Abstract
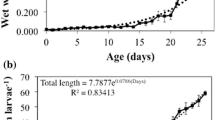
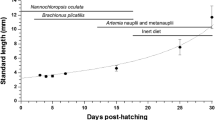
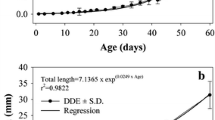
The evolution of the digestive enzyme equipment in seabream from hatching to 30 days old larvae was studied; there was a progressive increase in the activity of protease, amylase and acid and alkaline phosphatase from day 15 onwards. The use of specific inhibitors, and SDS-PAGE provided evidence to suggest that most of the proteases belonged to the serine group. A high α-amylase activity was also denoted. Zymograms of larval extracts indicated that exogenous food has more a qualitative than a quantitative role in the secretion of digestive enzymes in this species.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References cited
Baragi, V. and Lovell, R.T. 1986. Digestive enzyme activities in striped bass from first feeding through larva development. Trans. Am. Fish. Soc. 115: 478–484.
Bonnie, S.P., Lan, C.C. and Hung, T.Y. 1991. Changes in composition and proteolytic enzyme activities ofArtemia during early development. Comp. Biochem. Physiol. 100: 725–730.
Bradford, M.M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248–254.
Cahu, C.L. and Zambonino Infante, J.L. 1994. Early weaning of sea bass (Dicentrarchus labrax) larvae with a compound diet: effect on digestive enzymes. Comp. Biochem. Physiol. 109A: 213–222.
Clark, J., Murray, K.R. and Stark, J.R. 1986. Protease development in dover sole (Solea solea L.). Aquaculture 53: 253–262.
Corneille, S., Ollevier, F., Carrascosa, M., Pesci, F. and Rendon, A. 1989. Reduction of the use ofArtemia nauplii by early feeding of seabream larvae (Sparus aurata) with dry food. EAS Special Publ. no 10:73–74, n−n.
Corneille, S. and Delis, S. 1992. Impact of weaning feed on the marine fry production. Ioaninnian Fishfarming Conf. Greece.
Cousin, J.C.B., Baudin-Laurencin, F. and Gabaudan, J. 1987. Ontogeny of enzymatic activities in fed and fasting turobt,Scophthalmus maximus L. J. Fish Biol. 30: 15–33.
Dabrowski, K. and Glogowski, J. 1977. A study of the application of proteolytic enzymes to fish food. Aquaculture 12: 349–360.
García-Carreño, F.L. 1992. Protease inhibition in theory and practice. Biotech. Educ. 3: 145–150.
García-Carreño, F.L., Dimes, L.E. and Haard, N.F. 1993. Substrate-gel electrophoresis for composition and molecular weight of proteinases or proteinaceous proteinase inhibitors. Anal. Biochem. 214: 65–69.
García-Carreño, F.L. and Haard, N.F. 1993. Characterization of proteinase classes in langostilla (Pleuroncodes planipes) and crayfish (Pacifastacus astacus) extracts. J. Food Biochem. 17: 97–113.
Georgopoulou, U., Sire, M.F. and Vernier, J.M. 1985. Macromolecular absorption of proteins by epithelial cells of the posterior intestinal segment and their intracellular digestion in the rainbow trout. Ultrastructural and biochemical study. Biol. Cell 53: 269–282.
Govoni, J.J., Boehlert, G.W. and Watanabe, Y.L. 1986. The physiology of digestion in fish larvae. Env. Biol. Fish. 16: 59–77.
Hara, K., Arano, H. and Ishihara, T. 1984. Purification of alkaline protease of the rotiferBrachionus plicatilis. Bull. Jap. Soc. Sci. Fish. 50: 1605–1609.
Hjelmeland, K., Huse, I., Jorgensen, T., Molvik, G. and Raa, J. 1984. Trypsin and trypsinogen as indices of growth and survival potential of cod (Gadus morhua L.) larvae. Flodevigen Rapportser. 1: 189–201.
Kleinow, W. 1993. Biochemical studies onBrachionus plicatilis hydrolytic enzymes, integument proteins and composition of trophi. Hydrobiologia 255/256: 1–12.
Kolkovski, S., Tandler, A., Kissil, G.Wm. and Gertler, A. 1993. The effect of dietary exogenous enzymes on ingestion, assimilation, growth and survival of gilthead seabream (Sparus aurata, Sparidae, Linnaeus) larvae. Fish Physiol. Biochem. 12: 203–209.
Kunitz, M. 1947. Crystalline soybean trypsin inhibitor II. General properties. J. Gen. Physiol. 30: 291–310.
Laemmli, U.K. 1970. Cleavage of structural proteins during the assembly of the head of Bacteriophage T4. Nature, Lond. 227: 680–685.
Lauff, M. and Hofer, R. 1984. Proteolityc enzymes in fish development and the importance of dietary enzymes. Aquaculture 37: 335–346.
Letlier, M.E., Repetto, Y., Alduane, J. and Morello, A. 1985. Acid and alkaline phosphatase activity inTrypanosoma cruzi epimastigotes. Comp. Biochem. Physiol. 81: 47–51.
Moyano, F.J. and Sarasquete, M.C. 1993. A screening on some digestive enzyme activities of gilthead seabream (Sparus aurata) larvae. World Aquaculture'93. Special Publication No. 19. Torremolinos. Spain. n−n.
Munilla-Morán, R., Stark, J.R. and Barbour, A. 1990. The role of exogenous enzymes in digestion in culture of turbot larvae (Scophthalmus maximus L.). Aquaculture 88: 337–350.
Pedersen, B.H. and Andersen, K.P. 1992. Induction of trypsinogen secretion in herring larvae (Clupea harengus) Mar. Biol. 112: 559–565.
Pedersen, B.H. and Hjelmeland, K. 1988. Fate of trypsin and assimilation efficiency in larval herring (Clupea harengus) following digestion of copepods. Mar. Biol. 97: 467–476.
Pedersen, B.H., Nilssen, E.M. and Hjeldman, K. 1987 Variations in the content of trypsin and trypsinogen in larval herring (Clupea harengus) digesting copepod nauplii. Mar. Biol. 94: 171–181.
Robyt, J.F. and Whelan, W.J. 1988. nn.In Starch and its derivatives. pp. n−n. Edited by J.A.. Chapman and Hall, London.
Sarasquete, M.C., Polo, A. and Conzález de Canales, M.L. 1993. A histochemical and immunohistochemical study of digestive enzymes and hormones during the larval development ofSparus aurata L. Histochem. J. 24: 337–344.
Sarasquete, M.C., Polo, A. and Yufera, M. 1994. Histology and histochemistry of the development of the digestive system of larval gilthead seabream,Sparus aurata L. Aquaculture (In press).
Segner, H., Rosch, R., Schmidht, H. and Von Poeppinghausen, K.J. 1989. Digestive enzymes in larvalCoregonus lavaretus L. J. Fish Biol. 35: 249–263.
Stauffer, C. 1989. Enzyme Assays for Food Scientists. Van Nostand Reinhold/AVI, New York.
Stroband, H.W.J. and Dabrowski, K.R. 1979. Morphological and physiological aspects of the digestive system and feeding in fresh-water fish larvae.In Nutrition des Poissons. pp. 335–374. Edited by M. Fontaine. n−n.
Tanaka, M. 1972. Studies on the structure and function at the digestive system in teleost larvae. IV. Changes in the epithelium related to fat absorption in the anteromedian part of the intestine after feeding. Jap. J. Ichthyol. 19: 15–25.
Ueberschär, B. 1993. Measurement of proteolytic enzyme activity: significance and application in larval fish research.In Physiological and Biochemical Aspects of Fish Development. pp. n−n. Edited by B.T. Walther and H.J. Fuhn. n−n.
Walford, J. and Lam, T.J. 1993. Development of digestive tract and proteolytic enzymes activity in sea bass (Lates calcarifer) larvae and juveniles. Aquaculture 109: 187–205.
Walter, H.E. 1984. Proteinases: methods with hemoglobin, casein and azocoll as substrates.In Methods of Enzymatic Analysis. Vol. V, pp. 270–277. edited by H.J. Bergmeyer. Verlag Chemie, Weinham.
Yúfera, M., Pascual, E., Polo, A. and Sarasquete, M.C. 1993. Effect of starvation on the feeding ability of gilthead seabream,Sparus aurata L. larvae at first feeding. J. Exp. Mar. Biol. Ecol. 169: 259–272.
Zambonino Infante, J.L. and Cahu, C. 1994. Development and response to a diet of some digestive enzymes in sea bass (Dicentrarchus labrax) larvae. Fish Physiol. Biochem. 12: 399–408.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Moyano, F.J., Díaz, M., Alarcón, F.J. et al. Characterization of digestive enzyme activity during larval development of gilthead seabream (Sparus aurata). Fish Physiol Biochem 15, 121–130 (1996). https://doi.org/10.1007/BF01875591
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01875591