Abstract.
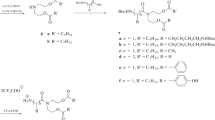
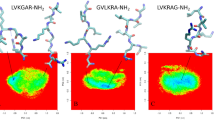
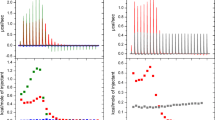
Lipopeptaibols are members of a novel group of naturally occurring, short peptides with antimicrobial activity, characterized by a lipophilic acyl chain at the N-terminus, a high content of the turn/helix forming α-aminoisobutyric acid and a 1,2-amino alcohol at the C-terminus. The amino acid sequences range from 6 to 10 residues and the fatty acyl moieties from 8 to 15 carbon atoms. The peptide portion of lipopeptaibols can be shorter than those of the nonlipidated peptaibols that range from 10 to 19 amino acid residues. The longest peptides fold into a mixed 310/α helix, whereas the shortest peptides tend to adopt a β-turn/sheet structure. Using solution methodologies, a series of analogues of trichogin GA IV was synthesized which allowed determination of the minimal lipid chain and peptide main-chain lengths for the onset of membrane activity and exploitation of a number of spectroscopic techniques aimed at determining its preferred conformation under a variety of conditions and investigating in detail its mode of interaction with, and its effect on, the phospholipid membranes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received 26 January 2001; received after revision 7 March 2001; accepted 15 March 2001
Rights and permissions
About this article
Cite this article
Toniolo, C., Crisma, M., Formaggio, F. et al. Lipopeptaibols, a novel family of membrane active, antimicrobial peptides. CMLS, Cell. Mol. Life Sci. 58, 1179–1188 (2001). https://doi.org/10.1007/PL00000932
Issue Date:
DOI: https://doi.org/10.1007/PL00000932