Abstract.
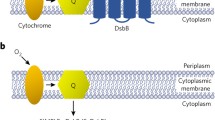
DsbD is a redox-active protein of the inner Escherichia coli membrane possessing an N-terminal (nDsbD) and a C-terminal (cDsbD) periplasmic domain. nDsbD interacts with four different redox proteins involved in the periplasmic disulfide isomerization and in the cytochrome c maturation systems. We review here the studies that led to the structural characterization of all soluble DsbD domains involved and, most importantly, of trapped disulfide intermediate complexes of nDsbD with three of its four redox partners. These results revealed the structural features enabling nDsbD, a ‘redox hub’ with an immunoglobulin-like fold, to interact efficiently with its different thioredoxin-like partners.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Received 3 February 2006; received after revision 1 March 2006; accepted 5 April 2006
Rights and permissions
About this article
Cite this article
Stirnimann, C.U., Grütter, M.G., Glockshuber, R. et al. nDsbD: a redox interaction hub in the Escherichia coli periplasm. Cell. Mol. Life Sci. 63, 1642–1648 (2006). https://doi.org/10.1007/s00018-006-6055-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-006-6055-1