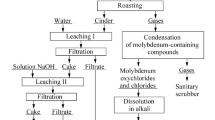
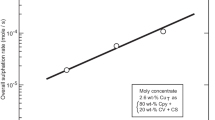
Abstract
Off-grade molybdenite concentrates, mainly derived from secondary sources as by-products of the processing of copper, uranium and tungsten ores, do not satisfy the purity requirements (percent MoS2) for the production of molybdenum bearing products. This investigation is a continuation of a successful effort to refine a low-grade molybdenite concentrate to prepare a high-grade MoS2 using a mixed acid (HF + HCl) leach procedure. Specifically, this investigation is concerned with an understanding of the separation chemistry underlying the removal of the oxide/silicate gangue and the base-metal sulfide associations present in the low-grade molybdenite by their selective dissolution in the two acids. Experiments were conducted on an off-grade concentrate containing 42.6% Mo. Results of the controlled dissolution tests indicate that both HCl and HF are suitable for the removal of the associated alumina and both work well for iron, nickel and copper. However, HF was found to be the most effective for dissolving silica/silicate and magnesia. An attempt was made to rank the leaching efficiencies of the acids used singly, sequentially or in a mixed mode. Scale-up (300 to 2,000 g) experiments were conducted to validate this approach. Excellent purification of the concentrate was possible by processing the off-grade concentrate with 10% HCl plus 15% HF at 363 K for 7.2 ks in open polypropylene reactors with internal stirring. These conditions leached practically all of the oxide and silicate gangue and 96% of the metallic impurities. Starting from the low-grade molybdenite concentrate (42.6% Mo; 71.1% MoS2) it was found feasible to obtain a refined product containing 97.8% MoS2, which is suitable for industrial use.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Forward, F.A., and Warren, I.H., 1960, “Extraction of metals from sulphides by wet methods,” Metallurgical Reviews, Vol. 5, pp. 137–164.
Jennings, P.H., Stanley, R.W., Ames, H.L., and Evans, J.I., 1974, “Developmentof a process for purifying molybdenite concentrate,” 2nd International Symposium on Hydrometallurgy, Published by AIME, New York, pp. 868–883.
Kumar, M., 2004, “Studies on the Upgrading (Refining) of a Low-grade Molybdenite Concentrate and Recovery of By-products,” PhD Thesis (Metallurgical Engineering), Institute of Technology, Banaras Hindu University, India, pp. 49–157.
Magyor, M.J., 2005, “Mineral Commodity Summaries,” U.S. Geological Survey, No. 1, pp. 113.
Rao, G.V., and Sastry, S.R.S., 1998, “Molybdenite recovery by column flotation in the byproduct recovery plant of UCIL, Jaduguda,” Metals Materials and Processes, Vol. 10, No. 2, pp. 119–126.
Mankhand, T.R., and Prasad, P.M., 1982, “Lime enhanced hydrogen reduction of molybdenite,” Metallurgical Transactions, Vol. 13B, pp. 275–282.
Natarajan, K.A., 1990, “Electrochemical aspects of bioleaching of base metal sulphides,” Chapter 4, in Microbial Mineral Recovery, Ehrilch, H.L., and Brierley, C.L., eds., McGraw-Hill Publishing Co., New York, pp. 79–106.
Prasad, P.M., Murthy, D.S.R., and Mankhand, T.R., 1999, “Project report on production of molybdenum carbide by direct synthesis from molybdenum sulphide,” submitted to The Council of Scientific and Industrial Research, Government of India, New Delhi.
Saha, A.K., Srinivasan, S.R., and Akrekar, D.D., 1985, “Removal of copper and nickel from low grade molybdenite concentrates,” NML Technical Journal, Vol. 27, pp. 40–45.
Ruiz, M.C., and Padilla, R., 1998, “Copper removal from molybdenite concentrate by sodium dichromate leachmg,” Hydrometallurgy, Vol. 48, No. 3, pp. 313–325.
Saha, A. K., Srinivasan, S.R., and Akrekar, D.D., 1985, “Acid treatment for purification and enrichment of low-grade molybdenite concentrate,” NML Technical Journal, Vol. 27, No. 3 and 4, pp. 46–55.
Saha, A.K., 1998, “Purification and enrichment of low grade molybdenite concentrates,” NML Technical Journal, Vol. 40, No. 1, pp. 17–21.
Spedden, H.R., Prater, J.D., Queneau, P.B., Foster, G.G., and Pickles, W.S., 1971, “Acid bake-leach-flotation treatment of offgrade molybdenite,” Metallurgical Transactions, Vol. 2, No. 9, pp. 3115–3122.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Prasad, P.M., Balasubramanian, K. & Mukhopadyay, R. Separation chemistry in the refining of an off-grade molybdenite concentrate by leaching with an acid mix. Mining, Metallurgy & Exploration 24, 97–104 (2007). https://doi.org/10.1007/BF03403364
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03403364