Summary
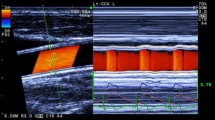
Wave intensity (WI) is a new hemodynamic index, which is defined as (dP/dt)(dU/dt) at any site of the circulation, where dP/dt and dU/dt are the time derivatives of blood pressure and velocity, respectively. Arterial WI in normal subjects has two positive sharp peaks. The first peak occurs during early systole when a forward-traveling compression wave is generated by the left ventricle. The magnitude of this peak increases markedly with an increase in cardiac contractility. The second peak, which occurs towards the end of systole, is caused by generation of a forward-traveling expansion wave by the ability of the left ventricle to actively stop aortic blood flow. The interval between the R wave of the ECG and the first peak of WI (R-lst peak interval) and the interval between the first and second peaks (lst–2nd interval) are approximately equal to the preejection period and left ventricular ejection time, respectively. Using a combined Doppler and echotracking system, we obtained carotid arterial WI non-invasively. We examined the characteristics of WI in 11 patients with mitral regurgitation (MR) before and after surgery, and 24 normal volunteers. In the MR group before surgery, the second peak was decreased and the (lst–2nd interval)/(R-R interval) ratio was reduced, compared with the normal group (140 ± 130 vs 750 ± 290mmHgm/s3, P < 0.0083; 20.7% ± 3.4% vs 26.7% ± 2.8%, P < 0.0083). There were no significant differences in the first peak between the normal group and the MR group before and after surgery. The second peak in the MR group was increased significantly (P < 0,016 vs before surgery) to 1150 ± 830mmHgm/s3 in the early period after surgery (stage I), and to 1090 ± 580mmHgm/s3 in the late period after surgery (stage II). These values did not differ significantly from that of the normal group. At stage I, the (R-1st peak interval)/(R-R interval) ratio was increased from 13.4% ± 2.7% to 2.6% ± 5.6% (P < 0.016 vs before surgery). At stage II, this ratio decreased to 16.2% ± 2.8% (P < 0.016 vs stage I), but was still significantly higher than that before surgery. The (1st–2nd inteval)/(R-R interval) ratio increased significantly after surgery (P < 0.016 vs before surgery) to values (27.0% ± 4.5% at stage I and 28.9% ± 2.6% at stage II) which did not differ significantly from that of the normal group. The recovery of the second peak after surgery suggests that the left ventricle had recovered the ability to actively stop aortic blood flow. Wave intensity is useful for analyzing changes in the working condition of the heart.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Parker KH, Jones CJH, Dawson JR, Gibson DG (1988) What stops the flow of blood from the heart? Heart Vessels 4:241–245
Parker KH, Jones CJH (1990) Forward and backward running waves in the arteries: analysis using the method of characteristics. ASME J Biomech Eng 112: 322–326
Jones CJH, Sugawara M, Davies RH, Kondoh Y, Uchida K, Parker KH (1994) Arterial wave intensity: physical meaning and physiological significance. In: Hosoda S, Yaginuma T, Sugawara M, Taylor MG, Caro CG (eds) Recent progress in cardiovascular mechanics. Harwood, Chur, pp 129–148
Sugawara M, Kondoh Y, Uchida K, Jones CJH (1995) Wave intensity: a new index of ventriculoarterial interacstion (in Japanese). BioMed Eng 9(7):22–27
Ramsey MW, Sugawara M (1997) Arterial wave intensity and ventriculoarterial interaction. Heart Vessels (suppl) 12:128–134
MacRae JM, Sun YH, Isaac DL, Dobson GM, Cheng CP, Little WC, Parker KH, Tyberg JV (1997) Wave-intensity analysis: a new approach to left ventricular filling dynamics. Heart Vessels 12:53–59
Koh TW, Pepper JR, DeSouza AC, Parker KH (1998) Analysis of wave reflections in the arterial system using wave intensity: a novel method for predicting the timing and amplitude of reflected waves. Heart Vessels 13:103–113
Sugawara M, Niki K (1999) A new hemodynamic index — wave intensity (in Japanese). Junkan Seigyo (Circ Control) 20:90–97
Barnett GO, Mallos AJ, Shapiro A (1961) Relationship of aortic pressure and diameter in the dog. J Appl Physiol 16:545–548
Patel DJ, de Freitas FM, Greenfield JC Jr, Fry DL (1963) Relationship of radius to pressure along the aorta in living dogs. J Appl Physiol 18:1111–1117
Sugawara M, Furuhata H, Kikkawa S, Suzuki S, Oonishi T, Takabayashi T, Suzuki N, Kurokawa T, Yoshimura S, Caro CG (1983) Development of a noninvasive method of measuring blood pressure wave (in Japanese). Iyodenshi to Seitaikogaku (Jpn J Med Electro Biol Eng) 21 (suppl): 429
Fujishiro K, Yoshimura S (1982) Development of ultrasonic quantitative blood flow measurement system (QFM). Jikeikai Med J 29:57–74
Jones CJH, Sugawara M (1993) “Wavefronts” in the aorta — implications for the mechanisms of left ventricular ejection and aortic valve closure. Cardiovasc Res 27:1902–1905
Sugawara M, Uchida K, Kondoh Y, Magosaki N, Niki K, Jones CJH, Sugimachi M, Sunagawa K (1997) Aortic blood momentum — the more the better for the ejecting heart in vivo? Cardiovasc Res 33:433–446
Sugawara M, Uchida K, Kondoh Y, Jones CJH (1996) Cardiac utilization of the momentum of blood. In: Sasayama S (ed) New horizons for failing heart syndrome. Springer, Berlin Heidelberg New York, Tokyo, pp 69–77
Tavel ME (1967) Clinical phonocardiography and external pulse recording. Year Book, Chicago, pp 277–278
Wanderman KL, Goldberg MJ, Stack RS, Weissler AM (1976) Left ventricular performance in mitral regurgitation assessed with systolic time intervals and echocardiography. Am J Cardiol 38:831–835
Schuler G, Peterson KL, Johnson A, Francic G, Dennish G, Utley J, Daily PO, Ashburn W, Ross J Jr (1979) Temporal response of left ventricular performance to mitral valve surgery. Circulation 59:1218–1231
Boucher CA, Bingham JB, Osbakken MD, Okada RD, Strauss HW, Block PC, Levine FH, Phillips HR, Pohost GM (1981) Early changes in left ventricular size and function after correction of left ventricular volume overload. Am J Cardiol 47:991–1004
Abe T, Tsukamoto T, Urita R, Komatsu S (1995) Reconstructive surgery for mitral valve regurgitation in 64 patients: postoperative results of each different types of surgical methods (in Japanese). Kyobu Geka 48:650–657
Harpole DH, Gall SA, Wolfe WG, Rankin JS, Jones RH (1996) Effects of valve replacement on ventricular mechanics in mitral regurgitation and aortic stenosis. Ann Thorac Surg 62:756–761
Gillebert TC, Sys SU, Brutsaert DL (1989) Influences of loading patterns on peak length-tension relation and on relaxation in cardiac muscle. J Am Coll Cardiol 13:483–490
Harada Y, Sugawara M, Beppu T, Higashidate M, Nakata S, Imai Y (1987) Principle of a noninvasive method of measuring Max(dP/dt) of the left ventricle: theory and experiments. Heart Vessels 3:25–32
van den Bos GC, Westerhof N, Randall OS (1982) Pulse wave reflection: can it explain the differences between systemic and pulmonary pressure and flow waves? A study in dogs. Circ Res 51:479–485
Author information
Authors and Affiliations
Additional information
K.N. and M.S. were supported by Grants-in-Aid for Scientific Research (B)-11695092 and (C)-11670713 from the Ministry of Education, Science, Sports and Culture of Japan.
Rights and permissions
About this article
Cite this article
Niki, K., Sugawara, M., Uchida, K. et al. A noninvasive method of measuring wave intensity, a new hemodynamic index: application to the carotid artery in patients with mitral regurgitation before and after surgery. Heart and Vessels 14, 263–271 (1999). https://doi.org/10.1007/BF03257237
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03257237