationships of nitrogen loadings, residential development, and physical characteristics with plant structure in New England salt marshes. Estuaries 26:1494–1504
Wigand C, McKinney R, Chintala M, Lussier S, Heltshe J (2010) Development of a reference coastal wetland set in Southern New England (USA). Environ Monit Assess 161:583–598
Woerner LS, Hackney CT (1997) Distribution of Juncus roemerianus in North Carolina tidal marshes: the importance of physical and biotic variables. Wetlands 17:284–291
Yarrow MM, Marín VI (2007) Toward conceptual cohesiveness: a historical analysis of the theory and utility of ecological boundaries and transition zones. Ecosystems 10:462–476
Zedler JB (1993) Canopy architecture of natural and planted cordgrass marshes: selecting habitat evaluation criteria. Ecol Appl 3:123–138
Acknowledgments
We thank the numerous CCU student volunteers that were involved in the collection and processing of samples, including A. Bevington, E. Dukes, C. Flick, L. Greenwood, M. Hicks, M. Kirschbaum, A. Mauldin, S. Rider, and L. Waite. Comments of two anonymous reviewers greatly improved the manuscript. This research was funded in part by NSF-MRI Program Grant OCE-116536 awarded to E. Koepfler.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Walters, K., Hutchens, J.J., Koepfler, E.T. et al. Local-scale characteristics of high-marsh communities next to developed and undeveloped shorelines in an ocean-dominated estuary, Murrells Inlet, SC. Aquat. Sci. 72, 309–324 (2010). https://doi.org/10.1007/s00027-010-0137-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00027-010-0137-8