Abstract.
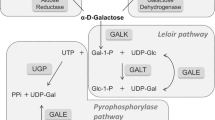
The conversion of beta-D-galactose to glucose 1-phosphate is accomplished by the action of four enzymes that constitute the Leloir pathway. Galactokinase catalyzes the second step in this pathway, namely the conversion of alpha-D-galactose to galactose 1-phosphate. The enzyme has attracted significant research attention because of its important metabolic role, the fact that defects in the human enzyme can result in the diseased state referred to as galactosemia, and most recently for its utilization via ‘directed evolution’ to create new natural and unnatural sugar 1-phosphates. Additionally, galactokinase-like molecules have been shown to act as sensors for the intracellular concentration of galactose and, under suitable conditions, to function as transcriptional regulators. This review focuses on the recent X-ray crystallographic analyses of galactokinase and places the molecular architecture of this protein in context with the extensive biochemical data that have accumulated over the last 40 years regarding this fascinating small molecule kinase.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Received 13 April 2004; accepted 7 June 2004
Rights and permissions
About this article
Cite this article
Holden, H.M., Thoden, J.B., Timson, D.J. et al. Galactokinase: structure, function and role in type II galactosemia. CMLS, Cell. Mol. Life Sci. 61, 2471–2484 (2004). https://doi.org/10.1007/s00018-004-4160-6
Issue Date:
DOI: https://doi.org/10.1007/s00018-004-4160-6