Abstract
Nitrogen (N) fixed by termites was evaluated as a N input to decomposition processes in two tropical forests, a dry deciduous forest (DDF) and the neighboring dry evergreen forest (DEF), Thailand. A diverse group of termite species were assayed by acetylene reduction method and only the wood/litter-feeding termites were found to fix N. More intensive samplings of two abundant species, Microcerotermes crassus and Globitermes sulphureus, were done across several seasons, suggesting N fixation rates of 0.21 and 0.28 kg ha−1 y−1 by termites in the DDF and DEF, respectively. Also, estimates of asymbiotic N fixation rates were 0.75 and 3.95 kg ha−1 y−1. N fixed by termites and by asymbiotic fixers is directly supplied to decomposers breaking down dead plant material and could be a major source of their N. N fixed by termites was 7–22% of that fixed by termites and asymbiotic fixers. Although N fixed by termites is a small input compared to other inputs, this N is likely important for decomposition processes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
INTRODUCTION
Nitrogen (N) is an element that affects not only primary production (Vitousek and Howarth 1991; Vitousek and others 2002), but also the decomposition of dead plant material (Hunt and others 1988; Vitousek and others 2002). In terrestrial ecosystems, N inputs occur through biological fixation and atmospheric deposition (Jordan 1985; Cleveland and others 1999; Son 2001) and N is lost primarily through leaching and gaseous losses (Vitousek and others 2002; Hall and Matson 2003). Biological N fixation occurs in plant symbiotic, free-living (asymbiotic), and xylophagous arthropod-gut symbiotic forms (Breznak 1975; Cleveland and others 1999; Nardi and others 2002); the latter two forms are expected to directly affect the decomposition process of dead plant material because they are distributed in the surface soil, litter, and dead wood. Despite the many quantitative studies of biological N fixation in the literature, few have considered the contribution of arthropods.
Termites are decomposers widely distributed in temperate, subtropical, and tropical regions. Many termite species utilize N-poor dead plant material and compensate by symbiotically fixing N in their guts (Benemann 1973; Breznak and others 1973; Sylvester-Bradley and others 1978; Bentley 1984; Tayasu and others 1994). Although N fixation rates by termites have been studied by many researchers, only a few studies quantified N fixation by termites at the ecosystem scale. Schaefer and Whitford (1981) showed that the subterranean termite, Gnathamitermes tubiformans, fix N at a rate of 0.066 kg ha−1 y−1 in a desert ecosystem, in New Mexico. Pandey and others (1992) estimated the N amount fixed by Reticulitermes spp. in Virginia. These studies, however, did not evaluate N fixation by termites at the ecosystem scale. Because termites are a major decomposer and are diverse and abundant in tropical regions (Wood and Sands 1978; Eggleton and Bignell 1995; Bignell and Eggleton 2000; Eggleton 2000) their contribution to ecosystem N inputs and decomposition processes in tropical ecosystems could be significant.
The aim of the present study was to quantify N fixation by termite assemblages and evaluate their importance by comparison with other biological N fixers in tropical forests. First, possible sources of error in the acetylene reduction assay were evaluated and conditions were chosen to minimize factors that might contribute to unrealistic rates. Second, we measured the N fixation rates of representative species of the termite assemblages in two neighboring forests in Thailand, across several seasons. Asymbiotic nitrogen fixation rates were also measured.
MATERIALS AND METHODS
Study Sites and Termites
Observations were carried out in two tropical forests, the dry deciduous forest (DDF) and dry evergreen forest (DEF), at Sakaerat Environmental Research Station (14°30′ N, 101°56′ E) at about 500 m above sea level, in the early rainy season (RsI: May–June), late rainy season (RsII: September), and late dry season (Ds: February) from 2002 to 2003. The two dominant species of wood/litter feeding termites, Microcerotermes crassus and Globitermes sulphureus, were investigated. In addition, as shown in Table 1, various termites were sampled not only in the DDF and DEF, but also at the campus of Kasetsart University in Bangkok and an orchard at Nong Sua near Bangkok (14°11′N, 100°54′E).
Only worker termites were assayed. Workers of Macrotermes gilvus were divided into two well-known types, namely major and minor workers, and assayed separately for each type. Because two types of workers (types-W and -Y) can be visually distinguished also for the genus Microcerotermes, each type was assayed separately. Type-W is characterized by a whitish head and larger head length (0.79 ± 0.03 mm); type-Y is characterized by a smaller (0.75 ± 0.03 mm) yellowish head. According to Roisin (2000), types-W and -Y are probably female and male, respectively.
Temperature in a Termite Nest
Temperatures in an epigeal nest of M. crassus were measured in the DDF using three thermistor SK-L200TH data loggers (Sato Keiryoki MFG, Tokyo, Japan) with one sensor probe each. The data loggers, of which the sensor probes were all capped with plastic tubes to avoid wetting and attack by termites, were set separately on the nest (ambient) and in the upper part as well as in the center of the nest (near the royal cell). Temperature data were logged at 15-min intervals for two weeks over September and October 2002.
Acetylene Reduction Assay
Laboratory Procedure
Termite nests at the two sites, Nong Sua and Kasetsart University, were removed and immediately returned to the laboratory. Twenty to one hundred workers (about 100–400 mg in weight) were taken from the nests. They were placed in glass screw-capped vials (12.6 ml in volume), in which wet filter paper was laid, with butyl rubber septa. Two to six duplicate samples were usually prepared for each nest. These sampling variations among nests reflect availability and population size of each termite species. About 2.5 ml of the headspace was replaced by the same volume of acetylene gas, resulting in a final atmosphere of 19% acetylene. This manipulation was done within 3 h after collection of the nests in the field. The samples were incubated for 2 h at a temperature between 25 and 30°C. During the incubation period, a 50-μl sample of the headspace of each vial was removed with a gas-tight syringe and injected into a GC—14B gas chromatograph (GC) (SHIMADZU, Kyoto, Japan) equipped with a flame ionization detector and a standard Porapak N column. Ethylene peak heights were calibrated against 202 and 505 ppm ethylene standards to confirm the concentration of acetylene in the headspace. N fixation rates were calculated as ethylene production rates and converted to rates based on N weight using a constant of acetylene reduction to N fixation (=3) (Hardy and others 1973; Bentley 1984).
Preliminary Tests
Because N fixation (acetylene reduction) activity in termites is known to be sensitive to unnatural conditions (Prestwich and others 1980; Lovelock and others 1985; Pandey and others 1992; Waller 2000) such conditions should be minimized. To determine the time over which acetylene production was linear, to test for the effects of temperature on acetylene reduction, and to determine the effect of a time delay between collection and incubation, we conducted the following experiments. Termite samples taken from the nest of Microcerotermes sp. collected at Kasetsart in September 2001 (Table 1) were incubated for 8 h, and the headspace gas was injected into a GC at 10-min intervals for the first 3 h and at the end of the incubation period. The ethylene concentration in the vial increased linearly for the first 3 h (R2 = 0.99, P < 0.001), whereas the concentrations were low at hour 8. For the nest of Microcerotermes sp. collected at Nong Sua in September 2002 (Table 1), six samples for each nest were incubated at 25 and 30°C. For the nests of Microcerotermes spp. collected at Nong Sua in March 2003 (Table 1), three samples for each nest were taken and incubated 2, 6 or 24 h after collection of the nests in the field.
Field Procedure
For termites in the DDF and DEF, acetylene reduction assays were performed in the field as follows. Prior to the assays, acetylene gas was stored in glass screw-capped vials with butyl rubber septa (acetylene vial) in the laboratory. About 3 ml of acetylene, drawn from the acetylene vial by injecting water using syringes, was injected into the incubation vial. The air in the incubation vial was homogenized by pumping several times, resulting in a final atmosphere of 19% acetylene. The air in the syringe (3 ml in volume and 19% acetylene) was immediately injected to an empty vial [gas sample vial (1): 12.6 ml in volume] screw-capped with a butyl rubber septum, and then the air in the gas sample vial (1) was homogenized by pumping several times [the rate of dilution = (final air volume in the syringe + the vial volume) / (initial air volume in the syringe)]. After a 2-h incubation in a closed container at a temperature between 25 and 30°C, 3-ml of the head space gas in the incubation vial was sampled by injecting water using syringes. This gas sample was immediately injected into an empty vial [gas sample vial (2)], and the air in the gas sample vial (2) was homogenized by pumping several times. Subsequently, the gas samples in the gas sample vial (1) and (2) were analyzed by GC within three days after the incubations. The concentrations of ethylene in the incubation vials were calculated with the rates of dilution.
Litter, Dead Wood, and Soil
A total of 18 pieces of dead wood were randomly sampled on a 50-m transect in each DDF and DEF in Ds and classified into three categories according to sizes: W1, W2, and W3 with diameters of 1–5, 5–10, and greater than 10 cm, respectively. Six surface soil cores (diameter = 2.2 cm, depth = 6 cm) were also sampled. Six heaps of litter were sampled only in the DEF. These samples were brought to the laboratory and investigated one day after collection. Each plant sample was broken down to small particles, and then 2 g of each plant sample was put in a glass vial (12.6 ml in volume). Each soil core was put in a glass vial (55 ml in volume). N fixation rates of litter, dead wood, and soil were determined in the laboratory by the same method as for termites. After the incubation, the small plant particles were oven-dried at 80°C for 48 h, and milled using a multi-bead shocker MB501(S) (YASUI KIKAI, Osaka, Japan). Using the milled samples, carbon (C) and N analyses were performed with a CHN analyzer (240C, Perkin Elmer, USA).
Statistical Treatment
Means ± standard deviations are shown for the obtained data. The detection limit of the N fixation rate was usually defined for each nest, because the weights of termites used for the measurements and also final concentrations of ethylene in the incubation vial differed from sample to sample. Accordingly, the detection limit was calculated as: (mean ethylene production rate of samples) − 1.96 × (standard error) > 0. Becasue standard deviations cannot be obtained for nests from which only one or two samples were taken, the detection limits were defined as follows: (mean of two samples or value of one sample) > 0.
RESULTS
N Fixation Rate Methodology
N fixation was not significantly affected by incubation temperature (rates were 317 ± 57.4 and 333 ± 41.6 nmol C2H4 g−1 h−1 at temperatures of 25 and 30°C, respectively). The temperatures of the M. crassus nest were 25.0 ± 3.8°C on the nest (ambient), and 25.3 ± 4.7 and 25.8 ± 2.0°C in the upper part and the center of the nest, respectively, indicating that the N fixation rates of M. crassus measured between 25 and 30°C likely reflect natural rates.
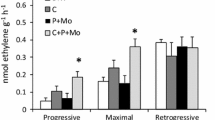
The N fixation rates 24 h after collection were significantly lower than those 2 and 6 h after collection (Figure 1). However, there was no significant difference in N fixation rate between 2 and 6 hours after collection by two-way ANOVA with Tukey’s post hoc test (ANOVA: for “nest”, F(2, 26) = 32.71, P < 0.01; for “time”, F(2, 26) = 133.33, P < 0.01; for “nest × time”, F(4, 26) = 0.97; Tukey’s post hoc test: P2h—24h < 0.01, P6h—24h < 0.01). Therefore, the undisturbed N fixation rates can be obtained when acetylene reduction assays are performed within 6 h after collection of the nest in the field.
Time courses of N fixation rates after collection in the field. Workers of Microcerotermes spp. were picked from nests 2, 6, and 24 h after collection of the nests in the field and incubated in 19% acetylene atmosphere. Different symbols indicate different nests. The error bars show standard deviation.
N Fixation Rates Observed
There was large variation in N fixation rates among species, ranging from an undetectable level to the highest level of 1130.8 nmol C2H4 g−1 h−1 for Microcerotermes sp. (Figure 2). This variation is usually related to functional groups. Three of the four species of fungus-growers and soil-feeders did not fix N at detectable levels. Only Odontotermes sp. fixed N at a low level (3.8 nmol C2H4 g−1 h−1). In contrast, fixation rates were usually higher than 20 nmol C2H4 g−1 h−1 for all non-fungus-growing wood/litter-feeding species except Speculitermes sp. and Schedorhinotermes medioobscurus.
Mean N fixation rates of termites. Species numbers are from Table 1. Each symbol indicates one nest or one caste of one nest.
For M. crassus and G. sulphureus, large intra-specific variation was observed among seasons within a single forest and between the DDF and DEF (Figure 3). N fixation rates were significantly different among nests of G. sulphureus in both the DDF and DEF in RsI (for DDF, F(2, 14) = 27.303, P < 0.01; for DEF, F(2, 14) = 47.420, P < 0.01, by one-way ANOVA). An inter-caste variation was observed in M. crassus, and N fixation rates of type-W were significantly higher than those of type-Y (d.f. = 5, T = 3.278–6.587, P < 0.05, by paired T-test), except for termites collected in the DEF in RsII and Ds. N fixation rates were significantly higher in the DDF than in DEF for both species (Table 2). A significant seasonal variation was detected from type-Y of M. crassus by two-way ANOVA (Table 2), and N fixation rates were higher in RsI than those in RsII and Ds.
The N fixation rates of workers of M. crassus, comprising types-W (McW) and -Y (McY), and of G. sulphureus (Gs) in the DDF and DEF. The means and standard deviations of six nests of each species are shown for the three seasons: RsI (May–June) (A), RsII (September) (B), and Ds (February) (C). n.t., not tested.
N Fixation Rates at the Ecosystem Scale
N fixation rates of termites are calculated at the ecosystem scale using their biomass which was previously obtained in the DDF (A. Yamada and others, unpublished manuscript) and in the DEF (Yamada and others 2003) and using the N fixation rates from this study. The biomasses of types-W and -Y of M. crassus and of G. sulphureus are 0.83, 0.83, and 0.23 g m−2, respectively, in the DDF, and 3.01, 3.01, and 1.50 g m−2, respectively, in the DEF, with the assumption that the ratio of the biomass of type-W to -Y is 1:1. Termites in the DDF and in the DEF fixed N at rates of 2.28 and 3.07 mmol C2H4 m−2 y−1, respectively, assuming that the length of each season is 4 months. For G. sulphureus, which was not measured in RsII, rates during that season were assumed equal to rates during RsI. The amounts of N fixed by termites were estimated to be 0.21 and 0.28 kg ha−1 y−1 in the DDF and DEF, respectively.
N Fixation Rates and C/N Ratios of Litter and Dead Wood
Table 3 shows N fixation rates and C/N ratios for litter and dead wood in the DDF and DEF, using biomass data from A. Yamada and others (unpublished manuscript) and Yamada and others (2003), respectively. The small pieces of dead wood (W1) showed higher N fixation rates in both the DDF (2.39 nmol C2H4 m−2 y−1) and the DEF (3.56 nmol C2H4 m−2 y−1) than the larger pieces (W2 and W3), whereas the N fixation rate of litter (Lt) in the DEF (the only site where it was measured) was apparently higher than in all other size classes of wood (4.68 mmol C2H4 m−2 y−1). The C/N ratios of W1 were usually lower than those of W2 and W3 in each DDF (W1 = 278, W2 and W3 = 356 and 198) and DEF (W1 = 75, W2 and W3 = 117 and 205). N fixation activity was not detected from the soil samples. Using these data, the N fixation rates of all the litter and dead wood at the ecosystems scale were calculated to be 8.1 and 42.3 mmol C2H4 m−2 y−1, which were converted to 0.75 and 3.95 kg N ha−1 y−1 in the DDF and DEF, respectively. Average values of the C/N ratio of all the litter and dead wood were also calculated to be 161.0 and 74.9 in the DDF and DEF, using data for the biomass of litter and dead wood in A. Yamada and others (unpublished manuscript) and Yamada and others (2003), respectively.
DISCUSSION
Factors Controlling N Fixation Rates of Termites
The N fixation rates of termites were obtained under conditions in which artificial underestimation was minimized by considering the sensitivity of N fixation activity to unnatural conditions reported in the literature (Prestwich and others 1980; Lovelock and others 1985; Pandey and others 1992; Waller 2000). Thus, it is probable that the rates represent natural ones.
Large intra- and inter-specific variation in N fixation rates of termites as shown in Figure 2 has been reported in several reviews (Breznak 1982; Waller and others 1989; Curtis and Waller 1998; Waller 2000). These reviews, however, report N fixation rates that were measured without considering the delay between collection and incubation. Others have shown that N fixation rates of termites decrease with N concentrations in their food (Breznak and others 1973; Rohrmann and Rossman 1980). In fact, for the smaller size classes of wood, the C/N ratio was higher in the DDF, where the N fixation rates of termites were also higher (Figure 3), than DEF (Table 3). Furthermore, the majority of termites distributed in dead wood have been found in such size classes of wood (unpublished data; Yamada and others 2003); therefore, the intraspecific variation in the N fixation rates of termites between the DDF and DEF (Figure 3) might be explained by wood C/N ratios. However, Curtis and Waller (1997) reported no clear correlation between the N fixation rate of Reticulitermes sp. and the N content of their food on the field scale. This is not a contradiction if the much higher N fixation rates of G. sulphureus and M. crassus compared to Reticulitermes sp. are more sensitive to and are strongly suppressed by the concentration and available N in foods.
Importance of N Fixed by Termites
To estimate the amounts of N fixed by termite assemblages in the DDF and DEF, we investigated two species, M. crassus and G. sulphureus, of non-fungus-growing wood/litter-feeding termites, for which N fixation activity was usually detected in significant levels (Figure 2). Although diverse non-fungus-growing wood/litter-feeders occur in the DEF, the two species are dominant and represented 86 and 96% of the biomass of non-fungus-growing wood/litter-feeders in the DDF and DEF, respectively (A. Yamada and others, unpublished manuscript; Yamada and others 2003).
Here we compare the amounts of N fixed by termites to that fixed by other biological N fixers, including plant symbionts and free-living (asymbiotic) fixers (Figure 4). The amounts of N mediated by plant-symbiotic N fixation have ranged from 0.5 to 60 kg ha−1 y−1 in tropical evergreen and deciduous forests (Cleveland and others 1999). Asymbiotic N fixation occurs on the surface of litter and dead wood and in the soil; the amounts of N fixed by asymbiotic N fixation were 0.75 and 3.95 kg ha−1 y−1 in the DDF and DEF, respectively, at the lower end of the range reported by Cleveland and others (1999) for tropical evergreen and deciduous forests (2.5 to 20.0 kg ha−1 y−1). Estimates of N in precipitation in tropical forests have ranged from 6.5 to 39.4 kg ha−1 y−1 (Jordan 1985; Edwards 1982; Strigel and others 1994) and are comparable to N inputs from either plant-symbiotic or asymbiotic N fixation.
Schematic of N inputs (kg N ha−1 y−1) to the DDF and DEF. Only non-fungus-growing wood/litter-feeders of termites fixed N, and asymbiotic N fixation activity was detected from litter and dead wood but not from the soil. * data for tropical forests in Jordan (1985), Edwards (1982), and Strigel and others (1994); † data for tropical evergreen and deciduous forests in Cleveland and others (1999).
The amounts of N fixed by termites were 0.21 and 0.28 kg ha−1 y−1 in the DDF and DEF, respectively. These values are much smaller than those derived through plant-symbiotic and asymbiotic N fixation and precipitation. Although the amount of N fixed by termites is likely a small proportion of the total N input to the DDF and DEF, nevertheless, N fixed by termites likely contributes significantly to the N dynamics of the ecosystems as described below.
Termites are calculated to be responsible for 7–22% of N inputs from fixation to decomposing plant material in the DEF and DDF, respectively (Figure 4). N from precipitation (Jordan 1985; Edwards 1982; Strigel and others 1994) and immobilization by fungal hyphae (Frey and others 2000) may also be potential sources of additional N to decomposing plant material. Taking these potential N sources into consideration, the amounts of N fixed by termites might be of minor importance in decomposition processes. However, from the spatial and temporal point of view, N fixed by termites is expected to contribute significantly to decomposition processes in different ways than the other sources of additional N. Many species of non-fungus-growing wood/litter-feeding termites, for example M. crassus, attack and break more or less freshly fallen branches and trunks, and feed on woody material in the center of the wood (Abe 1980) by utilizing fixed N in their guts. In contrast, additional N from asymbiotic fixation, precipitation, and fungus-mediated immobilization is expected be supplied mainly to surfaces of fallen branches and trunks. This is partly supported by the higher fixation rates and lower C/N ratios of smaller size classes of wood (Table 3), which usually have larger surface areas compared to their weights. Here, we suggest that N fixed by termites plays its role in the early decomposition processes and in different parts of the dead wood than the other sources of additional N. N fixed by termites would be excreted as feces out of their bodies. A stable isotope analysis has shown that feces of the termite Neotermes koshunensis contain atmospheric N (I. Tayasu, personal communication). It is most likely that at least a portion of feces containing fixed N is put into the center of the wood where the termites are feeding and/or nesting. Therefore, the feces containing N fixed by termites would be an exclusive source of additional N to the centers of freshly dead wood, promoting further decomposition. The present study is the first report on N fixed by termites in tropical forests on the ecosystem scale, in contrast to many excellent ecosystem-scale studies emphasizing the significant roles of C mineralization (Wood and Sands 1978; Martius 1994; Bignell and others 1997; Eggleton and others 1999; Konaté and others 2003).
Various animals prey on termites (Wood and Sands 1978), and thus N fixed by termites is expected to contribute to the N economy of the predators. Becasue termites are one of the most abundant animals in tropical forests (Fittkau and Klinge 1973; Collins 1980) and provide several times larger amounts of their bodies than the biomass to their predators per annum (Wood and Sands 1978), termites could be an important source of N to their predators.
In conclusion, N fixed by termites, though contributing weakly to total N input to the ecosystems, represents a significant part of N input to dead plant material (Figure 4), and importantly affects the decomposition process of dead plant material.
References
Abe T. 1980. Studies on the distribution and ecological role of termites in a lowland rain-forest of West Malaysia. 4. The role of termites in the process of wood decomposition in Pasoh-Forest-Reserve. Revue d’Ecologie et de Biologie du Sol 17:23–40
Benemann JR. 1973. Nitrogen fixation in termites. Science 181:164–5
Bentley BL. 1984. Nitrogen fixation in termites – fate of newly fixed nitrogen. J Insect Physiol 30:653–5
Bignell DE, Eggleton P. 2000. Termites in ecosystems. In: Abe T, Bignell DE, Higashi M, Eds. Termite: evolution, society, symbioses, ecology. Dordrecht: Kluwer Academic. p 363–87
Bignell DE, Eggleton P, Nunes L, Thomas KL. 1997. Termites as mediators of carbon fluxes in tropical forests: Budgets for carbon dioxide and methane emissions. In: Watt AD, Stork NE, Hunter MD, Eds. Forests and Insects. London: Chapman and Hall. p 109–34
Breznak JA. 1975. Symbiotic relationships between termites and their intestinal microbiota. In: Jennings DH, Lee DL, Eds. Symbiosis: 29th Symposium of the Society for Experimental Biology. Cambridge: Cambridge University Press. p 559–80
Breznak JA. 1982. Intestinal microbiota of termites and other xylophagous insects. Annu Rev Microbiol 36:323–43
Breznak JA, Brill WJ, Mertins JW, Coppel HC. 1973. Nitrogen fixation in termites. Nature 244:577–80
Cleveland CC, Townsend AR, Schimel DS, Fisher H, Howarth RW, Hedin LO, Perakis SS, Latty EF, Von Fischer JC, Elseroad A, Wasson MF. 1999. Global patterns of terrestrial biological nitrogen (N2) fixation in natural ecosystems. Glob Biogeochem Cycles 13:623–45
Collins NM. 1980. The distribution of soil macro fauna on the west ridge of Gunung Mount Mulu Sarawak. Oecologia 44:263–75
Curtis AD, Waller DA. 1997. Variation in rates of nitrogen fixation in termites: response to dietary nitrogen in the field and laboratory. Physiol Entomol 22:303–9
Curtis AD, Waller DA. 1998. Seasonal patterns of nitrogen fixation in termites. Funct Ecol 12:803–7
Edwards PJ. 1982. Studies of mineral cycling in a montane rain forest in New Guinea 5. Rates of cycling in throughfall and litter fall. J Ecol 70:807–28
Eggleton P. 2000. Global patterns of termite diversity. In: Abe T, Bignell DE, Higashi M, Eds. Termite: evolution, society, symbioses, ecology. Dordrecht: Kluwer Academic. p 25–51
Eggleton P, Bignell DE. 1995. Monitoring the response of tropical insects to changes in the environment: troubles with termites. In: Harrington R, Stork NE, Eds. Insects in a changing environment. London: Academic Press. p 478–97
Eggleton P, Homathevi R, Jones DT, MacDonald JA, Jeeva D, Bignell DE, Davies RG, Maryati M. 1999. Termite assemblages, forest disturbance and greenhouse gas fluxes in Sabah, East Malaysia. Philos Trans Roy Soc Lond B Biol Sci 354:1791–802
Fittkau EJ, Klinge H. 1973. On biomass and trophic structure of the central Amazonian rain forest ecosystem. Biotropica 5:2–14
Frey SD, Elliott ET, Paustian K, Peterson G. 2000. Fungal translocation as a mechanism of exogenous nitrogen inputs to decomposing surface residues in a no-tillage agroecosystem. Soil Biol Biochem 32:689–98
Hall JH, Matson PA. 2003. Nutrient status of tropical rain forests influences soil N dynamics after N additions. Ecol Monogr 73:107–29
Hardy RW, Burns RC, Holsten RD. 1973. Applications of the acetylene-ethylene assay for measurement of nitrogen fixation. Soil Biol Biochem 5:47–81
Hunt HW, Ingham ER, Coleman DC, Elliott ET, Reid CPP. 1988. Nitrogen limitation of production and decomposition in prairie mountain meadow and pine forest. Ecology 69:1009–16
Jordan CF. 1985. Nutrient Cycling in Tropical Forest Ecosystems: Principles and their Application in Management and Conservation. Chichester: John Wiley and Sons. 190 p
Konaté S, Le Roux X, Verdier B, Lepage M. 2003. Effect of underground fungus-growing termites on carbon dioxide emission at the point- and landscape-scales in an African savanna. Funct Ecol 17:305–14
Lovelock M, Obrien RW, Slaytor M. 1985. Effect of laboratory containment on the nitrogen metabolism of termites. Insect Biochem 15:503–10
Martius C. 1994. Diversity and ecology of termites in Amazonian forests. Pedobiologia 38:407–28
Nardi JB, Mackie RI, Dawson JO. 2002. Could microbial symbionts of arthropod guts contribute significantly to nitrogen fixation in terrestrial ecosystems? J Insect Physiol 48:751–63
Pandey S, Waller DA, Gordon AS. 1992. Variation in acetylene-reduction (nitrogen-fixation) rates in Reticulitermes spp. (Isoptera; Rhinotermitidae). VA J Sci 43:333–8
Prestwich GD, Bentley BL, Carpenter EJ. 1980. Nitrogen sources for Neotropical nasute termites Nasutitermes ephratae and Rhynchotermes perarmatus fixation and selective foraging. Oecologia 46:397–401
Rohrmann GF, Rossman AY. 1980. Nutrient strategies of Macrotermes ukuzii (Isoptera: Termitidae). Pedobiologia 20:61–73
Roisin Y. 2000. Diversity and evolution of caste patterns. In: Abe T, Bignell DE, Higashi M, Eds. Termite: evolution, society, symbioses, ecology. Dordrecht: Kluwer Academic. p 95–119
Schaefer DA, Whitford WG. 1981. Nutrient cycling by the subterranean termite Gnathamitermes tubiformans in a Chihuahuan desert ecosystem. Oecologia 48:277–83
Son Y. 2001. Non-symbiotic nitrogen fixation in forest ecosystems. Ecol Res 16:183–96
Strigel G, Ruhiyat D, Prayitno D, Sarmina S. 1994. Nutrient input by rainfall into secondary forests in East Kalimantan, Indonesia. J Trop Ecol 10:285–8
Sylvester-Bradley R, Bandeira AG, de Oliveira LA. 1978. Fixação de nitrogênio (redução de acetileno) em cupins (Insecta: Isoptera) da Amazônia Central. Acta Amazonica 8:621–7
Tayasu I, Sugimoto A, Wada E, Abe T. 1994. Xylophagous termites depending on atmospheric nitrogen. Naturwissenschaften 81:229–31
Vitousek PM, Cassman K, Cleveland C, Crews T, Field CB, Grimm NB, Howarth RW, Marino R, Martinelli L, Rastetter EB, Sprent JI. 2002. Towards an ecological understanding of biological nitrogen fixation. Biogeochemistry 57:1–45
Vitousek PM, Howarth RW. 1991. Nitrogen limitation on land and in the sea – How can it occur? Biogeochemistry 13:87–115
Waller DA. 2000. Nitrogen fixation by termite symbionts. In: Triplett EW, Ed. Prokaryotic nitrogen fixation: a model system for analysis of a biological process. Wymondham: Horizon Scientific Press. p 225–36
Waller DA, Breitenbeck GA, La Fage JP. 1989. Variation in acetylene-reduction by Coptotermes formosanus (Isoptera, Rhinotermitidae) related to nest source and termite size. Sociobiology 16:191–6
Wood TG, Sands WA. 1978. The role of termites in ecosystems. In: Brian MV, Ed. Production ecology of ants and termites. Cambridge: Cambridge University Press. p 245–92
Yamada A, Inoue T, Sugimoto A, Takematsu Y, Kumai T, Hyodo F, Fujita A, Tayasu I, Klangkaew C, Kirtibutr N, Kudo T, Abe T. 2003. Abundance and biomass of termites (Insecta: Isoptera) in dead wood in a dry evergreen forest in Thailand. Sociobiology 42:569–85
Acknowledgements
We thank the National Research Council of Thailand, the people who are working at Sakaerat Environmental Research Station, T. Johjima, Y. Hongoh, C. Boontong, S. Trakulnaleamsai, and N. Noparatnaraporn for their kind cooperation. We thank the chemical analysis section of RIKEN for the elemental analyses, M. B. Wamalwa, I. Tayasu, and two anonymous reviewers for their helpful comments on the manuscript, and A. Haraguchi and R. Araki for improving the English of the manuscript. This work was supported by Grant-in-Aid (09NP1501) for Creative Basic Research from Japan Ministry of Education, Culture, Sports, Science and Technology, and in part by grants from the Bioarchitect Research Program and the Eco Molecular Science Research Program from RIKEN. The first author (A. Y.) was also supported by a grant from the Junior Research Associate Program from RIKEN.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamada, A., Inoue, T., Wiwatwitaya, D. et al. Nitrogen Fixation by Termites in Tropical Forests, Thailand. Ecosystems 9, 75–83 (2006). https://doi.org/10.1007/s10021-005-0024-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-005-0024-7