Abstract
Pediatric pacemaker (PM) implants comprise less than 1 % of all PM implants. This study aimed to investigate permanent cardiac pacing among the pediatric population, identifying different indications and complications of pediatric cardiac pacing, especially focusing on the effect of the pacing sites, the PM lead type, and the indications for pacing. The current work is a cross-sectional study of 103 procedures for permanent PM insertion in pediatric patients between January 2001 and December 2010. The patients were followed up 1, 3, and 6 months after implantation, then every 6 months or as needed. Evaluation included routine clinical examination, electrocardiography, chest X-ray, echocardiography, and a full analysis of the pacing system measurements. The ages of the patients ranged from 0.09 to 12 years (median, 2.3 years). The most common indication for pacing was postoperative complete heart bock, noted in 54 patients (52.4 %). Transvenous endocardial PM insertion was performed in 92 procedures (89.3 %), whereas transthoracic epicardial insertion was performed in 11 procedures (10.7 %). The most common site of pacing was the right ventricular apex (n = 64, 62 %), followed by the right ventricular outflow tract (n = 25, 24.3 %). Transthoracic epicardial PM insertion was associated with a significantly higher percentage and greater severity of complications. In this study, 65 % of the patients with left ventricle (LV) dilation before pacing showed a significant improvement in LV dimensions and function after pacing. This was noted only in those with endocardially inserted PM leads in both the congenital and the postoperative groups regardless of the pacing site. Endocardial PM insertion in children is a safe procedure with fewer complications and a lower ventricular threshold than the epicardial route. Permanent single-chamber right ventricle pacing is safe and can lead to significant improvement in LV function and dimensions. However, long-term follow-up assessment is needed for further evaluation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Pacemakers (PMs) were introduced into clinical practice several decades ago and currently are used in a growing number of patients [12]. Since insertion of the first cardiac PM in 1958, vast changes have occurred in both the technology of the devices and their indications. Devices have evolved from single-lead and fixed-rate systems to multichamber rate-responsive systems with increasingly sophisticated software [22].
Pediatric PM implants comprise less than 1 % of all PM implants [13]. The indications for pacing in newborns and infants are divided predominantly into three groups: congenital abnormalities of the conduction system, acquired heart blocks after cardiac surgery for correction of congenital defects, and sinus node diseases [1]. After half a century of major progress in congenital heart disease management, atrioventricular (AV) conduction block continues to complicate 1–3 % of cardiac surgical procedures [9].
Chronic right ventricular (RV) apical pacing may have a detrimental effect on left ventricular (LV) function and could promote heart failure in adult patients with LV dysfunction [18]. However, in pediatric patients, with or without structural heart disease, this does not necessarily result in a decline of LV function [18]. Unfortunately, limited clinical pediatric studies have evaluated precise site-specific lead locations [11].
The implantation of permanent PMs in children and adolescents is a procedure with a generally favorable outcome [19]. Continued experience may lead to further improvements in cardiac pacing for children [4].
The current study aimed to investigate permanent cardiac pacing in the pediatric population, identifying different indications and complications of pediatric cardiac pacing and focusing especially on the effect of the pacing sites, the PM lead type, and the indications for pacing.
Patients and Methods
The current work is a cross-sectional study of consecutive pediatric patients who underwent PM insertion between January 2001 and December 2010. The following data were particularly stressed for the patients included in the current study:
-
Preinsertion of the PM: age, sex, weight, height, body surface area, preinsertion 12-lead electrocardiography (ECG), Holter monitoring, and echocardiography
-
PM insertion procedure details: route of lead insertion, mode of pacing, and primary pacing settings
-
Postinsertion: 12-lead ECG, Holter monitoring, and echocardiography
-
Follow-on pacing settings.
Implantation Procedure
All procedures were performed with the patient under general anesthesia. The endocardial pacing leads were inserted in the catheterization laboratory. The ventricular leads were positioned in the nonsystemic ventricular apex (LV in congenitally corrected transposition of the great arteries, levo transposition of the great artery (L-TGA), and in dextro-transposition of the great arteries after Senning palliation), from which adequate values of the pacing threshold (pulse amplitude, <1 V) and impedance (>300 and <1,000 ohms) were achieved.
Epicardial pacing leads were inserted in the operation theater by positioning the pacing lead in the RV or LV through sternotomy or thoracotomy (Fig. 1). The pacing threshold and impedance were evaluated during the implant procedure until adequate values were achieved. The voltage stimulation threshold and pacing impedance were rechecked on the second day, and patients were discharged from the hospital 4 to 6 days after implantation.
The pulse generator most commonly used for our patients was the Medtronic Sigma (48 patients, 46.6 %) followed by St. Jude Verity (23 patients, 22.3 %) and St. Jude Microny (19 patients, 18.4 %). Other types used were St. Jude Identity (6 patients, 5.8 %), Sorin (3 patients, 2.9 %), Biotronic (3 patients, 2.9 %), and Medtronic INSYNC III (1 patient, 0.97 %).
Pacing Leads
Either endocardial active-fixation steroid-eluting leads (model: St. Jude Medical, Tendril SDX 1688T/TC) or steroid-eluting bipolar epicardial pacing leads (model: Medtronic CapsureEpi 4968) were used in the study. Temporary pacing was used for some patients before permanent PM insertion of either the Medtronic single- or dual-chamber temporary PM.
Follow-up Evaluation
The patients were followed up 1, 3, and 6 months after implantation, then every 6 months or as needed. Evaluation included routine clinical examination, electrocardiography, chest X-ray, echocardiography, and a full analysis of the pacing system measurements.
Echocardiographic Follow-up Assessment
Echocardiographic evaluation was performed using the Vivid S5 echocardiograhic machines (GE-Vingmed, Horten, Norway). Parasternal M-mode images were used to measure the LV end-diastolic and end-systolic dimensions. Measurements were taken at the point of peak diastolic LV free-wall outward motion and peak systolic inward motion, respectively, and compared with the normal values of body weight-matched individuals using the z-score method. Left ventricular fractional shortening was calculated according to the following formula:
-
(LV end-diastolic dimension − LV end-systolic dimension)/LV end-diastolic dimension × 100.
Sinus pause or arrest was defined as absence of sinus P waves on the ECG for more than 2 s due to a lack of sinus nodal PM activity [5]. Pacing system implantation was defined as placement of a new PM generator and a lead. Replacement of a PM was defined as placement of only the PM generator without the insertion of new leads. Lead malfunction requiring a new pacing system implantation was defined as (1) exit block, (2) abnormal threshold increase with the need for high output values causing early battery depletion and/or partial loss of capture, (3) lead fracture, and (4) insulation break [20].
Statistical Analysis
Data were analyzed using the statistical package for social sciences version 15 (SPSS Inc., Chicago, IL). Numeric data are expressed as mean and standard deviation or median and range as appropriate. Qualitative data are expressed as frequency and percentage. For quantitative data, comparison between two groups was performed using the Mann–Whitney test (nonparametric t test). Comparison between three groups was performed using the Kruskal–Wallis test (nonparametric analysis of variance [ANOVA]) because the variables are not normally distributed. Comparison of two repeated measures before and after the procedure was performed using the Friedman test and the Wilcoxon signed-ranks test for quantitative variables and the McNemar test for qualitative variables. A p value lower than 0.05 was considered significant.
Results
The study involved 103 procedures for permanent PM insertion in 91 patients, with 10 patients undergoing two procedures for permanent PM replacement and one patient undergoing three procedures.
Patient Characteristics
Boys made up 65 % of the study population. Their characteristics are described in Table 1. The youngest patient was 16 days old and weighed 3.5 kg, whereas the youngest patient with an endocardial PM inserted was 56 days old and weighed 3.2 kg. The most common indication for pacing was postoperative complete heart block, noted in 54 (52.4 %) of the study group. Other indications included congenital heart block (33.2 %, n = 31), congenital sinus node dysfunction (3.8 %, n = 4), inappropriate sinus bradycardia (0.9 %, n = 1), and postoperative sinus node dysfunction (0.9 %, n = 1). The remaining 11 procedures were PM replacements.
The most common surgical intervention complicated by complete heart block and hence indicating a permanent PM implantation was VSD closure (26 patients, 48.15 %) followed by repair of Fallot’s tetralogy (11 patients, 20.35 %). Other surgeries complicated by complete heart block are shown in Table 2.
Mode of Pacing
The vast majority of our patients were paced using the VVIR (ventricular pacing, ventricular sensing, inhibition response and rate adaptive) mode devices (99 patients, 96.18 %). We used DDDR (Dual chamber pacing, Dual chamber sensing, Dual chamber response, rate response pacing) in two patients (1.94 %), DDD (Dual chamber pacing, Dual chamber sensing, Dual chamber response) in one patient (0.97 %), and multisite AV synchronous pacing (i.e., cardiac resynchronization therapy (CRT)) in one patient (0.97 %) with persistent heart failure functional class 4 after previous VVIR pacing with obvious myocardial impairment. No implantable cardioverter-defibrillators (ICDs) were included.
Unipolar pacing was used whenever a Microny PM was placed because it was the only available pacing mode of this PM. It also was used after high thresholds during bipolar pacing with other PMs.
Pace Polarity
Unipolar pacing was started in 19 patients (18.4 %) and bipolar pacing in 84 patients (81.6 %). Three of these patients (2.9 %) had lead problems (2 had breakage in one lead limb and 1 had a marked increase in the lead impedence) necessitating the shift to unipolar pacing.
Route of Insertion
Transvenous endocardial PM insertion was performed in 92 procedures (89.3 %), whereas transthoracic epicardial insertion was performed in 11 procedures (10.7 %).
Preinsertion Echocardiographic Findings
Among those with congenital heart block and congenital sinus node dysfunction, 81 % had abnormal echocardiographic findings, the most common of which was a dilated LV noted in 14 patients (37 %). The most common congenital heart defect was L-TGA, noted in 5 patients (13.5 %) (Table 3).
Prepacing Holter Recording
All the patients had prepacing 24-Holter recording. The slowest recorded heart rate was 35 bpm in a 2-year-old boy. Pauses defined as more than 2 s were present in six patients (5.8 %). The longest identified pause was 5 s. Other events are shown in Table 4.
Site of Pacing as Recorded From Postpacing ECG
The most common site of pacing used for PM lead insertion was the right ventricular apex (RVA) (n = 69, 66 %), followed by the right ventricular outflow tract (RVOT) (n = 25, 24.3 %). Left ventricular pacing was performed for four patients (3.9 %) (2 patients after the Senning operation with endocardial leads and 2 patients with epicardially inserted PMs). For five patients (4.8 %), L-TGA, pacing leads were inserted in the morphologic LV (i.e., the right-sided ventricle).
Combined Procedures
One patient underwent pulmonary artey banding in the same setting of epicardial permanent PM insertion. Another patient had a patent ductus arteriosis (PDA) closed by Amplatzer in the same session as endocardial PM insertion, and a third patient had his PDA closed by coil after PM was inserted in the same procedure.
Mortality
Three patients (27 %) died, all of them after an epicardial PM insertion. The first patient, who died intraoperatively, had a dilated myopathic heart and complete heart block associated with long QT syndrome. The second patient died after a mitral valve repair operation for severe mitral regurgitation. This patient had undergone previous corrective surgery for complete AV canal. In the same setting, his second PM had been epicardially inserted, and the procedure had been complicated by infective endocarditis. The third patient died shortly after epicardial multisite pacing (atrio-biventricular pacing) for cardiac resynchronization (to replace her old VVIR PM) because she had a severely dilated myopathic LV with refractory heart failure. None of the patients died after endocardial PM insertion.
Complications
Of the 103 procedures, 74 (72 %) resulted in a noncomplicated course. Transthoracic epicardial PM insertion was associated with a significantly higher percentage and greater severity of complications (Table 5).
Two procedures were difficult and lengthy, the one due to a double RV chamber and the other due to tricuspid regurgitation (TR). Seven patients required PM revision because of PM-related complications: 1 due to lead malfunction, 2 due to mediastinitis, 2 due to pocket infection, 1 due to PM malfunction, and 1 due to PM migration and skin necrosis. Three patients (3/7) with deep PM infections required system removal, whereas others were adequately treated with systemic antibiotics.
Three patients with postoperative complete heart block (5.7 % of postoperative patients with complete heart block) regained sinus rhythm more than 1 year after PM insertion. In two patients, congenital complete heart block became first-degree heart block with pacing independence most of the time more than 5 years after PM insertion.
LV Dilation
A statistically significant improvement occurred in the LV dimensions after pacing, with 11 patients experiencing LV dilation after pacing versus 32 patients before pacing (p < 0.001).
Improvement in LV dimensions was noted only in those patients with endocardially inserted PM leads (p < 0.001). However, the number of patients in the epicardially paced group was limited.
A statistically significant improvement in LV dimensions was found independently in the congenital complete heart block group (14 patients with LV dilation before pacing vs 6 patients after pacing; p < 0.001) and the postoperative group (18 patients with LV dilation before pacing vs 5 patients after pacing; p = 0.008), denoting improvement in LV dimensions after pacing in both groups.
A statistically significant improvement was found between the pre- and postpacing LV dimensions in patients who had RVA pacing (20 patients with LV dilation before pacing vs 3 patients after pacing; p < 0.001) and those who had RVOT pacing (10 patients with LV dilation before pacing vs 4 patients after pacing; p = 0.041), with significant improvement in LV functions (fractional shortening and ejection fraction) in both groups.
Postpacing QRS Width Compared in Different Routes of Insertion
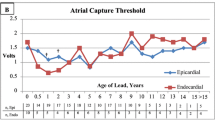
Patients with epicardially inserted PMs showed a significantly wider QRS complex (0.13 s) than the endocardial group (0.10 s) (p = 0.003).
Follow-up Period
The mean follow-up period was 7.16 years. The mean device longevity was 9–10 years for the St. Jude series and 7–8 years for the Medtronic and Biotronic series. We did not have to replace any lead due to malfunction (those that showed complications were switched from bipolar to unipolar pacing). The mean time until reoperation was 7.6 years.
Ventricular Output in the Last Programming Session
Comparison of the epicardially and endocardially inserted PMs in terms of ventricular pacing amplitude (output) showed higher outputs in the epicardial group than in the endocardial group (the mean and median outputs were respectively 3.06 and 2.5 V vs 2.3 and 2.00 V (p = 0.001).
Discussion
This study was a cross-sectional observational study of 103 procedures for permanent PM insertion in children from January 2001 to December 2010. Pacing in the pediatric patient is more difficult due to the size of the patient. Smaller pulse generators and steroid-eluting active fixation leads are a few of the technological advances that have made pacing in children easier, safer, and more durable. Continued experience may lead to further improvements in endocardial pacing for children [4].
In the current study, 92 procedures (89.3 %) were performed using the endocardial route and 11 (10.7 %) using the epicardial route. In children, especially in neonates and infants, the epicardial route was traditionally chosen until the advent of smaller generators and lead implantation techniques that allowed growth of the child without lead displacement.
In our study, the youngest patient with an endocardial PM inserted was 56 days old and weighed 3.2 kg, whereas the youngest patient with endocardially inserted PM reported by Nolasco et al. [14] was 1 month old and weighed 2.3 kg. Similarly, Kammeraad et al. [10] inserted an endocardial PM in a 2-day-old patient weighing 2.3 kg. Previously, the insertion of a transvenous PM in a 1.3-year-old patient that weighed 8.5 kg was considered an achievement [17].
In this study, postoperative complete heart block was the most common indication for PM implantation (52.4 % of the patients) followed by congenital heart block (33.2 %). Similarly, Çeliker et al. [4] showed that indications for PM implantation were more postoperative than congenital. Welisch et al. [24] reported that high-degree AV block (Mobitz II or complete AV block) was an indication for pacing in 65 % of their patients, 55 % of whom were postsurgery or postintervention cases. However, others have reported more congenital than postoperative indications for pacing [10, 21].
During the current study, three patient deaths were recorded. In all three cases, the patients had their PM inserted epicardially. Similarly, in a study of 148 children with a mean age of 8.2 years, a higher mortality rate was reported for the epicardial PM (5.4 %) than for the endocardial PM (0.7 %) [15]. An 18 % mortality rate was reported in a study including 138 epicardially placed PMs [6].
Three of our patients had lead malfunctions (2 with breakage of a limb of their epicardial lead and 1 with marked elevation of the endocardial ventricular lead impedance). Welisch et al. [24], reported an 18 % incidence of lead complications in their patients with an endocardial PM. This finding is congruent with several articles related to lead complications such as that by Fortescue et al. [7], who reported lead failure in 15 % of their patients, with significantly higher lead failure in epicardially placed leads. These authors found that epicardial leads were more likely to fail due to fracture or exit block, whereas transvenous leads failed more often due to insulation breaks or dislodgements. Also Post et al. [16], reported a 16 % ventricular lead failure rate.
Late recovery of AV conduction was defined as persistent recovery of AV conduction more than 14 days after the onset of complete heart block.
Atrioventricular conduction may return in a small but significant percentage of patients after PM implantation for complete heart block associated with congenital heart surgery [2]. In our study, three patients with postoperative complete heart block (~ 5 % of postoperative patients with complete heart block) regained sinus rhythm more than 1 year after PM insertion. The following higher rates of AV node recovery were reported in other studies: 14 % [24], 9.6 % [2], and 32 % [3].
When recovery of AV conduction occurs within the first months after surgery, it appears to be reliable, suggesting that lifelong cardiac pacing may not be necessary for these individuals. Although the results are encouraging, continued follow-up assessment is mandated for patients with late recovery of AV conduction due to the potential for paroxysmal AV block. Thus, they should be followed up like other PM-dependent patients. The follow-up assessment should include evaluation with electrocardiograms and Holter monitoring. The PM may be programmed to the lowest rate that would provide rate support in the event of complete heart block recurrence.
Recent studies demonstrating physiologic benefits of RV septal outflow or bundle of His pacing in deference to the apical implant site have gained interest for potential prevention of dysfunction and improvement of paced myocardial contractility. Unfortunately, clinical pediatric studies that evaluate precise site-specific lead locations have been limited [11]. However, chronic RV pacing in pediatric patients with or without structural heart disease does not necessarily result in a decline of LV function [18], as noted in the current study, because a significant improvement in LV dimensions was noted in 76 % of those with dilated LV prepacing after single-chamber RV pacing.
In our study, none of the patients who had normal LV dimensions before pacing developed LV dilation after pacing. Our results disagree with those reported by Gebauer et al. [8], who showed that the incidence of LV dilation and dysfunction in patients was found to increase significantly from 1.3 % before PM implantation to 13.4 % (11 of 82) patients after implantation at the last follow-up assessment. However, improvement in LV dimensions was noted only in those with endocardially inserted PMs.
Our comparison of the epicardially and endocardially inserted PMs in terms of ventricular pacing amplitude (output) showed higher outputs in the epicardial group than in the endocardial group. This is consistent with previous studies [15, 17, 20, 21, 23].
Conclusions and Recommendations
Endocardial PM insertion in children is a safe procedure with less complications and a lower ventricular threshold than epicardial PM insertion. Permanent single-chamber RV pacing is safe and can lead to significant improvement in LV function and dimensions. However, long-term follow-up assessment is needed for further evaluation.
References
Antretter H, Colvin J, Schweigmann U, Hangler H, Höfer D, Dunst K, Margreiter J, Laufer G (2003) Special problems of pacing in children. Indian Pacing Electrophysiol J 3:23
Batra AS, Wells WJ, Hinoki KW, Stanton RA, Silka MJ (2003) Late recovery of atrioventricular conduction after pacemaker implantation for complete heart block associated with surgery for congenital heart disease. J Thorac Cardiovasc Surg 125:1291–1293
Brockmeier K, Ulmer HE, Hessling G (2002) Termination of atrial reentrant tachycardias by using transesophageal atrial pacing. J Electrocardiol 35:159–163
Çeliker A, Başpınar O, Karagöz T (2007) Transvenous cardiac pacing in children: problems and complications during follow-up. Original investigation. Anadolu Kardiyol Derg 7:292–297
Doniger SJ, Sharieff GQ (2006) Pediatric dysrhythmias. Pediatr Clin North Am 53(1):85–105
Ector B, Willems R, Heidbüchel H, Gewillig M, Mertens L, Meyns B, Daenen W, Ector H (2006) Epicardial pacing: a single-centre study on 321 leads in 138 patients. Acta Cardiol 61:343–351
Fortescue EB, Berul CI, Cecchin F, Walsh EP, Triedman JK, Alexander ME (2004) Patient, procedural, and hardware factors associated with pacemaker lead failures in pediatrics and congenital heart disease. Heart Rhythm 1:150–159
Gebauer RA, Tomek V, Salameh A, Marek J, Chaloupecký V, Gebauer R, Matějka T, Vojtovič P, Janoušek J (2009) Predictors of left ventricular remodelling and failure in right ventricular pacing in the young. Eur Heart J 30:1097–1104
Gross GJ, Chiu CC, Hamilton RM, Kirsh JA, Stephenson EA (2006) Natural history of postoperative heart block in congenital heart disease: implications for pacing intervention. Heart Rhythm 3:601–604
Kammeraad JA, Rosenthal E, Bostock J, Rogers J, Sreeram N (2004) Endocardial pacemaker implantation in infants weighing < or = 10 kilograms. Pacing Clin Electrophysiol 27:1466–1474
Karpawich PP (2004) Chronic right ventricular pacing and cardiac performance: the pediatric perspective. Pacing Clin Electrophysiol 27:844–849
Kumor M, Baranowski R, Koźluk E, Walczak F (2010) Is the diagnostic function of pacemakers a reliable source of information about ventricular arrhythmias? Cardiol J 17:495–502
McLeod KA (2010) Cardiac pacing in infants and children. Heart 96:1502–1508
Nolasco RR, Avalos OM, Diez RG, Carrillo JC, Machuca RJ, De Haro S, Villacorta CH (2009) Transvenous pacing in children weighing less than 10 kilograms. Pacing Clin Electrophysiol 32:177–181
Odim J, Suckow B, Saedi B, Laks H, Shannon K (2008) Equivalent performance of epicardial versus endocardial permanent pacing in children: a single-institution and manufacturer experience. Ann Thorac Surg 85:1412–1416
Post MC, Budts W, Van de Bruaene A, Willems R, Meyns B, Rega F, Gewillig M (2011) Failure of epicardial pacing leads in congenital heart disease: not uncommon and difficult to predict. Neth Heart J 19(7–8):331–335
Sachweh JS, Vazquez-Jimenez JF, Schöndube FA, Daebritz SH, Dörge H, Mühler EG, Messmer BJ (2000) Twenty years experience with pediatric pacing: epicardial and transvenous stimulation. Eur J Cardiothorac Surg 17:455–461
Shalganov TN, Paprika D, Vatasescu R, Kardos A, Mihalcz A, Kornyei L, Szatmari A, Szili-Torok T (2007) Midterm echocardiographic follow-up of left ventricular function with permanent right ventricular pacing in pediatric patients with and without structural heart disease. Cardiovasc Ultrasound 5:13
Silvetti MS, Drago F (2006) Upgrading of VVIR pacemakers with nonfunctional endocardial ventricular leads to VDD pacemakers in adolescents. Pacing Clin Electrophysiol 29:691–696
Silvetti MS, De Santis A, Grovale N, Grutter G, Baccarini A, Drago F (2007) Ventricular pacing threshold variations in the young. Pacing Clin Electrophysiol 30:175–181
Soongswang J, Nana A, Laohaprasittiporn D, Durongpisitkul K, Chanthong P, KhaoSa-Ard B, Punlee K, Kangkakate C, Udompunturuk S (2005) Permanent cardiac pacing in pediatrics: experience in Thailand. J Med Assoc Thai 88:106–114
Toogood G (2007) Pacemaker therapies in cardiology. Aust Fam Physician 36:518–524
Udink ten Cate F, Breur J, Boramanand N, Crosson J, Friedman A, Brenner J, Meijboom E, Sreeram N (2002) Endocardial and epicardial steroid lead pacing in the neonatal and paediatric age group. Heart 88:392–396
Welisch E, Cherlet E, Crespo-Martinez E, Hansky B (2010) From pacing and clinical electrophysiology: a single-institution experience with pacemaker implantation in a pediatric population over 25 years. Pacing Clin Electrophysiol 33:1112–1118
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lotfy, W., Hegazy, R., AbdElAziz, O. et al. Permanent Cardiac Pacing in Pediatric Patients. Pediatr Cardiol 34, 273–280 (2013). https://doi.org/10.1007/s00246-012-0433-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-012-0433-2