Abstract
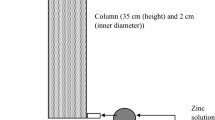
In this work, the efficiency of an adsorption process, in which Moroccan diatomite (ND) is used as a low-cost adsorbent to remove Congo red (CR) dye from contaminated waters in batch and column system, was examined. The influence of experimental conditions (pH, adsorbent dose and temperature) on the adsorption of CR onto the ND adsorbent was studied. A study of the adsorption kinetics for CR revealed that a pseudo-second-order model provided the best fit to the experimental kinetic data, and the equilibrium data were well described by the Langmuir isotherm model with an adsorption capacity of 6.07 mg/g using 15 g/L of ND, pH=6, contact time 3 h and 25 °C. On the other hand, the ND regeneration tests were investigated and showed that the desorption reaches at least 50% when using ethanol as eluent. In addition, the adsorption process in a continuous mode was studied. Breakthrough curves were properly represented by the Yoon — Nelson model. Hence, the adsorption capacity of 5.71 mg/g was reached using 0.114 g of adsorbent, CR concentration of 6 mg/L and a flow of 1 mL/min under 25 °C.
摘要
以摩洛哥硅藻土(ND)为低成本吸附剂,采用批式和柱式吸附法对污染水体中刚果红(CR)染料的去除效率进行研究。考察了实验条件(pH、吸附剂剂量和温度)对ND吸附CR的影响。实验结果表明,在15 g/L ND、pH=6、接触时间为3 h、温度为25 °C的条件下,CR的吸附量为6.07 mg/g,拟二级吸附模型与实验动力学数据吻合较好,Langmuir 等温线模型能较好地描述吸附平衡数据。再生实验结果表明,当以乙醇为洗脱剂时,ND的解吸率达到50%。此外,还研究了连续模式下的吸附过程。用Yoon−Nelson 模型可以很好地解释穿透曲线。在25 °C条件下,当吸附剂用量为0.114 g,CR浓度为6 mg/L,流量为1 mL/min时,吸附量可达5.71 mg/g。
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
EL MOSTAFA J. Social and ecological transformation in Morocco and across Africa [R]. Heinrich-Böll-Stiftung-Afrique du Nord, Rabat, 2017.
Business opportunities report for reuse of wastewater in morocco [R]. Commissioned by the Netherlands Enterprise Agency, 2018.
Moroccan climate change policy [R]. Ministry Delegate of the Minister of Energy, Mines, Water and Environment, in charge of Environment, 2014.
MANDIL, OUAZZANI N. Water and wastewater management in Morocco: Biotechnologies application [J]. Sustainable Sanitation Practice, 2013, 1(14): 9–16.
Morocco United Nations Environmental performance reviews [R]. New York and Geneva: United Nations Economic Commission for Europe in Cooperation with United Nations Economic Commission for Africa Office for North Africa. 2014.
OUAZZANI N, BOUHOUM K, MANDI L, et al. Wastewater treatment by stabilization pond: Marrakesh experiment [J]. Water Science and Technology, 1995, 31(12): 75–80. DOI: https://doi.org/10.2166/wst.1995.0462.
Ninth project of drinkable water and sanitation: Evaluation report established by the African bank of development [R]. Kingdom of Morocco, African Development Bank, 2006.
MAKHOKH M, BOURZIZA M. Country report for the expert consultation on wastewater management-Morocco-SEEE-ONEP [R]. Dubai, UAE, 2011.
The World Bank Managing Urban, International Bank for Reconstruction and Development Water scarcity in Morocco annexes to Sections 2 to 4 [R]. 2017.
POLLARD S J T, FOWLER G D, SOLLARS C J, et al. Low-cost adsorbents for waste and waste-water treatment: A review [J]. Science of the Total Environment, 1992, 116(1–2): 31–52. DOI: https://doi.org/10.1016/0048-9697(92)90363-W
HADRI M, CHAOUKI Z, DRAOUI K, et al. Adsorption of a cationic dye from aqueous solution using low-cost Moroccan diatomite: Adsorption equilibrium, kinetic and thermodynamic studies [J]. Desalination and Water Treatment, 2017, 75: 213–224. DOI: https://doi.org/10.5004/dwt.2017.20553.
MANE V S, VIJAY BABU P V. Kinetic and equilibrium studies on the removal of Congo red from aqueous solution using Eucalyptus wood (Eucalyptus globulus) saw dust [J]. Journal of the Taiwan Institute of Chemical Engineers, 2013, 44(1): 81–88. DOI: https://doi.org/10.1016/j.jtice.2012.09.013.
SAHA P D, CHAKRABORTY S, CHOWDHURY S. Batch and continuous (fixed-bed column) biosorption of crystal violet by Artocarpus heterophyllus (jackfruit) leaf powder [J]. Colloids and Surfaces B: Biointerfaces, 2012, 92: 262–270. DOI: https://doi.org/10.1016/j.colsurfb.2011.11.057.
KAUR S, RANI S, KUMAR V, et al. Synthesis, characterization and adsorptive application of ferrocene based mesoporous material for hazardous dye Congo red [J]. Journal of Industrial and Engineering Chemistry, 2015, 26: 234–242. DOI: https://doi.org/10.1016/j.jiec.2014.11.035.
DICHIARA A B, WEINSTEIN S J, ROGERS R E. On the choice of batch or fixed bed adsorption processes for wastewater treatment [J]. Industrial & Engineering Chemistry Research, 2015, 54(34): 8579–8586. DOI: https://doi.org/10.1021/acs.iecr.5b02350.
BHARATHI K S, RAMESH S T. Removal of dyes using agricultural waste as low-cost adsorbents: A review [J]. Applied Water Science, 2013, 3(4): 773–790. DOI: https://doi.org/10.1007/s13201-013-0117-y.
LAGERGREN S K. About the theory of so-called adsorption of soluble substances [J]. Sven Vetenskapsakad Handingarl, 1898, 24: 1–39.
OUSSALAH A, BOUKERROUI A, AICHOUR A, et al. Cationic and anionic dyes removal by low-cost hybrid alginate/natural bentonite composite beads: Adsorption and reusability studies [J]. International Journal of Biological Macromolecules, 2019, 124: 854–862. DOI: https://doi.org/10.1016/j.ijbiomac.2018.11.197.
LARGO F, HAOUNATI R, AKHOUAIRI S, et al. Adsorptive removal of both cationic and anionic dyes by using sepiolite clay mineral as adsorbent: Experimental and molecular dynamic simulation studies [J]. Journal of Molecular Liquids, 2020, 318: 114247. DOI: https://doi.org/10.1016/j.molliq.2020.114247.
HO Y S, MCKAY G. Pseudo-second order model for sorption processes [J]. Process Biochemistry, 1999, 34(5): 451–465. DOI: https://doi.org/10.1016/S0032-9592(98)00112-5.
KHALILZADEH SHIRAZI E, METZGER J W, FISCHER K, et al. Removal of textile dyes from single and binary component systems by Persian bentonite and a mixed adsorbent of bentonite/charred dolomite [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2020, 598: 124807. DOI: https://doi.org/10.1016/j.colsurfa.2020.124807.
WANG Zhong-min, GAO Ming-min, LI Xiao-juan, et al. Efficient adsorption of methylene blue from aqueous solution by graphene oxide modified persimmon tannins [J]. Materials Science and Engineering C, 2020, 108: 110196. DOI: https://doi.org/10.1016/j.msec.2019.110196.
SHAIDA M A, DUTTA R K, SEN A K. Removal of diethyl phthalate via adsorption on mineral rich waste coal modified with chitosan [J]. Journal of Molecular Liquids, 2018, 261: 271–282. DOI: https://doi.org/10.1016/j.molliq.2018.04.031.
SENTHIL KUMAR P, RAMALINGAM S, SENTHAMARAI C, NIRANJANAA M, VIJAYALAKSHMI P, SIVANESAN S. Adsorption of dye from aqueous solution by cashew nut shell: Studies on equilibrium isotherm, kinetics and thermodynamics of interactions [J] Desalination: 2010, 261: 52–60. DOI: https://doi.org/10.1016/j.desal.2010.05.032.
AYGÜN A, YENISOY-KARAKAŞ S, DUMAN I. Production of granular activated carbon from fruit stones and nutshells and evaluation of their physical, chemical and adsorption properties [J]. Microporous and Mesoporous Materials, 2003, 66(2–3): 189–195. DOI: https://doi.org/10.1016/j.micromeso.2003.08.028.
NAMASIVAYAM C, KAVITHA D. Removal of Congo red from water by adsorption onto activated carbon prepared from coir pith, an agricultural solid waste [J]. Dyes and Pigments, 2002, 54(1): 47–58. DOI: https://doi.org/10.1016/S0143-7208(02)00025-6.
OSTOLSKA I, WIŚNIEWSKA M. Application of the zeta potential measurements to explanation of colloidal Cr2O3 stability mechanism in the presence of the ionic polyamino acids [J]. Colloid and Polymer Science, 2014, 292(10): 2453–2464. DOI: https://doi.org/10.1007/s00396-014-3276-y.
SHAH B A, SHAH A V, MISTRY C B, et al. Surface modified bagasse fly ash zeolites for removal of reactive black-5 [J]. Journal of Dispersion Science and Technology, 2011, 32(9): 1247–1255. DOI: https://doi.org/10.1080/01932691.2010.505550.
DE O APOLINÁRIO F, PIRES A P. Oil displacement by multicomponent slug injection: An analytical solution for Langmuir adsorption isotherm [J]. Journal of Petroleum Science and Engineering, 2021, 197: 107939. DOI: https://doi.org/10.1016/j.petrol.2020.107939.
GUPTA V K, JAIN R, SIDDIQUI M N, et al. Equilibrium and thermodynamic studies on the adsorption of the dye rhodamine-B onto mustard cake and activated carbon [J]. Journal of Chemical & Engineering Data, 2010, 55(11): 5225–5229. DOI: https://doi.org/10.1021/je1007857.
NITZSCHE R, GRÖNGRÖFT A, KRAUME M. Separation of lignin from beech wood hydrolysate using polymeric resins and zeolites-Determination and application of adsorption isotherms [J]. Separation and Purification Technology, 2019, 209: 491–502. DOI: https://doi.org/10.1016/j.seppur.2018.07.077.
MESDAGHINIA A, AZARI A, NODEHI R N, et al. Removal of phthalate esters (PAEs) by zeolite/Fe3O4: Investigation on the magnetic adsorption separation, catalytic degradation and toxicity bioassay [J]. Journal of Molecular Liquids, 2017, 233: 378–390. DOI: https://doi.org/10.1016/j.molliq.2017.02.094.
LANGMUIR I. The adsorption of gases on plane surfaces of glass, mica and platinum [J]. Journal of the American Chemical Society, 1918, 40(9): 1361–1403. DOI: https://doi.org/10.1021/ja02242a004.
FREUNDLICH H M F. Over the adsorption in solution [J]. Physical Chemistry, 1906, 57: 385–470. DOI: https://doi.org/10.1515/zpch-1907-5723
HALL K R, EAGLETON L C, ACRIVOS A, et al. Pore- and solid-diffusion kinetics in fixed-bed adsorption under constant-pattern conditions [J]. Industrial & Engineering Chemistry Fundamentals, 1966, 5(2): 212–223. DOI: https://doi.org/10.1021/i160018a011.
WEBER T W, CHAKRAVORTI R K. Pore and solid diffusion models for fixed-bed adsorbers [J]. AIChE Journal, 1974, 20(2): 228–238. DOI: https://doi.org/10.1002/aic.690200204.
GUPTA V K, PATHANIA D, AGARWAL S, et al. Amputation of Congo red dye from waste water using microwave induced grafted Luffa cylindrica cellulosic fiber [J]. Carbohydrate Polymers, 2014, 111: 556–566. DOI: https://doi.org/10.1016/j.carbpol.2014.04.032.
MANE V S, VIJAY BABU P V. Kinetic and equilibrium studies on the removal of Congo red from aqueous solution using Eucalyptus wood (Eucalyptus globulus) saw dust [J]. Journal of the Taiwan Institute of Chemical Engineers, 2013, 44(1): 81–88. DOI: https://doi.org/10.1016/j.jtice.2012.09.013.
VIMONSES V, LEI Shao-min, JIN Bo, et al. Kinetic study and equilibrium isotherm analysis of Congo Red adsorption by clay materials [J]. Chemical Engineering Journal, 2009, 148(2–3): 354–364. DOI: https://doi.org/10.1016/j.cej.2008.09.009.
ANSARI R, SEYGHALI B, MOHAMMAD-KHAH A, et al. Highly efficient adsorption of anionic dyes from aqueous solutions using sawdust modified by cationic surfactant of cetyltrimethylammonium bromide [J]. Journal of Surfactants and Detergents, 2012, 15(5): 557–565. DOI: https://doi.org/10.1007/s11743-012-1334-3.
PURKAIT M K, MAITI A, DASGUPTA S, et al. Removal of Congo red using activated carbon and its regeneration [J]. Journal of Hazardous Materials, 2007, 145(1–2): 287–295. DOI: https://doi.org/10.1016/j.jhazmat.2006.11.021.
GHAEDI M, BIYAREH M N, KOKHDAN S N, et al. Comparison of the efficiency of palladium and silver nanoparticles loaded on activated carbon and zinc oxide nanorods loaded on activated carbon as new adsorbents for removal of Congo red from aqueous solution: Kinetic and isotherm study [J]. Materials Science and Engineering C, 2012, 32(4): 725–734. DOI: https://doi.org/10.1016/j.msec.2012.01.015.
OJEDOKUN A T, BELLO O S. Liquid phase adsorption of Congo red dye on functionalized corn cobs [J]. Journal of Dispersion Science and Technology, 2017, 38(9): 1285–1294. DOI: https://doi.org/10.1080/01932691.2016.1234384.
ALKAN N, FLUHR R, SHERMAN A, et al. Role of ammonia secretion and pH modulation on pathogenicity of Colletotrichum coccodes on tomato fruit [J]. Molecular Plant-Microbe Interactions: MPMI, 2008, 21(8): 1058–1066. DOI: https://doi.org/10.1094/MPMI-21-8-1058.
MADEJOVÁ J. Baseline studies of the clay minerals society source clays: Infrared methods [J]. Clays and Clay Minerals, 2001, 49(5): 410–432. DOI: https://doi.org/10.1346/ccmn.2001.0490508.
MUNAGAPATI V S, KIM D S. Adsorption of anionic azo dye Congo Red from aqueous solution by Cationic Modified Orange Peel Powder [J]. Journal of Molecular Liquids, 2016, 220: 540–548. DOI: https://doi.org/10.1016/j.molliq.2016.04.119.
SAIKIA B J, PARTHASARATHY G. Fourier transform infrared spectroscopic characterization of kaolinite from Assam and Meghalaya, northeastern India [J]. Journal of Modern Physics, 2010, 1(4): 206–210. DOI: https://doi.org/10.4236/jmp.2010.14031.
RATHEE G, AWASTHI A, SOOD D, et al. A new biocompatible ternary Layered Double Hydroxide Adsorbent for ultrafast removal of anionic organic dyes [J]. Scientific Reports, 2019, 9: 16225. DOI: https://doi.org/10.1038/s41598-019-52849-4.
IMAMURA K, IKEDA E, NAGAYASU T, et al. Adsorption behavior of methylene blue and its congeners on a stainless steel surface [J]. Journal of Colloid and Interface Science, 2002, 245(1): 50–57. DOI: https://doi.org/10.1006/jcis.2001.7967.
MALL I D, SRIVASTAVA V C, KUMAR G V A, et al. Characterization and utilization of mesoporous fertilizer plant waste carbon for adsorptive removal of dyes from aqueous solution [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2006, 278(1–3): 175–187. DOI: https://doi.org/10.1016/j.colsurfa.2005.12.017.
SHAH I, PRÉ P, ALAPPAT J. B. Steam regeneration of adsorbents: An experimental and technical review [J]. Chemical Science Transactions, 2013, 2(4): 1078–1088. DOI: https://doi.org/10.7598/cst2013.545.
YOON Y H, NELSON J H. Application of gas adsorption kinetics I. A theoretical model for respirator cartridge service life [J]. American Industrial Hygiene Association Journal, 1984, 45(8): 509–516. DOI: https://doi.org/10.1080/15298668491400197.
ZAITAN H, KORRIR A, CHAFIK T, et al. Evaluation of the potential of volatile organic compound (di-methyl benzene) removal using adsorption on natural minerals compared to commercial oxides [J]. Journal of Hazardous Materials, 2013, 262: 365–376. DOI: https://doi.org/10.1016/j.jhazmat.2013.08.071.
HAN Run-ping, WANG Yu, ZHAO Xin, et al. Adsorption of methylene blue by phoenix tree leaf powder in a fixed-bed column: Experiments and prediction of breakthrough curves [J]. Desalination, 2009, 245(1–3): 284–297. DOI: https://doi.org/10.1016/j.desal.2008.07.013.
AKSU Z, GÖNEN F. Biosorption of phenol by immobilized activated sludge in a continuous packed bed: Prediction of breakthrough curves [J]. Process Biochemistry, 2004, 39(5): 599–613. DOI: https://doi.org/10.1016/S0032-9592(03)00132-8.
AL-GHOUTI M A, AL-DEGS Y S, KHRAISHEH M A M, et al. Mechanisms and chemistry of dye adsorption on manganese oxides-modified diatomite [J]. Journal of Environmental Management, 2009, 90(11): 3520–3527. DOI: https://doi.org/10.1016/j.jenvman.2009.06.004.
KHRAISHEH M A M, AL-GHOUTI M A, ALLEN S J, et al. The effect of pH, temperature, and molecular size on the removal of dyes from textile effluent using manganese oxides-modified diatomite [J]. Water Environment Research, 2004, 76(7): 2655–2663.
ZHANG Chun-hui, JIANG Shan, TANG Jia-wei, et al. Adsorptive performance of coal based magnetic activated carbon for perfluorinated compounds from treated landfill leachate effluents [J]. Process Safety and Environmental Protection, 2018, 117: 383–389. DOI: https://doi.org/10.1016/j.psep.2018.05.016.
SILVA V C, ARAÚJO M E B, RODRIGUES A M, et al. Adsorption behavior of acid-treated Brazilian palygorskite for cationic and anionic dyes removal from the water [J]. Sustainability, 2021, 13(7): 3954. DOI: https://doi.org/10.3390/su13073954.
SRIRAM G, KIGGA M, UTHAPPA U T, et al. Naturally available diatomite and their surface modification for the removal of hazardous dye and metal ions: A review [J]. Advances in Colloid and Interface Science, 2020, 282: 102198. DOI: https://doi.org/10.1016/j.cis.2020.102198.
Author information
Authors and Affiliations
Corresponding author
Additional information
Contributors
HADRI Mohamed and HAMDAOUI Mustapha performed the experiments, analyzed and interpreted the data, writing the initial draft of the manuscript. ELMRABET Imane and CHAOUKI Zineb performed the experiments, analyzed and interpreted the data. DRAOUI Khalid analyzed and interpreted the data. DOUHRI Hikmat analyzed and interpreted the data. ZAITAN Hicham supervised, analyzed and interpreted the data, writing review & editing, conceptualization, funding acquisition, resources, validation.
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Hadri, M., El Mrabet, I., Chaouki, Z. et al. Valorization of natural diatomite mineral: Application to removal of anionic dye from aqueous solution in a batch and fixed-bed reactor. J. Cent. South Univ. 29, 2084–2098 (2022). https://doi.org/10.1007/s11771-022-5065-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11771-022-5065-y