Summary
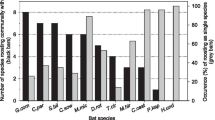
At a site in Costa Rica, three groups of 8–12 adult female vampire bats, Desmodus rotundus, utilize group-specific sets of hollow trees as day roosts. Long-term nonrandom associations between pairs of females, as measured by the proportion of time one bat spends roosting in the same tree with another bat over a 3 year period, occur even when preferences for particular trees are removed. Significant associations exist between both related and unrelated adult females. Adult male bats, however, show few associations with females or other males. By observing bats within trees and while foraging, and by monitoring feeding flights with radiotelemetry, the following potential benefits of association could be tested. Females roost together to (1) share a suitable microclimate, (2) avoid predators, (3) avoid ectoparasite infestations, (4) minimize travel to mobile prey animals, (5) respond to coercive males, (6) feed simultaneously from a bite, (7) remove ectoparasites by allogrooming, and (8) share food by regurgitating blood to other bats within roosts. The data do not indicate that any of the first five hypotheses provide significant benefits for long-term associations although predators and ectoparasite levels may cause occasional changes in roost sites. Simultaneous feeding was uncommon and apparently confined to females and their recent offspring. Allogrooming, although common, occurred independently of the presence of ectoparasites. Food sharing, however, occurred between both related and unrelated adult females with high levels of association and provides at least one selective advantage for maintaining cohesive female groups.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alexander RD (1974) The evolution of social behavior. Annu Rev Ecol Syst 5:325–383
Altmann J (1974) Observation study of behavior: sampling methods. Behaviour 49:227–267
Anderson DJ (1982) The home range: a new nonparametric estimation technique. Ecology 63:103–112
Axelrod R, Hamilton WD (1981) The evolution of cooperation. Science 211:1390–1396
Bekoff M (1981) Mammalian sibling interactions: genes, facilitative environments, and the coefficient of familiarity. In: Gubernick DJ, Klopfer PH (eds) Parental care in mammals. Plenum, New York, pp 307–346
Bradbury JW, Vehrencamp SL (1976) Social organization and foraging in emballonurid bats I. Field studies. Behav Ecol Sociobiol 1:337–381
Davis RB, Herreid II CF, Short HL (1962) Mexican free-tailed bats in Texas. Ecol Monogr 32:311–346
Eisenberg JF (1981) The mammalian radiations: an analysis of trends in evolution, adaptation and behavior. University of Chicago Press, Chicago, Ill
Fager EW (1957) Determination and analysis of recurrent groups. Ecology 38:586–595
Fleming TH, Hooper ET, Wilson DE (1972) Three Central American bat communities: structure, reproductive cycles and movement patterns. Ecology 53:555–569
Ford RG, Krumme DW (1979) The analysis of space use patterns. J Theor Biol 76:125–155
Frame LH, Frame GW (1976) Female African wild dogs emigrate. Nature 263:227–229
Frame LH, Malcolm JR, Frame GF, Lawick H van (1979) Social organization of African wild dogs (Lycaon pictus) on the Serengeti Plains, Tanzania, 1967–1978. Z Tierpsychol 50:225–249
Frankie GW, Baker HG, Opler PA (1974) Comparative phenological studies of trees in tropical wet and dry forests in the lowlands of Costa Rica. J Ecol 62:881–919
Greenhall AM, Schmidt U, Lopez-Forment W (1971) Attacking behavior of the vampire bat, Desmodus rotundus, under field conditions. Biotropica 3:136–141
Hamilton WD (1964) The genetical evolution of social behavior. J Theor Biol 7:1–52
Hayne DW (1949) Calculation of size of home range. J Mammal 30:1–18
Hollander M, Wolfe DA (1973) Nonparametric statistical methods. Wiley, New York
Holmes WG, Sherman PW (1983) Kin recognition in animals. Am Sci 71:46–55
Huey RB, Pianka ER, Pimm SL (1983) Temporal separation of activity and intraspecific dietary overlap. In: Huey RB, Pianka ER, Schoener TW (eds) Lizard ecology, studies of a model organism. Harvard University Press, Cambridge, Mass, pp 281–296
Janzen JH (1983) Costa rican natural history. University of Chicago Press, Chicago, Ill
Malcolm JR, Marten K (1982) Natural selection and the communal rearing of pups in African wild dogs (Lycaon pictus). Behav Ecol Sociobiol 10:1–13
Marshall AG (1982) Ecology of insects ectoparasitic on bats. In: Kunz TH (ed) The ecology of bats. Plenum, New York, pp 369–402
McCracken GF (1984) Communal nursing in Mexican freetailed bat maternity colonies. Science 223:1090–1091
McCracken GF, Bradbury JW (1981) Social organization and kinship in the polygynous bat, Phyllostomus hastatus. Behav Ecol Sociobiol 8:11–34
McFarland WN, Wimsatt WA (1969) Renal function and its relation to ecology of the vampire bat, Desmodus rotundus. Comp Biochem Physiol [A] 28:985–1006
McNab BK (1973) Energetics and distribution of vampire bats. J Mammal 54:131–144
Mills RS (1980) Parturition and social-interaction among captive vampire bats, Desmodus rotundus. J Mammal 61:336–337
Morrison DW (1978) Foraging ecology and energetics of the frugivorous bat Artibeus jamaicensis. Ecology 59:716–723
Morrison DW (1979) Apparent male defense of tree hollows in the fruit bat, Artibeus jamaicensis. J Mammal 60:11–15
Rood JP (1978) Dwarf mongoose helpers at the den. Z Tierpsychol 48:277–287
Rood JP (1983) The social system of the dwarf mongoose. In: Eisenberg JF, Kleiman DG (eds) Advances in the study of mammalian behavior. Spec Publ Am Soc Mammal 7:454–488
Sailler H von, Schmidt U (1978) Die sozialen Laute der Gemeinen Vampirfledermaus Desmodus rotundus bei Konfrontation am Futterplatz unter experimentellen Bedingungen. Z Säugetierkd 43:249–261
Schmidt U (1972) Social calls of juvenile vampire bats (Desmodus rotundus) and their mothers. Bonn Zool Beitr 23:310–316
Schmidt U (1973) Olfactory threshold and odour discrimination of the vampire bat (Desmodus rotundus). Period Biol 75:89–92
Schmidt U (1978) Vampirfledermäuse. Ziemsen, Wittenburg
Sokal RR, Rohlf FJ (1981) Biometry. Freeman, San Fransisco, Calif
Trivers RL (1971) The evolution of reciprocal altruism. Q Rev Biol 46:35–57
Turner DC (1975) The vampire bat. Johns Hopkins University Press, Baltimore, Md
Wade MJ (1978) A critical review of the models of group selection. Q Rev Biol 53:101–114
Ward P, Zahavi A (1973) The importance of certain assemblages of birds as “information centers” for food finding. Ibis 115:517–534
Wenzell RL, Tipton VJ, Kiewlicz A (1966) The streblid batflies of Panama (Diptera Calypterae: Streblidae). In: Wenzell RL, Tipton VJ (eds) Ectoparasites of Panama. Field Museum of Natural History, Chicago, Ill, pp 405–675
Wilkinson GS (1984) Reciprocal food sharing in vampire bats. Nature 309:181–184
Wilkinson GS (1985) The social organization of the common vampire bat. II. Mating system, genetic structure, and relatedness. Behav Ecol Sociobiol 17:123–134
Wilkinson GS, Bradbury JW (in press) Radio telemetry techniques for bats. In: Kunz TG (ed) Ecological and behavioral methods for the study of bats. Smithsonian Institution, Washington, DC
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wilkinson, G.S. The social organization of the common vampire bat. Behav Ecol Sociobiol 17, 111–121 (1985). https://doi.org/10.1007/BF00299243
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00299243