Abstract
The stem cell niche refers to a specific microenvironment where stem cells proliferate and differentiate to produce new specialized cells throughout an organism’s adulthood. Growth factors are crucial signaling molecules that diffuse through the extracellular space, reach the stem cell niche, and ultimately promote stem cell proliferation and differentiation. However, it is not well known how multiple growth factors, often with antagonistic activities, work together in the stem cell niche to select target stem cell populations and determine stem cell fate. There is accumulating evidence suggesting that extracellular matrix (ECM) molecules play an important role in promoting growth factor access and activity in the stem cell niche. In the adult brain neurogenic zone, where neural stem cells (NSCs) reside, there exist specialized ECM structures, which we have named fractones. The processes of NSC allow them to come into contact with fractones and interact with its individual components, which include heparan sulfate proteoglycans (HSPGs) and laminins. We have demonstrated that fractone-associated HSPGs bind growth factors and regulate NSC proliferation in the neurogenic zone. Moreover, emerging results show that fractones are structurally altered in animal models with autism and adult hydrocephalus, as demonstrated by changes in fractone size, quantity, or HSPG content. Interestingly, ECM structures similar to fractones have been found throughout β-amyloid plaques in the brain of patients with Alzheimer’s disease. Pathological fractones may cause imbalances in growth factor activity and impair neurogenesis, leading to inflammation and disorder. Generally speaking, these stem cell niche structures play a potentially vital role in controlling growth factor activity during both health and disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Stem cells proliferate and differentiate into specialized cells and eventually migrate to replace lost cells in all tissues and organs throughout an organism’s adulthood [1]. Differing from initial expectations, it is now known that even the adult heart and brain contain stem cells [2, 3]. However, stem cells are not found everywhere throughout the tissues and organs; rather they are located in niches. The stem cell niche refers to a microenvironment where stem cells reside, and where stem cell fate is determined [4, 5]. The question arises as to what makes the niche a specific nurturing environment for stem cells and their progeny. What are specific molecules in the niches, and how do these molecules interact together to regulate stem cell proliferation, differentiation, and migration?
There is accumulating evidence that stem cells respond to multiple growth factors. Growth factors induce stem cell proliferation, differentiation, and migration [6–12]. However, the mechanisms by which multiple growth factors are distributed (by diffusion) throughout the niche and work together to control stem cell proliferation, differentiation, and migration are still shrouded in mystery.
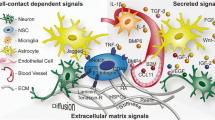
Extracellular matrix (ECM) molecules aggregate into organized structures next to stem cells and may be the key components of the stem cell niche. It has been long recognized that ECM molecules such as heparan sulfate proteoglycans (HSPGs) bind, store, and promote the biological activity of growth factors, which, in turn, determine cell fate and the production of new cells [13–17].
In the most active neurogenic zone of the adult brain, the subependymal layer (SEL) of the lateral ventricle, often referred as subventricular zone (SVZ), we have characterized a local specialized ECM, which coexists with neural stem cells and their immediate progeny (progenitor cells). We have named these structures fractones because of their fractal ultrastructure [18]. In this review, I will introduce fractones, illustrate their structural and ultrastructural organization, their function as binders and activators of growth factors in the adult neurogenic zone [19, 22], and their anatomical interactions with the meninges extending into the brain. Fractones capture growth factors from the brain ventricles, then bind and dispatch these molecules to the neurogenic zone or adjacent neural structures. The alterations of fractones in animal models of neural disorders will be presented and analyzed.
Growth factors in the stem cell niche
Hundreds of growth factors and cytokines have been characterized in the last 50 years. These signaling molecules induce cell proliferation, differentiation, migration, and growth [6–12] but also induce the production and release of other signaling cues, including hormones and neuromediators [23–26]. Growth factors and cytokines are diffusible proteins that can act in a paracrine mode near their site of production but also at a further distance following diffusion through the intercellular space [27]. In addition, growth factors and cytokines can function like hormones and circulate in the blood to target cells at long distance from the production sites. When their activity is unbalanced, growth factors and cytokines cause inflammation and disease [28–30].
Unfortunately, despite the biological importance of these molecules, the biological system involving the activity of growth factors and cytokines—coordinated throughout organs and tissues—has yet to be comprehensively characterized. This is in stark contrast to hormones that soon after their characterization was attached to a branch of biology named endocrinology.
Growth factors require HSPG to bind cognate cell surface receptors: a basic mechanism to regulate cell fate and function
Cell surface receptor binding is the basic principle by which growth factors, cytokines, and chemokines signal cells. However, these signaling molecules also bind HSPGs [13–17] and integrins at the cell surface [31, 32]. While it is well accepted that all these molecules interact to modulate and ultimately transduce the cellular response, it is not clear what defines the specificity of target cells. How do growth factors, cytokines, and chemokines, which diffuse throughout the extracellular space, identify specific cells in tissues and organs?
A large amount of growth factors and cytokines are heparin-binding molecules. This implies that HSPGs located in the ECM facilitate the binding of growth factors/cytokines to their cognate receptors at the surface of target cells [13–17]. Furthermore, integrins cooperate with HSPGs, and directly associate with receptors for growth factors in the plasma membrane to ultimately coordinate signal transduction and the growth factor-induced biological processes [31, 32]. HSPGs have been shown to store growth factors and provide a stable environment that prevents enzymatic degradation [33]. Therefore, the ECM, via HSPGs, plays a critical role in the storage and regulation of growth factor activity.
There is a biological potential that has hitherto been unexplored. The ECM is essentially “anchored” in between cells as basement membranes and interstitial matrix [34], whereas growth factors and cytokines are circulating cues. The ECM may serve as a system that turns the diffusible factors into bound molecules, serving as reservoirs of non-active molecules, while local activators regulate the biological activity of growth factors at the surface of target cells.
Supporting this hypothesis, stem cells, which have a key role in the maintenance and regeneration of tissues, always proliferate and differentiate on basement membranes [35–38] or on another special form of ECM, which colleagues and I have characterized as fractones [18, 39, 40].
Fractones differ from basement membranes. They are localized within either the parenchyma [18, 39] or connective tissues (extraparenchyma) [21, 41, 42], while basement membranes are located at the interface of the connective tissue with parenchyma [43] and through the blood vessel walls [9, 18, 44]. Fractones also differ from basement membranes morphologically as they are organized as puncta as viewed by light microscopy following immunolabeling for their ECM components [18, 21, 45]. This is in contrast to the sheath organization of basement membranes.
Characterization of the ECM fractones in the neurogenic zone of the adult brain
In the adult brain, a restricted zone contains neural stem cells, which proliferate and differentiate to continuously produce new neurons and glia throughout adulthood [3, 46–50]. The adult neurogenic zone has a complex anatomy [48] but is primarily located in the SEL (SVZ) or ventricular-SVZ (V-SVZ to name the whole ventricle wall) of the lateral ventricle [3, 46, 48] at the surface of the caudate nucleus. In this location, there are specialized ECM structures that we have named fractones [18]. Fractones can be visualized as series of puncta by light microscopy after immunolabeling for laminin or HSPG (Fig. 1) [18, 21, 39]. Figure 1 shows the most-active neurogenic zone along the SEL at the surface of the caudate nucleus. It is clear from this confocal image that proliferating neural stem and progenitor cells, visualized using bromoeoxyuridine (BrdU) immunolabeling, are located next to fractones, visualized using laminin immunolabeling. Therefore, fractones form a physical niche for neurogenesis in the most-active neurogenic zone of the adult brain. In further sections of this review, I will illustrate how fractones serve as a functional stem cell niche for adult neurogenesis, promote growth factor activity, and nurture stem cells and their immediate progeny to ultimately produce new neural cells.
Fractone niche for adult neurogenesis (adult mouse). Confocal micrograph showing proliferating stem and progenitor cells (bromodeoxyuridine immunofluorescence labeling, red) associated with fractones (laminin-immunoreactive puncta, green) in the most-active neurogenic zone of the adult brain in the SEL of the caudate nucleus (Ca). Vascular basement membranes are immunoreactive for laminin (green lines). Most proliferating cells are associated with fractones. Some cells proliferate next to blood vessels (white arrow). Inset image location and plane of sectioning, tangential to an anterior recess of the lateral ventricle (red rectangle). Immunofluorescence histochemistry was performed as indicated in [92]. Scale bar 50 µm
Fractone ultrastructure
The central nervous system comprises a complex network of cavities, namely, ventricles in the brain, and the spinal canal in the spinal cord. Specialized ECM structures are found throughout the walls bordering the ventricles and spinal canal, beneath the first layer of cells (the ependyma). We have named these ECM structures fractones because of their fractal ultrastructure [18, 39]. Fractal is a mathematical concept describing the structural properties of objects originally coined by Benoit Mandelbrot in 1975 [51]. Fractal objects can be visualized at different levels of magnification, as a repetition of “branching” patterns. As fractality of an object increases, contact surface area increases, while the volume/area ratio decreases. Fractones display a fractal ultrastructure, as shown in transmission electron microscopy [18, 39], although they merely appear as puncta using light microscopy after immunolabeling for their ECM components.
In adult vertebrates, including humans, fractones are found in series, visualized as individual structures all along the walls of the ventricle and spinal canal [18, 21, 39]. Figure 2 shows the utrastructure of individual fractones in the walls of the lateral ventricle. Fractones are electron-dense by transmission electron microscopy, without any immunolabeling (Fig. 2c, d, arrows). This is a common characteristic of fractones and basement membranes. However, fractones are distinct from basement membranes in many respects. Basement membranes are organized as sheets between the connective tissue and the parenchyma (“primarily working cells”) of an organ or tissue. In contrast, fractones are found within the parenchyma of tissues. Moreover, fractones are not derived from blood vessels and their basement membranes, but are present in the parenchyma of tissues long before the development of blood vessels, as attested by their presence in the neuroepithelium during brain morphogenesis [42]. Basement membranes come second in terms of developmental chronology (personal communication).
Fractone ultrastructure in the SEL of the lateral ventricle. a Left coronal sectioning 0.5-mm anterior to bregma; middle schematic coronal section showing the location (red rectangle) of schematics (b, e) and ultramicrographs (c, d); right semi-thin section showing ultramicrograph locations (c, d). cc the corpus callosum, Ca caudate nucleus, Ep ependymal, LV lateral ventricle. b Schematic of the ventricle wall beneath the corpus callosum; Ep (blue) line the ventricle; macrophages and astrocyte endfeet (gray) contact fractones (green). Note the respective location of neurons (axons and dendrites), astrocytes, and fractones. Arrows interstitial clefts allow cerebrospinal fluid to access fractones. c Ultramicrograph showing a fractone with its labyrinthine organization (arrow) beneath the cc; a fractone is typically electron-dense, but less than mitochondria (arrowhead); astrocyte processes (double arrow) contact fractones, axons (double arrowhead), and dendrites (asterisk). d Fractone (arrow) in the most active neurogenic zone of the adult brain, the SEL of the lateral ventricle. Neuroblasts (arrowheads) display electron-lucent bands (double arrowhead) and endfoot contacting fractones. e Schematic showing stem cells and fractones in the neurogenic zone. Arrows interstitial clefts. Scale bars 2 µm
Figure 2c shows the ultrastructure of an individual fractone, whose size in the adult brain is usually between 1 and 6 µm, and found beneath the corpus callosum, in the SEL of the lateral ventricle. In this zone, each fractone is contacted by multiple (>100) astrocytic endfeet (double arrow) that arise from the corpus callosum. These astrocytes tightly surround bundles of axons and dendrites (double arrowhead and asterisk). The organization reflected by this ultrastructure is shown in Fig. 2b, and the location of the ultramicrograph is shown in Fig. 2a. Interestingly, at least in this zone, neurons do not directly contact fractones; astrocytes always interface in between them. This is also the case for basement membranes, which reflects the general organization of tissues of the central nervous system [9, 44] (i.e., astrocytes in between neurons and the connective/vascular tissues, except in the neural lobe of the pituitary gland where astrocytes can physically retract and allow the passage of neurohormones to the vasculature).
Figure 2d shows the ultrastructural organization of fractones and their contacts with multiple cells of the neurogenic zone in the SEL covering the caudate nucleus. The cells present in this zone are neural stem cells and their immediate progeny, and neuroblasts and glioblasts. Several neuroblasts with the typical electron-lucent bands described by Doetsch et al. [29] appear in the ultramicrograph (arrowheads). Multiple cell processes originating from these cells contact the fractone (arrow). Additional descriptions of contacts between cells and fractones in the SEL neurogenic zone during adulthood are reported in [18]. These contacts are shown in Fig. 2e. Macrophages, which display characteristics similar to those of Kolmer cells, are associated with each fractone after originating from the choroid plexus [52]. These cells are putative producers of fractone-forming ECM molecules, as indicated by rich vesicular material at the contact areas of these macrophages and fractones [18]. This is also supported by the fact that macrophages produce HSPGs [53] and possess the components (enzymes and lysosomes) to both produce and degrade the ECM.
The fractal ultrastructure of fractones allows for the direct contact of an individual fractone with multiple cells, including ependymocytes, microglia, neural stem cells, neural progenitor cells, and astrocytes. Small terminals (endfoot processes) of the cells come into contact with fractones, while the cell body itself is located relatively far away from fractones. This is valid for both the neurogenic zone (Fig. 2d) and the zones covering white matter tracts (Fig. 2c). Therefore, the three-dimensional (fractal) organization of a single fractone allows for multiple contact points with cells of the SEL and of the neural/white matter tracts structures located nearby [18]. This is the exact definition of a fractal object: a large surface area allowing for multiple contact points, while occupying a small volume.
Another interesting feature of the ventricle wall organization is the presence of interstitial clefts, originally described by Brightman [54]. Interstitial clefts are narrow channels only visible through the ultrastructure, in between ependymocytes. Interstitial clefts allow the passage of cerebrospinal fluid (CSF) and CSF-borne signaling molecules between ependymocytes. These clefts terminate at the location of fractones.
Ependymocytes form a pseudoepithelium of cells attached to each other via tight junctions that serve as a barrier to the cerebrospinal fluid, with the exception of interstitial clefts, where the specific junctions allow the passage of molecules. Therefore, signaling molecules (e.g., growth factors and cytokines) produced in the choroid plexus, in the meninges and in neural structures can enter the ventricle wall, then reach and bind fractones. The growth factors that do not bind fractones, for example those that are not heparin-binding molecules such as epidermal growth factor (EGF) cross the SEL and diffuse through neural structures. Interstitial clefts, their junctions and their relationship to fractones are shown in [22]. Figure 2b shows a graphical representation of interstitial clefts leading to fractones in the apical surface of ependymocytes (arrows).
Fractone composition and arrangement as series along the ventricle wall
Chemically, fractones contain collagen-IV, laminin isoforms, nidogen, and heparan sulfate proteoglycans (HSPGs), such as collagen XVIII and perlecan [18, 21, 45]. This is similar to basement membranes. However, fractones are highly immunoreactive for N-sulfated-HSPGs (more so than basement membranes), a feature that may increase fractone capability of binding to signaling molecules, such as heparin-binding growth factors and cytokines. Figure 3 shows that fractones are distributed in series along the walls of the lateral ventricle. Interestingly, fractones located in the adult neurogenic niche of the SEL at the surface of the caudate nucleus are more immunoreactive for NS-HSPG (Fig. 3a, b, arrow) than those located at the surface of the fimbria hippocampus (Fig. 3b, arrowhead).
Fractones are organized in series along the ventricle walls. a Immunolabeling for N-sulfated heparan sulfate proteoglycans (NS-HSPGs, green) and laminin isoforms (red) in a brain section (adult mouse). Fractones (puncta) are arranged in series along the SEL (arrow). Fractones are larger in the SEL covering the caudate nucleus (Ca) (arrow) and are smaller in the SEL of the fimbria hippocampus (FH) (arrowhead). Immunoreactivity for NS-HSPGs is high in the meninge located beneath the FH (double arrow) and in the choroid plexus (double arrowhead) of the lateral ventricle (LV). Inset localization of the image a. b Magnification of the area indicated by an arrow in a showing that fractones are more immunoreactive for NS-HSPGs (green) in the SEL covering the Ca than in the SEL covering the FH. Inset laminin labeling (only the red color is displayed) is similar in the SVZ covering the FH (arrow) and Ca (arrowhead). Scale bars 50 µm
Fractones bind to heparin-binding growth factors
The majority of growth factors, cytokines, and chemokines are heparin-binding molecules. This implies that the signaling molecules require (or are facilitated by) heparan sulfates to recognize their cognate receptors. For example, the binding of a powerful neurogenic growth factor such as fibroblast growth factor-2 (FGF-2) to its FGFR receptors is facilitated by heparan sulfates present in HSPGs [16, 17, 55]. Thus, the HSPGs play a crucial role in intervening with ligand-receptor recognition and ultimately in the biological activity of the growth factors in the targeted cell (for example, stem cell proliferation and differentiation) by intervening in the ligand receptor recognition. This provides insight into the role of the ECM in the regulation of heparin-binding growth factor/cytokine activity. Sonic hedgehog (SHH), bone morphogenetic proteins (BMP), FGF isoforms, transforming growth factors (TGFs), amphiregulin, interferons (IFN), interleukins (IL), insulin-like growth factors (IGFs), tumor necrosis factor (TNF), heparin-binding epidermal growth factor (HB-EGF), and Leukemia inhibiting factor (LIF), only to list a few, are all heparin-binding molecules [13–17, 55–62], and these are potentially regulated by the HSPGs in the ECM. However, several growth factors, such as platelet-derived growth factor (PDGF), EGF, and brain-derived neurotrophic factor (BDNF), regulate cytogenesis in the adult brain and are not heparin-binding growth factors.
Consequently, we hypothesize that fractones and their HSPG capture heparin-binding growth factors circulating in the ventricle cavities. The choroid plexus, a meningeal derivative, is likely to produce a large amount of heparin binding growth factors, including BMP and FGF [63, 64], both active molecules in neurogenesis.
We have previously demonstrated that FGF-2 [20, 45], BMP-7 [19], and BMP-4 [22] bind to fractone-associated HSPGs in the SEL of the lateral ventricle, covering the caudate nucleus, quantitatively the most germinative zone in adulthood [40]. FGF-2 is a potent neurogenic factor, stimulating both proliferation and differentiation of neural stem cells. In contrast, BMP-4 and BMP-7 inhibit neural stem cell proliferation in the SEL neurogenic zone of the adult brain.
Figure 4a shows the binding of FGF-2 in the neurogenic SEL. Growth factor binding was assessed in vivo, using biotinylated-growth factors injected in the lateral ventricle or the rostral migratory stream, and the binding was visualized postmortem on frozen brain sections with streptavidin Texas red and imaging by immunofluorescence microscopy. The fact that FGF-2 binds to fractones in the neurogenic zone of the adult brain was also confirmed in vitro, using direct incubation of biotinylated-growth factors then visualization after incubation with streptavidin Texas red in brain tissue sections fixed with paraformaldehyde [21]. This new technique allowed for the detection of growth factor binding sites in situ by epifluorescence or confocal microscopy [21]. We have used the same technique to demonstrate that BMP-4 and BMP-7 bind to fractones in the SEL of the lateral ventricle [19, 22]. Interestingly, the binding of these growth factors is not limited to the neurogenic zone. Figure 4c shows the binding of BMP-7 to fractones and FGF-2 in the walls of the third ventricle, a non-neurogenic zone in adulthood.
Fractones bind growth factors in the adult neurogenic zone. a Biotinylated-FGF-2 binds to fractones (arrow) in the adult SEL after intracerebroventricular (ICV) injection. The binding of biotinylated-FGF-2 was visualized postmortem on frozen brain sections, using streptavidin Texas red and confocal microscopy. b N-sulfated-HSPG immunolabeling on the same section showing the location of fractones (arrow). c Biotinylated-morphogenetic protein-7 (BMP-7) binds to fractones (red puncta, arrow) in the SEL of the third ventricle (3V) after ICV injection. d Immunolabeling for N-sulfated-HSPG showing fractones on the same section. e Location of confocal images. f Schematic of fractones, growth factors, and growth factor receptors (FGFR) in the adult neurogenic zone. FGF-2 enters the neurogenic zone via interstitial clefts (red arrows), reaches fractones, and binds to fractone-associated HSPGs. g HSPG facilitates the binding of FGF-2 to FGFR, which transduces FGF-2 signaling and induces cell division. Scale bars 50 µm
For all growth factors investigated (FGF-2, BMP-4, and BMP-7), we have shown that they bind to fractones via HSPG [19–22]. This was demonstrated by performing in vivo experiments in which the growth factors were simultaneously injected with heparitinase-1, which cut the heparan sulfate chains of HSPG. In all cases, postmortem immunoreactivity for NS-HSPG was absent, showing the effectiveness of the enzyme’s abilities. Heparitinase-1 also prevented any form of binding, demonstrating that the growth factors bind with fractones via heparan sulfate chains [25–27, 37].
Growth factors must bind to fractone-associated HSPG to regulate cell proliferation in the SVZ neurogenic zone
Another hypothesis is that heparin-binding growth factors must bind to fractone-associated heparan to regulate cell proliferation in the neurogenic zone of the adult brain. The germinative (neurogenic) index was measured by BrdU immunoreactivity on serial frozen brain sections containing the SVZ after a short pulse of BrdU. Intracerebroventricular injection of FGF-2 increased cell proliferation, while injection of BMP-7 and BMP-4 decreased cell proliferation in the neurogenic zone [19, 20, 22]. When heparitinase-1 was coinjected with FGF-2, the mitotic effect of FGF-2 was drastically reduced [20]. Similarly, when heparitinase-1 was coinjected with BMP-7, the inhibitory effect of BMP-7 on cell proliferation in the neurogenic zone was greatly reduced [19]. This indicates that the binding of FGF-2 and BMP-7 to heparan sulfates is required to mediate the biological effect of these growth factors on cell proliferation. All these results indicate that heparan sulfates, especially those located in fractones in contact with stem cells, intervene in the mechanisms regulating neurogenesis.
The potential mechanisms by which fractones regulate adult neurogenesis are schematically shown in Fig. 4f, g. Heparin-binding growth factors circulate in the cerebrospinal fluid of the ventricles, diffuse through the interstitial clefts, and then bind to fractone-associated HSPG. Fractone-associated HSPGs store growth factors and, eventually, present them to their cognate receptors at the surface of the processes of neural stem and progenitor cells.
Connections between fractones and meninges: a potential system of communication
As demonstrated in our previous studies, a restricted set of blood vessels located near the neurogenic zone of the lateral ventricle, the meninges and choroid plexus bind with heparin-binding growth factors. Meninges, choroid plexus, and this restricted set of blood vessels are immunoreactive for N-sulfated-HSPG (Fig. 5b). We have previously demonstrated that an N-sulfated HSPG immunoreactive fibroblast–macrophage network extends through the meninges both near the surface of the brain and between major neural structures within the brain (Fig. 5a), before reaching both the choroid plexus and the SEL of the brain ventricles. Fractones form a continuum with this organization (Fig. 5) and [48]. At this stage, we can only speculate about the function of such an organization.
Fractones and meninges. a Top left Schematic of a brain section (bregma −2.7 mm, adult mouse) illustrating meninges and fractone (red puncta) anatomical relationships. Arrowhead meninges at the brain surface. Arrow meninges within the brain. 3V third ventricle, LVpr lateral ventricle, posterior; D3V third ventricle, dorsal. Bottom left Schematic at bregma −1.2 mm. Fractones (double arrow) form a continuum with meninges (double arrowhead). The inset indicates the location of image b. Right Organization of the LV wall, showing fractones in the neurogenic zone at the caudate nucleus (Ca) surface and near the white matter. BV blood vessel, CC corpus callosum, CP choroid plexus, FH fimbria hippocampus, Men meninges. b Continuum of N-sulfated HSPG immunolabeling showing fractones (arrows), meninges (double arrow) and CP. c Low magnification field showing the location of image b. Hip hippocampus
I hypothesize that, in adults, the meninges and its extensions within the brain (including the adventitia of the vascular wall and the choroid plexi) in conjunction with fractones throughout the ventricle walls form a physiological system that regulates the production, bioavailability, and biological activity of growth factors and cytokines, via specific HSPGs that are highly N-sulfated. Meninges and the choroid plexi produce highly diverse heparin-binding growth factors, including FGFs and BMPs [63–65]. Therefore, these structures may contribute to the expression and regulation of growth factors and cytokines in the central nervous system. The proximity to this system would provide an explanation for the location of neurogenic zones in the adult brain.
Interestingly, meninges may be a source of neural stem cells [66], comprise lymphatics [67], and produce growth factors, cytokines, chemokines, and an abundant ECM material. Meninges extend deeply into the brain (Fig. 5) [9, 40, 44, 48] potentially carrying signaling information to the brain [9]. Meninges, macrophages and an abundant brain ECM are present in all animals, including invertebrates [68]. In addition, meninges appear to control brain development [69] through multiple heparin-binding growth factors.
Alterations of fractones in disease
Neural tube defects, neurodevelopmental disorders, psychiatric disorders, and neurodegenerative diseases are associated with imbalanced expression of heparin-binding growth factors, including the inflammatory growth factors TNF and TGF, and the stem cell regulatory growth factors, such as FGF, BMP, and SHH. It is, therefore, possible that fractones, as potential captors of these signaling molecules, are implicated in brain disorders and malformations. To examine this possibility, we have investigated potential structural changes of fractones and meninges during disorders, in animal models with certain disorders [70, 71]. Figure 6 shows the structural alterations of fractones in the BTBR t+tf/J mouse, an animal model that displays agenesis of the corpus callosum [72]. This mouse is also known to have autism, because it displays the major symptoms associated with that disorder [73]. Fractones in the adult BTBR t+tf/J mouse are drastically reduced in size and number (compare fractones in images a and b). These fractone morphological alterations were observed throughout the lateral and third ventricle, but were more pronounced in the neurogenic zone at the surface of the caudate nucleus [70, 71]. The reduction in size and number of fractones is correlated with the NS-HSPG content. Therefore, alterations in fractones in the neurogenic niche are correlated with imbalanced neurogenesis.
Fractone alterations in the mouse model of autism (BTBR T+tf/J). a Control brain (B6 mouse) immunolabeled for laminin (red) and N-sulfated HSPGs (green). Fractones (puncta, arrow) are large and numerous in the neurogenic zone. CC corpus callosum, Ca caudate nucleus, CP choroid plexus, LV lateral ventricle. Inset location of images a and b (red rectangles); ec external capsule, BP bundles of Probst. b Fractones are reduced in size and numbers in the BTBR T+tf/J mouse. The loss of fractones is drastic in SEL facing the Ca (arrow) (neurogenic zone). Fractones are smaller, although to a lesser extent, along the BP. Scale bars 50 µm
Moreover, drastic malformations of the meninges were observed in this animal. The inter-hemispheric fissure (falx cerebri), a meninge joining the brain hemispheres, was elongated in BTBR t+tf/J mice [70]. These meningeal and fractone alterations may reflect neurogenic malformations that occur during development and persist into adulthood.
Interestingly, fractones were larger in size, as illustrated by their ultrastructure, in an animal model of adult hydrocephalus [74]. Adult hydrocephalus, which is clinically induced by neurocystercosis after a tapeworm infection, was induced by mechanically blocking the CSF pathway at the level of the fourth ventricle. This induces the enlargement of the ventricles [74]. Agenesis of the corpus callosum and adult hydrocephalus are drastically different disorders. Alterations in fractones may cause improper binding of growth factors, imbalance the biological activity of growth factors, disregulate neurogenesis, lead to inflammation, and cause several brain diseases.
Interestingly, it has been shown that laminin-immunoreactive puncta, similar to fractones, are associated with β-amyloid plaques in the cortex of patients with Alzheimer’s disease [75]. β-Amyloid, the hallmark of Alzheimer’s disease, is a heparin-binding growth factor that is highly expressed in the SEL of brain ventricles [76]. β-amyloid also regulates adult neurogenesis. HSPGs directly bind to β-amyloid associated with Alzheimer’s disease [77, 78]. A second biological marker of Alzheimer’s-disease, Tau tangles, is also related to HSPGs. It has been demonstrated that tau requires HSPGs to enter neurons and form the neuronal tau tangles typically associated with Alzheimer’s [79]. Together, these studies strongly support the view that fractones are implicated in the onset and progression of Alzheimer’s disease.
Moreover, inducible knock out of the enzyme EXT, responsible for the extension of glucosamines in the heparan sulfate chains, results in mice that display autistic behavior [80]. This illustrates another piece of evidence suggesting that HSPGs are involved in birth defects and in neurodevelopmental and neurodegenerative disorders. Holoprosencephaly type 3 and cyclopia, which are caused by SHH signaling deficiencies, may also implicate HSPGs because SHH is a heparin-binding growth factor.
A fractone system to control growth factor activity and thus plasticity throughout the brain
Signaling molecules such as hormones belong to a system of regulations that control homeostasis named the endocrine system. Since the discovery of nerve growth factor (also a heparin-binding molecule), many more growth factors and cytokines have been discovered. Currently, it is known that the level of growth factors present is distorted in most diseases and disorders, and that the inflammation process is involved. However, there is no system that describes the coordination of the actions of growth factors/cytokines in the general context of regulation (i.e., homeostasis, response to injury, healing, inflammation, and immune functions) nor a scheme that analyzes these phenomena in the context of diseases.
I propose that fractones in the adult brain regulate growth factor bioavailability and activation to control neurogenesis in the SEL of the lateral ventricle, and brain plasticity in non-neurogenic zones. Cytogenesis in the adult brain leads to the production of neurons oligodendrocytes, astrocytes, and the migration of these new cells toward zones that require cell replacement. The fractone system is located in an anatomically ideal position to control this aspect, and our results further support this idea, showing that neurogenic growth factors are processed by fractones. Therefore, fractones are a good candidate to explain what physically forms the stem cell niche in the adult brain.
Other possible functions of fractones, also expressed in the third [39] and fourth ventricles, include neuroendocrine regulation, synthesis and release of neurohormones and neuromediators, growth of neurons (neurotrophins are heparin-binding molecules), and chemoattraction of cells involved in immune functions. Chemokines, such as CXC and semaphorins are heparin-binding molecules [81]. Interestingly, fractone size increases with age [82]. However, the significance of this characteristic is unknown.
The particular organization of fractones in the SEL covering neural white matter tracts and neural structures demonstrates that multiple astrocyte endfeet reach fractones by their endfeet (Fig. 2). Astrocytes may “analyze” the information resulting from ECM/growth factor interactions at their contact with fractones, and ultimately relay the information to neurons in deep neural structures. Therefore, it is possible that astroglia act as an interface between fractones and neurons, modulating the function of neurons [44]. Moreover, oligodendrogenesis also occurs in the SEL at this location (sub-callosum zone) [50, 83]. The possibility that fractones, which are highly immunoreactive for multiple laminin isoforms [21], are implicated in oligodendrogenesis is supported by the fact that mice lacking α2 laminin show a reduction in the number of oligodendrocyte precursors [84]. The presence of HSPGs in the area [48] and the fact that perlecan is concentrated in fractones [21] and expressed by progenitor and mature oligodendrocytes suggest that fractones may regulate oligodendrogenesis [85, 86].
Mechanisms by which fractones may activate growth factors and control neurogenesis
The mechanisms by which growth factors bind to fractones and work together to determine which stem cells will proliferate and differentiate in the neurogenic zone are unknown. Also, the mechanism by which HSPG promotes growth factor action is not clearly understood. It has been proposed that heparan sulfate chains of HSPG cause a conformational change in growth factors, which then promotes binding to its cell-surface receptor (Fig. 4g). Another model suggests that HSPG, growth factors, and their high-affinity cell surface receptors bind simultaneously, acting as a ternary complex that triggers intracellular signaling and growth factor biological activity. The question arises as to how HSPGs specifically bind different growth factors within an individual fractone. We have found that BMP-7, BMP-4, and FGF-2 bind a vast majority of fractones in the adult neurogenic zone [19, 20, 22]. BMP-7 and BMP-4 inhibit neurogenesis, while FGF-2 stimulates the production of neurons.
The specific heparan sulfate motifs that are contained in fractone-associated HSPGs and that are responsible for binding specific growth factors are still unknown [40]. Supporting previous findings that N-sulfation intervenes in the binding of growth factors to their cell-surface receptors, we have found that growth factor binding occurs in highly N-sulfated HSPG immunolabeled fractones in the neurogenic zone [19–22, 42]. However, it has been shown that 2-O-sulfation and 6-O sulfation correlates with different binding affinities for growth factors [87]. The multiple enzymes responsible for glycosylation of HSPGs operate in the Golgi apparatus, but are also secreted to further operate in the extracellular space. Moreover, the different core proteins of fractone-associated HSPG include perlecan and collagen-XVIII [21, 45], which may further determine specificity for growth factor binding. It is still unknown which cells produce fractones. However, we have demonstrated that macrophages are always associated with fractones [18]. It is known that macrophages and microglia produce and degrade HSPGs and other ECM glycoprotein molecules [88–90]. Knowledge of the mechanisms underlying the production of fractones, their turnover, and their function is just emerging and merits further investigation.
Conclusion
In conclusion, there is increasing evidence to support the view that fractones in the adult brain are responsible for the binding and activation of growth factors, which arise from the brain ventricles. We have demonstrated that fractones form a niche for neural stem cells and neurogenesis in adulthood. However, the distribution of fractones throughout the ventricle system, in zones that are not neurogenic in adulthood, and the multiple biological activities of growth factors and cytokines suggest more functional possibilities in the regulation of brain function and plasticity, in both health and disease. Multiple experimental approaches, using transgenic animals, biochemistry, and glycomics (to identify heparan sulfates specific for growth factors), and ex vivo explants of the neurogenic zone [91] can be used to study the role of fractones, growth factors, and HSPGs in neurogenesis, and the physiology and pathology of the developing and adult brain in general.
References
Avashi S, Srivastava RN, Singh A, Srivastava M (2008) Stem cell: past, present and future—a review article. Intern J Med Update 3(1):22–30
Messina E, De Angelise L, Frati G, Morrone S, Chimenti S, Fiordaliso F, Salio M, Battaglia M, Latronico MVG, Coletta M, Vivarelli E, Frati L, Cossu G, Giacomello A (2004) Isolation and expansion of adult cardiac stem cells from human and murine heart. Circ Res 95:911–921
Doetsch J, Garcia-Verdugo JM, Alvarez-Buylla (1997) Cellular composition and three-dimensional organization of the subventricular germinal zone in the adult mammalian brain. J Neurosci 17:5046–5061
Ming GL, Song H (2001) Adult neurogenesis in the mammalian brain: significant answers and significant questions. Neuron 70(687):702
Rojas Rios P, Gonzalez-Reyes A (2014) The plasticity of stem cell niches: general properties behind homeostasis and repair. Stem Cells 32:852–859
Bragdon B, Moseychuk O, Saldanha S, King D, Julian J, Nohe A (2011) Bone morphogenetic proteins: a critical review. Cell Signal 23:609–620
Burgess AW (2015) Growth factors and cytokines. Rev Cell Biol Mol Med 1:104–126
Levi-Montalcini R, Angeletti PU (1964) Hormonal control of the NGF content in the submaxillary glands of mice. Int Ser Monogr Oral Biol 3:129–141
Mercier F, Hatton GI (2004) Meninges and perivasculature as mediators of CNS plasticity. In: Non-neuronal cells in the nervous system: function and dysfunction. Adv Mol Cell Biol 31:215–253
Nakagawa T, Schwartz JP (2004) Expression of neurotrophic factors and cytokines and their receptors on astrocytes in vivo. In: Non-neuronal cells in the nervous system: function and dysfunction. Adv Mol Cell Biol 31:561–273
Ornitz DM, Itoh M (2015) The fibroblast growth factor signaling pathway. Rev Dev Biol 4:215–266
Wachs FP, Winner B, Couillard-Despres S, Shiller T, Aigner R, Winkler J, Bogdahn U, Aigner L (2006) Transforming growth factor-beta1 is a negative modulator of adult neurogenesis. J Neuropathol Exp Neurol 65:358–370
Brickman YG, Ford MD, Small DH, Bartlett PF, Nurcombe V (1995) Heparan sulfates mediate the binding of basic fibroblast growth factor to a specific receptor on neural precursor cells. J Biol Chem 270:24941–24948
Gordon MY, Riley GP, Watt SM, Greaves MS (1987) Compartmentalization of a hemopoietic growth factor (GM-CSF) by glycosaminoglycans in the bone marrow microenvironment. Nature 326:403–405
Roberts R, Gallagher J, Spooncer E, Alien TD, Bloomfield F, Dexter TM (1988) Heparan-sulfate bound growth factors: a mechanism for stromal cell-mediated hematopoesis. Nature 332:376–378
Rapraeger AC, Guimond S, Krufka A, Olwin BB (1994) Regulation by heparin sulfate in fibroblast growth factor signaling. Methods Enzymol 245:219–240
Yayon A, Klagsbrun M, Esko JD, Leder P, Ornitz DM (1991) Cell surface, heparin-like molecules are required for binding of fibroblast growth factor to its high affinity receptor. Cell 64:841–848
Mercier F, Kitasako JT, Hatton GI (2002) Anatomy of the brain neurogenic zones revisited: fractones and the fibroblast/macrophage network. J Comp Neurol 451:170–188
Douet V, Arikawa-Hirasawa E, Mercier F (2012) Fractone-heparan sulfates mediate BMP-7-inhibition of cell proliferation in the adult subventricular zone. Neurosci Lett 528:120–125
Douet V, Arikawa-Hirasawa E, Mercier F (2013) Fractone-heparan sulphates mediate FGF-2 stimulation of cell proliferation in the adult subventricular zone. Cell Prolif 46:137–145
Kerever A, Schnack J, Vellinga D, Ichikawa N, Moon C, Arikawa-Hirasawa E, Efird JT, Mercier F (2007) Novel extracellular matrix structures in the neural stem cell niche capture the neurogenic factor FGF-2 from the extracellular milieu. Stem Cells 25:2146–2157
Mercier F, Douet V (2014) Bone morphogenetic protein-4 inhibits adult neurogenesis and is regulated by fractone-heparan sulfates in the subventricular zone. J Chem Neuroanat 57–58:54–61
Clemmons DR (2004) The relative roles of growth hormone and IGF-1 in controlling insulin sensitivity. J Clin Invest 113:25–27
Grau V, Wilker S, Lips KS, Hartman P, Rose F, Padberg W, Fehrenbach H, Wessler I, Kummer W (2007) Administration of keratinocyte growth factor down regulates the pulmonary capacity of acetylcholine production. Int J Biochem Cell Biol 39:1955–1963
Turnbull AV, Rivier CL (1999) Regulation of the hypothalamic-pituitary-adrenal axis by cytokines actions and mechanisms of actions. Physiol Rev 79:1–71
Yajima Y, Saito T (1984) The effect of epidermal growth factor on cell proliferation and prolactin production by GH3 rat pituitary cells. J Cell Physiol 120:249–256
Thorne RG, Hrabetova S, Nicholson C (2004) Diffusion of epidermal growth factor in the rat brain extracellular space measured by integrative optical imaging. J Neurophysiol 92:3471–3481
Connor B, Dragunow M (1998) The role of neuronal growth factors in neurodegenerative disorders of the human brain. Brain Res Rev 27:1–39
Steed DL (1997) The role of growth factors in wound healing. Surg Clinics 77:575–586
Wahl SM, Wong H, McCartney-Francis (1989) Role of growth factors in inflammation and repair. J Cell Biochem 40:193–199
Kim SH, Turnbull J, Guimond S (2011) Extracellular matrix and cell signaling: the dynamic cooperation of integrin, proteoglycan and growth factor receptor. J Endocrinol 209:139–151
Eliceri BP (2001) Integrin and growth factor receptor crosstalk. Circ Res 89:1104–1110
Coltrini D, Rusnati M, Zoppetti G, Oreste P, Isacchi A, Caccia P, Bergonzoni L, Presta M (1993) Biochemical bases for interactions of human basic fibroblast growth factor with glycosaminoglycans. New insights from trypsin digestion studies. Eur J Biochem 214:51–58
Farhquar MG (1991) The glomerular basement membrane. In: Hay ED (ed) Cell biology of the extracellular matrix. Plenum Press, New York, pp 365–418
Castro-Munozledo F (2013) Review: corneal epithelial stem cells, their niche and wound healing. Mol Vis 19:1600–1613
Fujiwara H, Ferreira M, Donati G, Marciano DK, Linton JM, Sato Y, Sekiguchi K, Reichardt F, Watt FM (2011) The basement membrane of hair follicle stem cells is a muscle cell niche. Cell 144:577–589
Kazanis I, ffrench-Constant C (2011) Extracellular matrix and the neural stem cell niche. Dev Neurobiol 71:1006–1017
Mackay-Sim A (2010) Stem cells and their niche in the adult olfactory mucosa. Arch Ital Biol 148:47–58
Mercier F, Kitasako JT, Hatton GI (2003) Fractones and other basal laminae in the hypothalamus. J Comp Neurol 455:324–340
Mercier F, Schnack J, Saint George Chaumet M (2011) Fractones: home and conductors of the neural stem cell niche. In: Seki T et al (eds) Neurogenesis in the adult brain, vol 1. Springer, Japan, pp 109–136
Bifari F, Beto N, Pino A, Kusalo M, Malpeli G, Di Chio M, Bersan E, Amato E, Scarpa A, Kamprepra M, Fumagalli G, Decimo I (2015) Meninges harbor cell-expressing neural precursor markers during development and adulthood. Front Cell Neurosci 9:383
Chyba M, Mercier F, Rader J, Douet V, Arikawa-Hirasawa E, Chow Kwon Y, Kodama R (2011) Dynamic mathematical modeling of cell-fractone interactions. J Math Ind 3:79–88
Mercier F, Mambie S, Hatton GI (2006) Brain macrophages: enigmas and conundrums. In: Dermietzel et al (eds) Blood-brain barriers—from ontogeny to artificial barriers. Wiley-VCH, Weinheim, pp 129–165
Mercier F (2004) Astroglia as a modulation interface between meninges and neurons. In: Hatton GI, Parpura V (eds) Glial/neuronal signaling, chapter 5. Kluwer Publishers, Amsterdam, pp 125–162
Kerever A, Mercier F, Nonaka R, de Vega S, Oda Y, Zalc B, Okada Y, Hottari N, Yamada Y, Arikawa-Hirasawa E (2014) Perlecan is required for FGF-2 signaling in the neural stem cell niche. Stem Cell Res 12:492–505
Das GD, Altman J (1970) Postnatal neurogenesis in the caudate nucleus and nucleus accumbens septi in the rat. Brain Res 21:122–127
Lois C, Alvarez-Buylla A (1994) Long distance neuronal migration in the adult mammalian brain. Science 265:1145–1148
Mercier F, Arikawa-Hirasawa E (2012) Heparan sulfate niche for cell proliferation in the adult brain. Neurosci Lett 510:67–72
Seki T (2002) Hippocampal adult neurogenesis occurs in a microenvironment provided by PSA-NCAM-expressing immature neurons. J Neurosci Res 69:772–783
Seri B, Herrera DG, Gritti A, Ferron S, Collado L, Vescovi A, Garcia-Verdugo JM, Alvarez-Buylla A (2006) Composition and organization of the SCZ. A large germinal layer containing neural stem cells in the adult mammalian brain. Cereb Cortex 16:103–111
Mandelbrot BB (1983) The fractal geometry of nature. WH Freeman, San Francisco
Ling EA (1998) Origin, nature and some functional considerations of intraventricular macrophages, with special reference to the epiplexus cells. Microsc Res Tech 41:43–56
Hada SH, Habuchi H, Kariya Y, Itoh N, Reddi AH, Kimata K (2004) Characterization of growth factor binding structures in heparin/heparan sulfate using an octasaccharide library. J Biol Chem 279:12346–12354
Brightman MW (2002) The brain’s interstitial clefts and their glial walls. J Neurocytol 31(2002):569–603
Ishihara M, Guo Y, Wei Z, Yang Z, Swiedler SJ, Orellana A, Hirsberg CB (1993) Regulation of biosynthesis of the basis fibroblast growth factor binding domains of heparan sulfate by heparan sulfate-N-deacetylase/N-sulfotransferase expression. J Biol Chem 268:20091–20095
Colak D, Mori T, Brill MS, Pfeifer A, Falk S, Deng C, Monteiro R, Mummery C, Sommer L, Gotz M (2008) Ault neurogenesis requires smad4-mediated bone morphogenetic protein signaling in stem cells. J Neurosci 28:434–446
Dierker T, Dreier R, Peterson A, Bordych C, Grobe K (2009) Heparan sulfate-modulated, metalloprotease-mediated sonic hedgehog release from producing cells. J Biol Chem 284:8013–8022
Gordts PL, Esko JD (2015) Heparan sulfate proteoglycans fine-tune macrophage inflammation via IFN-gamma. Cytokine 72:118–119
Khan SA, Nelson MS, Pan C, Gaffney PM, Gupta P (2008) Endogenous heparan sulfate and heparin modulate bone morphogenetic protein-4 signaling and activity. Am J Physiol Cell Physiol 294:1387–1397
Kishimoto S, Nakamura S, Hattori H, Nakamura SI, Oonuma F, Kanatani Y, Tanaka Y, Mori Y, Harada Y, Tagawa M, Ishihara M (2009) Human stem cell factor (SCF) is a heparin-binding cytokine. J Biochem 145:275–278
Lyon M, Rushton G, Gallagher JT (1997) The interaction of the transforming growth factor-betas with heparin/heparan sulfate is isoform specific. J Biol Chem 18:18000–18006
Thorne BA, Plowman GD (1994) The heparin-binding domain of amphiregulin necessitates the precursor pro-region for growth factor secretion. Mol Cell Biol 14:1635–1646
Johanson C, McMillan P, Tavares R, Spangenberger A, Duncan J, Silverberg G, Stopa E (2004) Homeostatic capabilities of the choroid plexus epithelium in Alzheimer’s disease. Cerebrospinal Fluid Res 1:3
Stopa EG, Berzin TM, Kim S, Song P, Kuo-Leblnc V, Rodriguez-Wolf M, Johanson CE (2001) Human choroid plexus growth factors: what are the implications for CSF dynamics in Alzheimer’s disease? Exp Neurol 167:40–47
Choe Y, Kozlova A, Graf D, Pleasure SJ (2013) Bone morphogenetic protein signaling is a major determinant of dentate development. J Neurosci 16:6667–6675
Bifari F, Decimo I, Chimulera C, Bersan E, Malpelli G, Johansson J, Lisi V, Bonetti B, Fumagalli G, Pizzolo G, Krampera M (2009) Novel stem/progenitor cells with neuronal differentiation potential reside in the leptomeningeal niche. J Cell Mol Med 9B:3195–3208
Louveau A, Smirnov I, Keyes TJ, Eccles JD, Rouhani SJ, Peske D, Derecki NC, Castle D, Mandell JW, Lee KS, Harris TH, Kipnis J (2015) Structural and functional features of central nervous system lymphatics. Nature 523:531–533
Mercier F, Weatherby TM, Harttine DK (2013) Meningeal-like organization of neural tissue sin calanoid copepods. J Comp Neurol 521:760–790
Siegenthaler JA, Pleasure SJ (2011) We have got you covered: how the meninges control brain development. Curr Opin Genet Dev 21:249–255
Mercier F, Cho-Kown Y, Douet D (2012) Hippocampus/amygdala alterations, loss of heparan sulfates, fractones and ventricle wall reduction in adult BTBR T+tf/J mice, animal model for autism. Neurosci Lett 506:208–213
Mercier F, Cho-Kwon Y, Kodama R (2011) Meningeal/vascular alterations and loss of extracellular matrix in the neurogenic zone of adult BTBR T+tf/J mice, animal model for autism. Neurosci Lett 498:173–178
Wahlsten D, Metten P, Crabbe JC (2003) Survey of 21 inbred mouse strains in two laboratories reveals that BTBR T+tf/J has severely reduced hippocampal commissure and absent corpus callosum. Brain Res 971:44–57
Moy SS, Nadler JJ, Young NB, Perez A, Holloway LP, Barbaro LP, Barbaro JR, West LM, Threadgill DW, Lauder JM, Magnuson TR, Crawley JN (2007) Mouse behavioural tasks relevant to autism: phenotype of 10 inbred mice. Behav Brian Res 176:4–20
Campos-Ordonez T, Herranz-Perez V, Chaichana KL, Riccon-Torroella J, Garcia-Verdugo JM, Quinines-Hinojosa A, Gonzalez-Perez O (2014) Long-term hydrocephalus alters the cytoarchitecture of the adult subventricular zone. Exp Neurol 261:236–244
Palu E, Liesi P (2002) Differential distribution of laminins in Alzheimer disease and normal human brain tissue. J Neurosci Res 69:243–256
Yasuoka K, Hirata K, Kuraoka A, Kawabuchi M (2004) Expression of amyloid precursor protein-like molecule in astroglial cells of he subventricular zone and rostral migratory stream of the adult rat forebrain. J Anat 205:135–146
Leveugle B, Scanameo A, Ding W, Fillit H (1994) Binding of heparan sulfate glycosaminoglycan to beta-amyloid peptide: inhibition by potentially therapeutic polysulfated compounds. NeuroReport 5:1389–1392
Scholefield Z, Yates EA, Wayne G, Amour A, McDowell W, Turnbull JE (2003) Heparan sulfate regulate amyloid precursor protein processing by BACE1, the Alzheimer’s β-secretase. J Cell Biol 163:97–107
Homes BB, DeVos SL, Kfoury N, Li M, Jacks R, Yanamandra K, Ouidja MO, Brodsky FM, Marasa J, Bagchi DP, Kotsbauer PT, Miller TM, Papy-Garcia D, Diamnon MI (2013) Heparan sulfate proteoglycans mediate internalization and propagation of specific proteopathic seeds. Proc Natl Acad Sci 110:E3138–E3147
Irie F, Badie-Mahdavi H, Yamaguchi Y (2012) Autism-like socio-communicative deficits and stereotypies in mice lacking heparan sulfate. Proc Natl Aced Sci 109:5052–5056
Campanella GSV, Em Lee, Sun J, Luster AD (2003) CXCR3 and heparin binding sites of the chemokine IP-10 (CXCL10). J Biol Chem 278:17066–17074
Kerever Yamada T, Suzuki Y, Mercier F, Arikawa-Hirasawa E (2015) Fractone aging in the subventricular zone of the lateral ventricle. J Chem Neuroanat 66–67:52–60
Gonzalez-Perez O, Alvarez-Buylla A (2011) Oligodendrogenesis in the subventricular zone and the role of epidermal growth factor. Brain Res Rev 67:147–156
Relucio J, Menezes M, Miyagoe-Suzuki MJ, Takeda S, Colognato H (2012) Laminin regulates postnatal oligodendrocyte production by promoting oligodendrocyte precursor survival in the subventricular zone. Glia 60:1451–1467
El Waly B, Macchi M, Cayre M, Durbec P (2014) Oligodendrogenesis in the normal and pathological central nervous system. Front Neurosci 8:145
Winkler S, Stahl RC, Carey DJ, Bansal R (2002) Syndecan-3 and perlecan are differentially expressed by progenitor and mature oligodendrocytes and accumulate in the extracellular matrix. J Neurosci Res 69:477–487
Pye DA, Vived RR, Turnbull JE, Hyde P, Gallagher JT (1998) Heparan sulfate oligosaccharide require 6-O-sulfation for promotion of basic fibroblast growth factor mitogenic activity. J Biol Chem 273:22936–22942
Sasaki N, Higashi N, Taka T, Nakajima M, Irimura T (2004) Cell surface localization of heparanase on macrophages regulates degradation of extracellular matrix heparan sulfate. J Immunol 172:3830–3835
Edwards IJ, Xu H, Obonuke JC, Goldberg IJ, Wagner WD (1995) Differentiated macrophages synthetize a heparan sulfate proteoglycan and an oversulfated chondrotine sulfate proteoglycan that bind lipoprotein lipase. Arterioscler Thromb Vasc Biol 15:400–409
Zhang GL, Zhang X, Wanfg XM, Li JP (2014) Towards understanding the role of heparan sulfate proteoglycans in Alzheimer’s disease. BioMed Res Int. doi:10.1155/2014/516028
Perez-Martin M, Grondona JM, Cifuentes M, Perez-Figares JM, Jimenez JA, Fernandez-Llebrez P (2000) Ependymal explants from the lateral ventricle of the adult bovine brain: a model system for morphological and functional studies of the ependyma. Cell Tissue Res 300:11–19
Mercier F, Hatton GI (2000) Immunocytochemical basis for a meningeo-glial network. J Comp Neurol 420:445–465
Acknowledgments
I thank C Alan Titchenal, Harry B Davis and Albert HW Jiang for proofreading this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mercier, F. Fractones: extracellular matrix niche controlling stem cell fate and growth factor activity in the brain in health and disease. Cell. Mol. Life Sci. 73, 4661–4674 (2016). https://doi.org/10.1007/s00018-016-2314-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-016-2314-y