Abstract
Herein we investigated the morphology, chemical characteristics, and source apportionment of fine particulate matter (PM2.5) samples collected from five sites in Jiaxing. Scanning electron microscopy (SEM) and transmission electron microscopy (TEM) showed that soot aggregates and coal-fired fly ash were generally the most abundant components in the samples. All the samples were analyzed gravimetrically for mass concentrations and their various compositions were determined. Our results revealed that the PM2.5 concentrations in the samples were in the following order: winter > spring > autumn > summer. The PM2.5 concentrations in winter and spring were higher than those in autumn and summer, except for inorganic elements. Carbonaceous species and water-soluble inorganic ions were the most abundant components in the samples, accounting for 26.17–50.44% and 34.27–49.6%, respectively. The high secondary organic carbon/organic carbon ratio indicated that secondary organic pollution in Jiaxing was severe. The average ratios of NO3−/SO42−, ranging from 1.01 to 1.25 at the five sites, indicated that mobile pollution sources contributed more to the formation of PM2.5 than stationary sources. The BeP/(BeP + BaP) ratio (0.52–0.71) in samples reflected the influence of transportation from outside of Jiaxing. The positive matrix factorization (PMF) model identified eight main pollution sources: secondary nitrates (26.95%), secondary sulfates (15.49%), secondary organic aerosol (SOA) (19.64%), vehicle exhaust (15.67%), coal combustion (8.6%), fugitive dust (7.7%), ships and heavy oil (5.23%), biomass burning, and other sources (0.91%). Therefore, PM2.5 pollution in Jiaxing during the winter and spring seasons was more severe than that in the summer and autumn. Secondary aerosols were the most important source of PM2.5 pollution; therefore, focus should be placed on controlling gaseous precursors.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Atmospheric particulate matter is a general term for various solid and liquid particulate matter present in the atmosphere. Fine particulate matter (PM2.5) (aerodynamic diameter ≤ 2.5 μm) has received widespread attention because of its harmful effects (Huang et al. 2015; Jung et al. 2009). PM2.5 containing complex chemical composition (Hu et al. 2014) originates from both natural sources and anthropogenic emissions (Kelly and Fussell 2012). In urban areas, PM2.5 is mainly produced from anthropogenic sources including both primary and secondary particles. The latter group is formed during the chemical conversion of gaseous precursor pollutants (Zhang et al. 2015a, b, c).
Since the 1980s, the USA and some European countries have conducted large-scale PM2.5 research, with the source apportionment of PM2.5 and its impact on human health emerging as important research focuses. Receptor models based on pollution sources and contaminated areas, such as the chemical balance model (CMB) and positive matrix factorization model (PMF), have been the most widely used PM2.5 source apportionment technique (Gibson et al. 2015). It is worth mentioning that, the PMF model is based on receptor chemical component data sets for source apportionment without source sample acquisition and is highly recommended when the sources were uncertain (Sharma et al. 2016). The source apportionment results in many parts of the world revealed that the main sources of PM2.5 are secondary inorganic aerosols and organic aerosols, coal combustion, industrial emissions, traffic emissions, biomass combustion, and soil dust (Almeida et al. 2005; Amato and Hopke 2012; Heo et al. 2009).
In the past few decades, with the acceleration of urbanization, the pollution due to PM2.5 in China has become severe, especially in the Pearl River Delta (PRD) (Ho et al. 2014), the Yangtze River Delta (YRD) (Li et al. 2011), and Beijing-Tianjin-Hebei regions (Zhao et al. 2013). For example, in the first quarter of 2013, China experienced extremely severe and persistent haze pollution, which affected a roughly 1.3-million-km2 region and 800 million people (Huang et al. 2015). In recent years, the air quality in China has improved. PM2.5 pollution has been reduced to a certain extent owing to the increased attention it has received. Chinese scholars have conducted a considerable amount of PM2.5 research and obtained many results on PM2.5 pollution characteristics (Sun et al. 2004), chemical composition (Cheng et al. 2015), and source apportionment (Wang et al. 2016; Jiang et al. 2018), the relationship between PM2.5 and other particulate matter (Wang et al. 2008), and the health risks of PM2.5 pollution (Hu et al. 2012). Studies in many regions of China showed that PM2.5 concentrations had obvious seasonal characteristics, with the highest concentration occurring in winter and the lowest in summer (Zhang et al. 2014; Zheng et al. 2005; Louie et al. 2005; Xu et al. 2012; Geng et al. 2006). The source apportionment results showed that the types of pollution sources were similar to those in Europe and the USA, but their contribution rates were different (Wang et al. 2013; Liu et al. 2015; Tao et al. 2014; Zhang et al. 2015a, b, c). However, in the past, few sampling sites and short sampling times have been utilized in research on the PM2.5 concentrations of various scale cities; furthermore, the analysis of samples has not been comprehensive enough.
Jiaxing, a medium-sized city with a population of over 3.5 million in the YRD, is located in the northeast of Zhejiang Province. It is less than 100 km away from cities such as Shanghai, Hangzhou, and Ningbo. The aerosol concentration in Jiaxing is affected by external sources of aerosols from cities around it. Meanwhile, the local pollution sources are complex. Therefore, it is necessary to determine the source apportionment of PM2.5 pollution to develop effective control policies. At present, there are few studies on the source apportionment of PM2.5 in Jiaxing. We, therefore, studied the differences between Jiaxing and surrounding cities, and focused on the differences between five sites in Jiaxing. In this work, we conducted multi-site, long-term sampling in Jiaxing. The morphology and comprehensive pollution characteristics of PM2.5 in Jiaxing were investigated at five sites from October 2016 to November 2017. The PMF model was applied for the source apportionment using chemical composition (organic carbon, elemental carbon, major secondary ions, inorganic elements) of PM2.5 mass.
Materials and method
Site description and PM2.5 sampling
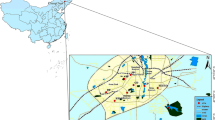
In this study, the PM2.5 samples were collected at Jiaxing. We carried out sampling at five sites: Canlian (CL), Heyuan (HY), Qinghe (QH), Gangqu (GQ), and Tongxiang (TX). Among them, the CL, HY, and QH are located in the Jiaxing city area. CL is located in the new urban area of Jiaxing, and is mainly surrounded by residential areas. QH is located in the business district of the old city and has a dense population and well-developed catering industry. Although HY is located downtown, its surroundings are very special: it is less than 500 m away from the highway in the east, less than 50 m away from the main traffic artery in the west, 1 km away from a coach station, and it is also close to a logistics center and a heavily used military airport. GQ is located in the Gangqu District and is close to the Zhapu Port. There are five polluting enterprises within 1.5 km. TX is located in the outskirts of Jiaxing. The arable land area of Tongxiang accounts for 52.8% of the total land area and is far away from sources of pollution. The location of each representative site is shown in Fig. 1.
During the period from October 2016 to November 2017, we used pairs of Ø90-mm quartz fiber filters to collect PM2.5 samples at five sites simultaneously with mid-volume samplers (Wuhan Tianhong TH Ш, China) at a rate of 100 L/min for a duration of 48 h. The heights of the sites for the CL, QH, HY, GQ, and TX were 20 m, 20 m, 5 m, 20 m, and 10 m, respectively. A total of 440 PM2.5 valid samples were collected. The filters were placed in a humidity chamber (temperature 25 ± 1 °C and relative humidity 50 ± 3%) for 48 h before and after sampling, and weighed with an electronic balance (0.01 mg precision, Sartorius BT 25S, Germany). Then, all sample filters were stored in a − 20 °C freezer until further chemical physical and chemical characterization.
Morphological observation of PM2.5
The morphology of PM2.5 was studied by scanning electron microscopy (SEM) and transmission electron microscopy (TEM). For SEM observation, PM2.5 filter of area 1 cm2 was pasted onto the surface of a double conductive adhesive fixed on an aluminum stub. Then, the sample was coated with gold and put into the sample compartment of the SEM (FEI, Nano 230, USA) for further analysis. For TEM analysis, a portion of the resuspended particles was placed onto carbon-coated copper grids and air-dried. The grids were analyzed with the TEM (FEI, Tecnai G2 spirit Biotwin, USA).
Inorganic elements and water soluble inorganic ions
To test for the presence of the inorganic elements and inorganic water-soluble ions in PM2.5, one eight of the sample filters were cut and ultrasonically extracted for 45 min with a 25-mL nitric acid solution (5 mL HNO3 + 20 mL Milli-Q purified water) and 25 mL Milli-Q purified water (specific resistivity = 18.2 MΩ cm−1; Millipore, Massachusetts, USA), respectively. Then, the mixture was filtered with a 0.45-μm aqueous filter. The presence of inorganic elements (Al, As, Ca, Cr, Fe, K, Mg, Mn, Na, Ni, Si, V, Zn) was determined using an inductively coupled plasma (ICP, Agilent 7600, USA). The anions (NO3−, SO42−) were detected by ion chromatography (ICS, ThermoFisher 5000+, USA), and the cations were detected by ion chromatography (ICS, ThermoFisher 900, USA). The analysis conditions of the instruments are listed in the Supplementary Materials (Table 1).
Carbon component
Carbonaceous aerosols in atmospheric particulate matter are mainly divided into two types: organic carbon (OC) and elemental carbon (EC). OC and EC concentrations on a 0.508 cm2 quartz filter were measured by using a thermal/optical carbon analyzer (DRI, Model 2015, USA). The sample was gradually heated in a pure helium atmosphere at 140 °C (OC1), 280 °C (OC2), 480 °C (OC3), and 580 °C (OC4), and then in a 2% O2/98% He atmosphere at 580 °C (EC1), 740 °C (EC2), and 840 °C (EC3) to release the carbon compounds. The carbon compounds were converted to CO2 by passing the volatilized compounds through an oxidizer (heated manganese dioxide, MnO2), and then the CO2 was quantified by a nondispersive infrared (NDIR) CO2 detector. The OC fraction of the sample might be underestimated, and the EC fraction might include some pyrolyzed OC (OCpyro) because of oxygen-free heating. According to the IMPROVE protocol (Chow et al. 2007), OC = OC1 + OC2 + OC3 + OC4 + OCpyro, EC = EC1 + EC2 + EC3 − OCpyro.
Polycyclic aromatic hydrocarbons
One-half of the sample filters were cut and smashed, and extracted with a mixture containing 20 mL of a dichloromethane and n-hexane mixture (CH3Cl2/C6H14, 1:1 v/v) for 60 min. The extract was then filtered and concentrated to 1 mL under a nitrogen atmosphere. Quantification of polycyclic aromatic hydrocarbons (PAHs) in the collected atmospheric aerosols was carried out by GC-MS (Agilent, 7890A-5975C) with a DB-5 ms capillary column (HP5-MS, 250 μm i.d., 30-m length, 0.25-μm film thickness) under the electron ionization (EI) mode (70 eV). One microliter of the sample was injected into a splitless injector (temperature 300 °C). Helium (purity = 99.999%) was used as the carrier gas at a constant flow rate of 1.0 mL min−1. The temperatures of the transfer line, ion source, and quadrupole were set to 290, 230, and 150 °C, respectively. The temperature program was as follows: heat the sample at 80 °C for 2 min, increase the temperature to 140 °C at a rate of 20 °C/min, increase the temperature to 305 °C at a rate of 3 °C/min, and then hold the temperature for 5 min. The mass spectrometry data was acquired in the full-scan mode (m/z 33–500). Analyses of the average mass spectra calculated at the chromatographic peak middle height were carried out with NIST 2014. The integrations were performed with ChemStation software (Agilent MSD).
PMF
PMF is an effective data analysis method proposed by Paatero and Tapper in 1993 (Paatero and Tapper 1993). The procedure is as follows: First, use the weights to calculate the error of various chemical components in the samples, and then use the least square method to resolve the main sources of contribution and contribution of atmospheric particles. The PMF model is a novel and effective method for the analysis of atmospheric particulate matter sources. Compared with other methods, the PMF model has the following advantages: there is no need to input local source spectrum information, one can use the standard deviation of the data to optimize, the element sharing rate in the decomposition matrix is non-negative, and the model can handle inaccurate data and missing data.
Assuming that X is an n × m matrix, whereby n is the number of samples, and m is the number of chemical components, then matrix X can be decomposed into matrix G and matrix F, where G is the source contribution matrix of the particle emission source of n × p, F is the spectrum matrix of the receptor points of p × m, and p is the number of major pollution sources. The definition is as follows:
where E is the residual matrix, which represents the difference between X and GF.
The purpose of PMF analysis is to minimize Q. Q is defined as
\( {\displaystyle \begin{array}{l}Q=\sum \limits_{i=1}^n\sum \limits_{j=1}^m{\left({e}_{ij}/{S}_{ij}\right)}^2\\ {}{e}_{ij}={\chi}_{ij}=\sum \limits_{k=1}^p{g}_{ik}{f}_{kj}\end{array}} \)
where sij is the uncertainty of the jth species concentration in sample i; and xij, gik, fkj, and eij are the elements of the X, G, F, and E matrices, respectively.
Under the constraints of gik ≥ 0 and fkj ≥ 0, an iterative minimization algorithm is used to solve Q. Then, the pollution source contribution value G (relative value) and pollution source spectrum F (chemical composition relative concentration value) can be determined simultaneously.
Results and discussion
Morphology of PM2.5
SEM and TEM results (Fig. 2) revealed that regular and irregular mineral particles, smooth and similar spherical particles, soot aggregates, etc., were common components of PM2.5. Mineral particles were mainly from resuspended soil and road and construction dust. Smooth spherical particles, which might originate mainly from coal burning, are called “coal-fired fly ash” (Shi et al. 2003). The surface of similar spherical particles is rough and might be a mixture of salt and organic matter (Buseck and Posfai 1999). Coal, motor vehicle exhaust, and biomass combustion are the main sources of soot aggregation (Yue et al. 2006). In Fig. 2c, the chain and cluster-like soot aggregates were gathered by a large number of round particles with a diameter of 30–50 nm. Dye et al. (2000) found that newly generated particles tend to have a chain-like distribution. With the aging, the soot aggregate particles tend to aggregate and had a clustered distribution or a more tightly aggregated distribution. We observed particle types from a variety of sources, indicating that the sources of PM2.5 in Jiaxing were diverse and their formation mechanism was complex.
Mass concentration of PM2.5
According to the experimental results, the maximum daily average PM2.5 concentration during the sampling period was 248.69 μg/m3 (December 5, 2016, GQ), and the lowest value was 16.17 μg/m3 (June 17, 2017, QH); the maximum monthly average value and lowest value of PM2.5 concentration were 132.41 μg/m3 (December 2016, GQ) and 26.74 μg/m3 (July 2017, CL), respectively. Furthermore, the monthly average mass concentrations of PM2.5 were all higher than the WHO average concentration standard (25 μg/m3).
The variation trend of the average PM2.5 concentration for each site was consistent with a “V” distribution (Fig. 3). The concentrations of PM2.5 at the five sites were in the following order: winter > spring > autumn > summer. This seasonal feature was similar to that of the surrounding cities, such as Hangzhou (Bao et al. 2010) and Ningbo (Xiao et al. 2012). In the summer, Jiaxing is affected by the plum rain season (special type of rainfall that mainly occurs in southern China) and the typhoon period, both of which bring about high-frequency and high-volume precipitation. This increases the wet deposition of atmospheric particles during this season, providing effective air self-purification; good atmospheric diffusion conditions also reduce the concentration of pollutants (Wang et al. 2013). However, in winter, the weather conditions are relatively stable, with more inversion temperatures and poor atmospheric diffusion, resulting in relatively high concentrations of particulate matter (Tai et al. 2010). Among the five sites, the PM2.5 levels of GQ and HY were significantly higher than those of other sites. This might be because GQ is located in an industrial zone and is close to a port, while HY is greatly affected by traffic sources. As TX is located in the outer suburbs and away from industrial and traffic sources, its PM2.5 concentration was low. The winter/autumn ratios for PM2.5 concentrations at CL and QH were quite different from those at other sites. In fact, as CL and QH are located in the main urban area, they were not affected by special sources like HY. Therefore, the concentrations of PM2.5 were lower than that at HY, especially in the summer and autumn, when the atmospheric diffusion conditions are relatively good.
Chemical characteristics of PM2.5
General characteristics and chemical composition
This study carried out detailed chemical composition analyses of atmospheric particulates in Jiaxing, with an emphasis on the composition of 13 inorganic elements; four water-soluble ions; OC and EC; and 17 PAH compounds. Studies have shown that the amount of organic matter (OM) in the atmosphere is 1.4–1.6 times that of OC in urban areas (Pathak et al. 2011; Yang et al. 2005). The factor of 1.6 is adopted in this work. The mass ratios of the main chemical components of PM2.5 and the composition of inorganic elements at the five sites were shown in Fig. 4. The total content of inorganic elements in PM2.5 at each site was only 3.67%–8.54%. The content of inorganic elements in GQ was significantly higher than those at other sites because of the severe dust problem in GQ: The roads are badly damaged, and there are several irregular coal yards, docks, and unhardened parking lots (http://www.jepb.gov.cn/News/171/06229b8e959c71b4afa57eaf0241d1854d8b1c25.html). Furthermore, the crustal elements from natural sources at each site were abundant. Ca, Na, Fe, K, and Si accounted for 80% to 88% of the inorganic element concentration, while Mg and Al presented an average of 12%. The unidentified components accounted for 11 to 19% of the total PM2.5 samples. In Fig. 5, except for inorganic elements, the concentrations of the main components at the five sites in winter and spring were all higher than those in autumn and summer. This feature was basically consistent with the seasonal distribution of PM2.5 mass concentration.
Water-soluble inorganic ions
It can be seen from Fig. 4 that the water-soluble ions at each site (except for HY) constituted the largest part of PM2.5, accounting for 34.27–49.6%. SNA (sum of sulfate, nitrate and ammonium) was mainly the product of a photochemical reaction of NOx, SO2, NH3, and other precursors (Squizzato et al. 2013). They were indicators of secondary inorganic aerosol (SIA). The high secondary ion content in PM2.5 at each site indicated that secondary inorganic ions were an important cause of atmospheric heavy pollution. The value of [NO3−]/[SO42−] is often used to characterize the relative contributions of mobile sources and stationary sources. If the ratio of [NO3−]/[SO42−] is greater than 1, it indicates that mobile sources, such as vehicle exhaust, contribute more than stationary sources, such as coal combustion (Arimoto et al. 1996). The average ratios of [NO3−]/[SO42−] at the five sites were greater than 1 (Fig. 6), indicating that the pollution from mobile sources in Jiaxing was severe. The average ratios of [NO3−]/[SO42−] at the five sites were also higher than that recorded for Shanghai (0.64) in 2005 (Wang et al. 2006a, b), indicating that mobile sources have become more and more dominant in the YRD.
Carbon components
Carbonaceous aerosols, including OC and EC, mainly come from industrial boilers/kilns and residential stoves, steel production, and the incomplete combustion of automotive fuel (Bond et al. 2007). Total carbonaceous aerosols (TCA), i.e., the sum of EC and OM, contributed 31%, 50%, 26%, 29%, and 35% to the PM2.5 mass concentrations in GQ, HY, CL, QH, and TX, respectively. In Nanjing (Li et al. 2015) and Shanghai (Cao et al. 2013), TCA accounts for 23% and 32% of the PM2.5 mass concentration, respectively. It can be seen that the ratio of TCA/PM2.5 in Jiaxing was significantly higher than that in Nanjing. Except in HY, the proportions of TCA in Jiaxing and Shanghai were similar. The ultra-high TCA/PM2.5 ratio of HY might be related to the severe vehicle exhaust problem in this location: close to highway, coach station, logistics center, and military airfield.
EC, which is a by-product of the incomplete combustion of carbonaceous materials or pollution migration in surrounding areas, has good chemical stability and is generally used as a tracer for human-made primary pollution sources (Turpin and Huntzicker 1995). The sources of OC are relatively extensive, ranging from primary organic carbon (POC) emitted directly from pollution sources to secondary organic carbon (SOC) generated by chemical reactions in the atmosphere. Therefore, the concentration ratio of OC to EC is often used to evaluate the contribution of secondary sources in atmospheric particles. Researchers compared OC/EC in cities around the world and found that OC/EC > 2 indicated the presence of secondary organic aerosol (SOA) (Chow et al. 1996; Turpin et al. 1990). The higher the ratio, the greater the SOA that have been generated. According to relevant research results (Song et al. 2007; Zhang et al. 2013), we could also adopt 1.6 for the conversion of SOA and SOC.
There is currently no direct way to separate POC from SOC. The EC tracer-based method is a widely applied approach, using EC as the tracer for primary sources and calculating POC in ambient samples with the OC/EC ratios obtained from primary emissions and measured EC levels (Turpin and Huntzicker 1995). However, under normal circumstances, the OC/EC ratio varies with different types of emission sources and can be influenced by many factors, such as the ambient temperature and the carbon element determination method. Therefore, the minimum ratio of test data ((OC/EC)min) can be used instead of the OC/EC ratio (Castro et al. 1999). The estimation equation is as follows:
Owing to the introduction of polar functional groups, such as oxygen and nitrogen, SOC has stronger polarity and hygroscopicity, and has an important influence on visibility reduction, ash formation, and climate change. Many studies have showed that SOC has become an important component of atmospheric particulates, especially fine particulates, and the proportion of SOC in organic carbon is 30%–52% (Cao et al. 2004; Lin and Tai 2001; Na et al. 2004). The SOC/OC ratio in each season at the five sites varied from 35% to 77% (Fig. 7), significantly higher than that in Shanghai (~ 30%) (Feng et al. 2009), indicating that the secondary organic pollution in Jiaxing was severe. Except in the autumn, the SOC/OC ratio at HY was significantly higher than those at other sites, which may be related to its location: close to highways, main roads, bus terminals, logistics centers, and military airports. The SOC/OC ratio at TX was significantly higher than those at other sites in the autumn. This may be due to the large area of cultivated land in Tongxiang District and the widespread burning of straw in the autumn. As shown in Fig. 7, the seasonal patterns of the SOC/OC ratios at each site were not consistent with other YRD cities where higher ratios were also found in the summer than in other seasons (Hou et al. 2011; Cao et al. 2007). The higher SOC/OC in winter and spring resulted partly from a lower (OC/EC)min than that in summer and autumn.
PAHs
PAHs are volatile fused ring compounds produced by incomplete combustion of organic matter such as coal, petroleum, tobacco, and wood. More than 200 species have been discovered so far. The US Environmental Protection Agency (EPA) has classified 16 compounds as superior-grade polycyclic aromatic hydrocarbons: naphthalene, acenaphthylene, acenaphthene, fluorene, phenanthrene, anthracene, fluoranthene, pyrene, benzo[a]anthracene, chrysene, benzo[b]fluoranthene, benzo[k]fluoranthene, benzo[a]pyrene, indeno[1,2,3-cd]pyrene, dibenzo[a,h]anthracene, and benzo[g,h,i]perylene.
We analyzed 16 kinds of superior-grade PAHs and benzo[e]pyrene (BeP), of which there are one, five, four, five, and two kinds of 2–6 rings PAHs. PAHs in the atmosphere can be gaseous and particulate, with low molecular weight PAHs with 2–3 ring mainly existing in gaseous form. The 2-ring PAHs, such as naphthalene, are extremely volatile. The distribution of 4-ring PAHs in gaseous and granular states is basically the same. The PAHs with 5–7 rings, owing to their large molecular weight, exist mostly in particulate form. In Fig. 8a, we focused on the data of PAHs with 4–6 rings at each site for comparison. The total concentrations of PAHs with 4–6 rings at the five sites in winter and spring were significantly higher than those in summer and autumn. This can be partly attributed to the high temperature and strong sunlight, which causes the PAHs to undergo a photochemical reaction, generating regeneration contaminants such as nitro and oxi PAHs (Bidleman et al. 1986; Schauer et al. 2003).
Of the 16 superior-grade PAHs, benzo[a]pyrene (BaP) is highly carcinogenic, so it is often used as a representative of PAHs (Yassaa et al. 2001). The concentrations of BaP at the five sites ranged from 0.07 to 1.95 ng/m3, with an average of 0.79 ng/m3, which was significantly lower than that in Hangzhou (1.6 ng/m3) (Feng et al. 2015). Only very few days exceeded the new air quality standard of China (1 ng/m3). BaP is unstable and prone to photochemical reactions. Most of the fresh exhaust gases had similar BaP and BeP content, the ratio of BeP/(BeP + BaP) could be regarded as an index of the aging of particles (Muránszky et al. 2011). In addition, the sources of atmospheric particles can be divided into two types: local generation and external transportation. Owing to long-distance transportation, foreign particles inevitably age compared to local particles. Therefore, during the same season, under similar weather conditions, the ratio of BeP/(BeP + BaP) could also reflect the relative contribution of foreign particles. In this study, the ratios of BeP/(BeP + BaP) in the summer were the highest at the five sites (Fig. 8b) because of the preferential loss of BaP through photo-degradation. The ratios of BeP/(BeP + BaP) during all the seasons were greater than 0.5, indicating that PM2.5 in Jiaxing had generally aged, and there was a certain degree of external transportation problems.
The source apportionment of PM2.5
In this study, PMF 5.0 issued by the US EPA was used to calculate the total of 440 valid sample data from five sites. The input sample data included 19 chemical components, PM2.5 mass concentration, and the uncertainty data for each component. Eight sources were obtained via minimized Q value with an optimal simulation result (Fig. 9).
Source 1 was designated as secondary nitrates because it was enriched with nitrate (NO3−) and ammonium (NH4+) ions, as well as some sulfate (SO42−) ions. Source 2 was identified as secondary sulfates because of the high abundance of SO42− and NH4+ in the source profile. These two sources could be collectively referred to as secondary inorganic aerosols (SIA). SNA is the main SIA component in PM2.5 and is mainly present in the form of (NH4)2SO4 and NH4NO3. Both are produced by neutralizing sulfuric acid (H2SO4) and nitric acid (HNO3) with ammonia (NH3), respectively (Squizzato et al. 2013; Stockwell et al. 2003).
OC had the highest contribution rate to source 3 and was much higher than EC, so source 3 was classified as SOA. Source 4 was vehicle exhaust, which was identified by the high contribution of EC. Many studies have shown that EC is a characteristic component of motor vehicle exhaust (Lin et al. 2015; Chan et al. 1999).
Source 5 represented coal combustion sources. The relative content of Cl− in this source was high. Yao et al. (2003) found that, in many cities in China, owing to coal burning, high concentrations of Cl− are often present in PM2.5 in winter. In addition, some studies also showed that Cl− was a tracer element of coal-fired sources (Xiu et al. 2004; Duan et al. 2006; Han et al. 2010; Yu et al. 2011). Coal still occupies a considerable proportion in the energy structure of cities in China. Therefore, coal-fired sources have always been an important source of urban particulate matter.
Source 6 was estimated to be fugitive dust because it contains high percentages of the elements Si, Ca, Mg, Al, Fe, Ca, and Mg, which are used to identify cement dust (Xue et al. 2010; Kim et al. 2004). Cement dust originates from cement, gypsum plants, and construction areas. Many studies have also confirmed that soil dust contains a large number of crustal elements, such as Si, Al, Fe, Ca, and Mg (Lee et al. 1999; Chueinta et al. 2000; Zhang et al. 2013; Kong et al. 2010). The average percentage of SiO2, Al2O3, Fe2O3, MgO, and CaO in soil are 57.8%–64.8%, 14.8%–16.7%, 4.6%–9.1%, 2.7%–4.4%, and 3.4%–7.5%, respectively.
Source 7 represented ships and heavy oil. Heavy oil is the part of crude oil that remains after gasoline and diesel oil are extracted. A significant source of heavy oil combustion is ship emissions and might also include land transport and industrial sources. Ni and V are common tracers of heavy oil combustion (Vallius et al. 2005). Yuan et al. used the PMF model to analyze the sources of PM10 in Hong Kong and also found a heavy oil combustion source with V as the marker, and considered it to be mainly from ship emissions (Yuan et al. 2006). As Jiaxing has a port and the river network is densely populated, ship and heavy oil pollution were common.
Source 8 had the highest characteristic value of K, and the characteristic values of Na, Zn, Cr, and Fe were also high. Owing to the use of potash fertilizer, crops are rich in K, and the particles released from combustion are rich in K, so K is considered an important tracer for biomass combustion (Simoneit et al. 1999). Some studies showed that the concentrations of Na (Yli-Tuomi et al. 2003; Begum et al. 2004) were high in the by-products of biomass burning in China. Zn, Fe, and Cr might be derived from garbage incineration, which is often carried out with open-air biomass combustion. They might also originate from other sources of pollution. Therefore, we classified factor 8 as biomass burning and others.
The source apportionment results in five sites were shown in Fig. 10: secondary nitrates (26.95%), secondary sulfates (15.49%), SOA (19.64%), vehicle exhaust (15.67%), coal combustion (8.6%), fugitive dust (7.7%), ships and heavy oil (5.23%), and biomass burning and other sources (0.91%), including three secondary sources and five primary sources.
Contribution rate of PM2.5 pollution sources in Jiaxing. Secondary nitrates, secondary sulfates, SOA, vehicle exhaust, coal combustion, fugitive dust, ships and heavy oil, biomass burning, and other sources were identified. Gangqu site (GQ), Tongxiang site (TX), Heyuan site (HY), Canlian site (CL), Qinghe site (QH)
First, the contribution of secondary aerosols (including SIA and SOA) to PM2.5 was generally large, from 43.33% to 69.76%. The contributions of SIA at the CL and QH sites were significantly higher than those at other sites, which was consistent with the results of the component analysis. The contribution of vehicle exhaust at the HY site was much higher than those in the CL and QH sites because HY is close to highways, trunk roads, automobile passenger terminals, military airports, and logistics centers. The contribution rate of the SOA at the HY site was also the highest. In fact, motor vehicles produce OC and EC (Lin et al. 2015). Based on the findings in this study, the high SOC/OC ratio indicated that OC contained a large amount of SOC. According to relevant research results (Song et al. 2007; Zhang et al. 2013), SOA can be estimated by multiplying SOC by a factor of 1.6. Hence, there was a strong correlation between vehicle exhaust and SOA. The contribution rate of dust at GQ was the highest among the five sites, which was consistent with the severe dust pollution in GQ. The contribution rate of coal combustion at GQ was the highest compared to those at other sites and was almost twice that of TX. This is probably because of the industrial coal consumption in Gangqu District, which in 2015 was nearly seven times that of Tongxiang District (Report on Source Release of Atmospheric Pollutants in Jiaxing City (2015)). Owing to the large number of rivers in Jiaxing, there are heavy oil pollution problems at each site, and there was little difference in contribution rates between the sites. The contributions due to ships and heavy oil on GQ was greater than those at other sites, as GQ is near a port. With respect to biomass burning and others, the contribution rates at all the sites were generally very low, and the contribution rate at TX was the highest, as TX is located in an ecological suburb and has a large area of cultivated land. A previous study found that, in 2010, biomass burning accounted for about 10% of the primary emission of PM2.5 in the YRD region (Fu et al. 2013). Therefore, our findings seem to suggest that the problem of biomass burning in Jiaxing has greatly improved in recent years, even in Tongxiang District, where the cultivated land is large.
Conclusion
Jiaxing City is located in the Yangtze River Delta economic circle. The local and surrounding pollution sources are complex and diverse. Therefore, many kinds of particulate matter were observed in the SEM and TEM images; however, the artificial sources represented by vehicle exhaust and coal combustion might be the main sources.
The seasonal variation in PM2.5 concentration in Jiaxing was as follows: winter > spring > autumn > summer, due to the climate characteristics of Jiaxing City. The PM2.5 concentration levels in GQ (closing to the port) and HY (closing to many traffic sources) were relatively high, indicating that a mobile source was an important source of fine particles. Carbon components and water-soluble ions exceeded three quarters of the total mass of PM2.5, and their seasonal variation was similar to that of PM2.5 concentration.
The [NO3−]/[SO42−] ratio of Jiaxing City was higher than 1 and higher than that of Shanghai in 2005, indicating that the influence of mobile sources in YRD region had increased in recent years and was already dominant. The TCA/PM2.5 ratio in Jiaxing was higher than that in Nanjing, which was equivalent to Shanghai, except for the HY. The SOC/OC ratio of Jiaxing was higher than that of Shanghai, and its seasonal characteristics were different from that obtained for the YRD region. The average annual concentration of BaP in Jiaxing (0.79 ng/m3) was much lower than that in Hangzhou (1.6 ng/m3), and lower than the Chinese national standard (1 ng/m3). The BeP/(BeP + BaP) ratio showed that PM2.5 in Jiaxing had generally aged and was affected by external sources.
The results of the source apportionment showed that secondary aerosols dominate; therefore, it is important to control the emissions of the gaseous precursors to reduce PM2.5 concentrations. Compared with the pollution sources at other sites, vehicle exhaust in HY, coal combustion and fugitive dust in the GQ, and biomass burning in TX contributed prominently to the PM2.5 concentrations.
In short, the pollution status of PM2.5 in Jiaxing had both commonalities with and was different from those in the surrounding cities. There were also differences at each site. Therefore, control policies in different regions should focus on different sources, and a refined control strategy should be formulated.
References
Almeida SM, Pio CA, Freitas MC et al (2005) Source apportionment of fine and coarse particulate matter in a sub-urban area at the Western European coast. Atmos Environ 39:3127–3138. https://doi.org/10.1016/j.atmosenv.2005.01.048
Amato F, Hopke PK (2012) Source apportionment of the ambient PM2.5 across St. Louis using constrained positive matrix factorization. Atmos Environ 46:329–337. https://doi.org/10.1016/j.atmosenv.2011.09.062
Arimoto R, Duce RA, Savoie DL, Prospero JM, Talbot R, Cullen JD, Tomza U, Lewis NF, Ray BJ (1996) Relationships among aerosol constituents from Asia and the North Pacific during PEM-West a. J Geophys Res Atmos 101:2011–2023. https://doi.org/10.1029/95JD01071
Bao Z, Feng YC, Jiao L et al (2010) Characterization and source apportionment of PM2. 5 and PM10 in Hangzhou. Environ Monit China 26(2):44–48
Begum BA, Kim E, Biswas SK, Hopke PK (2004) Investigation of sources of atmospheric aerosol at urban and semi-urban areas in Bangladesh. Atmos Environ 38:3025–3038. https://doi.org/10.1016/j.atmosenv.2004.02.042
Bidleman TF, Billings WN, Foreman WT (1986) Vapor-particle partitioning of semivolatile organic compounds: estimates from field collections. Environ Sci Technol 20:1038–1043. https://doi.org/10.1021/es00152a013
Bond TC, Bhardwaj E, Dong R, Jogani R, Jung S, Roden C, Streets DG, Trautmann NM (2007) Historical emissions of black and organic carbon aerosol from energy-related combustion, 1850-2000. Glob Biogeochem Cycles 21:1–16. https://doi.org/10.1029/2006GB002840
Buseck PR, Posfai M (1999) Airborne minerals and related aerosol particles: effects on climate and the environment. Proc Natl Acad Sci 96:3372–3379. https://doi.org/10.1073/pnas.96.7.3372
Cao JJ, Lee SC, Ho KF, Zou SC, Fung K, Li Y, Watson JG, Chow JC (2004) Spatial and seasonal variations of atmospheric organic carbon and elemental carbon in Pearl River Delta region, China. Atmos Environ 38:4447–4456. https://doi.org/10.1016/j.atmosenv.2004.05.016
Cao JJ, Lee SC, Chow JC, Watson JG, Ho KF, Zhang RJ, Jin ZD, Shen ZX, Chen GC, Kang YM, Zou SC, Zhang LZ, Qi SH, Dai MH, Cheng Y, Hu K (2007) Spatial and seasonal distributions of carbonaceous aerosols over China. J Geophys Res Atmos 112:1–9. https://doi.org/10.1029/2006JD008205
Cao J, Zhu C, Tie X et al (2013) Characteristics and sources of carbonaceous aerosols from Shanghai, China. Atmos Chem Phys 13(2):803–817. https://doi.org/10.5194/acp-13-803-2013
Castro LM, Pio CA, Harrison RM, Smith DJT (1999) Carbonaceous aerosol in urban and rural European atmospheres: estimation of secondary organic carbon concentrations. Atmos Environ 33:2771–2781. https://doi.org/10.1016/S1352-2310(98)00331-8
Chan YC, Simpson RW, Mctainsh GH, Vowles PD, Cohen DD, Bailey GM (1999) Source apportionment of PM and PM aerosols in Brisbane ( Australia ) by receptor modelling. Atmos Environ 33:3251–3268
Cheng Y, He K, Bin DZY et al (2015) Humidity plays an important role in the PM2.5 pollution in Beijing. Environ Pollut 197:68–75. https://doi.org/10.1016/j.envpol.2014.11.028
Chow JC, Watson JG, Lu Z, Lowenthal DH, Frazier CA, Solomon PA, Thuillier RH, Magliano K (1996) Descriptive analysis of PM2.5 and PM10 at regionally representative locations during SJVAQS/AUSPEX. Atmos Environ 30:2079–2112. https://doi.org/10.1016/1352-2310(95)00402-5
Chow JC, Watson JG, Chen LWA, Chang MCO, Robinson NF, Trimble D, Kohl S (2007) The IMPROVE_A temperature protocol for thermal/optical carbon analysis: maintaining consistency with a long-term database. J Air Waste Manage Assoc 57:1014–1023. https://doi.org/10.3155/1047-3289.57.9.1014
Chueinta W, Hopke PK, Paatero P (2000) Investigation of sources of atmospheric aerosol at urban and suburban residential areas in Thailand by positive matrix factorization. Atmos Environ 34:3319–3329. https://doi.org/10.1016/S1352-2310(99)00433-1
Duan FK, He KB, Ma YL et al (2006) Concentration and chemical characteristics of PM2.5 in Beijing, China: 2001-2002. Sci Total Environ 355:264–275. https://doi.org/10.1016/j.scitotenv.2005.03.001
Dye AL, Rhead MM, Trier CJ (2000) The quantitative morphology of roadside and background urban aerosol in Plymouth, UK. Atmos Environ 34:3139–3148. https://doi.org/10.1016/S1352-2310(99)00437-9
Feng Y, Chen Y, Guo H, Zhi G, Xiong S, Li J, Sheng G, Fu J (2009) Characteristics of organic and elemental carbon in PM2.5 samples in Shanghai, China. Atmos Res 92:434–442. https://doi.org/10.1016/j.atmosres.2009.01.003
Feng J, Hu J, Xu B, Hu X, Sun P, Han W, Gu Z, Yu X, Wu M (2015) Characteristics and seasonal variation of organic matter in PM 2.5 at a regional background site of the Yangtze River Delta region, China. Atmos Environ 123:288–297. https://doi.org/10.1016/j.atmosenv.2015.08.019
Fu X, Wang S, Zhao B, Xing J, Cheng Z, Liu H, Hao J (2013) Emission inventory of primary pollutants and chemical speciation in 2010 for the Yangtze River Delta region, China. Atmos Environ 70:39–50. https://doi.org/10.1016/j.atmosenv.2012.12.034
Geng H, Meng Z, Zhang Q (2006) In vitro responses of rat alveolar macrophages to particle suspensions and water-soluble components of dust storm PM2.5. Toxicol in Vitro 20:575–584. https://doi.org/10.1016/j.tiv.2005.09.015
Gibson MD, Haelssig J, Pierce JR, Parrington M, Franklin JE, Hopper JT, Li Z, Ward TJ (2015) A comparison of four receptor models used to quantify the boreal wildfire smoke contribution to surface PM2.5 in Halifax, Nova Scotia during the BORTAS-B experiment. Atmos Chem Phys 15:815–827. https://doi.org/10.5194/acp-15-815-2015
Han B, Kong S, Bai Z, du G, Bi T, Li X, Shi G, Hu Y (2010) Characterization of elemental species in PM2.5 samples collected in four cities of Northeast China. Water Air Soil Pollut 209:15–28. https://doi.org/10.1007/s11270-009-0176-8
Heo J-B, Hopke PK, Yi S-M (2009) Source apportionment of PM2.5 in Seoul, Korea. Atmos Chem Phys 9:4957–4971. https://doi.org/10.5194/acp-9-4957-2009
Ho KF, Engling G, Sai Hang Ho S, Huang R, Lai S, Cao J, Lee SC (2014) Seasonal variations of anhydrosugars in PM 2.5 in the Pearl River Delta region, China. Tellus Ser B Chem Phys Meteorol 66:22577. https://doi.org/10.3402/tellusb.v66.22577
Hou B, Zhuang G, Zhang R, Liu T, Guo Z, Chen Y (2011) The implication of carbonaceous aerosol to the formation of haze : revealed from the characteristics and sources of OC / EC over a mega-city in China. J Hazard Mater 190:529–536. https://doi.org/10.1016/j.jhazmat.2011.03.072
Hu X, Zhang Y, Ding Z, Wang T, Lian H, Sun Y, Wu J (2012) Bioaccessibility and health risk of arsenic and heavy metals (Cd, Co, Cr, Cu, Ni, Pb, Zn and Mn) in TSP and PM2.5 in Nanjing, China. Atmos Environ 57:146–152. https://doi.org/10.1016/j.atmosenv.2012.04.056
Hu G, Zhang Y, Sun J, Zhang L, Shen X, Lin W, Yang Y (2014) Variability, formation and acidity of water-soluble ions in PM2.5 in Beijing based on the semi-continuous observations. Atmos Res 145–146:1–11. https://doi.org/10.1016/j.atmosres.2014.03.014
Huang RJ, Zhang Y, Bozzetti C, Ho KF, Cao JJ, Han Y, Daellenbach KR, Slowik JG, Platt SM, Canonaco F, Zotter P, Wolf R, Pieber SM, Bruns EA, Crippa M, Ciarelli G, Piazzalunga A, Schwikowski M, Abbaszade G, Schnelle-Kreis J, Zimmermann R, An Z, Szidat S, Baltensperger U, Haddad IE, Prévôt ASH (2015) High secondary aerosol contribution to particulate pollution during haze events in China. Nature 514:218–222. https://doi.org/10.1038/nature13774
Jiang N, Li Q, Su F, Wang Q, Yu X, Kang P, Zhang R, Tang X (2018) Chemical characteristics and source apportionment of PM2.5 between heavily polluted days and other days in Zhengzhou, China. J Environ Sci (China) 66:188–198. https://doi.org/10.1016/j.jes.2017.05.006
Jung J, Lee H, Kim YJ, Liu X, Zhang Y, Gu J, Fan S (2009) Aerosol chemistry and the effect of aerosol water content on visibility impairment and radiative forcing in Guangzhou during the 2006 Pearl River Delta campaign. J Environ Manag 90:3231–3244. https://doi.org/10.1016/j.jenvman.2009.04.021
Kelly FJ, Fussell JC (2012) Size, source and chemical composition as determinants of toxicity attributable to ambient particulate matter. Atmos Environ 60:504–526. https://doi.org/10.1016/j.atmosenv.2012.06.039
Kim E, Hopke PK, Edgerton ES (2004) Improving source identification of Atlanta aerosol using temperature resolved carbon fractions in positive matrix factorization. Atmos Environ 38:3349–3362. https://doi.org/10.1016/j.atmosenv.2004.03.012
Kong S, Han B, Bai Z, Chen L, Shi J, Xu Z (2010) Receptor modeling of PM2.5, PM10, and TSP in different seasons and long-range transport analysis at a coastal site of Tianjin, China. Sci Total Environ 408:4681–4694. https://doi.org/10.1016/j.scitotenv.2010.06.005
Lee E, Chan CK, Paatero P (1999) Application of positive matrix factortization in source apportionment of particulate pollutants in Hong Kong. Atmos Environ 33:3201–3212. https://doi.org/10.1016/S1352-2310(99)00113-2
Li L, Chen CH, Fu JS, Huang C, Streets DG, Huang HY, Zhang GF, Wang YJ, Jang CJ, Wang HL, Chen YR, Fu JM (2011) Air quality and emissions in the Yangtze River Delta, China. Atmos Chem Phys 11:1621–1639. https://doi.org/10.5194/acp-11-1621-2011
Li B, Zhang J, Zhao Y, Yuan S, Zhao Q, Shen G, Wu H (2015) Seasonal variation of urban carbonaceous aerosols in a typical city Nanjing in Yangtze River Delta, China. Atmos Environ 106:223–231. https://doi.org/10.1016/j.atmosenv.2015.01.064
Lin JJ, Tai HS (2001) Concentrations and distributions of carbonaceous species in ambient particles in Kaohsiung City, Taiwan. Atmos Environ 35:2627–2636. https://doi.org/10.1016/S1352-2310(00)00444-1
Lin YC, Tsai CJ, Wu YC, Zhang R, Chi KH, Huang YT, Lin SH, Hsu SC (2015) Characteristics of trace metals in traffic-derived particles in Hsuehshan tunnel, Taiwan: size distribution, potential source, and fingerprinting metal ratio. Atmos Chem Phys 15:4117–4130. https://doi.org/10.5194/acp-15-4117-2015
Liu G, Li J, Wu D, Xu H (2015) Chemical composition and source apportionment of the ambient PM2.5 in Hangzhou, China. Particuology 18:135–143. https://doi.org/10.1016/j.partic.2014.03.011
Louie PKK, Watson JG, Chow JC et al (2005) Seasonal characteristics and regional transport of PM2.5 in Hong Kong. Atmos Environ 39:1695–1710. https://doi.org/10.1016/j.atmosenv.2004.11.017
Muránszky G, Ovari M, Virág I et al (2011) Chemical characterization of PM10 fractions of urban aerosol. Microchem J 98:1–10. https://doi.org/10.1016/j.microc.2010.10.002
Na K, Sawant AA, Song C, Cocker DR (2004) Primary and secondary carbonaceous species in the atmosphere of Western Riverside County, California. Atmos Environ 38:1345–1355. https://doi.org/10.1016/j.atmosenv.2003.11.023
Paatero P, Tapper U (1993) Analysis of different modes of factor analysis as least squares fit problems. Chemom Intell Lab Syst 18:183–194. https://doi.org/10.1016/0169-7439(93)80055-M
Pathak RK, Wang T, Ho KF, Lee SC (2011) Characteristics of summertime PM2.5 organic and elemental carbon in four major Chinese cities: implications of high acidity for water-soluble organic carbon (WSOC). Atmos Environ 45:318–325. https://doi.org/10.1016/j.atmosenv.2010.10.021
Report on Source Release of Atmospheric Pollutants in Jiaxing City (2015)
Schauer C, Niessner R, Pöschl U (2003) Polycyclic aromatic hydrocarbons in urban air particulate matter: decadal and seasonal trends, chemical degradation, and sampling artifacts. Environ Sci Technol 37:2861–2868. https://doi.org/10.1021/es034059s
Sharma SK, Mandal TK, Jain S, Saraswati, Sharma A, Saxena M (2016) Source apportionment of PM2.5 in Delhi, India using PMF model. Bull Environ Contam Toxicol 97:286–293. https://doi.org/10.1007/s00128-016-1836-1
Shi Z, Shao L, Jones TP, Whittaker AG, Lu S, Bérubé KA, He T, Richards RJ (2003) Characterization of airborne individual particles collected in an urban area, a satellite city and a clean air area in Beijing, 2001. Atmos Environ 37:4097–4108. https://doi.org/10.1016/S1352-2310(03)00531-4
Simoneit BRT, Schauer JJ, Nolte CG, Oros DR, Elias VO, Fraser MP, Rogge WF, Cass GR (1999) Levoglucosan, a tracer for cellulose in biomass burning and atmospheric particles. Atmos Environ 33:173–182. https://doi.org/10.1016/S1352-2310(98)00145-9
Song Y, Tang X, Xie S, Zhang Y, Wei Y, Zhang M, Zeng L, Lu S (2007) Source apportionment of PM2.5 in Beijing in 2004. J Hazard Mater 146:124–130. https://doi.org/10.1016/j.jhazmat.2006.11.058
Squizzato S, Masiol M, Brunelli A, Pistollato S, Tarabotti E, Rampazzo G, Pavoni B (2013) Factors determining the formation of secondary inorganic aerosol: a case study in the Po Valley (Italy). Atmos Chem Phys 13:1927–1939. https://doi.org/10.5194/acp-13-1927-2013
Stockwell WR, Kuhns H, Etyemezian V et al (2003) The Treasure Valley secondary aerosol study II: modeling of the formation of inorganic secondary aerosols and precursors for southwestern Idaho. Atmos Environ 37:525–534. https://doi.org/10.1016/S1352-2310(02)00895-6
Sun Y, Zhuang G, Wang Y, Han L, Guo J, Dan M, Zhang W, Wang Z, Hao Z (2004) The air-borne particulate pollution in Beijing—concentration, composition, distribution and sources. Atmos Environ 38:5991–6004. https://doi.org/10.1016/j.atmosenv.2004.07.009
Tai APK, Mickley LJ, Jacob DJ (2010) Correlations between fine particulate matter (PM2.5) and meteorological variables in the United States: implications for the sensitivity of PM2.5 to climate change. Atmos Environ 44:3976–3984. https://doi.org/10.1016/j.atmosenv.2010.06.060
Tao J, Gao J, Zhang L, Zhang R, Che H, Zhang Z, Lin Z, Jing J, Cao J, Hsu SC (2014) PM2.5 pollution in a megacity of Southwest China: source apportionment and implication. Atmos Chem Phys 14:8679–8699. https://doi.org/10.5194/acp-14-8679-2014
Turpin BJ, Huntzicker JJ (1995) Identification of secondary organic aerosol episodes and quantitation of primary and secondary organic aerosol concentrations during SCAQS. Atmos Environ 29:3527–3544. https://doi.org/10.1016/1352-2310(94)00276-Q
Turpin BJ, Cary RA, Huntzicker JJ (1990) An in situ, time-resolved analyzer for aerosol organic and elemental carbon. Aerosol Sci Technol 12:19–161. https://doi.org/10.1080/02786829008959336
Vallius M, Janssen NAH, Heinrich J, Hoek G, Ruuskanen J, Cyrys J, van Grieken R, de Hartog JJ, Kreyling WG, Pekkanen J (2005) Sources and elemental composition of ambient PM2.5 in three European cities. Sci Total Environ 337:147–162. https://doi.org/10.1016/j.scitotenv.2004.06.018
Wang X, Bi X, Sheng G, Fu J (2006a) Chemical composition and sources of PM10 and PM2.5 aerosols in Guangzhou, China. Environ Monit Assess 119:425–439. https://doi.org/10.1007/s10661-005-9034-3
Wang Y, Zhuang G, Zhang X, Huang K (2006b) The ion chemistry, seasonal cycle, and sources of PM 2.5 and TSP aerosol in Shanghai. Atmos Environ 40:2935–2952. https://doi.org/10.1016/j.atmosenv.2005.12.051
Wang H, Zhuang Y, Wang Y et al (2008) Long-term monitoring and source apportionment of PM2.5/PM10 in Beijing, China. J Environ Sci 20:1323–1327. https://doi.org/10.1016/S1001-0742(08)62228-7
Wang J, Hu Z, Chen Y, Chen Z, Xu S (2013) Contamination characteristics and possible sources of PM10 and PM2.5 in different functional areas of Shanghai, China. Atmos Environ 68:221–229. https://doi.org/10.1016/j.atmosenv.2012.10.070
Wang Y, Jia C, Tao J, Zhang L, Liang X, Ma J, Gao H, Huang T, Zhang K (2016) Chemical characterization and source apportionment of PM2.5 in a semi-arid and petrochemical-industrialized city, Northwest China. Sci Total Environ 573:1031–1040. https://doi.org/10.1016/j.scitotenv.2016.08.179
Xiao ZM, Bi XH, Feng YC et al (2012) Source apportionment of ambient PM10 and PM2.5 in urban area of Ningbo city. Res Environ Sci 25:549–555
Xiu G, Zhang D, Chen J, Huang X, Chen Z, Guo H, Pan J (2004) Characterization of major water-soluble inorganic ions in size-fractionated particulate matters in Shanghai campus ambient air. Atmos Environ 38:227–236. https://doi.org/10.1016/j.atmosenv.2003.09.053
Xu L, Chen X, Chen J, Zhang F, He C, Zhao J, Yin L (2012) Seasonal variations and chemical compositions of PM2.5 aerosol in the urban area of Fuzhou, China. Atmos Res 104–105:264–272. https://doi.org/10.1016/j.atmosres.2011.10.017
Xue YH, Wu JH, Feng YC, Dai L, Bi XH, Li X, Zhu T, Tang SB, Chen MF (2010) Source characterization and apportionment of PM10 in Panzhihua, China. Aerosol Air Qual Res 10:367–377. https://doi.org/10.4209/aaqr.2010.01.0002
Yang F, He K, Ye B, Chen X, Cha L, Cadle SH, Chan T, Mulawa PA (2005) One-year record of organic and elemental carbon in fine particles in downtown Beijing and Shanghai. Atmos Chem Phys 5:1449–1457. https://doi.org/10.5194/acpd-5-217-2005
Yao X, Lau APS, Fang M, Chan CK, Hu M (2003) Size distributions and formation of ionic species in atmospheric particulate pollutants in Beijing, China: 1—inorganic ions. Atmos Environ 37:2991–3000. https://doi.org/10.1016/S1352-2310(03)00255-3
Yassaa N, Meklati BY, Cecinato A, Marino F (2001) Particulate n-alkanes, n-alkanoic acids and polycyclic aromatic hydrocarbons in the atmosphere of Algiers City area. Atmos Environ 35:1843–1851
Yli-Tuomi T, Venditte L, Hopke PK, Basunia MS, Landsberger S, Viisanen Y, Paatero J (2003) Composition of the Finnish Arctic aerosol: collection and analysis of historic filter samples. Atmos Environ 37:2355–2364. https://doi.org/10.1016/S1352-2310(03)00164-X
Yu Y, Schleicher N, Norra S, Fricker M, Dietze V, Kaminski U, Cen K, Stüben D (2011) Dynamics and origin of PM2.5during a three-year sampling period in Beijing, China. J Environ Monit 13:334–346. https://doi.org/10.1039/c0em00467g
Yuan Z, Lau AKH, Zhang H et al (2006) Identification and spatiotemporal variations of dominant PM10 sources over Hong Kong. Atmos Environ 40:1803–1815. https://doi.org/10.1016/j.atmosenv.2005.11.030
Yue W, Li X, Liu J, Li Y, Yu X, Deng B, Wan T, Zhang G, Huang Y, He W, Hua W, Shao L, Li W, Yang S (2006) Characterization of PM2.5 in the ambient air of Shanghai city by analyzing individual particles. Sci Total Environ 368:916–925. https://doi.org/10.1016/j.scitotenv.2006.03.043
Zhang R, Jing J, Tao J, Hsu SC, Wang G, Cao J, Lee CSL, Zhu L, Chen Z, Zhao Y, Shen Z (2013) Chemical characterization and source apportionment of PM2.5 in Beijing: seasonal perspective. Atmos Chem Phys 13:7053–7074. https://doi.org/10.5194/acp-13-7053-2013
Zhang R, Jing J, Tao J, Hsu SC, Wang G, Cao J, Lee CSL, Zhu L, Chen Z, Zhao Y, Shen Z (2014) Erratum: to chemical characterization and source apportionment of PM2.5 in Beijing: seasonal perspective published in (atmospheric chemistry and physics (2013) 13 (7053-7074)). Atmos Chem Phys 14:175. https://doi.org/10.5194/acp-14-175-2014
Zhang F, wu WZ, rong CH et al (2015a) Seasonal variations and chemical characteristics of PM2.5 in Wuhan, Central China. Sci Total Environ 518–519:97–105. https://doi.org/10.1016/j.scitotenv.2015.02.054
Zhang N, Han B, He F, Xu J, Niu C, Zhou J, Kong S, Bai Z, Xu H (2015b) Characterization, health risk of heavy metals, and source apportionment of atmospheric PM2.5 to children in summer and winter: an exposure panel study in Tianjin, China. Air Qual Atmos Health 8:347–357. https://doi.org/10.1007/s11869-014-0289-0
Zhang R, Wang G, Guo S, Zamora ML, Ying Q, Lin Y, Wang W, Hu M, Wang Y (2015c) Formation of urban fine particulate matter. Chem Rev 115:3803–3855. https://doi.org/10.1021/acs.chemrev.5b00067
Zhao PS, Dong F, He D, Zhao XJ, Zhang XL, Zhang WZ, Yao Q, Liu HY (2013) Characteristics of concentrations and chemical compositions for PM2.5 in the region of Beijing, Tianjin, and Hebei, China. Atmos Chem Phys 13:4631–4644. https://doi.org/10.5194/acp-13-4631-2013
Zheng M, Salmon LG, Schauer JJ, Zeng L, Kiang CS, Zhang Y, Cass GR (2005) Seasonal trends in PM2.5 source contributions in Beijing, China. Atmos Environ 39:3967–3976. https://doi.org/10.1016/j.atmosenv.2005.03.036
Acknowledgments
This study was funded by the Municipal Environmental Protection Bureau of Jiaxing (JXSJ-2014-73), and Shanghai Natural Science Foundation of the Science and Technology Commission of Shanghai Municipal Government (No. 18ZR1418300).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Additional information
Responsible editor: Gerhard Lammel
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• The concentrations of PM2.5 in five sites were in accordance with the rule that winter > spring > autumn > summer.
• Secondary aerosols were the most important source of PM2.5 pollution in Jiaxing.
• Mobile sources contributed more to the formation of PM2.5 than stationary sources in Jiaxing.
Electronic supplementary material
ESM 1
(DOCX 15 kb)
Rights and permissions
About this article
Cite this article
Zhao, Z., Lv, S., Zhang, Y. et al. Characteristics and source apportionment of PM2.5 in Jiaxing, China. Environ Sci Pollut Res 26, 7497–7511 (2019). https://doi.org/10.1007/s11356-019-04205-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-04205-2