Abstract:
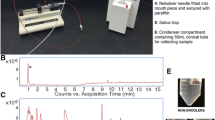
Objective: To qualitatively validate an enzyme immunoassay to measure leukotriene B4 in exhaled breath condensate. Exhaled breath condensate is a new non-invasive method to monitor airway inflammation.¶Subjects: Twenty-two subjects with different lung diseases attended the outpatient clinic on one occasion for exhaled breath condensate collection.¶Methods: Samples were pooled together and purified by reverse-phase high-performance liquid chromatography. The fractions eluted were assayed for leukotriene B4 by enzyme immunoassay.¶Results: A single peak of leukotriene B4-like immunoreactivity co-eluting with leukotriene B4 standard (retention time: 24 min) was identified by enzyme immunoassay. Reverse phase-high performance liquid chromatography peak of leukotriene B4 was clearly separated from those of 6-trans- leukotriene B4 (retention time: 14 min) and leukotriene B5 (retention time: 18 min) for which the antiserum used in the enzyme immunoassay had the highest cross-reactivity. Leukotriene B4 recovery was 64%.¶Conclusions: This study provides evidence for the presence of leukotriene B4 in the exhaled breath condensate and the specificity of the enzyme immunoassay used.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received 25 April 2002; returned for revision 7 August 2002; accepted by M. J. Parnham 1 October 2002
RID="*"
ID="*"Correspondence to: P. Montuschi
Rights and permissions
About this article
Cite this article
Montuschi, P., Ragazzoni, E., Valente, S. et al. Validation of leukotriene B4 measurements in exhaled breath condensate. Inflamm. res. 52, 69–73 (2003). https://doi.org/10.1007/s000110300003
Issue Date:
DOI: https://doi.org/10.1007/s000110300003