Abstract
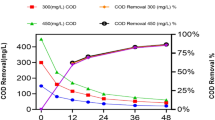
The removal of organic nitrogen compounds present in crude perroleum and shale oils poses a challenging problem in petroleum industries. The deleterious effect of nitrogen compounds on cracking catalysts and the indication that they contribute to gum formation in gasolines are some of these aspects. Pyridine, a representative nitrogen compound in gaavy oil—was degraded byRhodoccus sp. KCTC 3218 in a water-heavy oil two-phase system. The pyridine degradation rate was affected by the presence of hydrocarbons such as n-hexadecane. This microorganism formed flocs which could be a barrier to mass transfer between the cells in flocs and the pyridine dissolved in water. This problem could be overcome by the addition of a surfactant such as Triton X-100. The ratio of water to heavy oil was important to separate the heavy oil phase from the water phase after treating the heavy oil. The culture medium was emulsified by a sort of biosurfactant secreted by this microorganism. The emulsified oil phase returned to its natural state when the ratio of water to heavy oil was 1.5. Above this ratio, the emulsified oil phase remained an emulsion after decantation. Pyridine in heavy oil was completely degraded in 15 hr at this water to heavy oil ratio of 1.5 when the concentration of pyridine in heavy oil was 700 ppm and the cell concentration was 0.32 g DCW/L.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Benedik, M. J., P. R. Gibbs, P. R. Riddle, and R. C. Willson (1998) Microbial denitrogenation of fossil fuels.Tibtech. 16: 390–395.
Aislabie, J., N. K. Richards, and T. C. Lyttle (1994) Description of bacteria able to degrade isoquinoline in pure culture.Can. J. Microbiol. 40: 555–560.
Sims, G. K. and E. J. O'Loughlin (1989) Degradation of pyridine in the environment.Crit. Rev. Environ. Cotrol. 19: 309–340.
Shukla, O. P. and S. M. Kaul (1975) Succinate semialdehyde, an intermediate in the degradation of pyridine byBrevibacterium sp..Indian J. Biochem. Biophysics. 12: 321–330.
Arnold, E., S. Lenore, and D. Andrew (1992)Standard Methods for Examination of Water and Wastewater, 18thed., American Public Health Association, Washington DC, 4—94—4—97.
Rosenberg, M., D. Guntnick, and E. Rosenberg (1980) Adherence of bacteria to hydrocarbons: a sample method for measuring cell-surface hydrophobicity.FEMS Microbiol. Lett. 9: 29–33.
Britton, L. N. (1984) Microbial degradation of aliphatic hydrocarbons. p. 89–129 In:Microbial Degradation of Organic Compounds (Gibson, D. T. ed.). Marcel Dekker, NY, U.S.A.
Mimura, A., S. Watanabe, and I. Takeda (1971) Biochemical engineering analysis of hydrocarbon fermentation.J. Ferment. Technol. 49: 255–271.
Kurane, R., K. Takeda, and T. Suzuki (1986) Screening for and characteristics of microbial flocculants.Agric. Biol. Chem. 50: 2301–2308.
Marshall, K. C. and R. H. Cruickshank (1973) Cell surface hydrophobicity and the orientation of certain bacteria at interfaces.Arch. Microbiol. 91: 29–40.
Witholt, B., de Smet, M. J., Kingma, J. van Beilen, J. B., Kok, M., Lageveen, G., and G. Eggink (1980) Bioconversions of aliphatic compounds byPseudomonas oleovorans multiphase bioreactors: background and economic potential.Tibtech. 8: 46–51.
Blanch, H. W. and A. Einsele (1973) The kinetics of yeast growth on pure hydrocarbons.Biotechnol. Bioeng. 15: 861–877.
Zajic, J. E. and D. F. Gerson (1977) Emulsifying and surface active agent fromCorynebacterium hydrocarboclastu's.Biotechnol. Bioeng. 19: 1285–1301.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Do, J.H., Lee, W.G., Theodore, K. et al. Biological removal of pyridine in heavy oil byRhodococcus sp. KCTC 3218. Biotechnol. Bioprocess Eng. 4, 205–209 (1999). https://doi.org/10.1007/BF02931930
Issue Date:
DOI: https://doi.org/10.1007/BF02931930